Abstract
Background
Urological cancers occur worldwide. Many factors, among which the catechol-O-methyltransferase (COMT) Val158Met polymorphism, are said to be associated with the cancer risk. We conducted a meta-analysis to investigate the association between urological cancer susceptibility and COMT Val158Met in different genetic models.
Methods
This study was based on material obtained from the PubMed, HuGENet and Embase databases. Four models including dominant (AA + AG vs. GG), recessive (AA vs. AG + GG), codominant (AA vs. AG, AA vs. GG) and per-allele analysis (A vs. G) were applied. Odds ratios (OR) and the corresponding 95% confidence intervals (CI) were used to evaluate the power of the associations.
Results
Fourteen eligible studies comprising 3,285 cases and 3,594 controls were included. Although we could not detect a positive function of the COMT Val158Met polymorphism in urological cancers, the polymorphism might be significantly associated with bladder cancer risk (dominant model [AA + AG vs. GG]: OR = 0.736, 95% CI = 0.586-0.925, I2 = 0.00%; recessive model [AA vs. AG + GG]: OR = 0.822, 95%CI = 0.653-1.035, I2 = 6.30%; codominant model [AA vs. AG]: OR = 0.908, 95% CI = 0.710-1.161, I2 = 0.00%; codominant model [AA vs. GG]: OR = 0.693, 95% CI = 0.524-0.917, I2 = 30.20%; allele analysis [A vs. G]: OR = 0.826, 95%CI = 0.717-0.951, I2 = 30.20%). The same significant associations were not found for kidney cancer and prostate cancer risk in different ethnicities. There also seemed to be no distinct effect of the polymorphism on benign prostatic hyperplasia.
Conclusions
We suggest that bladder cancer but not prostate cancer and kidney cancer could be significantly associated with the Val158Met polymorphism. Interaction of COMT genetic and related environmental factors for urological cancers should not be ignored in future.
Introduction
Urological cancers are universal worldwide and include many different types; among these types, kidney cancer, bladder cancer and prostate cancer are probably the major causes of morbidity and mortality, causing massive loss to the economy and public health (1). It was estimated that worldwide >200,000 new cases with high death rate occurred every year (2). The high morbidity and mortality was also happened in the United States (3). Moreover, only in 2013, kidney cancer was diagnosed among 40,430 men and 24,720 women (4). Like other common types of urological cancer, bladder cancer and prostate cancer affect vast populations around the world (2, 5). Many risk factors such as lifestyle, environmental risks and genetic elements are considered among the etiologies for urological cancers; among these factors, genetic susceptibility plays a key role (5-6-7). Recently, studies have suggested that the catechol-O-methyltransferase (COMT) Val158Met polymorphism might be significantly associated with urological cancers (8). So this study was conducted to investigate this.
The COMT gene is located on 22q11.21 and encodes the enzyme mainly responsible for the degradation of catecholamines, such as dopamine and noradrenaline (9), catalyzing the O-methylation of 2-hydroxyestradiol to yield 2-ME2 (10). In addition, it is involved in the inactivation of potential carcinogenic compounds that produce inflammation and catechol estrogens, which have the ability to damage DNA and take part in tumorigenesis (11-12-13). As the major polymorphism, the Val158Met mutation of COMT was said to be associated with many diseases, especially cancers. In 2008, Hirata et al (14) suggested that the COMT Val158Met polymorphism might alter protein expression in endometrial cancer. Sazci et al (15) studied this polymorphism in breast cancer and discovered significant associations. Some studies also presented the potential function of COMT Val158Met polymorphism in the development of urological cancers. In 2006, Tanaka et al (8) investigated benign prostatic hyperplasia (BPH) and sporadic prostate cancer; the results suggested that COMT was associated with sporadic prostate carcinogenesis. They also identified a significant association between the COMT gene and renal cell carcinoma (16). However, the conclusions were controversial (17). On the basis of recent studies on the functions of the COMT gene and the inconsistent results obtained so far, this meta-analysis was conducted to investigate the association between cancer susceptibility and COMT Val158Met in different genetic models. Our study should give us a comprehensive view of the associations between the COMT Val158Met polymorphism and the 3 main urological cancers (kidney cancer, bladder cancer and prostate cancer), which could provide the theoretical basis for further studies.
Materials and methods
Study Collection and Literature Search
The literature retrieval was mainly based on 3 databases: PubMed, HuGENet and Embase. Comprehensive keywords were applied: “catechol-O-methyltransferase”, “COMT”, “renal carcinoma”, “kidney cancer”, “kidney carcinoma”, “renal cancer”, “renal cell carcinoma”, “RCC”, “Pca”, “prostate cancer”, “prostatic carcinoma”, “prostate carcinoma”, “bladder cancer”, “bladder carcinoma”, “carcinoma of urinary bladder”, “carcinoma of bladder”, “bladder neoplasm”. Additional studies were collected from the reference lists of identified articles. Language was restricted to English. Our study was updated in September 2015. We screened studies according to the following inclusion criteria: i) case-control studies; ii) studies investigating the association between the COMT Val158Met polymorphism and the 3 main urological cancers (kidney cancer, bladder cancer, prostate cancer). Studies were excluded for the following reasons: i) the study was designed as case-only, including only subjects with urological cancers and no other contrastive specimens; ii) the raw data for the number of every genotype of the COMT Val158Met polymorphism in case and control groups could not be extracted; iii) review papers; iv) duplicate publications.
To describe the features of eligible studies the following information was recorded: first author's name, year of publication, country of the subjects, ethnicity (categorized as Caucasian, Asian or mixed), types of cancer (kidney cancer, bladder cancer, prostate cancer), methods for experiments, characteristics of case and control groups.
Genotype Model Selection and Result Evaluation
Three common genotype models and allele analysis were applied in the study including a dominant model (AA + AG vs. GG), a recessive model (AA vs. AG + GG), a codominant model (AA vs. AG, AA vs. GG) and per-allele analysis (A vs. G). For the selection of effect models, the value of I2 was taken as the criterion. When I2 was <50, a fixed-effects model was selected; in all other cases a random-effects model was used. In the whole analysis, odds ratios (OR) and the corresponding 95% confidence intervals (CI) were used to evaluate the power of associations. The effects incorporated an estimate of the interstudy variance and provided wider 95% CIs if the results of the constituent studies differed among themselves. The chi-square-based Q statistic (p<0.10 as the standard) was applied to estimate the heterogeneity of results (18). The index represented the weighted sum of the squared difference in the overall effect sizes from each study. Subgroup analysis was carried out based on the same characteristics in every study, such as ethnicity, control sample, etc. The subjects of our eligible studies mainly came from 8 countries (Egypt, France, Italy, Turkey, Iran, Japan, UK and USA), and could be divided into Caucasian, Asian and mixed ethnicities.
Subgroup Analysis
Our study focused on the association between the COMT Val158Met polymorphism and 3 urological cancers: kidney cancer, bladder cancer and prostate cancer. Firstly, urological cancer was treated as an independent variable. According to the type of cancer, patients were divided into subgroups. The results would give us a view of the association between the COMT Val158Met polymorphism and the different urological cancers. After analysis, a significant association was found only in the bladder cancer subgroup. In 2009, Omrani et al (19) conducted a study about the association between COMT158 Val/Met polymorphism and prostate cancer and BPH, which suggested that activity of the COMT gene plays a possible role in the development of BPH but not prostate cancer. So, we speculated that the relationship between BPH and COMT would be one of confounding factors. As the other cause, the Hardy-Weinberg equilibrium (HWE) of every paper would influence the results. So, in order to reduce the heterogeneity, we tried to conduct a special analysis in which studies not satisfying the HWE were removed. Subjects with BPH in the control group were also removed to ensure that in all studies the cases were patients with prostate cancer and controls were healthy men or patients with diseases other than BPH. In this special analysis, 1 study was excluded for not satisfying HWE (19).
Funnel plots and Begg's unweighted regression test were used to assess the publication bias. All allele frequencies were calculated according to genotype data. Stata 9.0 (Stata Corporation) was used for statistical analysis, and all p values were 2-tailed.
Results
Eligible Studies
Using all the keywords, we searched the 3 databases (PubMed, HuGENet and Embase) comprehensively. A total of 202 papers came to our view; 36 studies were concerned with kidney cancer, 134 with prostate cancer and 32 with bladder cancer. Sixty-five duplicative studies were removed. After reading the abstracts we excluded 115 studies (23 on kidney cancer, 73 on prostate cancer and 19 on bladder cancer) because they provided no evidence of studying the association between COMT Val158Met polymorphism and the 3 urological cancers. Eight further studies were excluded for the following reasons: i) 2 studies about the association between COMT and bladder cancer lacked raw data on genotype frequencies (20, 21); ii) 1 study mainly studied the COMT polymorphism and prostate cancer among prostate cancer cases without any control group (22); iii) 3 papers were previously conducted meta-analyses (23-24-25); iv) 1 study investigated genetic variations and prostate cancer risk. However, no genotype data were used in our analysis (26); v) 1 study investigated the COMT polymorphism and renal cell carcinoma without available genotype frequencies for every variation (27). Finally, 14 studies about the association between COMT polymorphism and urological cancers comprising 3,285 cases and 3,594 controls remained for further analysis (8, 16-17, 19, 28-29-30-31-32-33-34-35-36-37).
Among these eligible studies, 4 were about bladder cancer (28-29-30-31), 9 about prostate cancer (8, 17, 19, 32-33-34-35-36-37) and 1 about renal cell cancer (16). Except for 1 study with a mixed population (35), 7 studies included subjects that were Caucasian (17, 28-29-30-31, 33, 34) and 6 included Asian subjects (8, 16, 19, 32, 36, 37). The cases were mainly defined as having histologically confirmed urological cancers (bladder, prostate or kidney cancer). Controls were healthy men or patients with diseases other than cancer. Only 2 studies had BPH patients in their control groups (8, 19). The characteristics are summarized in Table I.
The baseline features included in our study for the urological cancers (bladder cancer, prostate cancer, kidney cancer)
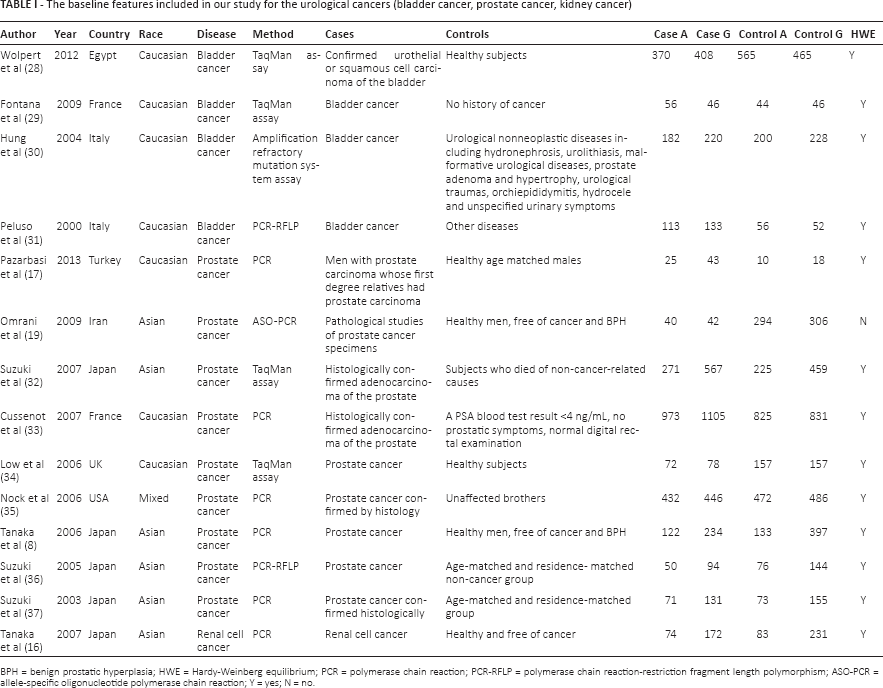
BPH = benign prostatic hyperplasia; HWE = Hardy-Weinberg equilibrium; PCR = polymerase chain reaction; PCR-RFLP = polymerase chain reaction-restriction fragment length polymorphism; ASO-PCR = allele-specific oligonucleotide polymerase chain reaction; Y = yes; N = no.
COMT Val158Met Polymorphism Associated with Bladder Cancer
In order to view the association between the COMT Val158Met polymorphism and urological cancers comprehensively, a pooled analysis was performed on the data for the 3 cancers (bladder, prostate and kidney) in which every type of cancer was treated as 1 subgroup. The results did not reveal a significant association between the COMT Val158Met polymorphism and urological cancers overall (dominant model [AA + AG vs. GG]: OR = 0.952, 95%CI = 0.855-1.059, I2 = 28.50%; recessive model [AA vs. AG + GG]: OR = 0.907, 95%CI = 0.800-1.027, I2 = 30.60%; codominant model [AA vs. AG]: OR = 0.935, 95%CI = 0.819-1.067, I2 = 10.00%; codominant model [AA vs. GG]: OR = 0.868, 95%CI = 0.749-1.005, I2 = 42.80%; allele analysis [A vs. G]: OR = 0.949, 95%CI = 0.885-1.018, I2 = 43.10%). In order to reduce the heterogeneity, subgroup analyses were performed. A negative relationship was also observed for prostate cancer and kidney cancer. However, the COMT Val158Met polymorphism was found to be associated with bladder cancer (dominant model [AA + AG vs. GG]: OR = 0.736, 95%CI = 0.586-0.925, I2 = 0.00%; recessive model [AA vs. AG + GG]: OR = 0.822, 95%CI = 0.653-1.035, I2 = 6.30%; codominant model [AA vs. AG]: OR = 0.908, 95%CI = 0.710-1.161, I2 = 0.00%; codominant model [AA vs. GG]: OR = 0.693, 95%CI = 0.524-0.917, I2 = 30.20%; allele analysis [A vs. G]: OR = 0.826, 95%CI = 0.717-0.951, I2 = 30.20%). So the bladder cancer risk might be influenced by the COMT Val158Met polymorphism (Fig. 1, Tab. II).
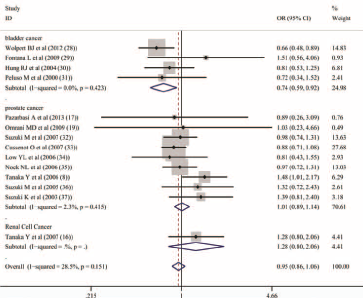
Meta-‐analysis with fixed effects and Dominant model (AA + AG vs GG) for the association between COMT Val158Met polymorphism and the urologic neoplasms. The first author and year of publication for each study was shown. In this analysis, three subgroups didvided by types of cancers were shown: bladder cancer, prostate cancer and renal cell cacner. OR and accompanying 95% CI were also presneted in this analysis.
Association between COMT Val158Met polymorphism and urological neoplasms with 4 different genotype models applied (dominant, recessive and codominant models and allele analysis)
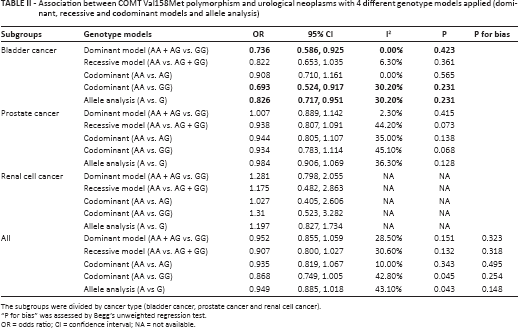
The subgroups were divided by cancer type (bladder cancer, prostate cancer and renal cell cancer).
“P for bias” was assessed by Begg's unweighted regression test.
OR = odds ratio; CI = confidence interval; NA = not available.
Prostate Cancer, BPH and COMT Val158Met Polymorphism
Considering the negative association between prostate cancer and the COMT Val158Met polymorphism and the confounding factors in a number of studies we included, further analysis was done. In this section, the HWE and compositions of subjects for every study were taken into consideration. After analysis, no association for prostate cancer risk was observed either among Caucasian or Asians (Fig. 2, Tab. III). On the basis of the conclusion that the COMT Val158Met polymorphism might affect the BPH risk (19), the genotype data of the healthy and BPH controls were extracted. However, we also could not discover any significant function of the COMT Val158Met polymorphism in BPH (dominant model [AA + AG vs. GG]: OR = 0.902, 95%CI = 0.578-1.404, I2 = 0.00%; recessive model [AA vs. AG + GG]: OR = 1.138, 95%CI = 0.469-2.761, I2 = 0.00%; codominant model [AA vs. AG]: OR = 1.192, 95% CI = 0.481-2.953, I2 = 0.00%; codominant model [AA vs. GG]: OR = 0.990, 95% CI = 0.378-2.596, I2 = 0.00%; allele analysis [A vs. G]: OR = 0.977, 95% CI = 0.757-1.260, I2 = 0.00%).
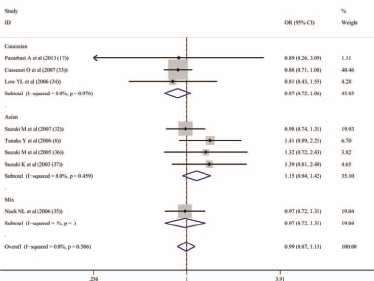
Meta-‐analysis with fixed effects and Dominant model (AA + AG vs GG) for the association between COMT Val158Met polymorphism and prostate cancer after removing the study unsatisfied Hardy Weinberg Equilibrium (HWE). The first author and year of publication for each study was shown. In this analysis, three subgroups were shown: Caucasuan, Asian and Mixes. OR and accompanying 95% CI were also presneted in this analysis.
Association between prostate cancer and COMT Val158Met polymorphism with 4 different genotype models (dominant, recessive and codominant models and allele analysis) after removing studies that did not satisfy the Hardy-Weinberg equilibrium and studies that included BPH subjects
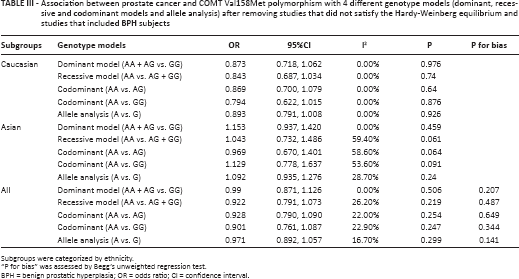
Subgroups were categorized by ethnicity.
“P for bias” was assessed by Begg's unweighted regression test.
BPH = benign prostatic hyperplasia; OR = odds ratio; CI = confidence interval.
Discussion
Urological cancers are terrible diseases affecting a large number of people in many populations. Etiological studies have been conducted recently in which genetic factors were found to play a key role. The COMT Val158Met polymorphism might be significantly associated with urological cancers (8), but there have been contrasting viewpoints. In order to investigate the real association, this study was performed. After comprehensive retrieval and analysis, our results showed that the COMT Val158Met polymorphism might be significantly associated with bladder cancer. No significant association was found in the other urological cancers studied. After further analysis, the polymorphism proved also not related to the BPH risk.
COMT is an important gene with 2 main functions. First, it is involved in the inactivation of the catecholamine neurotransmitters (9). Second, it can inactivate potentially carcinogenic compounds to some extent (11-12-13). Some cancers such as endometrial cancer (14) and breast cancer (15) were found to be associated with COMT Val158Met polymorphism. The association between urological cancers and the COMT Val158Met polymorphism has also been studied recently (8). Based on these studies, the present analysis was performed. The cancer group was divided into bladder cancer, prostate cancer and kidney cancer. To our delight, a significant function of the COMT Val158Met polymorphism in bladder cancer emerged, which suggested that this polymorphism might to some extent be a protective factor for bladder cancer. Although most studies about the COMT gene in bladder cancer did not detect any positive association (29, 30), Wolpert et al (28) in 2012 studied the relation of the COMT Val158Met polymorphism to bladder cancer in different sexes; the results suggested that the association was statistically significant for squamous cell carcinoma of the bladder. The authors found that intermediate- or low-activity enzyme (Val/Met [heterozygote: AG] or Met/Met [homozygous mutant: AA]) might be a protective factor in the development and progression of bladder cancer. As a vital gene, COMT encodes an enzyme that can catabolize estradiol metabolites into inactive compounds and affect normal biological function (38-39-40). In 2004, Eriksson et al (41) suggested that a low-activity genotype of COMT (Met/Met [homozygous mutant: AA]) was significantly associated with serum estradiol. In addition, it has been said that estrogens could protect against bladder cancer (42, 43). Meanwhile, among postmenopausal women who did not have high levels of estradiol with a low-activity COMT genotype, the significant association of COMT and bladder cancer was absent (28). Based on these findings, we believe that the COMT Val158Met polymorphism could increase the level of estradiol and protect against bladder cancer to some extent. Other factors, such as the environment, diet and hereditary factors, could also play a key role in the development of bladder cancer. So, further research should be focused on the interaction of estrogen, environment, diet, gender, histological types of cancer and other related gene polymorphisms, which would help us understand bladder cancer more comprehensively.
Considering the adequate numbers of studies on the association between prostate cancer and COMT genotypes, we performed further analysis to identify the real relation. Regrettably, no positive associations were detected, even when we removed those studies not satisfying HWE or including BPH subjects in the control group (8, 19). In the next analysis, the case group consisted of men with BPH and the control group were healthy men without any cancers. The results revealed no significance of COMT in BPH risk. These negative associations had also been shown in previous studies (17, 19, 32-33-34, 37), with samples from Turkey, Iran, Japan, France and the UK (including Asians and Caucasians). Two of these studies suggested a weak association between the COMT polymorphism and prostate cancer (17, 37), and 2 studies on Asians suggested that the COMT polymorphism might be related to prostate cancer risk (8, 36). Although a significant association was discovered in Asians recently, we also believed the COMT polymorphism might be correlated with prostate cancer in different ethnicities, considering its function. COMT can catalyze the methylation of the hydroxyl compounds of estrogens. The COMT Val158Met polymorphism would lead enzyme activity to be low, which would induce a high-estrogen environment (23). In 2015, Mishra et al (24) suggested that estrogen and estrogen receptor alpha could promote the development and progression of prostate cancer. Antiestrogens or estrogen receptor blockage would repress the tumorigenicity of prostate cancer (25). So, the significant function of the COMT Val158Met polymorphism in prostate cancer could also not be ignored. Compared with a previous meta-analysis that had found a significant association between the COMT Val158Met polymorphism and prostate cancer (44), our study might present more comprehensive results. Firstly, we updated the paper recently, with more studies included. Additionally, a study conducted by Omrani et al (19) did not satisfy the HWE, which might have influenced the results; however, this factor was not taken into account. Although there have been positive results before, we did not repeat the conclusion after considering all the confounding factors. According to our results, there was a weak association between the COMT Val158Met polymorphism and prostate cancer. However, the positive association was not obvious, which could be related to other confounding factors such as the number of samples, the age of the study participants, geographical areas, working environment, and so on. Further research should therefore focus on the interactions of COMT gene variants and environmental factors for the prostate cancer risk especially.
Limitations
Some limitations should be pointed out in order to evaluate our study comprehensively: i) there might be additional eligible studies in the other databases, except for the PubMed, HuGENet and Embase applied in this study, which would induce some basis for the results; ii) we could not provide any clear conclusion in relation to kidney cancer because there was only 1 eligible study included; iii) we have drawn our conclusions on the basis of the COMT gene function, recent studies and our own results. The speculation might have some limitations.
Conclusions
Urological cancers are ubiquitous worldwide, affecting many populations. The COMT Val158Met polymorphism might be one of the genetic factors involved. The results of our meta-analysis suggest that bladder cancer instead of prostate cancer and kidney cancer could be significantly associated with the Val158Met polymorphism of the COMT gene. Further research should focus on the interaction of COMT genetic and related environmental factors with the prostate cancer risk.
Footnotes
Financial support: This study was supported by The Science and technology development plan of Guangxi (2013BC26299).
Conflict of interest: The authors have no financial interest related to this study to disclose.
