Abstract
Background:
There is contradicting evidence on vitamin D levels and cancer mortality rates. In this study, we aimed to evaluate the impact of baseline vitamin D level on the outcome in patients with epidermal growth factor receptor (EGFR)-mutant advanced non–small-cell lung cancer (NSCLC) who received either gefitinib or gefitinib with chemotherapy (pemetrexed and carboplatin) as first-line therapy in a prospective randomized study.
Methods:
This was a post hoc analysis of a phase III randomized trial comparing gefitinib with gefitinib with carboplatin and pemetrexed in patients with advanced NSCLC with activating EGFR mutations in the first-line setting. As a part of regular practice, baseline vitamin D levels were measured using circulating 25(OH) levels in blood. We included 334 patients who had baseline vitamin D levels in the study and evaluated the effect of the vitamin D level on oncologic outcomes.
Results:
There were 136 (40.7%) patients with a sufficient (>20 ng/mL) baseline vitamin D level, and 198 (59.3%) patients who were deficient in vitamin D (<20 ng/mL). The median progression-free survival (PFS) in patients with normal vitamin D levels was 17 months, whereas that in patients with deficient vitamin D levels was 15 months, with a hazard ratio of 1.45 (95% confidence interval [CI] = 1.03-2.06). The median overall survival (OS) in patients with normal vitamin D levels was 28.6 months, whereas that in patients with deficient vitamin D levels was 28.5 months, with a hazard ratio of 1.17 (95% CI = 0.81-1.68). On multivariate analysis, only 2 factors impacted the PFS, the baseline vitamin D level, and the treatment regimen; other factors like age, sex, disease stage, and performance status did not.
Conclusions:
Baseline vitamin D levels have a significant impact on PFS, whereas OS is not affected by the baseline vitamin D levels on patients receiving targeted therapy for EGFR-mutant lung cancer.
Trial registration:
The trial was prospectively registered with the Clinical Trial Registry of India, registration number CTRI/2016/08/007149. The date of the registration was 5 August 2016.
Introduction
Vitamin D, commonly known to be an essential nutrient, is a precursor of a potent steroid hormone, calcitriol, which regulates a broad spectrum of physiological processes. In addition to the traditional roles of vitamin D like bone metabolism, studies have suggested that vitamin D may play a vital role in prevention and treatment of cancer.1-4 Several cancer cell studies and animal studies have shown that biological activities of vitamin D might slow or prevent the development of cancer. 5
Vitamin D deficiency has been implicated as a risk factor for prostate cancer. 6 In various cell culture studies, vitamin D metabolites have had protective action against cancer development. 7 Studies have shown vitamin D levels to have an inverse relation with cancer mortality, whereas others have regarded it a potential risk factor. 8 With regard to breast cancer-specific outcome, Rose et al 9 previously reported that women with vitamin D deficiency were at an increased risk of distant recurrence and death. In this study, this correlation was confirmed, although the effect was less significant.
Certain studies have shown that vitamin D supplementation could decrease cancer mortality, but the evidence is weak. A meta-analysis done by Keum et al 10 reported that vitamin D supplementation was associated with 13% reduction in cancer mortality over 3 to 10 years of follow-up, but the results were not confirmatory.
Lung cancer is the most common cancer worldwide, accounting for 1.2 million new cases annually and responsible for 18% of all cancer deaths. 11 In vitro studies have demonstrated that similar to other cancer cell lines, vitamin D and various analogs are capable of inhibiting lung cancer cell proliferation.12, Pilz et al 13 demonstrated that increased circulating 25-hydroxyvitamin D (25[OH]D) was associated with improved survival in patients with lung cancer. A study done by Kilkkinen et al 11 showed that although there was no overall association between 25(OH)D level and lung cancer risk, women, and younger participants with a higher circulating level of vitamin D had a lower risk of lung cancer. In a study done by Tadashi Akiba, the 5-year relapse-free survival (RFS) in the vitamin D and placebo groups was 65% and 57%, respectively, and the 5-year overall survival (OS) in the vitamin D and placebo groups was 76% and 78%, respectively. 14
Thus, there is contradictory evidence on the impact of vitamin D levels, both on cancer risk and prognosis. 15 Through this study, we aimed to evaluate the impact of baseline vitamin D level on the outcome in patients with epidermal growth factor receptor (EGFR)-mutant advanced non–small-cell lung cancer (NSCLC) who received either gefitinib or gefitinib with chemotherapy (pemetrexed and carboplatin) as a first-line therapy in a prospective randomized study. 16
Methodology
This was a post hoc analysis of the vitamin D levels of the patients from the trial titled, “A randomized, open-label, phase III study compared gefitinib with gefitinib with carboplatin and pemetrexed in patients with advanced NSCLC with activating EGFR mutations in the first-line setting.” We assessed the baseline vitamin D levels and compared its effect on the outcome of the patients enrolled in the trial. All patients who had vitamin D levels examined were included in this study. As a part of regular practice, baseline vitamin D levels were measured using circulating 25(OH) levels in blood by the automated chemiluminescence micro-particle immunoassay (CMIA) method.
The details of the original study have already been published. 16 Briefly, it was a randomized, open-label, phase III study comparing gefitinib with gefitinib with carboplatin and pemetrexed in patients with advanced NSCLC with activating EGFR mutations in the first-line setting. The study was approved by the Institutional Ethics Committee and monitored by the Data Safety Monitoring Subcommittee. The study was conducted according to the principles laid down by the International Conference on Harmonization Good Clinical Practice guidelines, the Declaration of Helsinki, the New Drugs and Clinical Trials Rules, 2019 and the guidelines established by the Indian Council of Medical Research. The study was prospectively registered with the Clinical Trial Registry of India, registration number CTRI/2016/08/007149. All patients provided written informed consent.
Patient population
Patients of age group 18 years and above and had histologically or cytologically confirmed NSCLC with EGFR mutations in exons 19, 21, or 18 and an Eastern Cooperative Oncology (ECOG) performance status (PS) of 0 to 2 were enrolled. Patients in the study either received gefitinib (250 mg orally daily) or gefitinib (250 mg orally daily) + pemetrexed (500 mg/m2 intravenously on day 1 every 21 days) + carboplatin (area under the curve 5, on day 1 every 21 days); pemetrexed and carboplatin were administered for 4 cycles, followed by maintenance pemetrexed in patients who did not have disease progression. For the purpose of this post hoc analysis, all patients who had vitamin D level examined were included in this study. Vitamin D levels had not been not measured for 9 male patients and 7 female patients. Hence, these patients were not included for analysis.
As a part of the routine procedure, baseline vitamin D levels were measured for the patients using 25(OH)D levels in circulation using the automated CMIA method. Based on the previous literature, 20 ng/mL was considered the normal level for vitamin D in our population. This cut-off value was used to divide patients into 2 groups. The decision regarding supplementation of vitamin D was at the discretion of treating physician.
Statistical analysis
Statistical analysis was done using IBM SPSS Statistics version 21 (IBM Corp, Armonk, New York) and R (version 4.1; R Foundation for Statistical Computing).
Descriptive statistics was used for demographic data analysis. The Kaplan-Meier curve was plotted for progression-free survival (PFS) and OS for both groups of patients.17,18 These curves were compared between the 2 group by the log rank test. P < .05 was considered statistically significant. We used the Cox regression analysis for performing the multivariate analysis.19,20 We assessed the effect of various factors including age (<60 years vs 60 years and over), sex (male vs female), PS (0-1 vs 2), treatment regimen (gefitinib vs gefitinib + pemetrexed + carboplatin), disease stage (III vs IV), and baseline vitamin D level (sufficient vs deficient) using the Cox regression analysis for PFS.
Results
Between August 2016 and August 2018, 350 patients were recruited; 174 patients were assigned to the gefitinib with carboplatin and pemetrexed arm, and 176 were assigned to the gefitinib arm. As a part of regular practice baseline vitamin D levels were measured using circulating 25(OH) levels in blood for 334 patients (baseline vitamin D level was not measured for 6 patients assigned to gefitinib with carboplatin and pemetrexed therapy, and 10 patients assigned to gefitinib therapy). There were 136 (40.7%) patients with a sufficient baseline vitamin D level, and 198 (59.3%) patients who were deficient in vitamin D. The median baseline vitamin D level was 17.5 ng/mL (interquartile range = 11.6-25.9).
Vitamin D supplementation was administered to 25 patients (12.6%) who had vitamin D levels below 20 ng/mL and to 6 patients with levels between 20 and 30 ng/mL. No vitamin D supplementation was administered to 173 patients (87.4%) who had deficient baseline vitamin D levels. .
Demographic and baseline clinical characteristics
The patients’ baseline characteristics are listed in Table 1.
Baseline characteristics.

The median age of the patients was 55 years, ranging from 27 to 78 years. We included 172 (51.5%) male patients and 162 (48.5%) female patients in this analysis. Vitamin D supplementation was administered to 29 patients; 230 patients did not receive vitamin D supplementation and 73 patients for whom the vitamin D supplementation was not applicable.
Progression-free survival
There were 228 (68.2%) patients who had an event for PFS. The median PFS in patients with normal vitamin D level was 17 months (95% confidence interval [CI] = 15-21), whereas the median PFS of patients with deficient vitamin D level was 15 months (95% CI = 13-17); hazard ratio (HR) of 1.45 (95% CI = 1.03-2.06) as seen in Figure 1. On a multivariate analysis, we found that only the treatment regimen and the baseline vitamin D level had a significant impact on PFS, whereas age, sex, stage, and PS did not (Figure 2).
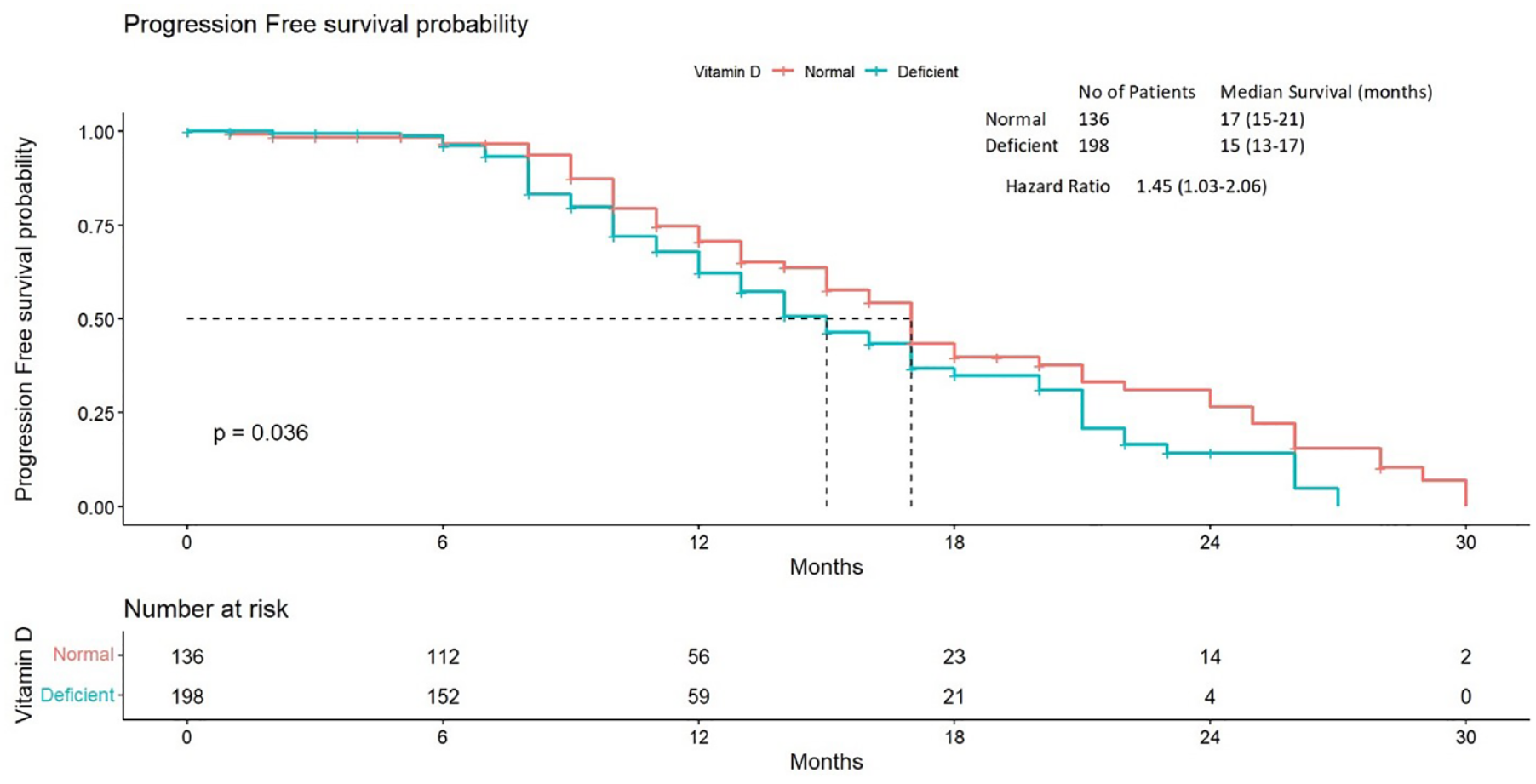
Kaplan-Meier plot for progression-free survival comparing normal and deficient levels of vitamin D using the log rank test.
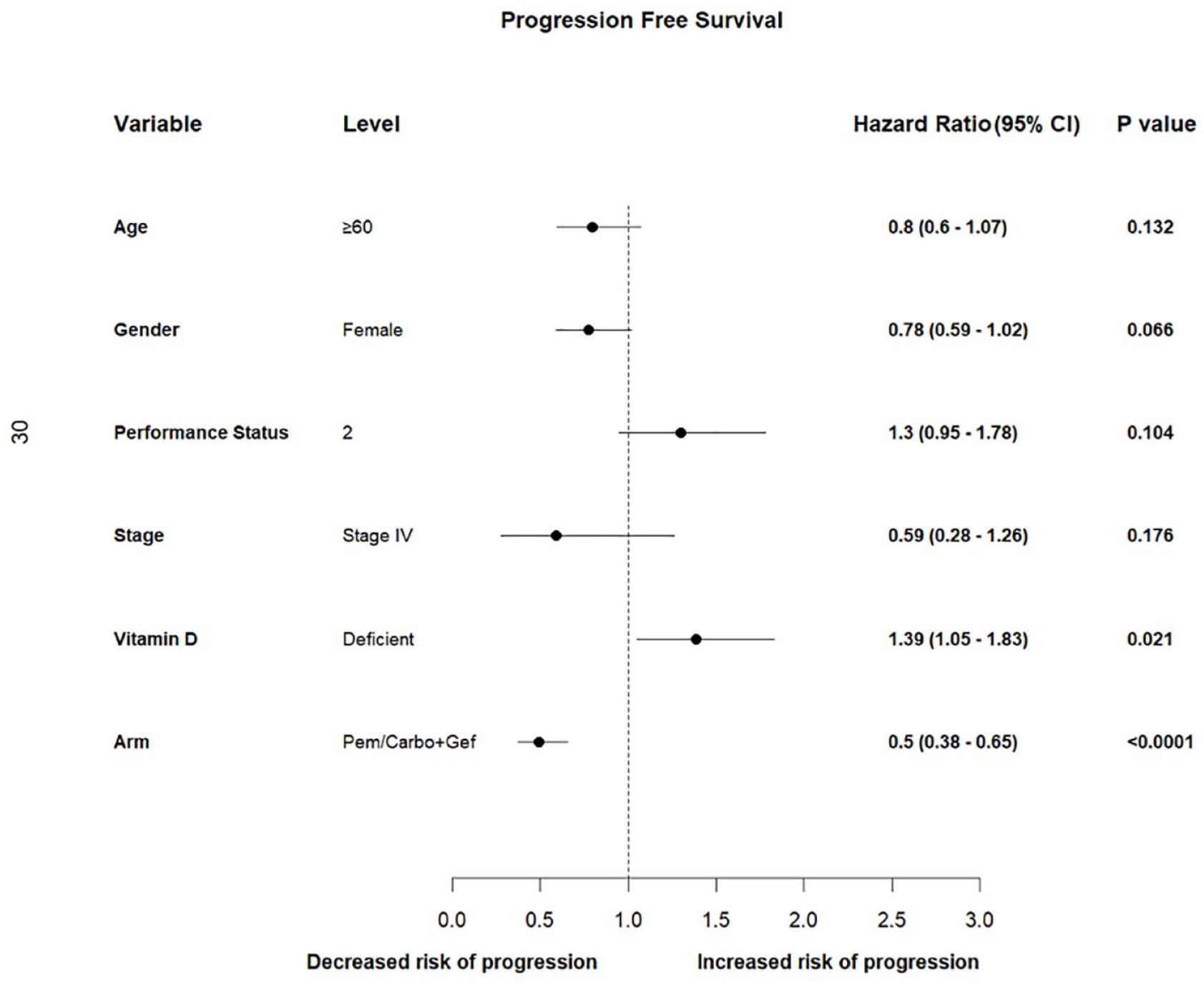
A multivariate analysis of progression-free survival for the variables age, gender, performance status, stage, vitamin D, and arm.
Overall survival
There were 117 (35.04%) patients who had an event for OS. The median OS in patients with normal vitamin D level was 28.6 months (95% CI = 19.73-37.5), whereas the median OS in patients with deficient vitamin D levels was 28.5 months (95% CI = 16.91-40.06); HR of 1.17 (0.81-1.68) as seen in Figure 3.
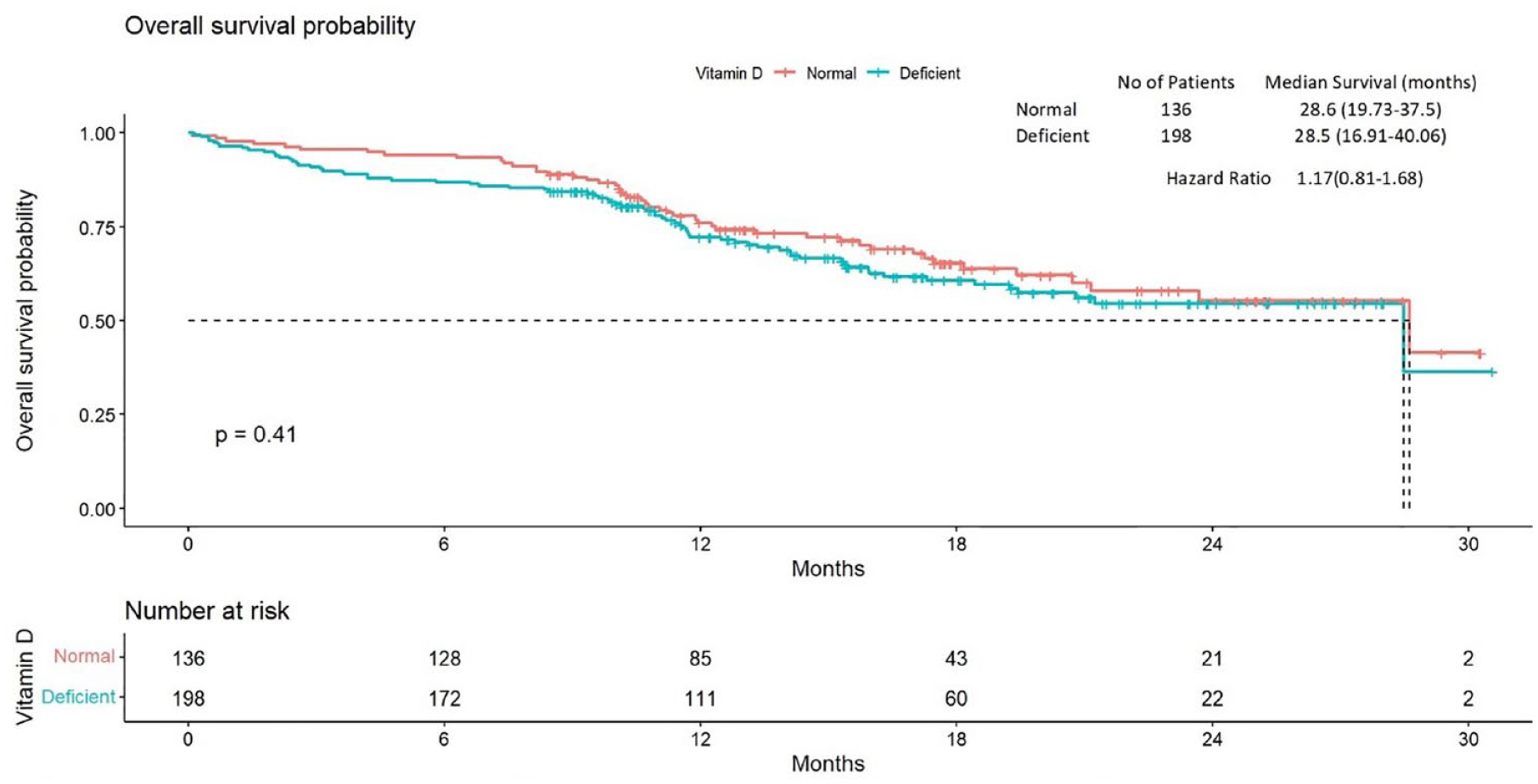
Kaplan-Meier plot for overall survival comparing 2 groups of vitamin D using the log rank test.
Vitamin D supplementation
We analyzed the relation between vitamin D supplementation on OS and found no significant association between the vitamin D supplementation and the outcome.
Discussion
In this study, we observed the PFS in patients with lower vitamin D levels was shorter than in the patients with normal vitamin D levels. The median PFS in patients with normal vitamin D level was 17 months (95% CI = 15-21), whereas the median PFS of patients with deficient vitamin D level was 15 months (95% CI = 13-17), which was a statistically significant difference. The impact of the baseline vitamin D level on PFS retained statistical significance in a multivariate analysis, along with the treatment regimen, whereas other factors tested, including age, sex, disease stage, and PS, did not significantly impact PFS. Our findings were in line with the results of the meta-analysis by Liu et al, 21 in which higher 25(OH)D levels correlated with a better prognosis.
There was no statistically significant difference in the OS in patients with normal vitamin D levels and deficient vitamin D levels. The median OS in patients with normal vitamin D level was 28.6 months (95% CI = 19.73-37.5), whereas the median OS in patients with deficient vitamin D levels was 28.5 months (95% CI = 16.91-40.06).
As there was no previous literature regarding correction of vitamin D deficiency to improve oncologic outcomes, hence it was left to the physician’s discretion. This being a post hoc analysis including the patients who were EGFR mutant and treated with Gefitinib-targeted therapy or Gefitinib-targeted therapy along with pemetrexed and carboplatin in a study setting, the general association between vitamin D levels in other patients with lung cancer receiving treatment for the same needs to be further studied. The significance of correction of vitamin D in a prospective randomized study also needs to be done for confirming the hypothesis. Ours was a retrospective analysis, and vitamin D receptors had not been evaluated in the study. We were, therefore, unable to assess the connection between vitamin D receptors and survival; this was a limitation of our study.
Conclusions
Baseline vitamin D levels have a significant impact on PFS, whereas OS is not affected by the baseline vitamin D levels in patients receiving Gefitinib-targeted therapy for EGFR-mutant lung cancer. However, the impact of vitamin D needs to be further studied in other lung cancer patients in prospective setting along with correction for deficiency for a clearer decision on clinical practice.
Footnotes
Acknowledgements
None.
Correction (June 2024):
This article has been updated with minor grammatical or style corrections since its original publication.
Author Contributions
VN and KP were responsible for the conception and design of the study. All authors were responsible for acquisition of the data. VN and KP were responsible for analysis of the data. All authors were responsible for interpretation of the data. VN drafted the article, and all other authors revised it critically for important intellectual content.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The individual de-identified data will be available on request to the corresponding author, Dr KP (
Consent for publication
Not applicable.
Ethics Approval and Consent to Participate
The trial was conducted at Tata Memorial Center (TMC), an academic tertiary oncology hospital in Mumbai, India. The study was approved by the Institutional Ethics Committee of TMC (study number 204) and monitored by the Data Safety Monitoring Subcommittee. The study was conducted as per Good Clinical Practice, following Indian and International regulations and guidelines. All participants provided written informed consent before enrollment on this study. The CTRI number of the study is CTRI/2016/08/007149. The date of the registration was 5 August 2016.
