Abstract
Background:
There have been few studies on the efficacy of tyrosine kinase inhibitors in lung carcinomas. The purpose of this study was to evaluate the effect of gefitinib as a first-line therapy in patients with advanced non-small cell lung cancer (NSCLC) who were positive for epidermal growth factor receptor (
Methods:
This prospective analysis included 120 patients with advanced NSCLC with
Results:
The ORR and DCR were 59.2% and 95.8%, respectively. The median PFS was 14.5 months and the median OS was 33 months. The longer OS was statistically significant in women and non-smokers, and the patients had a good PS. Adverse events occurred in 59.2% patients, but most of them were grade 1 and 2 events.
Conclusion:
This study conducted in Vietnam suggests the effectiveness of gefitinib as a first-line treatment option in patients with advanced NSCLC and positive
Background
Epidermal growth factor receptor (
Methods
Patients
A total of 120 patients with advanced NSCLC with
Methods
Study design
This was a prospective real-world study.
Study protocol
The study protocol involved clinical examination, paraclinical testing, chest radiography, chest computed tomography, and tumor biopsy. Tissue samples were obtained after biopsy as well as post-surgery. The pathologist localized the malignant cells and then sent a specimen for genetic mutation testing. Following this,
The selection criteria for targeted therapy included: (1) patients with stage IIIB, IIIC, and IV NSCLC; (2) patients >18 years old; (3) had received no prior systemic treatment for advanced NSCLC; (4) duration of treatment was at least 3 months at the time of data analysis; (5) patients provided consent for treatment with gefitinib; and (6) sufficient information on treatment was available. The exclusion criteria were as follows: (1) patients with stage I, II, and IIIA NSCLC; (2) patients with small-cell lung cancer; and (3) patients did not provide consent for treatment with gefitinib.
The patients were followed up every 3 months or when the patients had symptoms of progressive disease. Tumor response was evaluated using the Response Evaluation Criteria in Solid Tumors 1.1. Toxicity was evaluated using the National Cancer Institute Common Terminology Criteria for Adverse Events version 4.03. 2010. The main criteria for the analysis were the progression-free survival (PFS) and overall response rate (ORR). The secondary criteria were the disease control rate (DCR), toxicity, overall survival (OS), and OS rate at 12, 24, 36, 48, and 60 months. The treatment was discontinued if the disease progressed, if severe adverse events occurred, if combined treatment still did not relieve the symptoms, or when the patients disagreed on treatment. The endpoint for data analysis was May 2020.
Ethics approval and consent to participate
The study was approved by the ethics committee of the 108 Military Central Hospital, Vietnam at 2524/HĐĐĐ, 28 May 2019. All patients who participated in this study voluntarily provided their informed consent.
Statistical analysis
The Statistical Package for the Social Sciences software version 22.0 was used for all statistical computations. Differences were considered statistically significant when
Results
Clinical and paraclinical characteristics of the patients treated with gefitinib
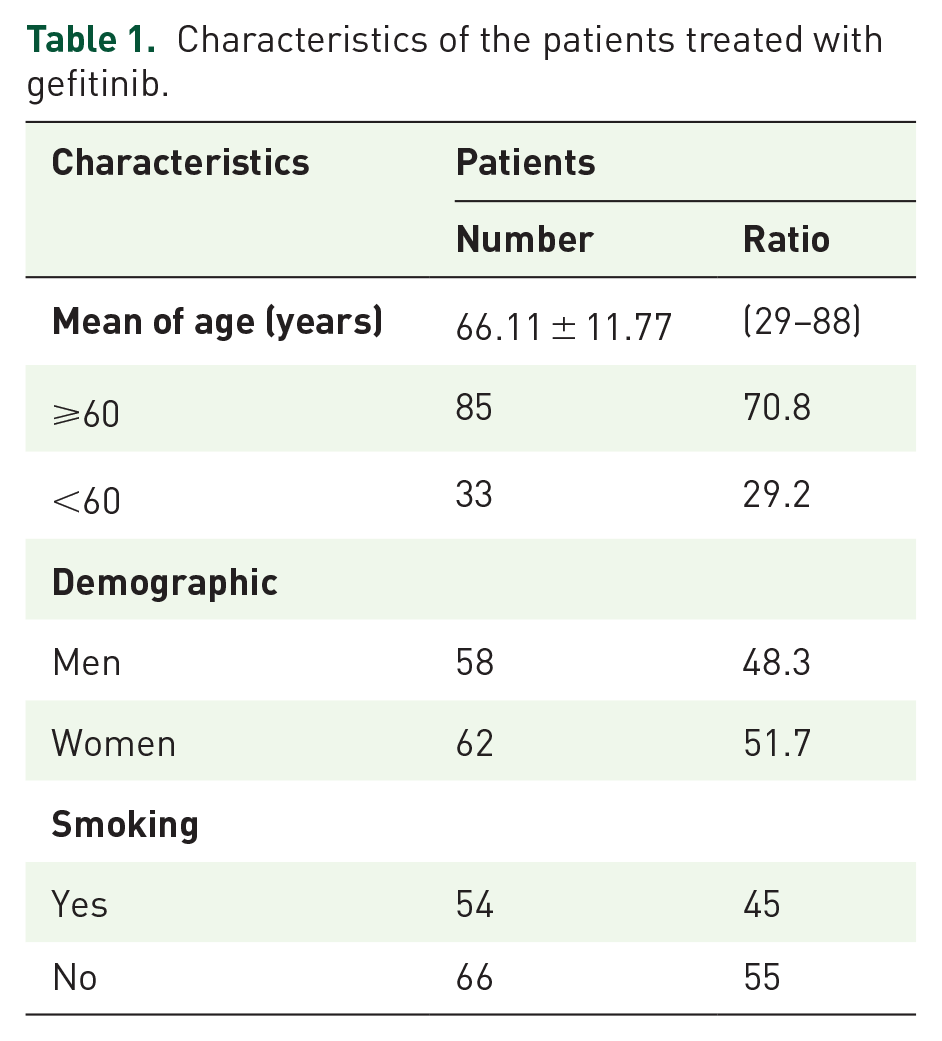
The median age of the patients treated with gefitinib in this study was 67.5 years and 11.7 months, with a maximum age of 88 and a minimum of 29 years. The men/women ratio was nearly 1/1. Most patients did not have a smoking history. The characteristics of the patients treated with gefitinib are detailed in Table 1.
Characteristics of the patients treated with gefitinib.
Effects of targeted therapy using gefitinib
The median follow-up time was 15.5 months, with a minimum of 3 months and a maximum of 63 months. In the third month, the ORR was 59.2% (complication rate and partial response rate were 2.5% and 56.7%, respectively), and the DCR was 95.8%.
The median PFS was 14.5 ± 0.86 months [confidence interval (CI) 95%:12.8–16.1] (Figure 1).
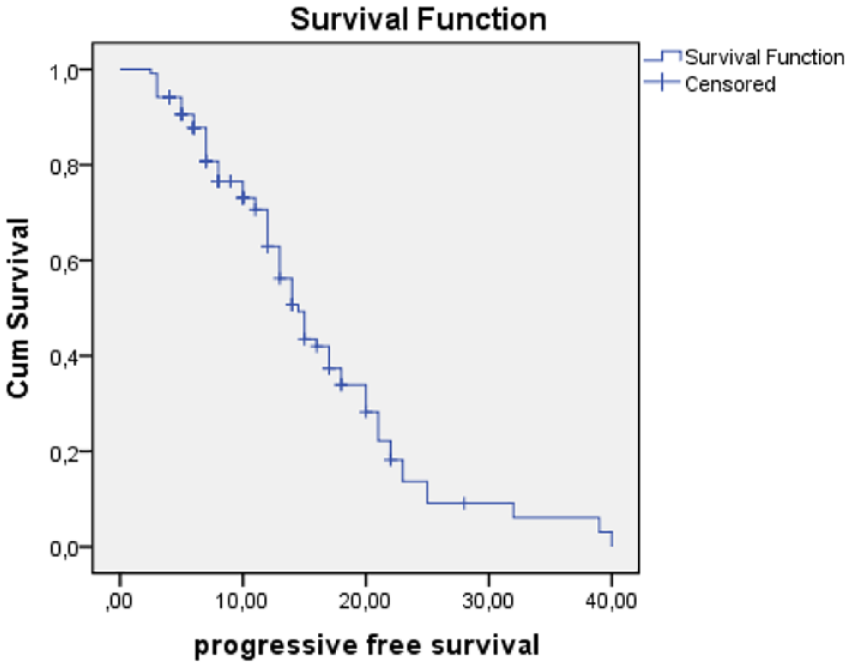
Progression-free survival.
The number of events was 26. The median OS was 33 months (95% confidence intervals [CI]: NR–NR).The OS rates at 12, 24, 36, 48, and 60 months were 63.3%, 25%, 5.8%, 2.5%, and 1.7%, respectively (Figure 2, Table 2).

Overall survival.
Overall survival rates.

OS, overall survival.
The median OS in women was longer than that in men, and the difference was statistically significant (
Relationship of PFS and OS with some characteristics.

OS, overall survival; PFS, progression-free survival; PS, performance status.
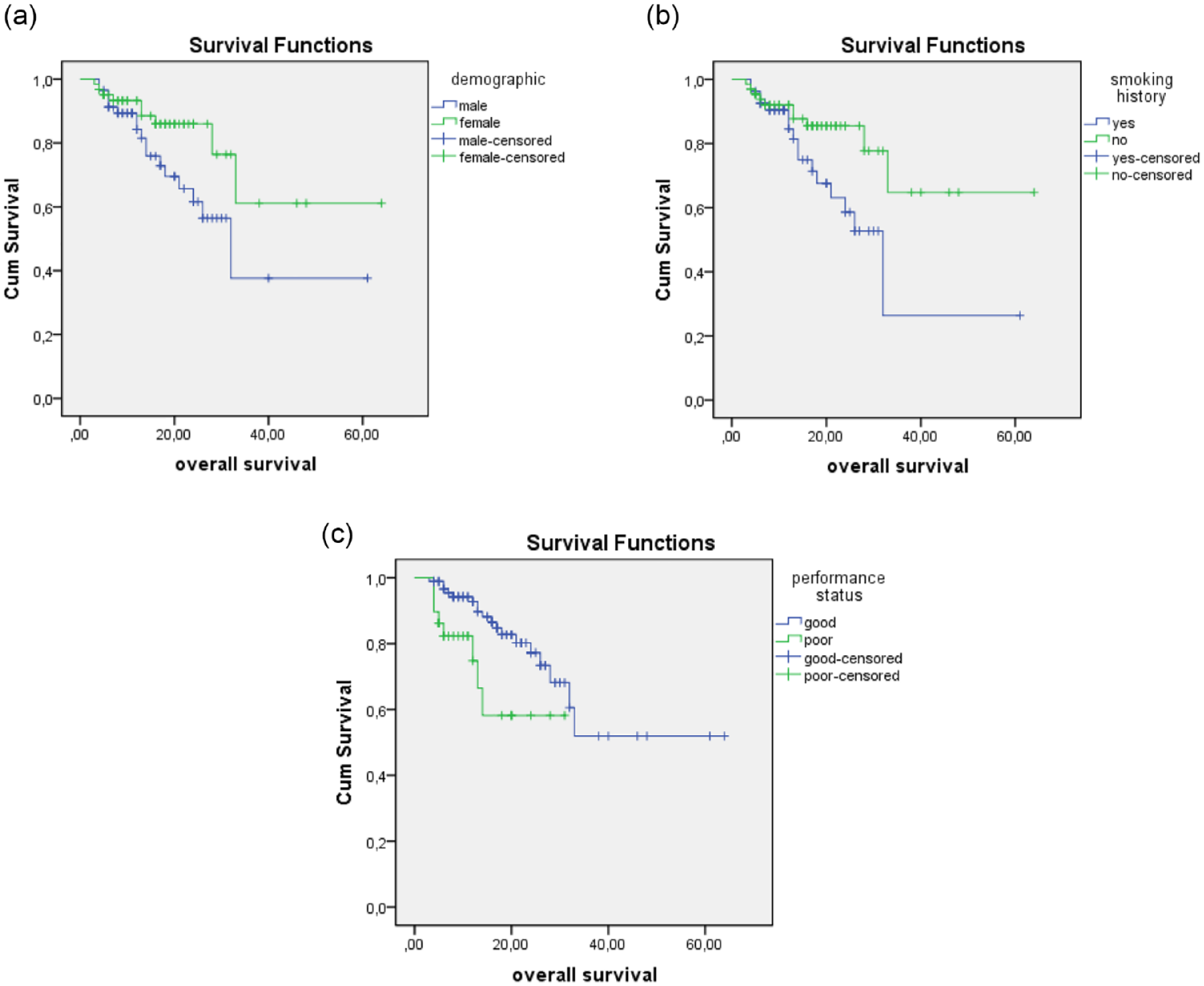
Relationship of overall survival (OS) with some patient characteristics. (a) OS and sex, (b) OS and smoking history, (c) OS and performance status (PS) of patients.
Toxicity was observed in 59.2% of the patients, the majority of which comprised rash and acne, accounting for 41.7%. However, these toxicities were commonly grade 1 and grade 2 toxicities. These adverse events were followed by other toxicities, such as paronychia, anorexia, and diarrhea. Two patients exhibited an increase in the transaminase level of grade 3 by 1.7%. No patients showed a reduction in the red blood cells, white blood cells, or platelets in the bone marrow. There were no data available on interstitial lung disease (ILD) due to gefitinib (Figure 4).

Adverse events.
Discussion
Researchers from around the world including from Vietnam have demonstrated that older age is one of the risk factors for lung cancer.8–10 In our study, the mean age of the patients was 66.11 years, with a maximum of 88 and a minimum of 29 years, and most patients were >60 years old (70.8%). Although the mean age in this study was similar to that in other studies, this study also shows the age rejuvenation of lung cancer patients. This rejuvenation has been explained by an increase in the smoking habit among young people. According to statistics, up to 85–90% of lung cancer patients have a history of smoking. Environmental pollution is also one of the factors that increases the risk of lung cancer.
8
In our study, there were more women (51.7%) than men, and the proportion of non-smokers (55%) was also higher than that of smokers (45%). Studies have shown that men and smokers have a higher incidence of lung cancer compared with women and non-smokers. However, in this study, we evaluated the effect of EGFR-TKI treatment on advanced NSCLC with
The efficacy of EGFR-TKIs in patients with NSCLC harboring
In terms of monitoring and evaluating the response of patients who were treated with TKIs, clinical trials of gefitinib have been conducted at intervals of 6 to 8 weeks, and have demonstrated that the PFS in patients using targeted therapy were 9.8 months and 10.8 months in the IPASS and NEJ002 trials, respectively.2,3 In the FLAURA study, which compared the efficacy of osimertinib with first-generation TKIs in patients with advanced NSCLC harboring
In this study, we also analyzed the relationship between PFS and OS with respect to clinical and paraclinical factors such as sex, smoking history, and PS of patients, which has rarely been mentioned in previous studies, especially the PS of patients. The results demonstrated that compared with men and smokers, the OS was significantly longer in women and non-smoking patients (
Another concern about targeted therapy in patients with NSCLC who are positive for
This is the first study on the use of gefitinib as a first-line treatment in Vietnamese patients with advanced NSCLC harboring
The limitations of this study are that it had a limited number of patients and it was conducted in a single center. We plan to continue treatment and follow-up, as well as an analysis of a larger number of patients, including patients with brain metastases.
Conclusions
Gefitinib is an effective first-line treatment in Vietnamese patients with advanced NSCLC who are positive for
Footnotes
Authors’ contributions
-PVL, Medical Doctor in the Department of Respiratory Medicine, 108 Military Central Hospital, Vietnam: formal analysis, principal investigation, methodology, resources, writing original draft, review, and editing.
-NDT, Associate Professor, Head of Department of Respiratory Medicine, 108 Military Central Hospital, Vietnam: Research member, formal analysis, and methodology.
-NMH, PhD, Vice Head of Department of Respiratory Medicine, 108 Military Central Hospital, Vietnam: Research member and provided resources.
-NDaoT, PhD, Vice Head of Department of Respiratory Medicine, 108 Military Central Hospital, Vietnam: Research member and provided resources.
-TTD, Medical Doctor in the Department of Respiratory Medicine, 108 Military Central Hospital, Vietnam: Research member and provided resources.
All authors have read and approved the manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval and consent to participate
The study was approved by the ethics committee of the 108 Military Central Hospital, Vietnam. All patients who participated in this study voluntarily provided their consent.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
