Abstract
Background:
The risk of first recurrence of hepatocellular carcinoma (HCC) within years 5 to 10 after curative hepatectomy remains unknown. We aimed to assess the incidence and prognostic factors for very late recurrence among patients who achieved 5 years’ recurrence-free survival (RFS) after primary resection.
Methods:
We retrospectively analyzed 337 patients with early-stage HCC underwent primary tumor resection and achieved more than 5 years’ RFS.
Results:
A total of 77 patients (22.8%) developed very late recurrence. The cumulative very late recurrence rate increased from 6.9% and 11.7% to 16.6% at 6, 7, and 8 years, respectively. Patients stopped smoking had a higher rate of very late RFS.
Conclusions:
The high rates of very late recurrence in HCC indicate that patients warrant continued surveillance, even after 5 recurrence-free years. Moreover, smoking is a risk factor for very late HCC recurrence, and quitting smoking may reduce the risk of very late recurrence.
Introduction
Hepatocellular carcinoma (HCC) is the fifth most common cancer worldwide and the second most frequent cause of cancer-related deaths.1,2 Multiple risk factors, such as hepatitis B virus (HBV) or hepatitis C virus (HCV) infections, alcohol abuse, smoking, and metabolic syndrome are considered be related to the development of HCC. 3 In Taiwan, the major cause of HCC is HBV infection, followed by HCV infection—with similar observations reported in many other Asian areas.4-6
Hepatectomy remains the most effective treatment for patients with early-stage HCC who have well-preserved liver function. Nevertheless, the overall survival (OS) rate after curative resection remains unsatisfactory due to the high rate of recurrence. The 5-year survival rate after curative resection is around 50% to 70%.7-9 Previous studies used the timepoint of 24 months after curative hepatectomy as a cutoff to categorize HCC recurrence as early or late recurrence10,11 and showed that early recurrence after resection is associated with lower OS in HCC.11,12 Several risk factors—including viral infection status, tumor stage, performance status, liver reserve evaluation, alcohol consumption, and smoking status—have been associated with the prognosis of patients with HCC after curative hepatectomy. However, in contrast to other types of cancers that have minimal risks of recurrence after 5 years without recurrence, such as stomach or colon cancer, de novo HCC may develop even after a long recurrence-free period (eg, 5 years) due to the progression of underlying chronic liver disease over time. 13 This indicates that long-term follow-up for recurrence is necessary in HCC, even for patients with long recurrence-free periods.
Smoking is so common that it can be difficult to grasp the severity of its health impact. However, tobacco kills more than 8 million people each year. More than 7 million of those deaths are the result of direct tobacco use, and around 1.2 million are the result of non-smokers being exposed to second-hand smoke. 14 Smoking has been proven to contribute to cardiovascular disease, chronic lung disease, and diabetes. Smoking is also related to the incidence and prognosis of several types of cancer, including carcinomas of the lung, stomach, esophagus, bladder, pancreas, kidney, and colon.15,16 In some countries, smoking is widespread and is associated with lung tumors.17,18 Recently, cigarette smoking has not only be regarded as a predisposing factor for the occurrence of HCC, but it has also been investigated as an independent predictor of mortality and been shown to increase the risk of development of HCC in people with HCV or HBV infections and individuals with alcoholism.19-21 However, the association between cigarette smoking and recurrence after curative resection in HCC has not yet been well investigated and is still controversial.
To date, there is little evidence of the factors associated with the survival of patients with more than 5 years’ recurrence-free survival (RFS) after curative resection for early-stage HCC. Therefore, we aimed to identify the characteristics, rates of very late recurrence, and factors associated with very late recurrence in patients with early-stage primary HCC treated with curative resection, with a specific focus on patients who did not experience recurrence for at least 5 years.
Patients and Methods
Ethics statement
This study complied with the standards of the Declaration of Helsinki and current ethical guidelines, and approval was obtained from the Ethics Committee of Chang Gung Memorial Hospital (Institutional Review Board [IRB] approval number: 201901103B0). The requirement for informed consent for this study was waived by the IRB, and all data were analyzed anonymously.
Patients and analysis
We reviewed a total of 2137 patients with HCC who underwent surgical resection between January 2001 and April 2016 at Kaohsiung Chang Gung Memorial Hospital. This hospital is a tertiary referral center that covers the south of Taiwan. The exclusion criteria were as follows: (1) Barcelona Clinic Liver Cancer (BCLC) stage C or B (n = 918); (2) prior treatment for HCC before surgical resection (n = 234); (3) recurrence in the 5 years after surgery (n = 476); (4) died within the 5 years after surgery (n = 37); (5) liver transplantation after surgery (n = 98); and (6) loss to follow-up (n = 37). Finally, 337 patients with BCLC stage 0 or A HCC who underwent primary curative resection were enrolled in this study (Figure 1).

Patient selection flow diagram.
Data on baseline demographics, smoking status (never, former, or current smoker), serum biochemistry, tumor burden, and antiviral therapy before any forms of definite treatment were comprehensively recorded. Patients ware defined as smokers if they smoked ⩾100 cigarettes prior to surgery. After curative liver resection, they were under regular outpatient department follow-up. Physicians record smoking status after surgery in their medical records. Patient were defined as former smokers when they had quit smoking for more than 5 years after surgery based on the medical records. The diagnosis of cirrhosis was documented in the pathologic reports for resected non-tumor tissues. Blood tests were performed within 1 week before resection. Recurrence-free survival of very late recurrence was defined as the period from 5 years after resection until the detection of recurrence. Overall survival was defined as the period from 5 years after tumor removal by resection to death, last contact, or April 2022.
Definition
The diagnosis of HCC was based on the criteria of the American Association for the Study of Liver Disease (AASLD) and the European Association for the Study of the Liver (EASL)22,23 and confirmed by histological analysis, if available. Early stage of HCC was defined as BCLC stage 0 or A. The diagnosis of HCC recurrence was based on the typical imaging findings or continual elevation of serum alpha-fetoprotein (AFP). Biopsies were performed to obtain histopathologic evidence of recurrence in patients with controversial imaging or laboratory findings.
Statistical analysis
Statistical analyses were performed using SPSS 23.0 statistical package (SPSS, Inc, Chicago, Illinois). The chi-square test and the Fisher exact test were used to compare categorical variables; the
Results
Baseline characteristics of the patients
Table 1 presents the baseline characteristics of the study cohort. There were 267 (79.2%) men and 70 (20.8%) women, with a mean age of 56.1 ± 12.1 years at enrollment. Fifty-five patients (16.3%) had diabetes mellitus (DM), 114 patients (33.8%) had smoked, and 79 patients (23.4%) consumed alcohol. Liver cirrhosis was observed in 132 patients (39.3%). The etiologies of hepatitis were hepatitis B (192/337; 57%), hepatitis C (81/337; 24%), hepatitis B + C (16/337; 4.7%), and non-B non-C (NBNC) (48/337; 14.3%). Moreover, 111 patients had Child-Pugh grade A and 21 had Child-Pugh grade B cirrhosis. The mean level of total bilirubin was 0.83 ± 0.34 g/dL, and the mean level of albumin was 3.76 ± 0.6 g/dL. Overall, 133 (39.5%) patients had elevated alpha-fetoprotein (AFP) (>10 ng/mL) and 149, 176, and 10 patients had ALBI (albumin-bilirubin) grade I, II, and III, respectively. The mean level of FIB-4 (Fibrosis-4) is 0.79 ± 0.53.
Clinicopathological features of the 337 patients with early-stage HCC who were alive without recurrence at 5 years after primary resection.
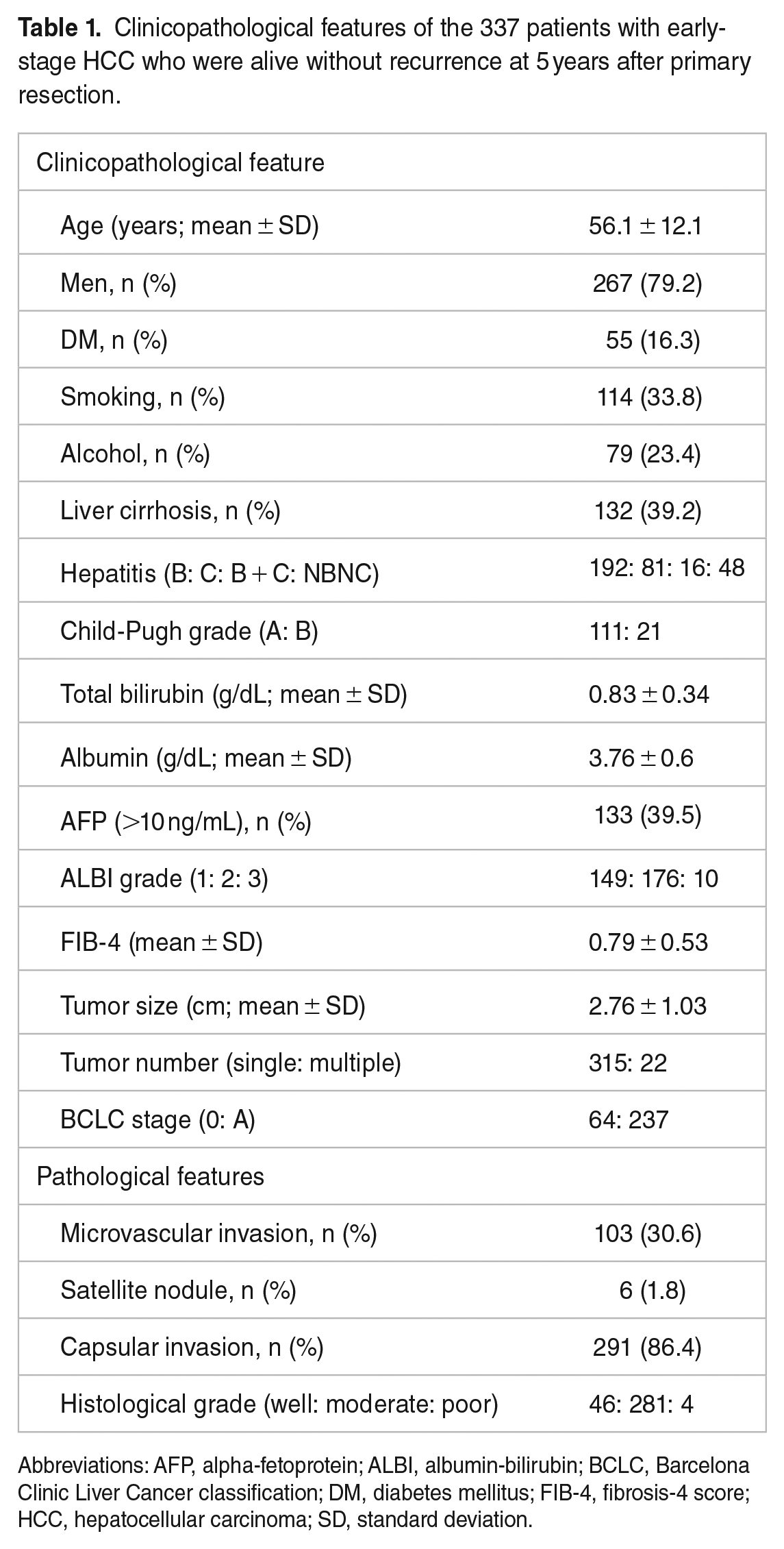
Abbreviations: AFP, alpha-fetoprotein; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer classification; DM, diabetes mellitus; FIB-4, fibrosis-4 score; HCC, hepatocellular carcinoma; SD, standard deviation.
All patients in the study cohort had BCLC stage 0/A HCC (64/237). The mean tumor size was 2.76 ± 1.03 cm, and 22 patients had multiple tumors. In pathological features, 103 patients (30.6%) had microvascular invasion, 6 (1.8%) had satellite nodules, and 291 (86.4%) had capsular invasion. Forty-six, 281, and 4 patients had well, moderately, and poorly differentiated tumors, respectively.
Analysis of very late recurrence and survival for patients who were alive without recurrence at 5 years
After the median follow-up of 126 months, 77 patients (22.8%) had developed recurrence. The cumulative very late recurrence rate increased from 6.9% and 11.7% to 16.6% at 6, 7, and 8 years, respectively (Figure 2A). The OS rates were 98.8%, 96.0%, and 91.7% at 6, 7, and 8 years after resection (Figure 2B).
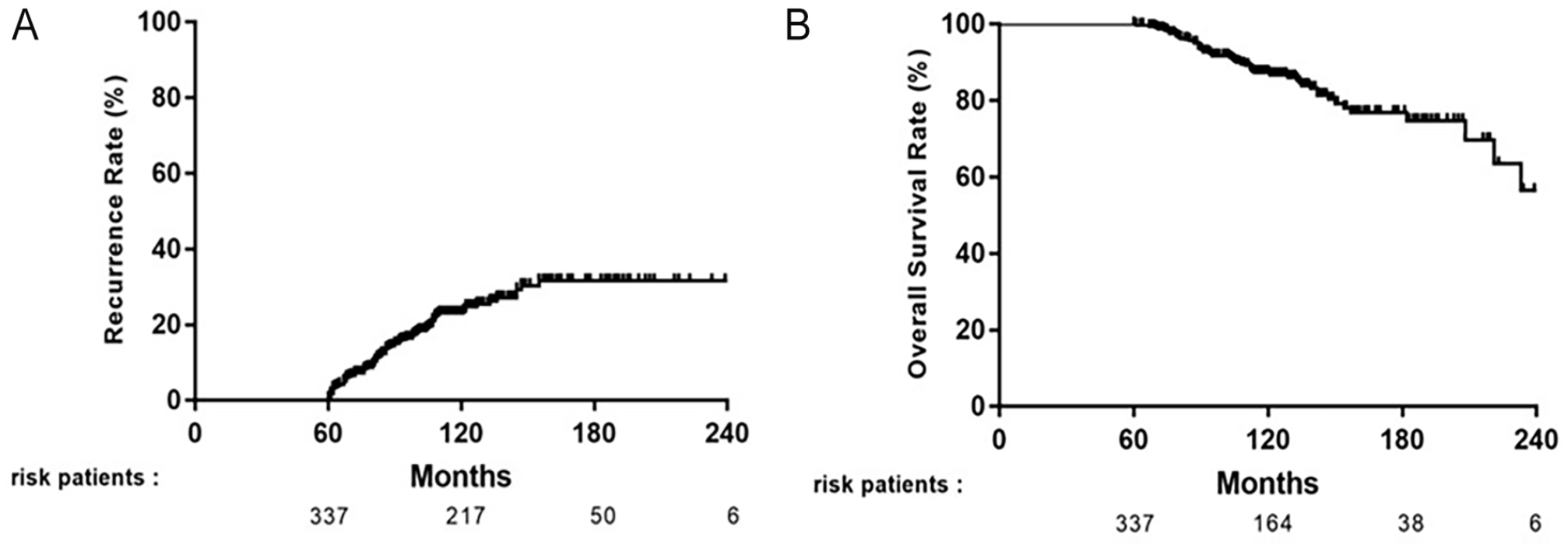
Kaplan-Meier analysis of very late recurrence (A) and overall survival (B) for patients with early-stage HCC who were alive without recurrence at 5 years after primary resection.
As shown in Table 2, smoking, tumor number, and serum AFP at 5 years after surgery were significantly associated with very late HCC recurrence in univariate analysis. In multivariate analysis, smoking (hazard ratio [HR] = 2.14; 95% confidence interval [CI] = 1.28-3.58;
Univariate and multivariate analysis of very late recurrence for patients with early-stage HCC who were alive without recurrence at 5 years after primary resection.

Abbreviations: AFP, alpha-fetoprotein; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; FIB-4, fibrosis-4 score; HCC, hepatocellular carcinoma; HR, hazard ratio.
In analysis of OS, age >60 years, smoking, DM, Child-Pugh grade, FIB-4, antiviral therapy, and serum AFP at 5 years after surgery were significantly associated with mortality in univariate analysis. The multivariate Cox proportional hazards model revealed that age >60 (HR = 3.23; 95% CI = 1.60-6.55;
Univariate and multivariate analysis of mortality for patients with early-stage HCC who were alive without recurrence at 5 years after primary resection.

Abbreviations: AFP, alpha-fetoprotein; ALBI, albumin-bilirubin; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; FIB-4, fibrosis-4 score; HCC, hepatocellular carcinoma; HR, hazard ratio.
Analysis of very late recurrence stratified by smoking status
Notably, patients who quit smoking postoperatively had better outcomes. Thus, we performed subgroup analyses to achieve a better understanding of how smoking status affects very late recurrence in HCC; 223 patients were categorized as non-smokers and 114 patients as smokers at enrollment. Among the smokers, 46 were subdivided into current smokers, and 68 patients were categorized as former smokers based on the outpatient records 5 years after surgery. Kaplan-Meier analysis revealed non-smokers had a higher rate of very late RFS than current smokers and those who had quit smoking, with linear trends observed (
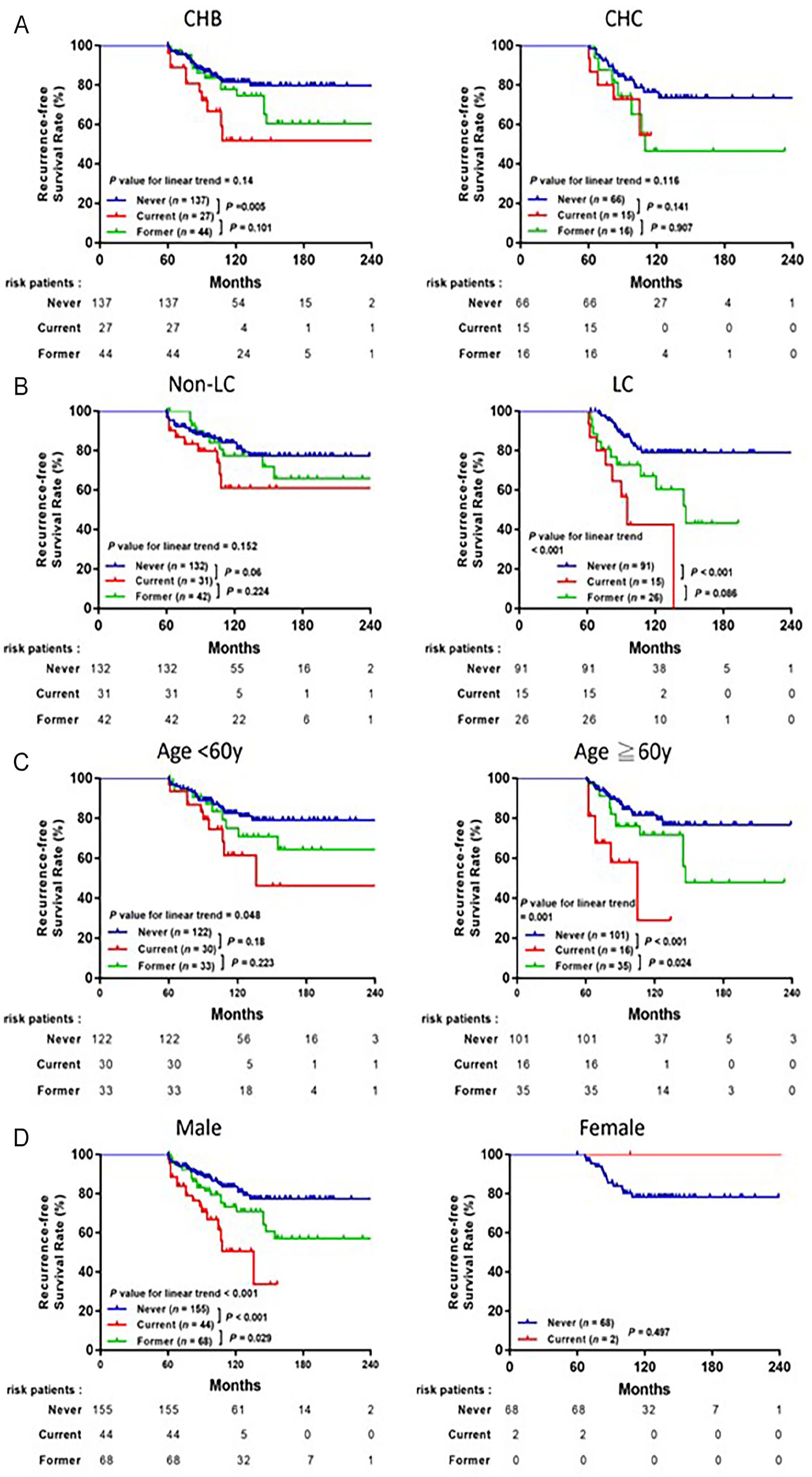
Kaplan-Meier analysis of very late recurrence-free survival (A) and overall survival (B) for patients who were alive without recurrence at 5 years after primary resection stratified by smoking status.
We further investigated the impact of smoking status on very late RFS in patient subgroups stratified by common clinicopathological factors. Kaplan-Meier plots showed significant differences in very late RFS based on smoking status (non-smoker, current smoker, and former smoker) in subgroup analyses of liver cirrhosis (

Kaplan-Meier analysis of cumulative very late recurrence-free survival based on smoking status for the subgroups of patients stratified by (A) hepatitis, (B) liver cirrhosis, (C) age, (D) sex, and (E) serum AFP.
Associations between clinical characteristics and smoking habits
Subgroup analyses revealed smokers were predominantly men (98% vs 69.5%,
Comparison of the clinical characteristics of patients with HCC who were alive without recurrence at 5 years after primary resection based on smoking status.

Abbreviations: DM, diabetes mellitus; HCC, hepatocellular carcinoma; SD, standard deviation.
Discussion
In this long-term follow-up study, we evaluated the factors associated with very late RFS and OS among patients with early-stage HCC who achieved 5 recurrence-free years after curative resection. Our first major finding is that the rate of very late recurrence is high. Second, we found that smoking is an independent risk factor for very late HCC recurrence and mortality, and quitting smoking may reduce this risk. These results indicate that intensive, long-term surveillance is needed for patients with HCC, even after a 5-year recurrence-free period. In addition, smokers diagnosed with HCC should quit smoking as soon as possible to reduce the risk of very late recurrence and mortality.
In most types of cancer, such as stomach or colon cancer, the risk of recurrence after 5 years without recurrence is minimal.24,25 In gastric cancer, the cumulative recurrence rate after curative resection is 3.7% at 10 years, with almost all recurrences occurring between 6 months and 5 years after resection. 26 In colon cancer, the rates of local recurrence at 5 to 10 years after primary surgery are very low (2.9%) compared with the 1-year recurrence rate (2.3%). 27 Hence, long-term intensive surveillance is not recommended for these types of cancer after 5 recurrence-free years. However, HCC is different from other cancers, as de novo HCC may develop after a long recurrence-free period due to underlying chronic liver disease. Very late HCC recurrence, ie, >5 years after resection, is infrequently reported in the literature. However, the results of this study indicate that the cumulative recurrence rates for patients with 5 recurrence-free years increase from 6.9% and 11.7% to 16.6% at 6, 7, and 8 years, respectively. Among patients who were alive and free of tumor recurrence at 5 years after curative resection, we found the very late recurrence rate steadily increased by approximately 6% to 7% per year between 5 and 10 years after resection. These results are consistent with a previous study in Korea, 28 in which the cumulative recurrence rate in the 5 to 10 years after primary resection was 27.0%. Therefore, our results confirm that patients with HCC require continued intensive surveillance, even after a 5-year recurrence-free period.
Currently, the health insurance policy in Taiwan provides additional insurance benefits for patients with cancer for the first 5 years, depending on the type of cancer. However, these benefits end after 5 years if no further recurrence is detected. However, this study suggests that patients with HCC should continue to receive intensive surveillance and extension of the health insurance benefits should be considered, even after 5 recurrence-free years.
We observed smoking was associated with very late recurrence and mortality in HCC. Notably, in subgroup analysis, patients who had stopped smoking cigarettes achieved better RFS than current smokers (
The liver really regenerates34,35; however, carcinogenesis is a complex and complicated process. Previous studies had revealed immunity and certain mediators play a role in the process of carcinogenesis, as well as the effects of viruses, chemical substances (aflatoxins), and radiation.36,37 Recently, several mechanisms have been hypothesized to explain tobacco-induced hepatic carcinogenesis. A variety of chemicals present in tobacco smoke, including 4-aminobiphenyl (4-ABP) and polycyclic aromatic hydrocarbons (PAHs), can be metabolized into reactive carcinogenic agents in the liver 38,39 and lead to degeneration of the liver parenchyma. 40 Various tobacco components such as nitrosamines and benzenes can be activated into procarcinogens via CYP2E1. 41 Moreover, exposure to tobacco smoke promotes the release of pro-inflammatory cytokines such as interleukin (IL)-33, IL-1β, and tumor necrosis factor alpha (TNFα).42,43 Smoking can also lead to fibrosis and progression to more advanced stages of liver diseases with various etiologies.44,45 In line with these findings, chronic exposure to smoke is associated with hepatocarcinogenesis.46,47
In subgroup analyses, we observed significant differences in smoking status in the subgroups of patients with liver cirrhosis (linear trend;
There are some limitations to this study. First, this was not a prospective study. However, we believe that the risk of bias is low as the patients were followed-up by the same physicians throughout the course of their disease and underwent standard clinical and laboratory assessments and HCC screening based on ultrasonography every 3 to 6 months. However, we could not assess the relationships between the dosage and duration of smoking exposure on HCC recurrence, as this information was not available in the data retrospectively collected from the medical records. Therefore, properly conducted observational studies are needed to further confirm the dose-effect and time-effect relationships between smoking and very late recurrence in HCC.
Conclusions
Our retrospective analysis suggests patients who have survived without recurrence for 5 years after curative hepatectomy should still undergo intensive surveillance, as the very late recurrence rate for this group of patients increases from 6.9% and 11.7% to 16.6% at 6, 7, and 8 years, respectively. Moreover, smoking is a risk factor for very late HCC recurrence among this group of patients. Smoking cessation counseling should be provided to encourage smokers with HCC, especially younger patients, to stop smoking to improve their prognosis.
Footnotes
Acknowledgements
The authors appreciated the Biostatistics Center, Kaohsiung Chang Gung Memorial Hospital for statistics work. The authors also thank the service provided by the Cancer Registration of Cancer Center Department, Kaohsiung Chang Gung Memorial Hospital, especially the help by the cancer registers.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants CMRPG8F0661 and CMRPG890161 from Chang Gung Memorial Hospital, Taiwan.
Author Contributions
M-CT contributed to conception and design. W-RC contributed to manuscript writing. C-CW, C-CL, Y-HY, C-HC, Y-HK, C-CY, C-HH, P-YH, M-JT contributed to collection and assembly of data. W-RC, A-CL, and M-CT contributed to data analysis and interpretation. All authors approved the final version of the manuscript.
Consent for Publication
Not applicable.
Availability of Data and Material
All analyzed data are included in this published article. The original data are available on reasonable request to the corresponding author.
Ethics Approval and Consent to Participate
This study complies with the standards of the Declaration of Helsinki and current ethical guidelines, and approval was obtained from the Ethics Committee of Chang Gung Memorial Hospital (IRB approval number: 201901103B0). The requirement for informed consent for this study was waived by the Ethics Committee of Chang Gung Memorial Hospital, and all the data were analyzed anonymously.
