Abstract
Backgroud:
The tumor immune microenvironment influences the efficiency of concurrent chemoradiotherapy (CCRT) in high-grade glioma (HGG). This study investigated peripheral blood T lymphocyte subsets as clinical indicators of therapeutic response and prognosis in pediatric high-grade glioma (pHGG).
Methods:
This retrospective study included 77 patients with postoperative pHGG who were treated concurrently with temozolomide and external beam radiotherapy between January 1, 2012, and December 31, 2018. The median follow-up was 26 (range: 5-106) months. Peripheral venous blood samples were collected before and after CCRT. The proportions of peripheral blood T lymphocytes and their association with treatment outcome and survival were determined.
Results:
Sixty-four (83.1%) patients achieved complete remission, partial remission, and stable disease, and 13 (16.9%) patients had progressive disease. Higher CD3+ T cell, CD4+ T cell, and CD8+ CD28+ T cell ratios were predictive of better response, while a higher CD8+ CD28− T cell ratio was predictive of poorer response. Binary logistic regression analysis showed that the CD8+ CD28+ T cell ratio was a significant independent predictor of CCRT response (odds ratio [OR] = 53.521, 95% confidence interval [CI] = 4.294-667.119, P = .002). Univariate and multivariate analysis of prognostic factors associated with survival showed that the CD8+ CD28+ T lymphocyte ratio was a significant independent predictor of progression-free survival (hazard ratio [HR] = 1.80, 95% CI = 1.06-3.08, P = .03), but none of the subsets were significantly associated with overall survival.
Conclusion:
Peripheral blood T lymphocytes have potential as predictors of CCRT response and prognosis in pHGG.
Introduction
Pediatric high-grade glioma (pHGG), classified as World Health Organization (WHO) grade III and IV gliomas, are the most aggressive primary tumors of the central nervous system. 1 At present, the methods used to treat glioma, including surgery, radiotherapy, and chemotherapy with temozolomide (TMZ) have progressed, but the prognosis of pHGG patients remains dismal, with the 5-year survival rate being only 15% to 20%. 2 The developments in immunotherapy and research on the tumor immune microenvironment (TIME) have helped achieve favorable therapeutic effects in the case of a growing number of solid tumors, such as melanoma and non-small-cell lung cancer (NSCLC), and improve the survival of patients.3,4 However, it has poor therapeutic effect in the case of HGG because the average mutational load of gliomas is lower than that of other solid tumors. 5 In addition, the poor therapeutic effect of immunotherapy on HGG is also related to the presence of abundant regulatory T cells or T reg cells, as well as tumor-associated macrophages and marrow-derived suppressor cells, in the tumor stroma of high-grade gliomas (HGGs). 6 These immune cells secrete certain suppressive cytokines and directly suppress the amplification of CD8+ T cells and CD4+ T cells to restrain the antitumor immune response. Therefore, one of the popular current research topics is how to effectively reverse the suppressive TIME of HGGs.
Concurrent chemoradiotherapy (CCRT), especially radiotherapy, in addition to its mechanism of cell killing through direct and indirect damage to DNA, also induces immunogenic cell death via the release of damage-associated molecular pattern molecules, such as calreticulin, HMGB1, and adenosine triphosphate (ATP), by irradiated tumors. 7 These molecules attract and activate dendritic cells and induce them to engulf tumor cells and antigens and to present the processed tumor antigens to T cells to stimulate tumor-specific immune responses.8,9 They also induce the production of inflammatory molecules and their release into the TIME. 10 Therefore, CCRT has the potential to initiate adaptive and innate immune responses and produce a systemic antitumor effect within and outside of the radiation field. 11 However, changes in the TIME and peripheral blood immune cells during CCRT vary considerably between patients, as do their clinical responses to therapy. 12 Based on all these findings, we tried to determine whether enhancing the peripheral immune response can result in prolonged tumor control in pHGG, by investigating the peripheral blood immune response to CCRT and determining whether specific changes in immune cells were correlated with clinical outcome. Our previous findings showed that a decrease in the CD8+/CD28− cell subset of immunosuppressive T regs was a predictive biological marker of the efficacy of adoptive cellular immunotherapy for various solid tumors.13-16 We also observed that measuring peripheral blood T reg, IL-6 and IL-10 levels could be valuable for predicting radiotherapeutic responses of pediatric medulloblastoma and germ cell tumors. 17 Other articles have reported similar changes in the peripheral blood lymphocyte subsets of HGG patients receiving CCRT: an elevated T reg cell proportion accompanied by decreased proportions of peripheral blood CD8+ T cells and CD4+ T cells was detected after radiotherapy. 18 These findings indicate that in HGG patients, CCRT can induce changes in the antitumor immune environment. However, the findings of these studies are limited by their small sample sizes. Therefore, in this study, we tried to explore whether the outcome of CCRT was associated with changes in peripheral blood immune cells in a large cohort by analyzing 77 peripheral blood samples from pHGG patients before and after CCRT.
Materials and Methods
Study population
This study enrolled 77 pediatric patients with newly diagnosed WHO grade III and IV glioma at the Beijing Shijitan Hospital, between January 1, 2012, and December 31, 2018. The inclusion criteria were (1) ⩽21 years of age, (2) diagnosis of WHO grade III and grade IV glioma confirmed by pathology, (3) no previous malignant tumor or second primary tumor, (4) no previous radiotherapy or chemotherapy, (5) no central nervous system metastasis, and (6) sufficient clinical information and follow-up data. The exclusion criteria were (1) clinical evidence of acute infection, (2) autoimmune diseases or hematological diseases, and (3) glucocorticoid therapy before and during CCRT.
This pilot study was approved by the Beijing Century Altar Ethics Committee (2018-64). The study was conducted in accordance with the ethical standards of the Declaration of Helsinki of the World Medical Association. As it was a retrospective design, the institutional review committee waived off the need for informed consent. To protect the privacy of the patients, we removed all identifying information from our records.
Data collection and flow cytometry analysis
Approximately 3 mL of peripheral venous blood was collected in an ethylenediaminetetraacetic acid (EDTA)-containing tube after surgery but before the start of RT/TMZ combination therapy and 4 weeks after the completion of RT/TMZ therapy. In each test tube, 100 µL of whole blood was stained with 10 µL of fluorescence-labeled antibodies. Each tube was incubated for 15 min at 4°C in the dark. Erythrocyte lysis was induced by red blood cell lysis buffer and protected from light at room temperature for 10 min (Solarbio life sciences, Beijing, China). Then, the samples were centrifuged at 1500 rotations/min for 5 min and the supernatant was discarded. Following this, the pellet (containing the cells) was resuspended in 250 µL of phosphate-buffered saline (PBS). All the samples were analyzed with a 4-color staining flow cytometer (Cytomics FC500; Beckman Coulter, Brea, California, USA), and data were obtained and analyzed using the CXP analysis software. T lymphocytes were gated on CD3+ cells, and the analysis was set to collect 5000 gated events (Supplementary Figure 1). In addition, cell surface staining was performed with the appropriate monoclonal antibodies (Beckman Coulter, Brea, California) targeting the T-lymphocyte subsets CD3+, CD3+ CD4+, CD3+ CD8+, CD8+ CD28-, CD8+ CD28+, and CD4+ CD25+ CD127low/− (Regular T cells, T regs).
Characteristics and treatment of the patients
Patients who were newly diagnosed with primary pHGG underwent surgical resection followed by 6 weeks of chemoradiotherapy comprising radiotherapy delivered at a median dose of 58 Gy (range: 40-62 Gy, for 5 days/week) and TMZ at a dosage of 75 mg/m2 daily, as well as 6 to 12 additional cycles of TMZ (150-200 mg/m2, for 5 days per month). 19 Intensity-modulated radiation therapy (IMRT) was used to treat postoperative tumor bed areas. The patients received 40-62 Gy (1.8-2 Gy per fraction, 5 days per week) of the 95% prescribed dose of planning target volume (PTV). For all patients, a highly conformal IMRT plan was designed using Eclipse (Varian Medical Systems, Palo Alto, California), to achieve optimal sparing of critical structures. The current standard for assessing brain tumors treatment response is defined by the RANO (response assessment in neuro-oncology), which is based on a 2-dimensional measurements of T2 and contrast-enhanced T1 magnetic resonance imaging (MRI) sequences (T1-CE). Stable disease (SD) is defined as enhanced tumor size increased by less than 25% at T1, no significant increase of T2/FLAIR nonenhanced lesions. Partial response (PR) is defined as a reduced of more than 50% in enhanced tumors or a significant decrease in nonenhanced lesions. Progressive disease (PD) is defined as an increase of more than 25% in enhanced tumors or a significant increase in nonenhanced lesions. In RANO response criteria, true progression cannot be defined within 12 weeks of RT unless there is pathologic confirmation or new out-of-field enhancement. The primary aim was to correlate changes in various T lymphocyte subsets during CCRT with progression-free survival (PFS) and overall survival (OS). The secondary aim was to assess the relationship between the changes in T lymphocyte subsets and the efficacy of CCRT in patients with pHGGs.
Statistical analysis
According to the RANO criteria, the treatment response of the patients was evaluated, and they were divided into the CR + PR + SD group (CR = complete remission, PR = partial remission, SD = stable disease) (n = 64) and the PD (progressive disease) group (n = 13). 20 Because the T lymphocyte subsets in both groups exhibited normal distribution, the paired-sample t-test was used to analyze pre- and post-CCRT changes and the chi-square test was used to analyze differences in variables between the 2 groups. The Kaplan-Meier method was used to analyze survival. Univariate analysis of the correlation between survival and experimental data was conducted using the log-rank test. The significant variables were entered into a multivariate Cox regression model to identify the significant variables associated with survival by step-wise backwards elimination. P values <.05 were considered to indicate statistical significance. All the statistical analyses were performed using the Graph Pad Prism software program 7.0.
Results
Clinical response to CCRT
The median follow-up time of this cohort was 26 (range: 5–106) months, and the last day of follow-up was March 31, 2022. The patients’ clinical characteristics and treatments are outlined in Table 1. At 4 weeks after CCRT, 13 of the 77 patients had PD, including 6 patients with grade III glioma and 7 patients with glioblastoma. The PR, CR, and SD patients were assigned to one group that included 39 cases of grade III glioma and 25 cases of glioblastoma. Two children diagnosed with pseudoprogression by routine MRI watch-and-wait methods were not included in the PD group. The overall clinical control rate was more than 83%.
Demographics and baseline characteristics of patients (n = 77).

Abbreviations: IDH, isocitrate dehydrogenase; MGMT, O6-methylguanine-DNA methyltransferese; PTV, planning target volume; RT, radiotherapy; SD, standard deviation.
We analyzed the age, gender, histopathological grade, surgical resection status, radiotherapy dose, PTV volume, isocitrate dehydrogenase (IDH) mutation status, and O6-methylguanine-DNA methyltransferese (MGMT) methylation status and evaluated their correlation with CCRT efficacy (Table 2). However, none of these variables were significantly associated with CCRT efficacy.
Univariate analysis of clinical pathological variables in response to concurrent chemoradiation.

Notes: Data were presented as mean values ± standard deviation (SD). P values were calculated by Mann–Whitney U test.
Abbreviations: CR, complete remission; IDH, isocitrate dehydrogenase; MGMT, O6-methylguanine-DNA methyltransferese; PD, progressive disease; PR, partial remission; PTV, planning target volume; RT, radiotherapy; SD, stable disease.
Changes in peripheral blood T lymphocyte subsets after CCRT
A significant increase in the percentage of CD8+ T cells (P = .02), CD8+ CD28− T cells (P = .02), CD8+ CD28+ T cells (P < .001), and T reg cells (P = .01) was observed after CCRT (Figure 1A). In contrast, the percentage of CD3+ T cells (P = .03) and CD4+ T cells (P < .001) was significantly lower after CCRT (Figure 1A).

Peripheral blood T lymphocyte subsets in HGG patients and differences among clinical efficacies. (A) T lymphocyte subsets in HGG receiving CCRT from preradiation to postradiation. (B) Comparison of the ratios (post-/pre-RT) of T lymphocyte subsets between CR + PR + SD group and PR group. CCRT indicates concurrent chemoradiotherapy; CR, complete remission; HGG, high-grade glioma; PD, progressive disease; PR, partial remission; RT, radiotherapy; SD, stable disease; NS: no statistical difference between the two groups (P >.05).
Correlation of variables with CCRT efficacy
To determine whether the CCRT outcome was associated with changes in the T-lymphocyte subsets, the ratio of the percentage of each T lymphocyte subpopulation after CCRT to the percentage of the T lymphocyte subpopulation before CCRT was calculated and compared between the CR + PR + SD group and PD group (Figure 1B): The ratio of CD3+, CD4+, and CD8+ CD28+ T cells was significantly higher in the CR + PR + SD group than in the PD group, while the ratio of CD8+ CD28- T cells was significantly higher in the PD group.
Binary logistic regression models were used to identify the factors associated with CCRT efficacy. Univariate analysis showed that the ratios of CD3+ T cells, CD4+ T cells, CD8+ CD28− T cells, and CD8+ CD28+ T cells were significantly correlated with CCRT response. Multivariate analysis of these subsets revealed that the ratio of CD8+ CD28+ T cells was an independent predictor of CR, PR, and SD (P = 0.002, Figure 2).
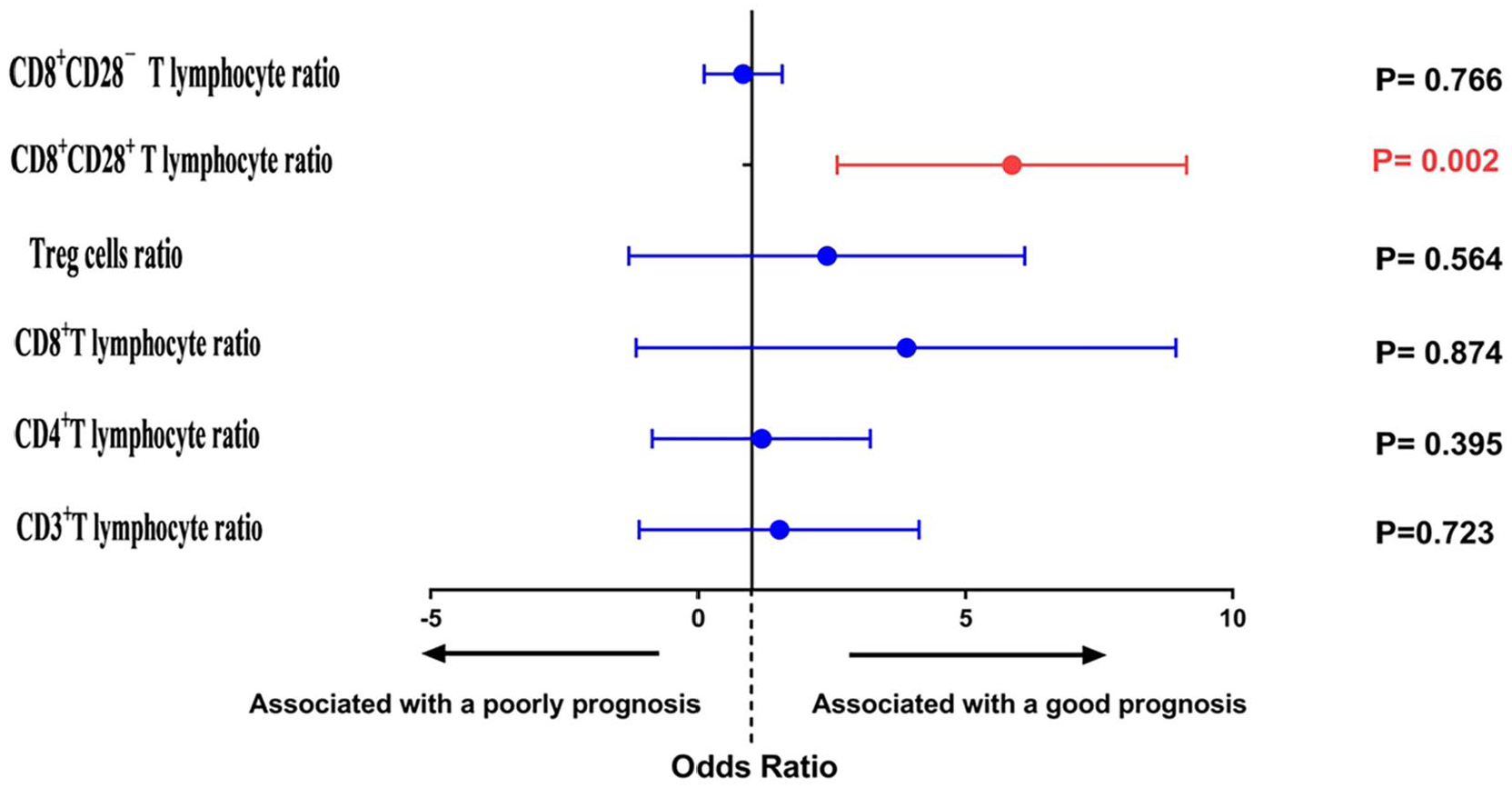
Multivariable logistic regression analysis of variables for prediction of concurrent chemoradiotherapy response.
CD8+ CD28+ T lymphocyte ratio was a significant predictor of prognostic
For the entire cohort, the median PFS and 5-year PFS rates were 1.25 (range: 0.17-5.67) years and 5.5%, respectively, and the median OS and 5-year OS rates were 2.08 (range: 1.83-7.17) years and 21.6%, respectively.
None of the clinicopathological variables were significantly associated with the PFS of the pHGG patients in our cohort. With regard to the T lymphocyte subset ratios, univariate analysis showed that IDH mutation status, CD8+ T cell ratio, CD8+ CD28- T cell ratio, and CD8+ CD28+ T cell ratio were correlated with PFS when the significance level was set at P < .05 (Figure 3), and multivariate analysis of these variables showed that the CD8+ CD28+ T lymphocyte ratio was a significant predictor of PFS (hazard ratio [HR] = 1.80, 95% confidence interval [CI] = 1.06-3.08; P = .03, Table 3).
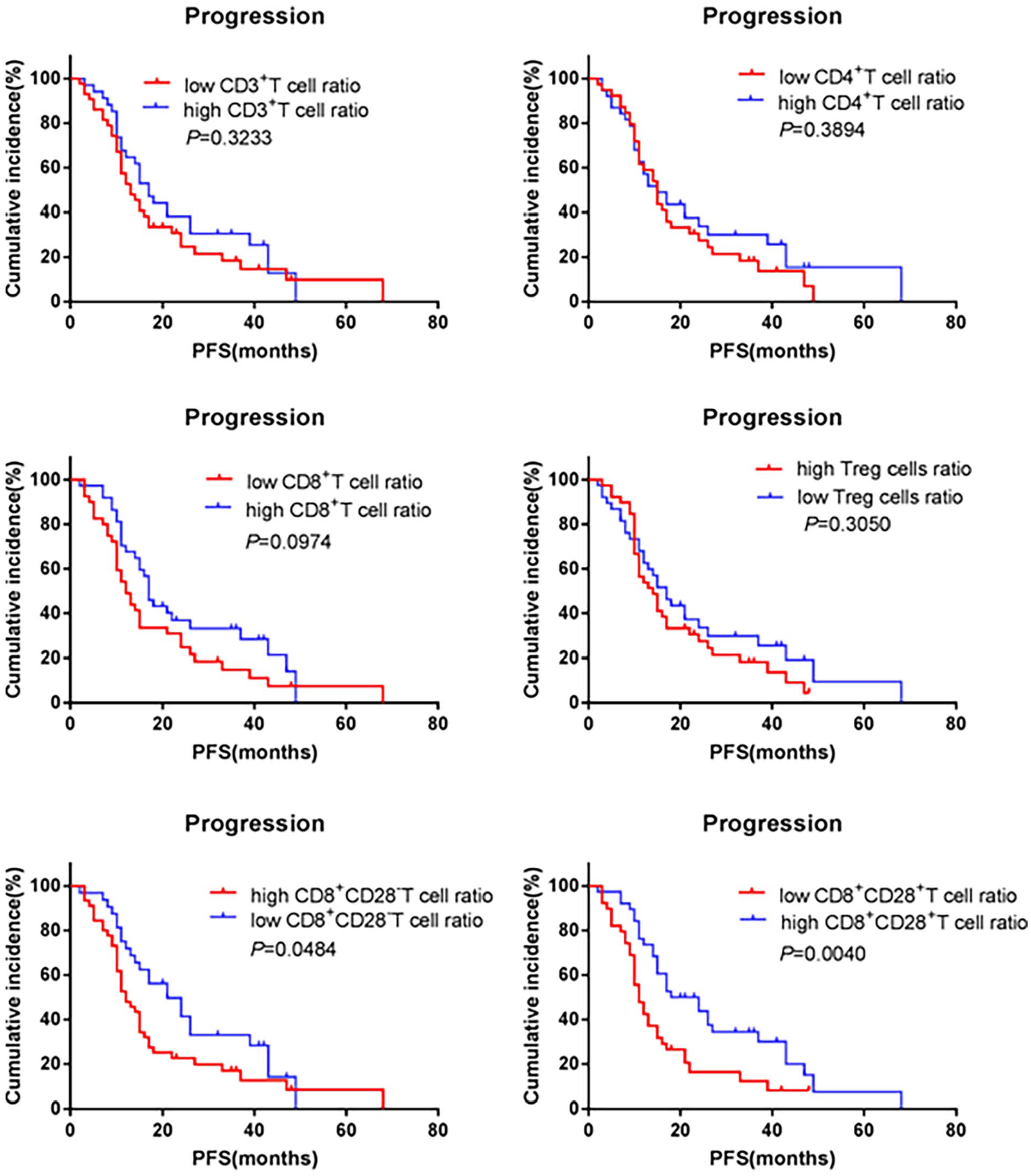
Prognostic effects of T lymphocytes subsets on PFS. PFS indicates progression-free survival.
Multivariable Cox regression analysis of PFS and OS.

Abbreviations: CI, confidence interval; HR, hazard ratio; IDH, isocitrate dehydrogenase; OS, overall survival; PFS, progression-free survival.
None of the clinicopathological variables were associated with OS either. With regard to the T lymphocyte subset ratios, the CD8+ CD28- T cell (P = .03), CD8+ CD28+ T cell (P = .01), and T reg (P = .06) ratios were correlated with OS in the univariate analysis (Figure 4), but none of these were independent predictors of OS in the multivariate analysis.

Prognostic effects of T lymphocytes subsets on OS. OS indicates overall survival.
Discussion
The application of immunotherapy in many malignant tumors (including children’s nervous system malignant tumors) can get lower toxicity and curative effect, which leads to the need to better understand the response of immune cells to these malignant tumors and their treatment. In addition, there is little research on the relationship between treatment response and immune system in pHGG.
In this cohort of 77 pediatric patients, comparison of the ratio of individual lymphocyte subset proportions before treatment to their proportions after treatment showed that the ratios of CD3+ T cells, CD4+ T cells, and CD8+ CD28+ T cells were significantly higher, while the ratio of CD8+ CD28- T cells was significantly lower, in the CR + PR + SD group than in the PD group. Binary logistic regression analysis of these 4 lymphocyte subsets showed that in HGG patients receiving CCRT, elevation in the proportion of CD8+ CD28+ T cells is a predictor of better prognosis. In line with our findings, CD8+ CD28+ T cells have been found to play a vital role as antitumor immune cells in multiple tumors.21,22
A study revealed that exhausted CD8+ T cells are rescued by PD-1-targeted therapies in a CD28-dependent manner. 23 In contrast, the loss of CD28 expression causes CD8+ T cells to lose cytotoxic function and inhibits T cell proliferation. 24 The CD28 costimulatory receptor is expressed on all T lymphocytes at birth. 25 Binding of the CD28 receptor on T cells provides an essential second signal alongside T cell receptor ligation for naïve T cell activation. The resulting CD28 signaling has diverse effects on T cell function, including orchestrating membrane raft trapping at the immunological synapse, transcriptional changes, downstream posttranslational modifications, and actin cytoskeletal remodeling. 26 This leads to intracellular biochemical events such as activation of survival and proliferation signals, induction of IL-2, activation of telomerase, stabilization of mRNA for several cytokines, increased glucose metabolism, and enhanced T cell migration and homing.27,28 Therefore, the elevation of CD8+ CD28+ T cells by CCRT might have various regulatory effects on the antitumor immune microenvironment that promote the amplification and activation of surrounding CD4+ T cells and CD8+ T cells and eventually enhance the antitumor immune response and prolong survival. Thus, the enhanced antitumor immune activation induced by CD8+ CD28+ T lymphocytes could contribute to the therapeutic effect of CCRT. 29 Our center has previously studied the dynamic changes of peripheral blood lymphoid subsets after dendritic cell-cytokine induced killer treatment of gastric cancer and pancreatic cancer. We found that the increase of CD3+ CD28+ T cells after treatment often indicates a better prognosis.14,16 One study discussed the prognostic value of peripheral blood CD8+ CD28+ T cells in patients with NSCLC undergoing chemo(radio)therapy. The study found that an increase in the level of CD8+ CD28+ T cells independently predicted a favorable OS and PFS in patients with lung adenocarcinoma. 30 Another study investigated the prediction of the efficacy and safety of PD-1/PD-L1 inhibitors in cancer patients by peripheral blood CD8+ CD28+ T lymphocytes. The results showed that patients who responded to PD-1/PD-L1 inhibitors had significantly higher circulating CD8+ CD28+ T cell counts. Elevated levels of circulating CD8+ CD28+ T cells are a potential biomarker for immunotherapy response and better prognosis. 31 CD28 is involved as the receptor for the second signal of T-cell activation and is a marker for an effector memory population of T cells following adoptive transfer. 14
To further understand the role of T lymphocyte subsets in the therapeutic effects of CCRT, we analyzed differences in survival according to the T lymphocyte subset ratios. The results indicated that an elevated CD8+ CD28+ T cell ratio was associated with markedly prolonged survival, whereas an elevated CD8+ CD28− T cell ratio was associated with reduced survival. In contrast, the other lymphocyte subsets did not have a significant influence on survival. The findings for PFS and OS were consistent. To exclude the effect of confounders and confirm the findings, we conducted COX regression analysis using T lymphocyte subset ratios and IDH status, and the findings verified that the proportion of CD8+ CD28+ T cells was significantly associated with PFS. However, none of the lymphocyte subsets were significantly associated with OS. This difference in the findings for OS and PFS could be related to subsequent treatments and the life cycle of CD8+ CD28+ T cells.
Pseudoprogression constitutes a major challenge for response assessment in various pediatric and adult brain tumors, which may result in discontinuing effective treatment, unnecessarily exposing these patients to additional salvage protocols, or inappropriately enrolling these patients in clinical trials, as well as great anxiety for patients and families. The absolute criterion for distinguishing pseudoprogression from true progression is histological diagnosis. However, due to the associated risks, surgical intervention to obtain tissue is rarely performed in children. Therefore, diagnosis is usually based on retrospective imaging as well as markers in the blood and cerebrospinal fluid.32,33 In our study, the effect of pseudoprogression on peripheral blood T lymphocyte subsets could not be analyzed due to the rapid brain cells repair and a few cases of pseudoprogression in children, which was difficult to constitute an effective statistical analysis.
Some important limitations of our study need to be mentioned. First, the analysis was limited to peripheral blood immune cells, rather than the cellular composition within the tumor tissue itself. Therefore, it is unclear whether similar changes induced by radiotherapy also occur at the tumor site, and this needs to be examined in future investigations. Second, there is a possibility of a selection bias because of this was a single-center retrospective study. Third, the clinical treatment of glioma is guided by the updated WHO classification guideline now; however, the patients were enrolled in this cohort from 2012 to 2018. To guarantee the uniform case data, we applied the classification standard based on a version of the WHO classification predating 2016. This set of data lacks the predictive value of recent features for this particular patient population. To verify the potential of circulating T-lymphocyte subsets as new prognostic variables in clinical practice, a well-designed multicenter large-scale prospective study on pHGG patients applying the updated WHO classification standard is required.
Conclusion
In this study, we have investigated the significance of peripheral blood T lymphocyte subsets as clinical predictors of treatment outcome after CCRT in pHGG patients. Our findings indicated that the proportions of T reg cells, CD8+ T cells, and CD8+ CD28− T cells were elevated after treatment, especially the proportion of CD8+ CD28+ T cells was a significant predictor of prognostic. Concurrent chemoradiotherapy is known to have a myelosuppressive effect that results in the suppression of lymphocyte production. However, our findings indicate that in pHGG patients, CCRT resulted in a decrease in the proportion of CD3+ T cells and an increase in the proportion of T lymphocytes that are associated with antitumor immune responses. Thus, the present findings demonstrate that CCRT had an effect on the antitumor immune response in patients with HGG.
Supplemental Material
sj-docx-1-onc-10.1177_11795549241227421 – Supplemental material for Peripheral Blood CD8+ CD28+ T Cells as an Independent Predictor of Treatment Response and Survival After Concurrent Chemoradiotherapy in Pediatric High-Grade Glioma Patients
Supplemental material, sj-docx-1-onc-10.1177_11795549241227421 for Peripheral Blood CD8+ CD28+ T Cells as an Independent Predictor of Treatment Response and Survival After Concurrent Chemoradiotherapy in Pediatric High-Grade Glioma Patients by Shuo Wang, Xiaofeng Mu, Xiaoli Wang, Li Chen, Changyu Lu and Linan Song in Clinical Medicine Insights: Oncology
Footnotes
Acknowledgements
The authors thank Xiaoguang Qiu for valuable discussion and Jiangping Wu for assistance with the experiments.
Author Contributions
Conceived and designed the analysis: LS and SW.
Collected the data: XW and LC.
Contributed data or analysis tools and performed the analysis: XM.
Wrote the article: LS, CL, and SW.
Funding acquisition: LS.
Study supervision: CL and SW.
Technical support: CL.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Youth Fund (2018-q14) and Open Research Funding (2019-KF03) of Beijing Shijitan Hospital, Capital Medical University, Wu Jieping Medical Foundaion (320.6750.2022-22-2).
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Ethics Statement
This pilot study was approved by the Beijing Century Altar Ethics Committee (2018-64). The study was conducted in accordance with the ethical standards of the Declaration of Helsinki of the World Medical Association.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
