Abstract
Background:
Hepatocellular carcinoma (HCC) patients have a poor prognosis after radio-frequency ablation (RFA), and investigating the risk factors affecting RFA and establishing predictive models are important for improving the prognosis of HCC patients.
Methods:
Patients with HCC undergoing RFA in Shenzhen People’s Hospital between January 2011 and December 2021 were included in this study. Using the screened independent influences on recurrence and survival, predictive models were constructed and validated, and the predictive models were then used to classify patients into different risk categories and assess the prognosis of different categories.
Results:
Cox regression model indicated that cirrhosis (hazard ratio [HR] = 1.65), alpha-fetoprotein (AFP) ⩾400 ng/mL (HR = 2.03), tumor number (multiple) (HR = 2.11), tumor diameter ⩾20 mm (HR = 2.30), and platelets (PLT) ⩾ 244 (109/L) (HR = 2.37) were independent influences for recurrence of patients after RFA. On the contrary, AFP ⩾400 ng/mL (HR = 2.48), tumor number (multiple) (HR = 2.52), tumor diameter ⩾20 mm (HR = 2.25), PLT ⩾244 (109/L) (HR = 2.36), and hemoglobin (HGB) ⩾120 (g/L) (HR = 0.34) were regarded as independent influences for survival. The concordance index (C-index) of the nomograms for predicting disease-free survival (DFS) and overall survival (OS) was 0.727 (95% confidence interval [CI] = 0.770-0.684) and 0.770 (95% CI = 0.821-7.190), respectively. The prognostic performance of the nomograms was significantly better than other staging systems by analysis of the time-dependent C-index and decision curves. Each patient was scored using nomograms and influencing factors, and patients were categorized into low-, intermediate-, and high-risk groups based on their scores. In the Kaplan-Meier survival curve, DFS and OS were significantly better in the low-risk group than in the intermediate- and high-risk groups.
Conclusions:
The 2 prediction models created in this work can effectively predict the recurrence and survival rates of HCC patients following RFA.
Introduction
Hepatocellular carcinoma (HCC) was one of the top 3 causes of cancer death in many countries. 1 Although surgery is considered the primary treatment for early HCC,2 some patients cannot tolerate surgical treatment due to conditions such as cirrhosis. Ablation therapy, which has been widely used, can achieve similar efficacy to surgical resection in some patients with early stage HCC.3-6 Radio-frequency ablation (RFA) is a commonly used ablation modality for minimally invasive treatment of HCC, which has the advantages of convenient operation, short hospitalization time, precise efficacy, and controllable ablation range. 7 Despite advances in HCC treatment, the mortality and recurrence rates of HCC patients remain significant. Worldwide, HCC patient mortality rates are 11.1% and 43.8% at 1 and 5 years, respectively, while recurrence rates are as high as 28.9% and 64.8% at 1 and 5 years. Patients with HCC have a mortality rate of 39.8% at 5 years after RFA, and the recurrence rate is much higher at 74.8%.8,9
The high mortality and recurrence rates of HCC patients after RFA have a significant impact on clinical treatment outcomes and patient quality of life. Nevertheless, there are currently no reliable ways to predict survival and recurrence in HCC patients after RFA. In this study, we got the risk variables for HCC recurrence and survival following RFA by gathering imaging and laboratory data from patients at Shenzhen People’s Hospital. We tried to develop 2 risk prediction models for HCC recurrence and survival after RFA to help doctors perform timely preoperative interventions and tailor the treatment plan and postoperative follow-up to improve the prognosis of HCC patients.
Materials and Methods
Study design and participants
Patients with HCC undergoing RFA in Shenzhen People’s Hospital between January 2011 and December 2021 were included in this study.
Treatment method
The included patients were treated with computed tomography (CT)-guided RFA with a Siemens SOMATOM. ART scanner, a Radionics Cool-tip RFA system (USA), and various types of ablative treatment needles. The radio-frequency needle type and puncture route are chosen based on the size and location of the tumor, with the puncture site selected under CT guidance. The lesion is pierced with a matching RFA needle after disinfection, towel laying, and local anesthesia. The RFA treatment needle is then used to puncture the lesion with a radio-frequency electrode needle, fixing the liver in the respiratory state. The electrodes are opened for RFA once a CT scan shows a successful puncture, ensuring complete thermal coagulation and necrosis of the tumor tissue as well as at least 1 cm of surrounding normal liver tissue, with complete RFA of the shunt tract as the target area. A dynamic enhancement CT, multiparametric magnetic resonance imaging (MRI) scan, or ultrasonography will be examined about a month following ablation to assess the ablation’s impact. In addition, it is important to monitor dynamic changes in serological tumor markers.
Postoperative follow-up
From the start of therapy through the date of death or the final follow-up, the overall survival (OS) rate was determined. From the date of treatment through the date of tumor recurrence or the last date of follow-up, the disease-free survival (DFS) rate was determined. New nodules in the liver, metastatic lesions inside or outside the liver, or the development of previously treated lesions are all considered postoperative recurrence. Following complete ablation, patients receive serum tumor markers and imaging examinations every 2 to 3 months for the first 3 years and then every 6 months after 3 years. 11 Patients who had newly acquired hepatic lesions or local reoccurring lesions received another ablation treatment, which is ended when the patient dies. The follow-up period will conclude on December 31, 2022.
Statistical analysis
The Shapiro-Wilk test and Levene’s test were used to identify all continuous variables. Non-normally distributed continuous variable is expressed as median (quartile distance, interquartile range [IQR]); categorical data were expressed as number. Use X-tile 3.6.1 software to determine the best cutoff value for the relevant metric. Independent prognostic factors for DFS and OS were determined by Cox regression analysis; variables with P < .05 for univariate analysis were substituted into the multivariate analysis; recurrence and survival nomogram models incorporate independent factors obtained from multivariate Cox regression analysis. Two nomogram prediction models were developed, and calibration curves were used to test the model’s consistency with the actual results. Predictive accuracy of the model at different time points was assessed by time-dependent area under the receiver operating characteristic (ROC) curve and the concordance index (C-index, calibration of C-index by means of 1000 boot-strap resamples); decision curve analysis (DCA) was used to evaluate the clinical validity of models; finally, using X-tile software, the total scores generated by these models were classified as low-risk, intermediate-risk, and high-risk, and Kaplan-Meier (K-M) survival analysis was used to compare DFS and OS between the subgroups. Seven conventional liver cancer staging systems (Barcelona clinic liver cancer [BCLC] stage, Hong Kong liver cancer [HKLC] stage, China liver cancer [CNLC] stage, Cancer of the Liver Italian Program [CLIP], Chinese University Prognostic Index [CUPI] stage, Okuda score, and French score) were compared with the validation process of the aforementioned models. The above data were statistically analyzed by R software (version 4.2.3) and X-tile software (version 3.6.1 ).
Results
Patient characteristics
This study included 201 patients, including 163 men and 38 women, with a median age of 57.0 (48.7-66.0) years, and 117 with a diagnosis of cirrhosis (Table 1), the DFS at 1, 3, and 5 years for patients without cirrhosis was 59.5%, 51.7%, and 40.8%, respectively, whereas the OS at 1, 3, and 5 years for patients with cirrhosis was only 43.1%, 27.5%, and 18.3%. Median DFS and OS follow-up times were 9.5 (4.0-27.0) months and 31.0 (17.0-48.0) months, respectively. Before the study’s conclusion, 117 patients had tumor recurrence and a total of 77 patients had died.
Preoperative clinical characteristics.

Abbreviations: AFP, alpha-fetoprotein; AKP, alkaline phosphatase; ALB, albumin; ALBI, albumin-bilirubin; CR, creatinine; Fg, fibrinogen; GGTP, gamma-glutamyl transpeptidase; GOT, glutamic-oxaloacetic transaminase; GPR, gamma-glutamyl transpeptidase-to-platelet ratio; GPT, glutamic-pyruvic transaminase; HBV, hepatitis B virus; HGB, hemoglobin; INR, International standard ratio; LMR, lymphocyte-to-monocyte ratio; NE, neutrophilic granulocyte; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PLT, platelets; PNI, prognostic nutritional index; PT, prothrombin time; SII, systemic immune-inflammation index; TBIL, total bilirubin; TP, total protein; WBC, white blood cells.
Cox regression analyses and construction of nomogram models
To increase the uniformity of the data and to avoid the exclusion of meaningful influencing factors, this study stratified the utilization of continuous data according to the clinical significance of the influencing factors and the X-tile, and the stratified results were subjected to K-M survival analysis, which suggested that there was a statistically significant difference in DFS and OS of the groupings of influencing factors. Using the Cox risk regression model analysis, we included the factors with P < .05 in the results of the univariate analysis in the multivariate analysis and then included the factors with P < .05 in the multivariate analysis in the modeling, which showed that cirrhosis (hazard ratio [HR] = 1.60; 95% confidence interval [CI] = 1.09-2.36), alpha-fetoprotein (AFP) ⩾400 ng/mL (HR = 1.97; 95% CI = 1.10-3.35), tumor number (multiple) (HR = 2.11; 95% CI = 1.46-3.03), tumor diameter ⩾20 mm (HR = 2.30; 95% CI = 1.51-3.50), and platelets (PLT) ⩾244 (109/L) (HR = 2.37; 95% CI = 1.28-4.39) were risk factors for recurrence of patients after RFA (Table 2). Recurrence nomogram model was constructed for the screened variables, which predicted DFS (Figure 1A). On the contrary, AFP ⩾400 ng/mL (HR = 2.48; 95% CI = 1.39-4.44), tumor number (multiple) (HR = 2.52; 95% CI = 1.52-4.18), tumor diameter ⩾20 mm (HR = 2.25; 95% CI = 1.17-4.31), PLT ⩾244 (109/L) (HR = 2.36; 95% CI = 1.15-4.87), and hemoglobin (HGB) ⩾120 (g/L) (HR = 0.34; 95% CI = 0.19-0.61) were regarded as risk factors for survival of patients after RFA (Table 3). Survival nomogram model was constructed for the screened variables, which predicted OS (Figure 1B). In the 2 predictive nomogram models, each influencing factor is assigned a corresponding score. By summing the scores for each variable, we can calculate the predicted probability of DFS and OS for each patient.
Results of recurrence Cox regression analysis.

Abbreviations: AFP, alpha-fetoprotein; AKP, alkaline phosphatase; ALB, albumin; ALBI, albumin-bilirubin; APTT, activated partial thromboplastin time; CI, confidence interval; CR, creatinine; Fg, fibrinogen; GGTP, gamma-glutamyl transpeptidase; GOT, glutamic-oxaloacetic transaminase; GPR, gamma-glutamyl transpeptidase-to-platelet ratio; GPT, glutamic-pyruvic transaminase; HBV, hepatitis B virus; HGB, hemoglobin; HR, hazard ratio; INR, International standard ratio; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-lymphocyte ratio; PLT, platelets; PNI, prognostic nutritional index; PT, prothrombin time; SII, systemic immune-inflammation index; TACE: transcatheter arterial chemoembolization; TBIL, total bilirubin; TP, total protein.

Preoperative nomograms for DFS (A) and OS (B) at 1, 2, and 3 years for HCC patients after ablation. For each individual patient, the corresponding value is located on each variable axis, and a line is drawn upward to determine the number of points received for each variable value. Linear predictor (LP) is obtained by adding the products of the independent variables and their regression coefficients (β), and after obtaining the LP, the prediction scores can be calculated. Total point axis represents the sum of these numbers, and 3 lines are drawn downward to the1-, 2-, and 3-year DFS (A) and OS (B). AFP indicates alpha-fetoprotein; DFS, disease-free survival; HCC, hepatocellular carcinoma; HGB, hemoglobin; PLT, platelets; OS, overall survival.
Results of survival Cox regression analysis.

Abbreviations: AFP: alpha-fetoprotein; AKP: alkaline phosphatase; ALB: albumin; ALBI: albumin-bilirubin; APTT: activated partial thromboplastin time; CI: confidence interval; CR: creatinine; Fg: fibrinogen; GGTP: gamma-glutamyl transpeptidase; GOT: glutamic-oxaloacetic transaminase; GPR: gamma-glutamyl transpeptidase-to-platelet ratio; GPT: glutamic-pyruvic transaminase; HBV: hepatitis B virus; HGB: hemoglobin; HR: hazard ratio; INR: International standard ratio; LMR: lymphocyte-to-monocyte ratio; NLR: neutrophil-to-lymphocyte ratio; PLR: platelet-lymphocyte ratio; PLT: platelets; PNI: prognostic nutritional index; PT: prothrombin time; SII: systemic immune-inflammation index; TACE: transcatheter arterial chemoembolization. TBIL: total bilirubin; TP: total protein.
Calibration and verification of nomogram
Calibration curves of nomogram models demonstrated excellent agreement between forecasts and actual observations for DFS and OS in 1, 3, and 5 years (Figure 2). The C-index of the nomogram models predicting DFS and OS were 0.727 (95% CI = 0.770-0.684) and 0.770 (95% CI = 0.821-7.190), respectively. By analyzing the time-dependent C-index, the nomogram models’ prognostic performance at different time points was much superior than that of previous staging methods (Figure 3). The analysis of decision curves revealed that the 2 nomograms greatly outperformed other staging systems, offering the greatest net benefit within a reasonable threshold probability (Figure 4). The total points of nomograms were classified into the low-, intermediate-, and high-risk groups by X-tile analysis. In the K-M survival curves, DFS (low-risk group, 65.2%, 51.5%, and 38.8%; intermediate-risk group, 22.8%, 12.7%, and 6.8%; high-risk group 10.6%, 0%, and 0 %, P < .001) and OS (low-risk group, 95.4%, 91.1%, and 82.9%; intermediate-risk group, 82.2%, 71.5%, and 62.7%; and high-risk group 41.4%, 24.3%, and 24.3 %, P < .001) at 1-, 3-, and 5-year were significantly higher in the low-risk group than in the intermediate- and high-risk groups (Figure 5).
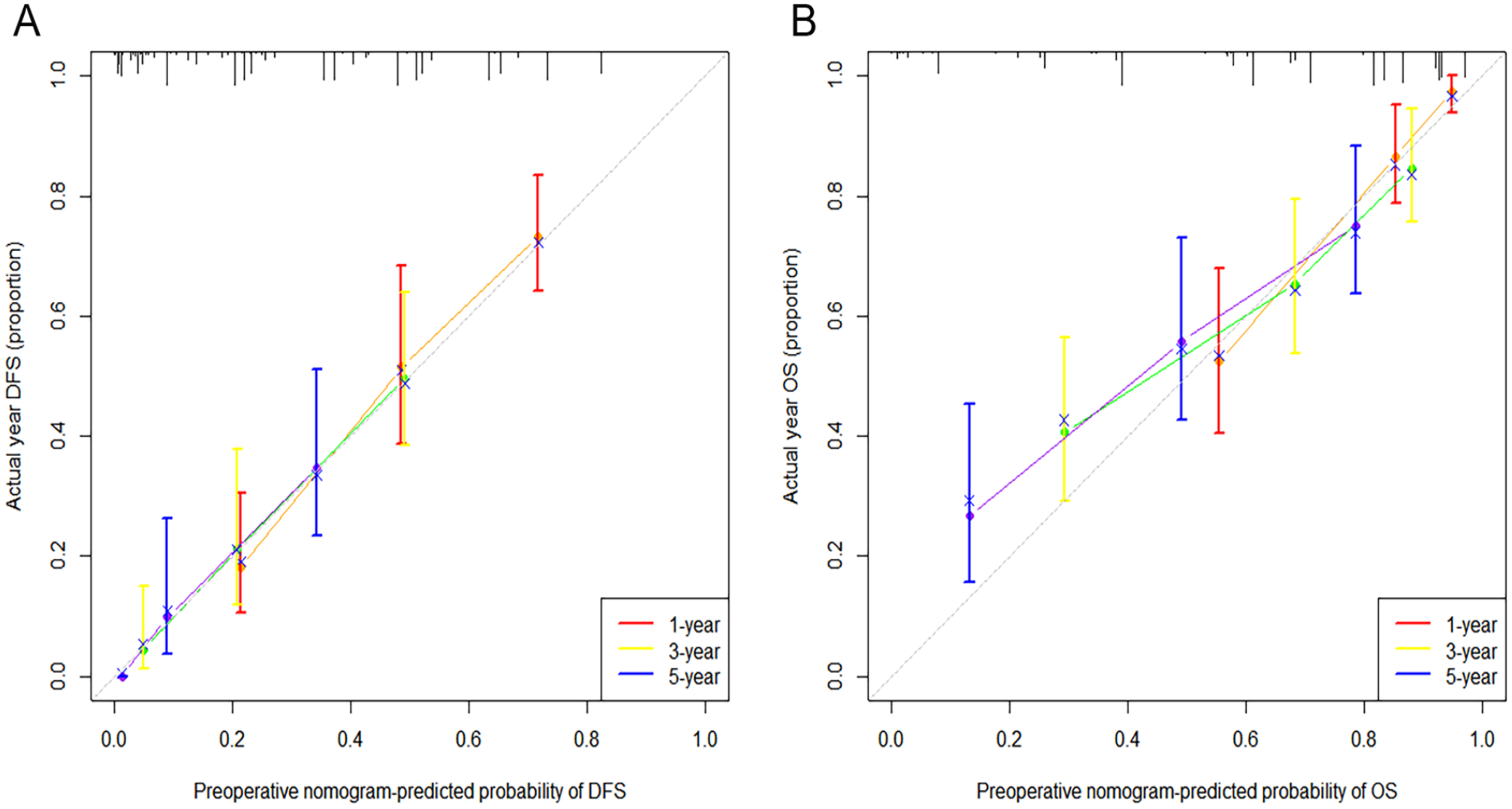
Calibration curves of preoperative nomograms for DFS (A) and OS (B) at 1, 3, and 5 years. DFS indicates disease-free survival; OS, overall survival.

Time-dependent C-index of DFS (A) and OS (B) for the established nomograms and other staging systems. BCLC indicates Barcelona clinic liver cancer; C-index, concordance index; CLIP, Cancer of the Liver Italian Program; CNLC, China liver cancer; CUPI, Chinese University Prognostic Index; DFS, disease-free survival; HKLC, Hong Kong liver cancer; OS, overall survival.

Decision curves of DFS (A to C) and OS (D to F) at 1, 2, and 3 years for the established nomograms and other staging systems. BCLC indicates Barcelona clinic liver cancer; CNLC, China liver cancer; CLIP, Cancer of the Liver Italian Program; CUPI, Chinese University Prognostic Index; DCA, decision curve analysis; DFS, disease-free survival; HKLC, Hong Kong liver cancer; OS, overall survival.

Kaplan-Meier analysis of OS (A) and DFS (B) in different risk strata of 2 nomograms; the prediction of OS and DFS according to the nomogram points divided into low-, intermediate-, and high-risk groups by X-title software. The lightly stained areas on both sides of the curve are 95% CI ranges. CI indicates confidence interval; DFS, disease-free survival; OS, overall survival.
Discussion
Radio-frequency ablation is one of the radical treatments for HCC. It is less dangerous and intrusive than surgery, and it can ameliorate the tumor microenvironment and provide effective local tumor control. However, it still has a high rate of recurrence and death after RFA.12-14 It is unclear which risk factors affect patients undergoing RFA, and there are not enough reliable prognostic models available to provide clinical guidance. In this study, we constructed 2 nomograms on prognostic models for patients with HCC after RFA. Mortality and recurrence rates are the main indicators for assessing the prognosis. Both of these indicators have gotten a lot of attention.
Many studies have shown that preoperative indicators, such as AFP, tumor diameter, multifocal HCC, and cirrhosis, influence postoperative recurrence in HCC patients.15-18 Hemoglobin levels are also considered to be important factor influencing the prognosis of HCC patients. 19 In addition, patients with cancer frequently have thrombocytosis, and numerous studies have shown that PLT play a crucial role in the angiogenesis process during malignancy. Tumor cells also trigger platelet activation and subsequent aggregation through both direct and indirect methods.20,21
In our research, we found that AFP, tumor diameter, tumor number, and PLT all had independent effects on HCC recurrence and survival after RFA. These influencing factors have been mentioned in previous studies.16,17 We stratified the above influences according to clinical significance as well as X-tile software and performed a K-M survival curve analysis between the groups. Our study also discovered that cirrhosis was a separate risk factor for recurrence, and HGB <120 (g/L) was a well-representative independent influence on the nomogram of the OS prediction model, in addition to the previously mentioned risk factors.
Because AFP is frequently observed to rise to exceptionally high levels during the development of HCC, it is used in HCC screening. AFP levels more than 400 ng/mL are considered to be strongly suggestive of HCC in the standard for diagnosis and treatment of primary liver cancer (2022 edition) when other conditions that can boost AFP levels have been ruled out. 10 Lu et al 22 discovered that preoperative AFP level more than 200 ng/mL in HCC patients had significantly poorer survival rates than those with low AFP, indicating that AFP represents a separate risk factor for recurrence following RFA. Alpha-fetoprotein decreased the activity of dendritic cells, natural killer cells, and T lymphocytes and enhanced cell proliferation, cell motility, and invasive characteristics of HCC cell lines.23,24 We stratified AFP at 400 ng/mL in this study based on the X-tile stratification results and included it in the Cox survival regression model analysis of recurrence and survival, which showed that AFP ⩾400 ng/mL was an independent risk factor not only for recurrence but also for survival. This result further demonstrates that high AFP levels have an important impact on the prognosis of HCC patients treated with RFA. In addition, the association of preoperative PLT and other coagulation-related indicators with the tumor microenvironment has received increasing attention.25-28 Platelets are nucleated cell fragments produced by megakaryocytes, whose main function is to participate in coagulation and hemostasis. A growing number of recent clinical studies have shown that PLT are involved in and enhance tumor metastasis and recurrence.29-31 It has been demonstrated that the development of tumors causes an increase in PLT and their aggregation, which hinders the immune system’s ability to detect and destroy cancer cells or encourages endothelial cell obstruction, which in turn helps cancer cells survive and proliferate. 32 In this study, PLT ⩾244 (109/L) was identified by X-tile stratification as a common risk factor for recurrence and survival, and patients with PLT ⩾244 (109/L) had significantly increased recurrence and mortality rates after RFA.
Tumor diameter and tumor number are often included in many international staging of HCC, including the CNLC stage and BCLC stage. Tumor diameter is closely linked to the prognosis of HCC patients. Microvascular infiltration is tightly correlated with tumor diameter. Liang et al 33 demonstrated that the risk of cancer microvascular invasion increases with increasing tumor diameter. Many studies have shown that multifocal HCC is a risk factor for HCC.34-37 Hepatocellular carcinoma develops into multifocal HCC due to microscopic metastases of unicentric origin or multicentric origin, but microscopic metastases in tumors are difficult to detect and do not allow for timely intervention, resulting in more patients relapsing early after RFA.36,37 Tumor cells were shown to be more prevalent in the blood in patients with multifocal HCC than in those with monofocal HCC in a study from 2021, which may be connected to the fact that multifocal HCC is more likely to metastasize. 38 This study was analyzed using X-tile software, stratifying tumor diameter by 20 mm, and found that patients with tumor diameter ⩾20 mm and tumor number (multiple) had significantly lower 1-, 3-, and 5-year DFS and OS after RFA.
Cirrhosis is caused by various mechanisms of liver injury that result in necrosis and fibrous formation; histologically, it is distinguished by the use of nodal regeneration surrounded by dense fibrous septa, followed by parenchymal loss and collapse of the liver structure. Hypoproteinemia, impaired liver function, and gastrointestinal infections, which are typically strongly associated with an elevated recurrence rate in these patients, are disorders that can be brought on by cirrhosis.39-41 The cirrhosis in the patients in our study was in the compensated phase, as determined by symptoms and imaging, with a Child-Pugh grade A liver function score. By using data from our acquired study, we discovered that the DFS of patients with cirrhosis who underwent RFA was significantly lower than that of noncirrhotic individuals (Figure 6). In addition, low preoperative HGB levels have been shown to have a significant impact on the prognosis of patients with tumors, and our investigation reflects this finding.42,43 Our study’s findings indicate that HGB ⩾120 (g/L) is a significant influencing factor for reducing postoperative mortality in HCC patients at 1-, 3-, and 5-year, OS: HGB <120 (g/L), 69.6%, 54.6%, and 49.2%, P < .001; HGB ⩾120 (g/L): 86.5%, 80.7%, and 73.2%, P < .001. Patients’ low HGB levels should be actively corrected before RFA to improve prognosis.

Kaplan-Meier survival analysis and log-rank test in recurrence and survival nomogram based on DFS (A to E) and OS (F to J); yellow curves represent low-risk groups, and blue curves represent high-risk groups; the lightly stained areas on both sides of the curve are 95% CI ranges. The lightly stained areas on both sides of the curve are 95% CI ranges. AFP indicates alpha-fetoprotein; CI, confidence interval; DFS, disease-free survival; HGB, hemoglobin; OS, overall survival; PLT, platelets.
Undoubtedly, there are some shortcomings in our study. Since there was no external validation of the data used in this investigation, patient selection bias could not be totally eliminated. Furthermore, the conclusions drawn from this study need to be validated and optimized by further prospective, multicenter, large-sample, and long-term studies.
Conclusions
In conclusion, this study effectively identified independent prognostic factors for recurrence and survival, and developed and validated credible nomograms that can accurately predict DFS and OS in patients after RFA. Our nomograms not only use complete and accurate clinical data, but also outperform 7 internationally widely used HCC scoring systems in predicting postoperative recurrence and survival. Our prognostic models allow for efficient patient distinction and preoperative assessment, which can provide more effective interventions and therapeutic measures for different patients. On one hand, we can provide timely intervention to patients with preoperative risk factors, such as lowering preoperative PLT levels and increasing preoperative HGB levels; on the other hand, for patients with high AFP levels, cirrhosis, large tumor diameter, or multifocal HCC, we can try other treatment modalities (such as surgical resection) or combine with transcatheter arterial chemoembolization (TACE) after RFA to improve the patient’s prognosis.
Footnotes
Acknowledgements
The authors acknowledge Mr He (Yongzhu He, The First Affiliated Hospital of Nanchang University) for providing high-quality advice to researchers.
Author Contributions
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Shenzhen Science and Technology Innovation Commission Foundation (JCYJ20210324113008022) and Guangdong Basic and Applied Basic Research Foundation (2021A1515220059).
Data Availability Statement
The data sets generated and analyzed during this study are not publicly available due to privacy and ethical concerns, but are available from the corresponding author on reasonable request.
Ethical Approval
This study was a retrospective study. Approval of the research protocol: All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study was approved by the ethics committee of the Shenzhen People’s Hospital (Ethics number: LL-KY-2023064-01/2023-04-03). Individual consent for this retrospective analysis was waived.
