Abstract
Background:
The treatment response to radiotherapy of hepatocellular carcinoma (HCC) primary tumors and different types of tumor thrombus under identical radiation doses remains uncertain.
Objectives:
This multicenter study aimed to evaluate and compare the radiotherapy responses of primary tumors (PT), portal vein tumor thrombus (PVTT), and hepatic vein tumor thrombus (HVTT) in HCC patients and to establish a prediction model for treatment response.
Design:
This multicenter retrospective cohort study analyzed the treatment response of 242 HCC patients with macrovascular tumor thrombus who received radiotherapy combined with systemic therapy from five hospitals.
Methods:
The objective response rate (ORR) was evaluated using Response Evaluation Criteria in Solid Tumors (RECIST) V1.1 and modified RECIST (mRECIST) criteria. Independent factors of treatment response were identified by logistic regression analysis and used to create a nomogram. Model performance was evaluated through the area under the receiver operating characteristic curve (AUC), calibration curves, and decision curve analysis (DCA).
Results:
Among the entire cohort, the ORRs evaluated by RECIST were 51.65% for PT, 56.32% for PVTT, and 81.93% for HVTT; the ORRs evaluated by mRECIST were 55.79% for PT, 62.63% for PVTT, and 87.95% for HVTT. Both criteria demonstrated that HVTT exhibited significantly higher response rates than PT and PVTT (p < 0.001). Multivariate logistic regression analysis identified irradiated primary tumor size, biologically effective dose, and tumor thrombus type as independent predictors of response in the radiation-field lesions. The developed nomogram showed good discriminative ability with an AUC of 0.788 in the training cohort and 0.736 in the external validation cohort. Calibration and DCA indicated that the model provided reliable predictions and substantial clinical benefit.
Conclusion:
Under identical radiation doses, macrovascular tumor thrombus, especially HVTT, is associated with a higher response rate to radiotherapy compared to HCC primary tumors. The nomogram demonstrated good predictive performance, but prospective validation in larger cohorts is still warranted.
Introduction
Primary liver cancer is the sixth most prevalent malignancy and the third leading cause of cancer-related mortality worldwide, 1 and hepatocellular carcinoma (HCC) accounts for 75%–85% of primary liver cancer. 2 During its progression, HCC frequently invades the vasculature, leading to the formation of tumor thrombus, primarily portal vein tumor thrombus (PVTT) and hepatic vein tumor thrombus (HVTT), with a clinical detection rate ranging from 44% to 62.2%3,4 and 12.5% to 26.2%,4,5 respectively. The presence of major vascular invasion is a hallmark of advanced-stage HCC and is classified as Barcelona Clinic Liver Cancer stage C. Current clinical guidelines recommend systemic therapy as the primary treatment strategy for these patients. However, the efficacy of systemic therapy remains limited, with objective response rate (ORR) ranging from 2% to 27.3%,6,7 and the prognosis of patients with major vascular invasion remains poor.
Radiotherapy is one of the local treatment modalities for HCC, offering effective tumor control, with reported 2-year local control rates ranging from 83.8% to 94.6%.8,9 Previous retrospective studies suggest that vascular tumor thrombus may be more radiosensitive than the primary tumors (PT), with HVTT exhibiting a higher response rate compared to PVTT.10–13 While some studies have reported no significant difference in response rates between the primary tumors and vascular tumor thrombus.14–16 These studies vary in terms of inclusion criteria of patients, treatment regimens, prescribed radiation doses, and evaluation criteria of response, making direct comparisons challenging. To date, there is a lack of studies directly comparing the therapeutic response of HCC primary tumors and different types of vascular tumor thrombus to the same radiation dose. Whether primary tumors and tumor thrombus exhibit differential treatment response under equivalent radiotherapy doses remains inconclusive. Clarifying the sensitivity of different lesions to radiotherapy can provide a reference for clinical physicians to make treatment decisions for such patients.
The primary objective of this study is to analyze the differential treatment response of primary HCC tumors and macrovascular tumor thrombi to identical radiation doses, and develop a prediction model for the treatment response of radiotherapy in HCC with macrovascular tumor thrombus.
Patients and methods
Study design
This study is a retrospective study, and the reporting of this study adheres to the STROBE statement 17 (Table S1).
Patients
HCC with macrovascular tumor thrombus were enrolled from five hospitals (Mengchao Hepatobiliary Hospital of Fujian Medical University, First Affiliated Hospital of Fujian Medical University, Fujian Medical University Cancer Hospital, Zhangzhou Affiliated Hospital of Fujian Medical University, and First Affiliated Hospital of Xiamen University) between January 1, 2018, and August 31, 2024. The sample size of this study is determined by the number of cases diagnosed and treated in each hospital during the study period.
The inclusion criteria were as follows: (1) HCC with PVTT or HVTT, diagnosed according to the Guidelines for the Diagnosis and Treatment of Primary Liver Cancer in China (2022 edition), 2 (2) both primary tumor and tumor thrombus received radiotherapy, (3) received tyrosine kinase inhibitor (TKI) plus immune checkpoint inhibitor (ICI) as systemic treatment, and (4) completion of at least two evaluations of treatment response. The exclusion criteria were as follows: (1) combined HCC with intrahepatic cholangiocarcinoma, (2) presence of other malignancies, or (3) incomplete clinical data or missing follow-up records.
Clinical data were collected from electronic medical records. Clinical data include sex, age, hepatitis B virus (HBV) infection status, cirrhosis, Child-Pugh grade, Eastern Cooperative Oncology Group score, tumor number, primary tumor size, irradiated primary tumor size, PVTT size, HVTT size, metastasis status, alpha-fetoprotein (AFP), protein induced by vitamin K absence or antagonist-II (PIVKA-II), tumor thrombus type, previous treatment, prescribed dose of radiotherapy, and biologically effective dose (BED). HBV infection was defined as hepatitis B surface antigen (HBsAg) or HBV-DNA positive. Cirrhosis was confirmed by histopathology or clinical diagnosis. The primary tumor size was defined as the maximum diameter of intrahepatic lesions measured by imaging. Irradiated primary tumor (IRPT) size was defined as the maximum diameter of the irradiated part of the intrahepatic primary tumor that received radiotherapy. The PVTT size and HVTT size are defined as the maximum diameters of PVTT and HVTT measured radiographically, respectively. PVTT and HVTT were categorized using Cheng’s classification.18,19 Previous treatment includes surgical resection, radiofrequency ablation, microwave ablation, transcatheter arterial chemoembolization (TACE), hepatic arterial infusion chemotherapy (HAIC), and systemic treatment. The prescribed dose of radiotherapy is represented as D95%, which means the dose for 95% of the planning target volume (PTV). Other dose parameter of radiotherapy includes near-maximum dose (D2%), median dose (D50%), and near-minimum dose (D98%). To make the radiation doses comparable, the total dose was converted to BED using an L-Q model with an HCC α/β ratio of 10 Gy.
Treatment
All patients received image-guided intensity-modulated radiotherapy (IMRT) with a linear accelerator. The target area contained the primary tumor and tumor thrombus, and the prescribed dose of both was the same. Radiotherapy was combined with systemic therapy at the same time, and combined therapy was defined as a time interval of less than 1 month between systemic therapy and radiotherapy. The regimens of systemic therapy were TKI plus ICI, in which TKI includes sorafenib, lenvatinib, donafenib, regorafenib, and apatinib, and ICI includes camrelizumab, tislelizumab, and sintilimab. Systemic therapy was divided into first-line and second-line or more lines. Prior to radiotherapy, patients underwent four-dimensional contrast-enhanced computed tomography (CT) simulation to capture respiratory motion. Magnetic resonance imaging (MRI) or PET-CT was incorporated if necessary for accurate target delineation. The gross tumor volume (GTV) includes both the primary tumor and tumor thrombus. The primary tumor was defined as the tumor volume that enhanced in the arterial phase. Tumor thrombus was defined as the tumor volume that was shown as a filling defect in the venous phase. For patients with large tumor volume, multiple or diffuse lesions, to ensure the irradiation dose of the normal liver, only the tumor thrombus and partial intrahepatic lesions were irradiated. The internal target volume was delineated considering the tumor movement for every respiratory phase. The clinical tumor volume (CTV) was expanded to 5 mm from the GTV. The PTV included a 5 mm margin in the anterior–posterior and left–right directions and a 10 mm margin in the cranial–caudal direction around the CTV. The D95% ranged from 24 to 60 Gy, with daily fraction doses ranging from 2 to 8 Gy, administered 5 days per week. The dose constraints for the organs at risk (OARs) were as follows: mean dose of liver ⩽24 Gy; maximum dose of stomach/small bowel ⩽54 Gy and V50 ⩽10 mL; maximum dose of spinal cord ⩽40 Gy; left kidney, V20 ⩽20%, right kidney, V20 <30%. However, the final prescribed dose was adjusted based on the dose constraints for OARs. Cone beam CT was commonly utilized for online repositioning prior to treatment.
Assessment and follow-up
Hematological parameters, including complete blood counts, biochemical profiles, and coagulation tests, were assessed within 3 days prior to initiating radiotherapy and within 2 weeks following its completion. In addition, AFP, PIVKA-II, chest CT, and liver contrast-enhanced CT or MRI were conducted at intervals of every 6–8 weeks. The images were evaluated by two physicians with 5 and 8 years of experience; discrepancies in opinions were resolved through discussion to achieve consensus. Follow-up for this study was censored on February 28, 2025.
Study outcomes
The primary outcome was ORR. The secondary outcome was risk factors of treatment response and progression-free survival (PFS) time. Tumor response was evaluated based on enhanced CT or MRI findings according to the Response Evaluation Criteria in Solid Tumors (RECIST) V1.1 20 and modified Response Evaluation Criteria in Solid Tumors (mRECIST). 21 Complete response (CR) is defined as “the disappearance of any intratumoral arterial enhancement in all target lesions”, partial response (PR) is defined as “at least a 30% decrease in the sum of diameters of viable (enhancement in the arterial phase) target lesions, taking as reference the baseline sum of the diameters of target lesions”, stable disease (SD) is defined as “any cases that do not qualify for either partial response or progressive disease”, progressive disease (PD) is defined as “an increase of at least 20% in the sum of the diameters of viable (enhancing) target lesions, taking as reference the smallest sum of the diameters of viable (enhancing) target lesions recorded since treatment started”. The ORR was defined as the sum of CR + PR, whereas the disease control rate (DCR) was defined as CR + PR + SD. PFS was defined as the time from radiotherapy or systemic treatment to tumor progression or death.
Statistical analysis
Data were analyzed via Rstudio (Posit Software, version 4.2.2) using “readxl,” “readr,” “Table 1,” “survminer,” “survival,” “ggplot2,” “ggpubr,” “lubridate,” “magrittr,” “glmnet,” “broom,” “tidyverse,” “autoReg,” “dcurves,” “rmda,” and “pROC” packages. Categorical variables were reported as numbers (percentages), and differences between groups were evaluated using the chi-square test or Fisher’s exact test. Continuous variables were expressed as the median [Q1, Q3] and compared using Student’s t test or the Mann–Whitney U test. Kaplan–Meier survival curves were used to assess PFS. Univariate and multivariate regression analyses in the training cohort were applied to identify risk factors of treatment response. Odds ratios (ORs) as estimates of the relative risk with 95% confidence intervals (CIs) were obtained for each risk factor. Variables with statistical significance in univariate analysis were included in multivariate analysis, and variables that reached statistical significance in multivariate analysis were used as predictors for constructing a nomogram. Model fit was assessed by calibration plots via 1000 bootstrap resamples. The clinical utility of the models was evaluated by decision curve analysis (DCA). p < 0.05 was considered statistically significant.
Baseline characteristics.

AFP, alpha-fetoprotein; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis B virus; HVTT, hepatic vein tumor thrombus; PIVKA-II, protein induced by vitamin K absence or antagonist-II; PVTT, portal vein tumor thrombus.
Results
Baseline characteristics of the cohorts
A total of 242 patients were included in this study, of which 174 cases from Mengchao Hepatobiliary Hospital of Fujian Medical University were selected as the training cohort, and 68 patients from the other 4 hospitals were selected as the external validation cohort. The median follow-up time was 26.3 (19.67–32.33) months. In the entire cohort, 88.8% of patients were male, 89.3% had HBV infection, 82.2% presented with cirrhosis, 80.2% were classified as Child-Pugh class A, 68.2% exhibited multiple tumors, and 36.0% with metastatic lesions. The median prescribed radiation dose was 45 Gy, corresponding to a median biologically effective dose at α/β = 10 Gy (BED10) of 58.5 Gy. Baseline characteristics are shown in Table 1.
Treatment response of the entire cohort
The entire cohort analysis showed that the ORR of all lesions, PT, PVTT, HVTT, and radiation-field lesions evaluated by RECIST were 51.24%, 51.65%, 56.32%, 81.93%, and 59.50%, respectively (Table 2). The ORR of all lesions, PT, PVTT, HVTT, and radiation-field lesions evaluated by mRECIST were 55.37%, 55.79%, 62.63%, 87.95%, and 63.22%, respectively. Whether evaluated by RECIST or mRECIST, HVTT showed significantly higher ORR than PT and PVTT (all p < 0.001). Although the ORR of PVTT appeared higher than that of PT, the difference did not reach statistical significance (all p > 0.05).
Treatment response of HCC with macrovascular tumor thrombus.

RECIST: PT vs PVTT (p = 0.382), PT vs HVTT (p < 0.001), PVTT vs HVTT (p < 0.001);.
mRECIST: PT vs PVTT (p = 0.168), PT vs HVTT (p < 0.001), PVTT vs HVTT (p < 0.001).
DCR, disease control rate; HCC, hepatocellular carcinoma; HVTT, hepatic vein tumor thrombus; IRPT, irradiated primary tumor; mRECIST, modified RECIST; ORR, objective response rate; PVTT, portal vein tumor thrombus; RECIST, Response Evaluation Criteria in Solid Tumors.
Treatment response of HCC with PVTT
In patients with PVTT (Table S2), the ORR of PVTT evaluated by RECIST was slightly higher than PT, but there was no significant difference (56.60% vs 48.43%, p = 0.178). The ORR of PVTT evaluated by mRECIST was significantly higher than PT (62.89% vs 49.69%, p = 0.024).
Treatment response of HCC with HVTT
In patients with HVTT (Table S3), the ORR of HVTT was significantly higher than PT, no matter evaluated by RECIST (90.38% vs 69.23%, p = 0.013) or mRECIST (94.23% vs 76.92%, p = 0.023).
Treatment response of HCC with both PVTT and HVTT
In patients with both PVTT and HVTT (Table S4), the ORR of HVTT evaluated by RECIST was significantly higher than PT (67.74% vs 38.71%, p = 0.041), while the ORR of HVTT evaluated by mRECIST was slightly higher with borderline significance (77.42% vs 51.61%, p = 0.062). However, there was no statistical difference between PT and PVTT (both p > 0.05).
Association of treatment response and PFS
To explore the relationship between treatment response and PFS, we selected ORR and PFS of radiation-field lesions for analysis. Survival analysis showed that the PFS rate of patients with a response was significantly higher than non-responders, whether evaluated by RECIST or mRECIST (both p < 0.001). The median PFS time of patients with response evaluated by the mRECIST criterion was 8.26 (95% CI = 2.3–19.18) months, while that of patients with non-response was 4.49 (95% CI = 1.51–15.11) months (Figure 1(a)). The median PFS of patients with response evaluated by the RECIST criterion was 8.49 (95% CI = 2.3–19.18) months, while that of patients with non-response was 4.72 (95% CI = 1.54–15.11) months (Figure 1(b)).

PFS of different treatment responses evaluated by mRECIST (a) and RECIST (b).
Subgroup analysis for treatment response
To learn about the difference in ORR of different subgroups, we conducted subgroup analysis. Whether RECIST and mRECIST criteria were used for assessment, HVTT shows a higher ORR in all subgroups, with a significant difference, except for the subgroups of female, HBV negative, Child-Pugh B, cirrhosis, single tumor, without previous anti-tumor treatment, which showed a tendency toward statistical significance (Tables S5 and S6). Tables S7 and S8 show the ORR of the training cohort and external validation cohort evaluated by RECIST and mRECIST criteria, respectively. The ORR of each lesion in the training cohort was consistent with that in the external validation cohort, with no significant difference between the two cohorts. In addition, we conducted a comparison of the size and dose among IRPT, PVTT, and HVTT. The results show that there were no significant differences in size and dose among the lesions (Table S9), indicating the uniformity and comparability of dose among the lesions.
The risk factors of treatment response in the training cohort
To facilitate clinical applicability, we chose the ORR of the radiation-field lesions evaluated by mRECIST as the predictive outcome. Univariate analysis showed that cirrhosis, IRPT size, tumor thrombus types, D2%, D50%, D95%, D98%, and BED10 were associated factors of ORR. Due to the collinearity among D2%, D50%, D95%, D98%, and BED10, hence, only BED10 was included in the multivariate analysis. Multivariate analysis showed that IRPT size was an independent risk factor of ORR (OR = 0.744, 95% CI = 0.576–0.939, p = 0.017), tumor thrombus type (OR = 4.14, 95% CI = 1.42–14.4, p = 0.015), and BED10 (OR = 1.063, 95% CI = 1.031–1.098, p < 0.001) were independent protective factors of ORR (Table 3).
Risk factors of treatment response in the training cohort.
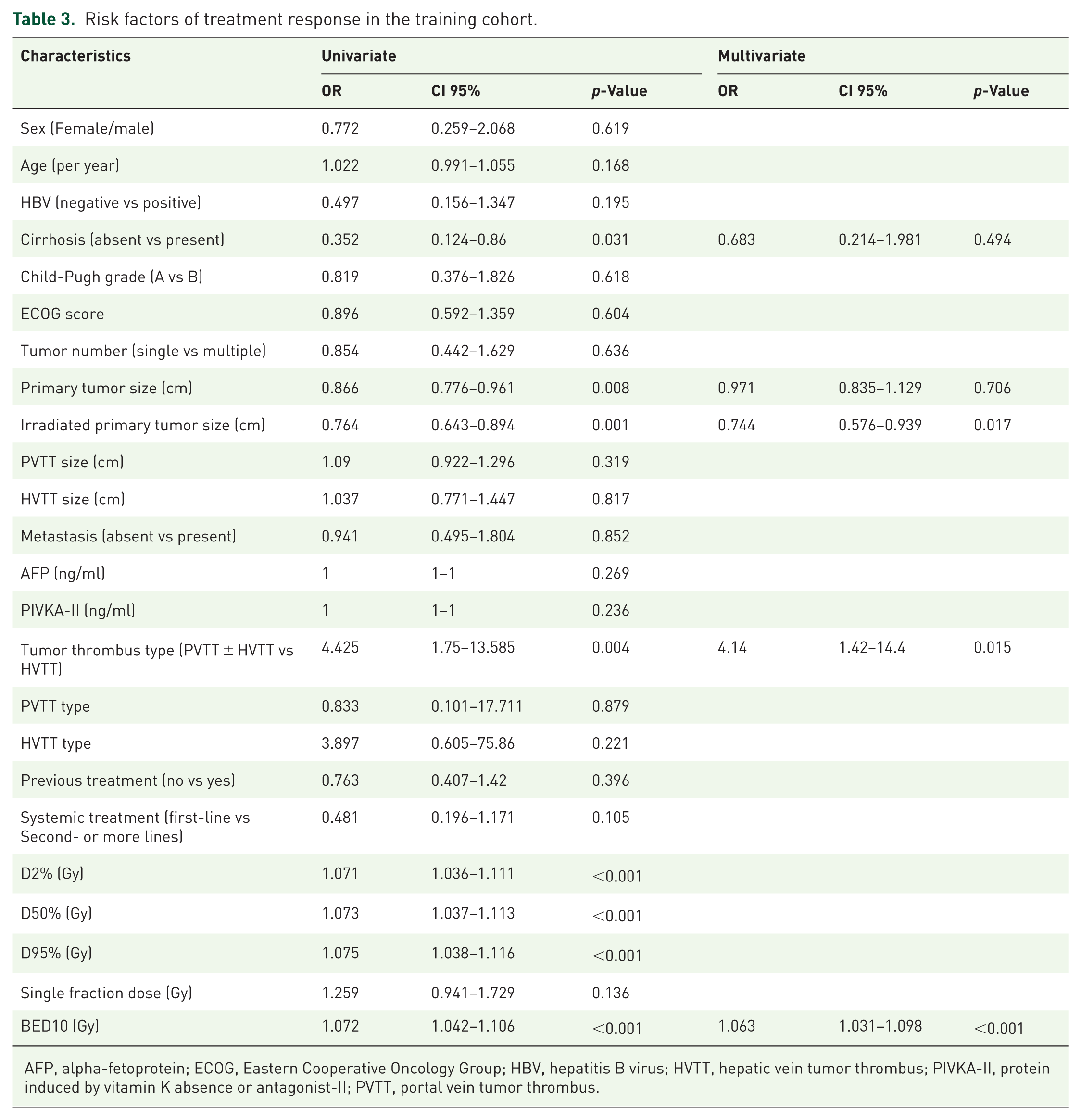
AFP, alpha-fetoprotein; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis B virus; HVTT, hepatic vein tumor thrombus; PIVKA-II, protein induced by vitamin K absence or antagonist-II; PVTT, portal vein tumor thrombus.
Prediction model of treatment response and validation
We used the variables obtained from multivariate analysis as predictors to construct a nomogram (Figure 2). The area under the receiver operating characteristic curve (AUC) of the nomogram was 0.788 (95% CI = 0.72–0.856) in the training cohort (Figure 3(a)), and 0.736 (95% CI = 0.611–0.861) in the external validation cohort (Figure 3(b)).

A nomogram for predicting treatment response was developed in the training cohort.
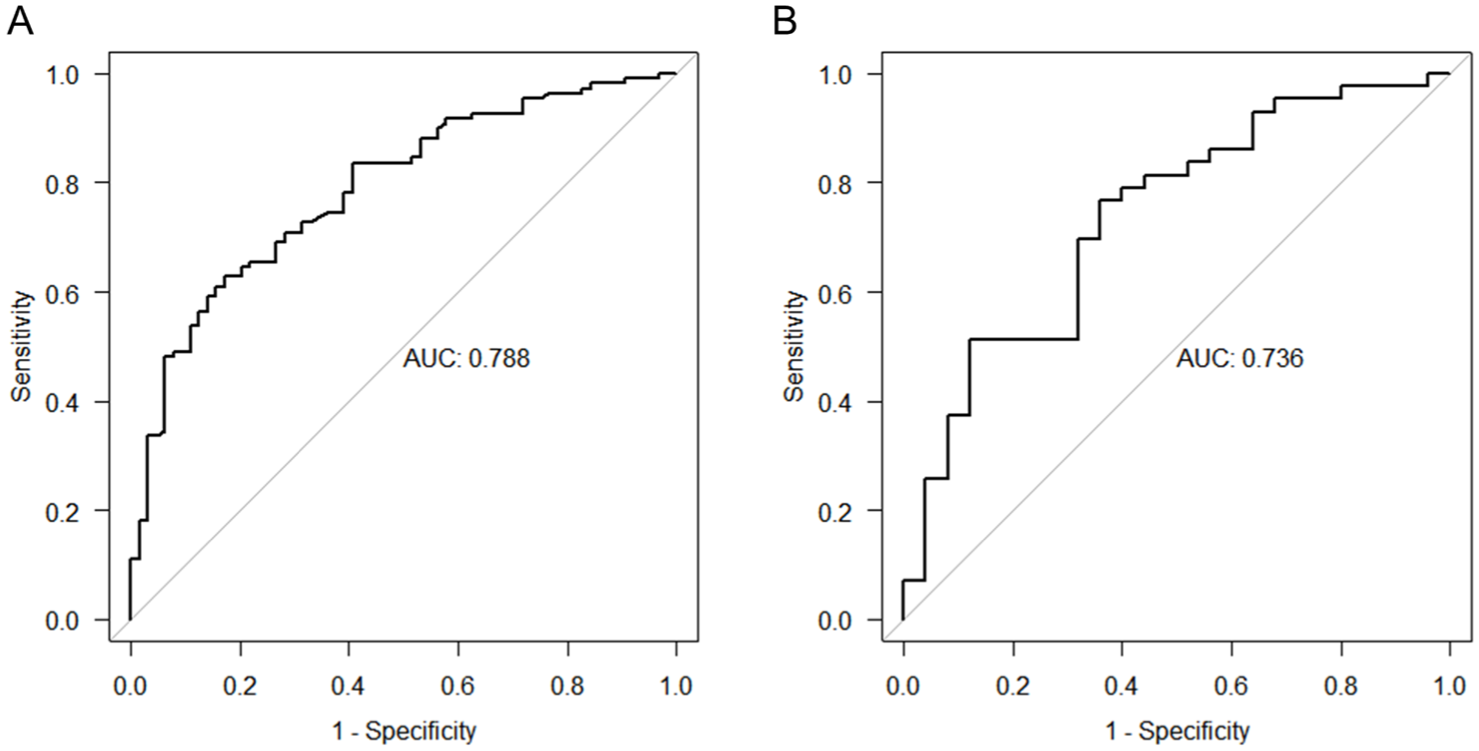
Receiver operating characteristic curves of models for treatment response in training (a) and validation (b) cohorts.
Prediction model performance
The calibration curves showed good consistency between the nomogram in both the training (Figure 4(a)) and validation cohorts (Figure 4(b)). DCA demonstrated that the model provided considerable clinical net benefit (Figure 4(c) and (d)).

Predictive performance of models. Calibration curves of the nomogram on the training cohort (a) and validation cohort (b). Decision curve analysis for the nomogram in the training cohort (c) and validation cohort (d).
Discussion
The findings of this study reveal that under the same dosage of radiation, the sensitivity of macrovascular tumor thrombus to radiotherapy surpasses that of the HCC primary tumor, which suggests that HCC with macrovascular tumor thrombus may derive greater benefits from radiotherapy. Furthermore, the prediction model we constructed offers a quantitative tool for personalized treatment, facilitating the selection of patients who are more suitable candidates for radiotherapy in clinical practice.
From the results of treatment response, radiotherapy is an effective local treatment for HVTT, with ORR and DCR reaching 81.93% and 100%, respectively. The radiotherapy sensitivity of PVTT is lower than that of HVTT, but it still has advantages compared to PT. Further research is needed to explore ways to improve the efficacy of radiotherapy in PVTT patients, such as optimizing dosage and fractionated schemes. However, the effectiveness of radiotherapy for PT is still unsatisfactory, suggesting that it may be necessary to increase the radiation dose or combine other treatment methods (such as TACE/HAIC or radiosensitizers) to improve the efficacy.
Prior studies have documented heterogeneous radiotherapy response rates between the HCC primary tumor and different types of tumor thrombus. In HCC with tumor thrombus receiving radiotherapy, the ORR of primary tumor lesions was 16%–50%,11,16 PVTT was 45.7%–65.2%,12,22 and HVTT was 61.7%–96%.10,13 Yet, these studies exhibit critical methodological limitations. First, in some studies, the tumor volume that received radiotherapy was only the tumor thrombus, thereby precluding comparative analysis of radiosensitivity between primary lesions and tumor thrombus. Second, these studies employed discordant evaluation criteria (including RECIST, mRECIST, and WHO classifications), which fundamentally impeded meta-analysis of treatment response across studies. Third, the literature primarily reported radiotherapy response rates for individual entities (HVTT, PVTT, or primary tumor) or pairwise comparisons, with no systematic comparative analysis encompassing all three lesions. Although direct comparison is not feasible, data from previous literature indicate that there are indeed differences in the response rates to radiotherapy among HVTT, PVTT, and PT, which is consistent with our findings.
We speculate that the differential radiosensitivity between HVTT and PVTT may be attributed to multifactorial mechanisms involving hemodynamic characteristics, tumor biological behavior, and distinct microenvironmental influences. From a hemodynamic perspective, the intravascular pressure of the hepatic vein system is lower than that of the portal vein system. 23 HVTT typically exhibits anterograde growth along physiological blood flow direction, whereas PVTT demonstrates retrograde propagation against portal venous flow. This pressure gradient disparity and flow-directional growth patterns may facilitate enhanced oxygen and nutrient delivery to HVTT, potentially contributing to their superior radiation response rates. Another potential influencing factor is the difference in the tumor microenvironment. The hepatic venous blood flow is processed through the liver, containing hepatic metabolic products, while the portal venous blood flow primarily originates from the gastrointestinal tract, containing metabolites from the gastrointestinal system and gut microbiota, 24 which may affect the response of the tumor to radiotherapy. Furthermore, compositional heterogeneity between HVTT and PVTT could influence radiosensitivity. Current evidence regarding tumor thrombus component disparities remains scarcely documented, particularly in terms of extracellular matrix composition, hypoxia-inducible factor expression, and vascular endothelial cell proportion. These potential biological distinctions merit further investigation through proteomic and transcriptomic profiling to elucidate their roles in radiosensitivity.
The prediction model includes IRPT size, BED, and tumor thrombus type. Tumor size reflects tumor burden and tumor hypoxia, affects the size of the radiotherapy target area and the dose constraints of OARs, as well as the prescription dose of radiotherapy, and has a crucial impact on treatment response. BED10, as a unified standard after dose adjustment, reflects the equivalent therapeutic intensity under different fractionation schemes and is the core variable for predicting radiotherapy response. 25 The sensitivity differences among tumor thrombus types have been consistently validated in multiple subgroup analyses of this study, demonstrating clear predictive value. Currently, there are few studies on predictive models for radiotherapy response in HCC. Lausch et al. 25 used variables such as BED, tumor volume, and liver function to predict local control rates, with an AUC of approximately 0.70. However, this study did not involve vascular thrombus and did not undergo external validation. He et al. 26 analyzed the prognosis of patients with advanced HCC who received intensity-modulated IMRT plus anti-PD1 treatment, including 102 patients with a total response rate of 56.99%, which was similar to this study. They constructed a nomogram to predict OS but did not predict treatment response, with a small sample size and a single-center design. Our study combines tumor-related parameters and radiotherapy-related parameters and has undergone external validation.
This study has several limitations that warrant consideration. First, as a retrospective cohort study, there was unavoidable selection bias; although all patients received TKI + ICI as systemic treatment, the systemic treatment regimens varied, and further stratified analysis was limited due to the sample size. Second, although multicenter data are used, the sample size of this study is still insufficient, and the prediction model may be overfit; the AUC decreased to 0.736 in the external validation cohort. This decrease suggests that the predictive performance of the model may be weakened in external populations. Therefore, caution should be exercised in the clinical application of this predictive model. Third, the patients are all Chinese people, predominantly with HBV background; the applicability of these findings to patients with different etiologies of liver disease requires validation through larger-scale, multicenter studies.
Conclusion
Under identical radiation doses, macrovascular tumor thrombus, especially HVTT, is associated with a higher response rate to radiotherapy compared to HCC primary tumors. The nomogram demonstrated good predictive performance, but prospective validation in larger cohorts is still warranted.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251392117 – Supplemental material for Treatment response and prediction model of radiotherapy for hepatocellular carcinoma with macrovascular tumor thrombus: a multicenter retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359251392117 for Treatment response and prediction model of radiotherapy for hepatocellular carcinoma with macrovascular tumor thrombus: a multicenter retrospective study by Qizhen Huang, Jing Yang, Xiaohong Zhong, Yaqi Zhong, Xingte Chen, Zongren Ding, Haoming Xia, Shaoxing Chen, Yufei Zhou, Lingdong Shao, Kongying Lin, Shaoguang Liao, Yongyi Zeng and Jinsheng Hong in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
