Abstract
Background:
Recent studies have shown that inflammatory indicators are closely related to the prognosis of patients with hepatocellular carcinoma, and they can serve as powerful indices for predicting recurrence and survival time after treatment. However, the predictive ability of inflammatory indicators has not been systematically studied in patients receiving transarterial chemoembolization (TACE). Therefore, the objective of this research was to determine the predictive value of preoperative inflammatory indicators for unresectable hepatocellular carcinoma treated with TACE.
Methods:
Our retrospective research involved 381 treatment-naïve patients in 3 institutions, including the First Affiliated Hospital of Soochow University, Nantong First People’s Hospital, and Nantong Tumor Hospital, from January 2007 to December 2020 that received TACE as initial treatment. Relevant data of patients were collected from the electronic medical record database, and the recurrence and survival time of patients after treatment were followed up. Least absolute shrinkage and selection operator (LASSO) algorithm was used to compress and screen the variables. We utilized Cox regression to determine the independent factors associated with patient outcomes and constructed a nomogram based on multivariate results. Finally, the nomogram was verified from discriminability, calibration ability, and practical applicability.
Results:
Multivariate analysis revealed that the levels of aspartate aminotransferase-to-platelet ratio index (APRI) and lymphocyte count were independent influential indicators for overall survival (OS), whereas the levels of platelet-to-lymphocyte ratio (PLR) was an independent influential index for progression. Nomograms exhibited an excellent concordance index (C-index), in the nomogram of OS, the C-index was 0.753 and 0.755 in training and validation cohort, respectively; and in the nomogram of progression, the C-index was 0.781 and 0.700, respectively. The time-dependent C-index, time-dependent receiver operating characteristic (ROC), and time-dependent area under the curve (AUC) of the nomogram all exhibited ideal discrimination ability. Calibration curves significantly coincided with the standard lines, which indicated that the nomogram had high stability and low degree of over-fitting. Decision curve analysis revealed a wider range of threshold probabilities and could augment net benefits. The Kaplan-Meier curves for risk stratification indicated that the prognosis of patients varied significantly between risk categories (P < .0001).
Conclusions:
The developed prognostic nomograms based on preoperative inflammatory indicators revealed high predictive accuracy for survival and recurrence. It can be a valuable clinical instrument for guiding individualized treatment and predicting prognosis.
Introduction
Hepatocellular carcinoma (HCC) has been considered as a worldwide health concern; it has risen to the fourth leading cause of cancer-related death in the world.1,2 At present, liver transplantation and surgical resection are still radical therapeutic regimen for the treatment of HCC; however, due to the concealment of HCC, some patients have missed the optimal timing of liver transplantation and surgical resection, and then turn to palliative treatments such as ablation, transarterial chemoembolization (TACE) and systemic therapy.3,4 Transarterial chemoembolization has been shown to improve prognosis and is defined as the first-line treatment in palliative therapy of HCC.3,5-8 Nevertheless, due to the heterogeneity of HCC, the effectiveness of patients treated with TACE varies significantly, and not all patients could benefit from TACE treatment; 9 therefore, identifying the individuals who can truely benefit from TACE remains a topic of signifcant discussion. 10 Currently, there are some screening criteria for TACE ideal candidates including up-to-7 criteria, 11 6- and 12-criteria, 12 Cancer of the Liver Italian Program (CLIP) score, 13 hepatoma arterial-embolization prognostic (HAP) scoring system, 14 and Barcelona clinic liver cancer (BCLC) staging system. 15 These models are mainly composed of liver function and tumor burden for screening patients who are suitable for TACE treatment; 16 nevertheless, these models do not allow for individualized prediction, and some are for the treatment of HCC rather than for TACE alone.17,18 Furthermore, thecomplexity of these standards can lead to inconveniences in the practical applications of clinical diagnosis and treatment. 19 Therefore, it is important to find biomarkers that are effective, easily accessible and highly predictive to identify patient subgroups most likely to benefit from TACE treatment.
Most instances of HCC originated from gradual progression of chronic liver disease, include nonalcoholic fatty liver disease (NAFLD), alcoholism, and hepatitis virus infection.20-22 Long-term persistent inflammation of the liver could lead to impaired liver immune system, allowing tumor cells to avoid immune monitoring readily. 23 According to current research, the inflammatory response of the organism is essential for tumor development and is substantially connected with prognosis;24-26 this negative effect can be linked to the mitochondrial dysfunction of antitumor immune cells. 27 A previous meta-analysis study found that patients with high levels of aspartate aminotransferase-to-platelet ratio index (APRI had a worse overall survival (OS) time after TACE treatment and a higher rate of long-term recurrence, 28 and this was also consistent with the results of Tang et al. 29 In addition to APRI, studies about other inflammatory indicators such as platelet-to-lymphocyte ratio (PLR) and neutrophil-to-lymphocyte ratio (NLR) in peripheral blood have also shown that patients with high levels of NLR and PLR have a poor prognosis after TACE treatment;30-32 hence, these previous studies have suggested that serological inflammatory markers can predict the prognosis of TACE treatment. However, these inflammatory indices can be easily calculated by blood routine examination and biochemical analysis. However, current research on the association between inflammatory indicators and patient prognosis in TACE treatment is insufficient; these studies only revealed the possible relationship between inflammatory indices and prognosis, but did not explore the potential predictive ability, and which index has the most predictive ability is still unclear.28,29,31,33-37
Nomogram is a graphical calculator that allows personalized predictions in clinical practice, and it can help doctors select appropriate regimen based on benefit-risk assessments.38,39 For example, a recent study constructed a novel nomogram to determine patients who may benefit from TACE treatment, and the authors screened factors such as albumin-bilirubin (ALBI) grade and tumor response to construct the model, the results showed that it exhibited ideal predictive performance. 40 However, several factors could potentially impact the efficacy of TACE and the prognosis of patients; therefore, the purpose of this study was to further explore the predictive ability of preoperative inflammatory indicators for TACE patients, by integrating inflammatory indicators to develop clinical nomogram and validate it in external cohort.
Methods
Study design
This study retrospectively collected and analyzed the clinical data of patients with unresectable HCC who received TACE as the initial treatment in 3 institutions from January 2007 to December 2020, including the First Affiliated Hospital of Soochow University, Nantong First People’s Hospital, and Nantong Tumor Hospital. When the patient was admitted to the hospital, through multidisciplinary consultation, the size, number, location of the tumor, and the general condition of patient were strictly evaluated to determine whether the patient was suitable for surgical resection, or met the criteria for liver transplantation, or adopted palliative treatment. This research protocol was approved by the institutional review committees of all 3 participating institutions and conformed to Helsinki Declaration. In view of the observational characteristic of the research, we have waived the requirement of written informed consent. The diagnosis of HCC was based on the criteria of the American Association for the Study of Liver Diseases (AASLD) or the European Association for the Study of the Liver (EASL).3,6 Each clinical decision was collaboratively evaluated by multidisciplinary experts.
Inclusion criteria include (1) Eastern Cooperative Oncology Group (ECOG) = 0 or 1; (2) Child-Turcotte-Pugh grade A or B; (3) treatment-naïve individuals with unresectable HCC, patients have not received treatment for HCC previously, including ablation, systemic therapy, surgical treatment, and so on; and (4) patients ⩾18 years old.
Patients were eliminated for the reasons listed below: (1) severe and uncorrectable coagulation disorders; (2) comorbidity with other malignancies; (3) vascular invasion or extra-hepatic spread; and (4) incomplete clinical data.
Treatment protocol
Through enhanced computed tomography (CT) or magnetic resonance imaging (MRI) within 1 week prior to TACE to evaluate the condition of tumor. Four interventional radiologists with at least 10 years of expertise conducted the operations. All patients treated with conventional TACE therapy. Transarterial chemoembolization operation was conducted through femoral artery approach under regional anesthesia, lidocaine in the amount of 5 mL was injected into the subcutaneous tissue of the groin to establish regional anesthesia. Diagnostic angiography of the superior mesenteric artery and celiac trunk was used by 5-Fr RH catheters (COOK, Bloomington, IN, USA), then we used 2.7-Fr microcatheters (Progreat, Terumo, Japan) for superselective catheterization of tumor-feeding artery, to minimize harm to normal liver tissue and get the maximum potential therapeutic effect. 41 Lipiodol (Laboratoire Guerbet, France) mixed with oxaliplatin (85 mg/m2) and doxorubicin (20-40 mg/m2) was injected intra-arterially under fluoroscopy guidance; polyvinyl alcohol particles (Alicon, Hangzhou, China) were then injected to achieve complete embolization of the tumor-feeding arteries. The end point of embolization was defined that the blood flow in the tumor-feeding artery was stagnant within at least 5 cardiac cycles under angiography. Angiography was repeated after 5 min to ensure that the relevant arteries were fully embolized and there was no residual enhancement of tumor.
The outcomes of laboratory examination and follow-up
From electronic medical databases of 3 medical institutions, we have collected baseline data of patients, including biochemical analysis, blood routine examination, epidemiological data, demographic data, and postoperative initial tumor response. The following formula was used to compute inflammatory indicators: prognostic nutritional index (PNI) = 5 × lymphocyte (109/L) + serum albumin (g/L), systemic immune inflammation index (SII) = platelet × neutrophil/lymphocyte, neutrophil-to-lymphocyte ratio (NLR) = neutrophil/lymphocyte, platelet-to-lymphocyte ratio (PLR) = platelet/lymphocyte, aspartate aminotransferase-to-platelet ratio index (APRI) = 100 × (aspartate aminotransferase [U/L]/upper limit of normal value [U/L]/platelet), aspartate aminotransferase-to-lymphocyte ratio index (ALRI) = aspartate aminotransferase (U/L)/lymphocyte, and aspartate aminotransferase-to-neutrophil ratio index (ANRI) = aspartate aminotransferase (U/L)/neutrophil. Variables were divided into different categories based on the appropriate cut-off points automatically calculated by X-Tile software (version 3.6.1). 42 The patient’s follow-up status was determined by querying the patient’s electronic case or calling the patient’s family members.
Imaging follow-up was performed 4 to 60 weeks after TACE treatment. Enhanced CT/MRI was used to evaluate whether there were new lesions or residual tumor tissue, and the need for further embolization was determined according to the principle of “on-demand” treatment.43,44 Two radiologists with more than 7 years about abdominal imaging diagnostic expertise evaluated the radiological response of the TACE-treated target lesion. In cases of disagreement, the final judgment was made by the third radiologist with 15 years of expertise. All of them were unaware of patient-related treatment protocol. The classification of the response assessment was based on the modified response evaluation criteria in solid tumors (mRECIST). 45
Overall survival was the primary endpoint, and it was defined as the time period from the initial diagnosis of HCC to the death of patients due to various causes or to the end of the last follow-up. Progression-free survival (PFS) was the secondary endpoint, and it was calculated from diagnosis of HCC until radiologic tumor progression or death; 46 those patients who were alive and had not progressed during the last follow-up period were considered censored. The follow-up data of patients were obtained by telephone interview with the patient’s relatives or from the electronic medical record systems.
Statistical analysis
Quantitative variables were represented as mean value ± standard deviation or median (interquartile range); qualitative variables were represented as frequencies and percentages. The statistical method of quantitative variables was t-test or the Wilcoxon test, and the statistical method of qualitative variables was the chi-square test or the Fisher test. Least absolute shrinkage and selection operator (LASSO) algorithm was applied for preliminary screening of variables. Then, variables screened by LASSO were integrated into univariate and multivariate Cox regression to eventually determine independent influencing factors; finally, the nomogram was constructed based on the results of multivariate Cox regression. The nomogram was assessed from distinguishing ability, calibration ability and clinical applicability, Harrell concordance index (C-index) of the model was calculated, and exhibited time-dependent C-index curves, plotted time-dependent receiver operating characteristic (ROC) and time-dependent area under the curve (AUC) curves at different time points. Calibration curves were applied for assessing calibration capability of constructed nomogram, which were derived by computing the anticipated probability at different time points and comparing them with the observed outcomes. Decision curve analysis (DCA) was applied for evaluating clinical applicability and net benefits at various threshold probabilities. The total risk score of each patient was calculated, and the Kaplan-Meier curve was plotted to analyze the difference between groups. The statistical methods involved above were defined as statistically significant according to P < .05. All statistical analysis and plotting were performed using R software (version 4.1.3); the R packages used in this study were as follows: “survival,” “survivalROC,” “survminer,” “glmnet,” “mass,” “rms,” “pec,” “timeROC,” and “ggplot2.”
Results
Baseline characteristics
Our research enrolled 381 individuals, the training cohort consisted of 232 patients from the First Affiliated Hospital of Soochow University was used to develop nomogram, and a total of 149 patients from Nantong First People’s Hospital and Nantong Tumor Hospital composed the external validation cohort, which was applied for validating the predicting accuracy of nomogram. Figure 1 depicted the process of the method about patient selection and allocation procedure in the research, as well as the nomogram construction strategy. The median follow-up time was 10.8 months (range = 0.7-91.2 months) for training cohort, whereas the validation cohort was 19.0 months (range = 1.3-86.3 months). Table 1 summarized the basic characteristics of the patients recruited, and Figure 2 were the Kaplan-Meier curves for Child-Pugh classification in each cohort.

Flow chart of this study.
The clinic laboratory data and demographic features of training cohort and validation cohort.

Abbreviations: AFP, alpha-fetoprotein; ALP, alkaline phosphatase; ALRI, aspartate aminotransferase-to-lymphocyte ratio index; ALT, alanine transaminase; ANRI, aspartate aminotransferase-to-neutrophil ratio index; APRI, aspartate aminotransferase-to-platelet ratio index; AST, aspartate transaminase; BCLC, Barcelona Clinic Liver Cancer; Hb, hemoglobin; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; mRECIST, modified response evaluation criteria in solid tumors; NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; PNI, prognostic nutritional index; PT, prothrombin time; SII, systemic immune inflammation index; TBIL, total bilirubin.

Kaplan-Meier curves for Child-Pugh classification in each cohort. (A) and (B) OS in training cohort and validation cohort. (C) and (D) PFS in training cohort and validation cohort.
Preliminary screening of variables
The LASSO regression analysis coefficient diagrams for candidate variables were plotted based on the training cohort, and variables corresponding to lambda.min were selected as screening results. In the OS analysis, tumor size, spleen enlargement, BCLC, peritumor capsule, aspartate transaminase (AST), total bilirubin (TBIL), prealbumin, prothrombin time (PT), neutrophil, lymphocyte, APRI, and mRECIST were screened (Figure 3A). In the PFS analysis, age, tumor size, spleen enlargement, BCLC, peritumor capsule, alanine transaminase (ALT), TBIL, prealbumin, alkaline phosphatase (ALP), alpha-fetoprotein (AFP), hemoglobin (Hb), PT, neutrophil, lymphocyte, platelet, PLR, and mRECIST were screened (Figure 3B).

Dimensionality reduction and elements selection of OS (A) and PFS (B) using the LASSO regression models.
Construction and external validation of nomograms
We incorporated the OS-related variables screened by the LASSO algorithm into the univariate Cox regression, and the eligible parameters in the univariate Cox regression were then incorporated into multivariate Cox regression according to the standard of P value less than 0.2. Multivariate analysis indicated that tumor size, spleen enlargement, BCLC, peritumor capsule, TBIL, PT, lymphocyte, APRI, and mRECIST were independent influencing factors for OS (Table 2), and Figure 4A exhibited the constructed nomogram based on the results of multivariate results.
Univariate and multivariate Cox regression analysis for OS.

Abbreviations: APRI, aspartate aminotransferase-to-platelet ratio index; AST, aspartate transaminase; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; mRECIST, modified response evaluation criteria in solid tumors; OS, overall survival; PT, prothrombin time; TBIL, total bilirubin.

The nomogram of OS. (A) Nomogram for predicting the 6-, 12-, 18-, and 24-month probability of survival. Time-dependent C-index curves in training cohort (B) and validation cohort (C).
The nomogram exhibited an ideal discriminating capacity, the C-index was 0.753 [95% confidence interval (CI) = 0.720-0.786] in training cohort, and 0.755 [95% CI = 0.708-0.802] in external validation cohort. Time-dependent C-index curves exhibited excellent consistency at all time points (Figure 4B and C), and the bootstrapping method was used to perform 1000 internal repeated samplings for further internal validation, and the results indicated that the constructed predictive model had significantly stable characteristics (Figure 4B). The calibration curves in Figure 5 showed excellent consistency between the predicted probabilities and the actual occurrence frequencies at different time points; calibration curves greatly coincided with the standard line, and this indicated that the nomogram had high stability, excellent predictive performance, and low degree of over-fitting.
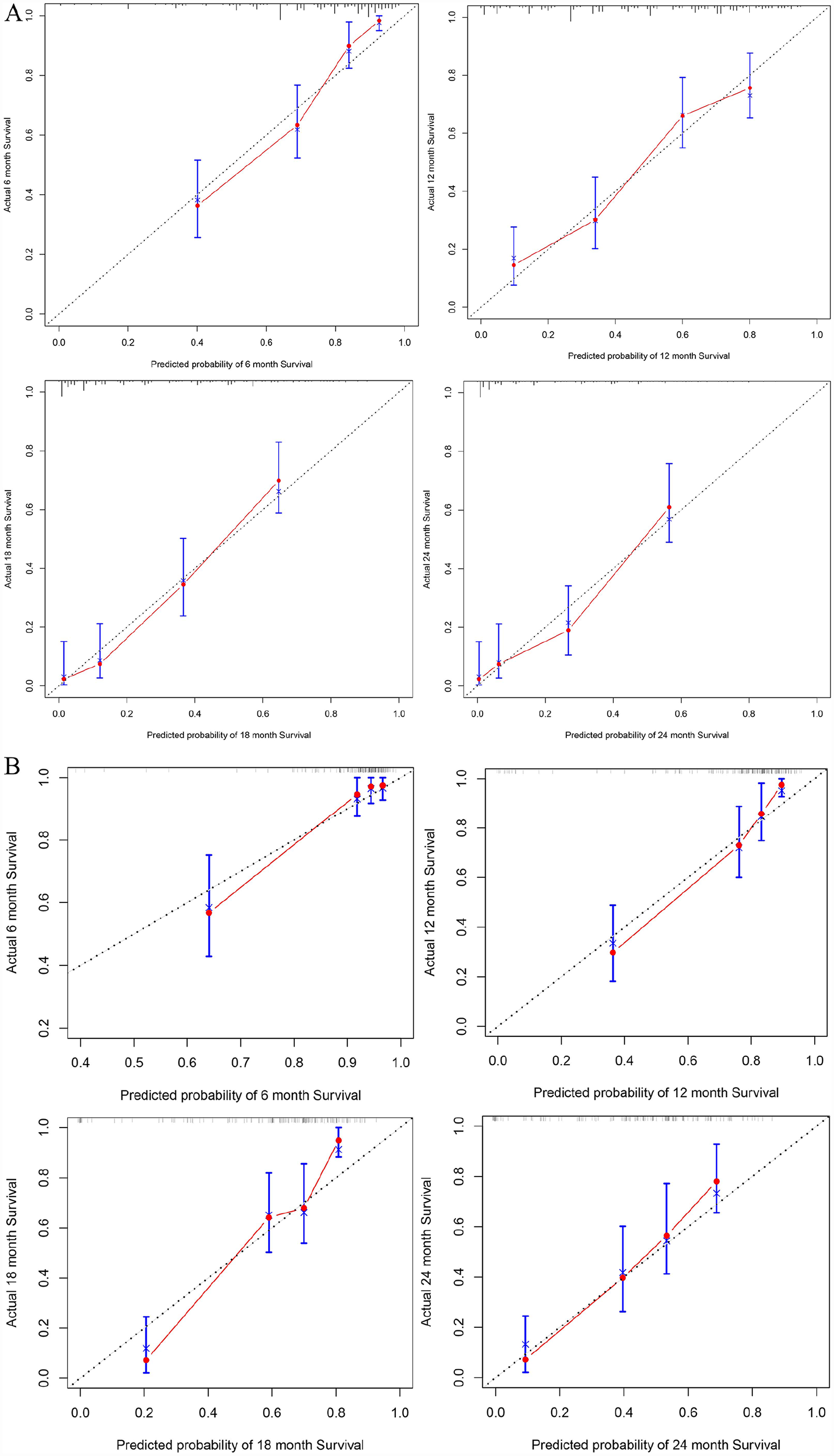
Calibration curves of nomogram for predicting the 6 month, 12 month, 18 month, and 24 month probability of OS in training cohort (A) and validation cohort (B).
Similarly, in the PFS analysis, the variables screened by the LASSO algorithm were incorporated into the univariate and multivariate Cox regression, and the results showed that tumor size, spleen enlargement, peritumor capsule, ALT, TBIL, ALP, PLR, and mRECIST were independent influencing factors for PFS (Table 3), and Figure 6A exhibited the constructed nomogram based on the results of multivariate results.
Univariate and multivariate Cox regression analysis for PFS.

Abbreviations: AFP, alpha-fetoprotein; ALP, alkaline phosphatase; ALT, alanine transaminase; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; mRECIST, modified response evaluation criteria in solid tumors; PFS, progression-free survival; PLR, platelet-to-lymphocyte ratio; TBIL, total bilirubin.

The nomogram of PFS. (A). Nomogram for predicting the 6-, 12-, 18-, and 24-month probability of PFS. Time-dependent C-index curves in training cohort (B) and validation cohort (C).
The C-index of nomogram was 0.781 [95% CI = 0.750-0.812] in training cohort and 0.700 [95% CI = 0.655-0.745] in external validation cohort. Similarly, the curve of time-dependent C-index also exhibited excellent consistency at all time points (Figure 6B and C), bootstrapping method was used to perform 1000 internal repeated samplings for further internal validation, and significant stability was presented (Figure 6B). The calibration curves in Figure 7 showed that the prediction probabilities at different time points after treatment exhibited excellent consistency, high stability, and low degree of over-fitting; the obtained nomogram could successfully predict the PFS probability after TACE treatment.

Calibration curves of nomogram for predicting the 6-, 12-, 18-, and 24-month probability of PFS in training cohort (A) and validation cohort (B).
Time-dependent receiver operating characteristic and time-dependent area under the curve
Figure 8 exhibited the ROC curves of OS and PFS of training cohort and validation cohort at different time points. In the prediction model of OS, the nomogram showed excellent discrimination (AUROC6-month = 0.846, AUROC12-month = 0.800, AUROC18-month = 0.885, AUROC24-month = 0.878) and exhibited good generalizability in the validation cohort (AUROC6-month = 0.876, AUROC12-month = 0.870, AUROC18-month = 0.859, AUROC24-month = 0.817). In the nomogram of PFS, excellent AUC was shown in both sets (training cohort: AUROC6-month = 0.823, AUROC12-month = 0.824, AUROC18-month = 0.850, AUROC24-month = 0.873 and validation cohort: AUROC6-month = 0.874, AUROC12-month = 0.765, AUROC18-month = 0.781, AUROC24-month = 0.717).

Time-dependent ROC of OS (A) and PFS (B) in training cohort and validation cohort at different time points.
The time-dependent AUC curves were plotted to further verify the discriminability of the nomogram, and the results exhibited good stability and sufficient discriminant ability that could predict the probabilities of survival and progression in patients receiving TACE (Figure 9).

Time-dependent AUC in training cohort and validation cohort for predicting OS (A) and PFS (B).
Risk stratification and clinical decision analysis
The total risk score of each patient was calculated, and the stratification was conducted through the median risk score of all patients. Survival curves were drawn by the Kaplan-Meier method, and then we performed the log-rank test to determine the difference between groups. The results suggested that the nomogram exhibited excellent discrimination in 2 cohorts (Figure 10).

The analysis of risk stratification. Risk stratification in training cohort (A) and validation cohort (B) for OS. Risk stratification in training cohort (C) and validation cohort (D) for PFS.
Finally, we evaluated the potential clinical applicability of nomograms. The solid gray arc represented the clinical benefit of TACE in all patients, and the horizontal line represented the benefit for all patients without intervention; vertical and horizontal axes exhibited net benefit degree and threshold probability, respectively. Clinical decision curves demonstrated that nomograms could indicate a broader range of threshold probability and increase net benefit (Figures 11 and 12).

Decision curve analysis in training cohort (A) and validation cohort (B) for predicting the 6-, 12-, 18-, and 24-month probability of OS.

Decision curve analysis in training cohort (A) and validation cohort (B) for predicting the 6-, 12-, 18-, and 24-month probability of PFS.
Discussion
In our research, the predictive value of inflammatory indicators in predicting the prognosis of TACE patients was first systematically elucidated. The nomograms were constructed by LASSO regression algorithm and Cox regression method, and each model was evaluated from discrimination, calibration, and clinical applicability through internal and external cohort. Finally, our research demonstrated that clinical predictive models based on inflammatory indicators exhibited ideal predictive efficiency. Elevated levels of APRI and decreased lymphocyte counts were independent prognostic factors for survival, whereas high levels of PLR were independent dangerous factor for disease progression. Both the nomogram of OS that incorporated APRI and lymphocyte count and the nomogram of PFS that contained PLR exhibited excellent predictability and stratification capabilities.
In the nomogram of OS, APRI and lymphocyte count were strongly associated with survival, which suggested that these 2 indices had significant clinical application value and could be used as reliable predictors of OS after TACE. Our research revealed that elevated level of APRI was an independent influential indicator for decreased survival period, a meta-analysis by Zhang et al 28 explored the relationship between serum APRI levels and the prognosis of HCC patients, and the research revealed that elevated serum APRI was a significant risk factor for OS and preoperative APRI levels could be used to predict the survival of patients, Tang et al 29 suggested that preoperative APRI levels exhibited strong predictive potential for estimating long-term survival probability. These results were consistent with our findings; however, the potential mechanism of APRI related to survival is still unclear. According to the findings of the present studies, APRI plays an important role in determining the degree of cirrhosis, advanced liver fibrosis, and hepatitis,47,48 APRI is constituted of AST and platelet, which might represent the severity of hepatocyte inflammation and liver reserve capacity, respectively.49,50 Overall, APRI may be a sign of both the inflammatory hepatic milieu that favors tumor invasion and the presence of persistent hepatocyte injury. 51 In addition, most of the patients included in the research had hepatitis virus infection, liver fibrosis, and cirrhosis; long-term persistent inflammatory state of the liver leads to hepatocyte injury and decreased liver reserve capacity, which ultimately affected the OS of patients, and the corresponding changes in serological indicators may be an increase in APRI levels. At the same time, previous clinical study has also confirmed the rationality of the above inference; a high level of APRI was identified as a significantly relevant factor for predicting the mortality of HCC patients in the setting of cirrhosis. 52 In addition, we also identified that low levels of peripheral blood lymphocyte count was an essential risk factor for the survival time of patients; previous research by Daly et al 53 demonstrated an association between surgical patients’ shorter life spans and lower lymphocyte counts. As an essential component of adaptive immune system, lymphocytes serving as the biological foundation for immunologic editing and monitoring against malignancy; 54 through the production of cytokines, regulation of immune response, and modification of the microenvironment, lymphocytes could suppress the growth of tumors. 55 Conversely, a decrease in lymphocytes may be a sign of a weakened host immunologic response to cancer cells.56,57
In the nomogram of PFS, we have found that PLR was an independent indicator of tumor recurrence after TACE treatment. The meta-analysis by Li et al 32 explored the association between PLR and the poor prognosis after TACE treatment, and the findings demonstrated that preoperative elevated levels of PLR was a poor prognostic indicator for individuals receiving TACE therapy. Specifically, our research indicated that patients with preoperative serum PLR > 138 had a higher preference of tumor progression, which was consistent with the results of Nicolini and his colleagues; 58 according to its findings, patients with a preoperative PLR > 150 had a higher probability of tumor progression after receiving TACE. According to the basic characteristics of patients enrolled in our research, patients generally had a large tumor burden, and the tumor diameter of most patients was more than 5 cm; our hypothesis was supported by some early studies, that was, PLR may represent the tumor burden of patients. Xue and his co-workers 34 found that patients receiving TACE with large tumors and high levels of PLR had a poor prognosis, and the results of the Song trial and the meta-analysis by Li et al 32 both verified this tendency, which exhibited that the rise in PLR was proportional to tumor size. 59 Therefore, we assumed that high levels of PLR may be a comprehensive manifestation of tumor burden; increased tumor burden leads to decreased lymphocytes, which was indicative of a weakened host immune response.56,57 However, many previous research works have also shown that platelets could promote tumor metastasis60-63 and protect tumor cells from natural killer cells during immune escape; 64 under the combined action of 2 aspects, the high levels of PLR may be more prone to tumor recurrence and metastasis.
There are limitations in our research. First, the retrospective features of this study inevitably lead to bias in the patient selection procedure. Second, some patients may be excluded from the cohort due to insufficient follow-up or baseline data. Third, the heterogeneity of HCC was that the efficacy of patients with the same type varied significantly; this study attempted to screen out the participants who are suitable for TACE through preoperative inflammatory indicators. Although the results exhibited that the efficacy of patients after screening was excellent, our research only elaborated on the aspect of inflammatory indicators, which still needs to be further explored by clinical trials in a larger population in the future to identify other potential factors that may affect the efficacy of TACE treatment.
Conclusions
The as-obtained prognostic nomograms based on preoperative inflammatory markers demonstrated ideal accuracy and excellent predictive efficiency in predicting survival and recurrence after TACE. It can be a valuable clinical instrument for guiding individualized treatment and predicting prognosis.
Footnotes
Acknowledgements
None.
Author Contributions
All authors contributed to the study conception and design. (1) Material preparation and data collection were performed by DZ, WX, LX, WD, and AX. (2) Data analysis was performed by DZ, YZ, and ZH. (3) The first draft of the manuscript was written by DZ and WX. (4) WD, AX, ZH, and CN provided critical opinions and revised the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The trial was conducted in accordance with the Declaration of Helsinki, and has been reviewed and approved by The First Affiliated Hospital of Soochow University, Nantong First People’s Hospital, and Nantong Tumor Hospital (approval ID: 2023072 and approval data: February 27, 2023). The study is retrospective cohort research, and only clinical information of patients will be collected without interfering with patients’ treatment plans or posing physiological risks to patients. All research procedures were in compliance with the relevant guideline and regulations. The ethics committee waived the requirement of written informed consent for participation.
