Abstract
Background:
The B-type rafkinase (BRAF) V600E gene mutation plays an important role in the pathogenesis, diagnosis, and prognosis of thyroid carcinoma. This study was conducted to investigate the rate of the BRAF V600E mutation, the relationships between the BRAF V600E gene mutation and some immunohistochemical markers, and recurrence rate in patients with differentiated thyroid cancer.
Method:
The study was conducted by a descriptive and longitudinal follow-up method on 102 thyroid carcinoma patients at 103 Military Hospital, Hanoi, Vietnam. All patients were identified with the BRAF V600E gene mutation by real-time polymerase chain reaction.
Results:
The rate of BRAF V600E gene mutation in patients with thyroid cancer was 60.8%. Patients with BRAF V600E gene mutation had a significantly higher rate of positive cyclooxygenase 2 (COX-2) and Ki67 markers than those without the mutation (COX-2: odds ratio [OR] = 2.93; 95% confidence interval [CI] = 1.27-6.74, P = .011; Ki67: OR = 3.41; 95% CI = 1.31-8.88, P = .01). A statistically significant relationship was identified between the rate of BRAF V600E mutation and the rate of positive Hector Battifora mesothelial 1 (HBME-1) (B = −1.040; P = .037) and COX-2 (B = −1.123; P = .023) markers. The recurrence rate in patients with BRAF V600E gene mutation was significantly higher than that in those without the mutation (P = .007). The mean of the recurrence time of patients with BRAF V600E mutation was significantly lower than that in those without the mutation (P = .011).
Conclusions:
A high prevalence of BRAF V600E gene mutation was found in thyroid carcinoma patients. The rates of positive HBME-1, COX-2, and Ki67 markers were significantly correlated to BRAF V600E gene mutation. Patients with BRAF V600E gene mutation showed a significantly higher relapse rate and earlier relapse time than those without the mutation.
Introduction
Thyroid carcinoma is the most common type of cancer. In 2019, an epidemiological study of thyroid cancer in the United States based on the American National Cancer Database showed that the incidence of thyroid cancer increased from 7.1 per 100 000 in 2000 to 17.4 per 100 000 in 2013—a 2.5-fold increase. 1 Thyroid carcinoma is divided into two types, including differentiated and undifferentiated types; primarily in the differentiated type, including papillary, follicular, and papillary-follicular combined.2-4 Because differentiated thyroid carcinoma develops slowly and is similar to other benign diseases of the thyroid gland for the clinical manifestations, it is easy to confuse the diagnosis. Thyroid carcinoma usually may develop locally or invade the thyroid cortex, surrounding tissues (esophagus, trachea, larynx, skin infiltrates, and so on), or metastasize to cervical lymph nodes or distant metastases depending on the histopathological type and the time of the disease detection. 5
Thyroid cancer can be diagnosed based on ultrasound-guided core biopsy and confirmed histopathologically with conventional hematoxylin plus eosin staining. 6 However, some cases have insufficient laboratory information to distinguish between benign and malignant lesions with conventional hematoxylin plus eosin staining alone. 7 Although most thyroid carcinomas can be identified preoperatively by cytology, approximately 20% to 30% of cases may not be identifiable by conventional tests. 8 Many studies have shown that the pathological status of the thyroid gland can be clearly diagnosed using immunohistochemistry with specific antigen-antibody markers.9-11 In recent years, several studies reported the role of B-type rafkinase (BRAF) V600E mutations in the diagnosis and prognosis of papillary thyroid carcinoma.12,13 Position mutation T1799A (V600E) on BRAF gene sequence increases enzyme kinase activity and activates the mitogen-activated protein kinase (MAPK) signaling pathway; somatic BRAF gene mutation was found in tumor cells with different tumor sizes and with detection frequency ranging from 28.8% to 69%. 12 In vitro studies have shown that thyroid carcinoma cell lines with the BRAF V600E gene mutation have a much faster growth rate than wild-type thyroid cell lines,14,15 and it is hypothesized that the BRAF V600E mutation acts as an oncogene. 16 The BRAF V600E gene mutation was reported as a very valuable molecular marker in the diagnosis and the following of thyroid carcinoma. 17 The BRAF V600E mutation is only presented in thyroid carcinoma cells but not in benign thyroid cells. Identifying mutations in the BRAF V600E gene will help to limit diagnoses missing of thyroid carcinoma and to improve the quality of medical examination, treatment, following, and patient management. 18 However, previous studies mainly focused on analyzing the value of BRAF V600E mutations in diagnosis and prognosis.17,18 Previously, there were some studies investigating the relationships between BRAF V600E mutations and clinical and subclinical characteristics and recurrence status in patients with thyroid carcinoma.19-21 From the aforementioned fact, this study was conducted to investigate the rate of BRAF V600E mutation and determine the association of BRAF V600E gene mutation with some clinical, subclinical, and recurrence characteristics in patients with differentiated thyroid cancer.
Materials and Methods
Study population and design
The study subjects included patients who were operated on and diagnosed by postoperative histopathology with thyroid carcinoma at 103 Military Hospital, Hanoi, Vietnam, from July 2013 to December 2016. Exclusion criteria included patients with non-differentiated carcinoma of the thyroid gland, thyroid cancer secondary to metastases from other sites, and extensively invasive differentiated thyroid cancer without total thyroidectomy. 22 All patients have explained the study procedure and agreed to participate in the study.
The study was conducted by a descriptive and longitudinal follow-up method. A total of 102 patients were recruited in the present study. All patients who met the inclusion and exclusion criteria were selected during the study period.
Clinical and biochemical assessments
The characteristic information (name, age, gender, address, and occupation), medical history and current diseases, examination of clinical signs and symptoms, laboratory tests, and BRAF gene mutation tests were collected on the patient’s record.
The stage of thyroid cancer was identified based on the TNM (Tumor-Node-Metastase) classification of the American Cancer Society (1998). 23 Serum levels of free triiodothyronine (FT3) and free thyroxine (FT4) were quantified by an automatic chemiluminenescence system. Tg concentration was quantified by an immunoradiometric assay technique at the Department of Biochemistry, 103 Military Hospital.
All patients were tested for BRAF gene mutation by the real-time polymerase chain reaction technique through candle-molded tumor specimens. The primers and blockers used in the study were ordered from IDT, USA, with the following sequence:
Forward primers (no. 1): 5′-CTGTTTTCCTTTACTTACTACACCTCAGAT-3′
Reverse primers (no. 2): 5′-CAACTGTTCAAACTGATGGG-3′
Allele-specific reverse primers (no. 3): 5′-CCCACTCCATCGAGATTTCT-3′
BRAF-blocker: 5′-CATCGAGATTTCACTGTAGCTAGA-PO4-3′
All postoperative specimens were stained for immunohistochemistry, with markers Hector Battifora mesothelial 1 (HBME-1), cytokeratin 19 (CK19), rearrangement during transfection (RET), P53, Ki67, and cyclooxygenase 2 (COX-2). Immunohistochemical staining using the avidin-biotin complex method 24 was conducted at the Department of Pathology, Military Hospital 103 (Supplemental Document).
After surgery, the patient was re-examined at 1 month and received adjuvant treatment with iod-131. The patient was then followed up and evaluated for recurrence status. The time of recurrence was counted from the date of surgery to the date of examination and detection of recurrent disease.
Evaluation of recurrence
After adjuvant treatment with 131I, patients were followed up every 3 months. At the time of re-examination, the patients were examined with clinical checking, ultrasound, thyroid function measurement, thyroglobulin (Tg) concentration, and thyroid scintigraphy. The results of the assessment of recurrence are carried out according to the guidelines of the Ministry of Health of Vietnam, 25 specifically as follows:
No recurrence: After adjuvant therapy with 131I, the patient was determined to be disease-free during follow-up visits (complete thyroid tissue, negative whole-body scintigraphy, and Tg concentration < 10 ng/ mL) until the last follow-up visit.
Relapse: After adjuvant therapy with 131I, the patient was determined to be free of the disease in the next follow-up (thyroid tissue was gone, whole-body scintigraphy was negative, and Tg concentration < 10 ng/mL) but then re-emergence at a follow-up visit (based on clinical or scintigraphy findings) in the neck or distant metastases, confirmed by fine needle aspiration (FNA) or other appropriate methods.
Existing disease: After adjuvant therapy with 131I, it was still determined that the disease has not been cured in follow-up visits (thyroid tissue was still present, whole-body scintigraphy shows lymph node invasion or distant metastases, the concentration of Tg > 10 ng/mL). The disease persisted at the last follow-up.
Ethical statement
All participants provided written informed consent and agreed to join our study. The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethical Review Committee of Vietnam Military Medical University (reference no.: 168/2014/IRB-VMMU).
Statistical analysis
All results are presented as mean (standard deviation [SD]) or, if biased, as median (interquartile range) for continuous variables and as percentages for categorical variables. Differences between groups were tested by t-test. The chi-square test or Fisher’s exact test was used to assess whether there was a relationship between two categorical variables. The association between gene mutation status and immunological marker test results was determined using multivariable logistic regression, using the variance inflation factor (VIF) coefficient to assess the presence or absence of multicollinearity between variables. P < .05 was considered to be statistically significant. All data were processed using SPSS software version 26 (64-bit) for Windows (SPSS Inc., Chicago, IL).
Results
The demographic and characteristics of the patients were shown in Table 1. The mean ± SD age of the patients was 45.1 ± 13.4 years. No significant difference in age was observed between men and women (P > .05).
The demographic and characteristics of the patients.

Abbreviations: BRAF, B-type rafkinase; CK19, cytokeratin 19; COX-2, cyclooxygenase 2; FT3, free triiodothyronine; FT4, free thyroxine; HBME-1, Hector Battifora mesothelial 1; RET, rearrangement during transfection; SD, standard deviation; Tg, thyroglobulin.
Fisher’s exact test.
Chi-square test.
Student’s t-test.
The rate of BRAF V600E gene mutation was 60.8%, and there was no difference in the rate of BRAF V600E gene mutation between the sexes (P > .05). Fifty-three patients (52% of the total patients) were in the first stage of the cancer; 45 patients (44.1%) were in the second stage; and there were only 4 patients (3.9%) in the third stage. Most patients (84.3%) had the papillary type in histopathology. Increased Tg and FT3 levels were found in 35.3% and 15.6% of the total number of patients, respectively. All patients had the normal FT4 level. No significant differences for the disease stage, histopathology, the concentration of hormones (Tg, FT3, FT4), and immune markers were shown between the sexes. Eleven patients (21.2%) were recrudesced after a treatment. A borderline significant difference in the rate of recurrence was found between men and women (Table 1).
The demographic characteristics were compared between the patients with and without BRAF V600E gene mutation. The results were shown in Table 2. There were no significant differences in age, sex, disease stage, histopathology, or hormone concentration (Tg, FT3, FT4) between the patients with and without BRAF V600E gene mutation (P > .05) (Table 2).
Comparison of demographic characteristics between the patients with and without BRAF V600E gene mutation.
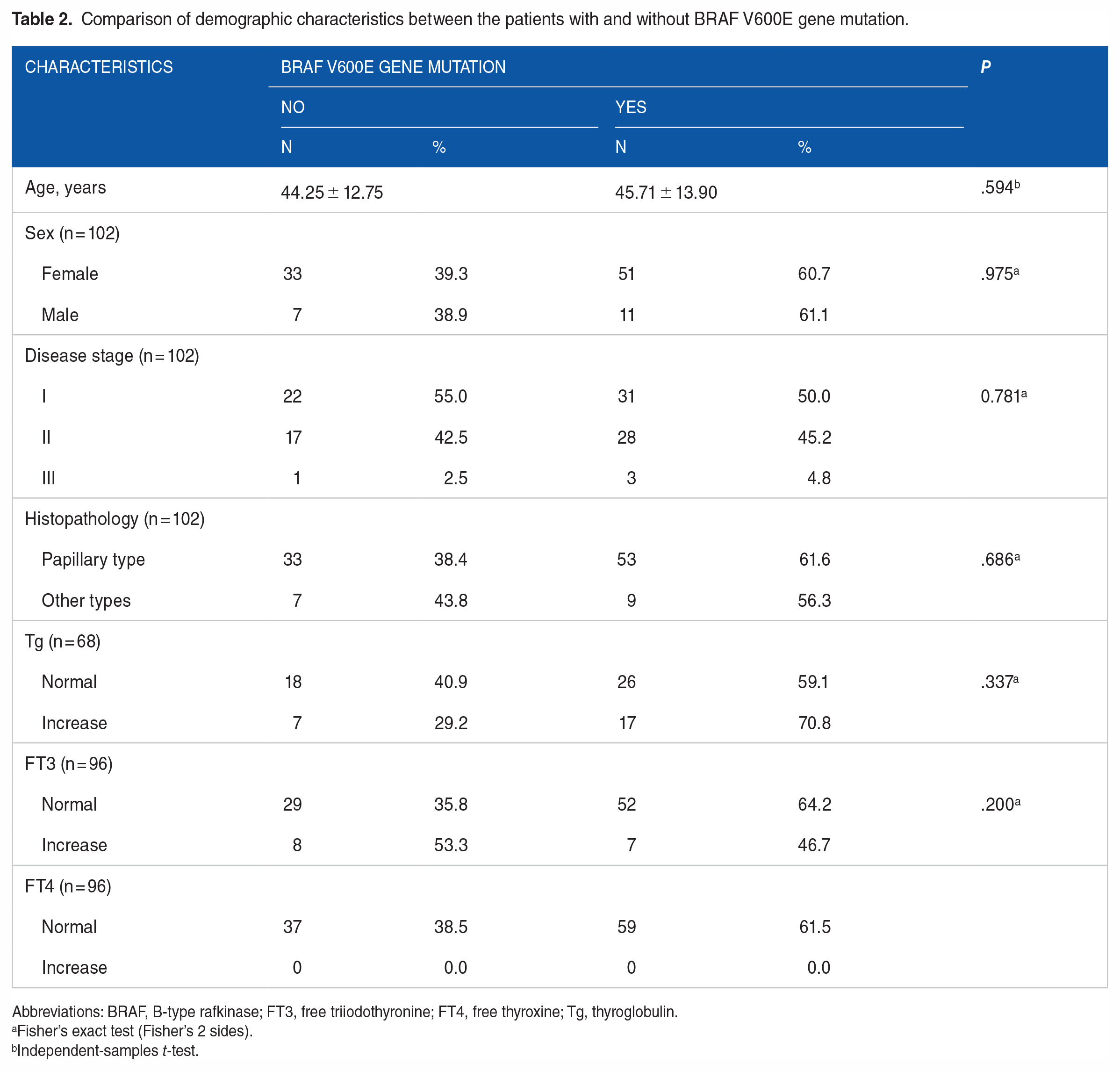
Abbreviations: BRAF, B-type rafkinase; FT3, free triiodothyronine; FT4, free thyroxine; Tg, thyroglobulin.
Fisher’s exact test (Fisher’s 2 sides).
Independent-samples t-test.
The rates of the positive immune markers were compared between the patients with and without BRAF V600E gene mutation and were shown in Table 3. The rates of positive COX-2 and Ki67 markers were significantly higher in the patients with BRAF V600E gene mutation than those in patients without the mutation. Of all, 72.6% of patients with BRAF V600E mutation had a positive COX-2 marker, while the rate of the positive COX-2 marker in the non-mutant group was 47.5% (P = .011; odds ratio [OR] = 2.93; 95% confidence interval [CI] = 1.27-6.74), and 41.9% of patients with BRAF V600E mutation had a positive Ki67 marker, while the positive Ki67 marker rate in the non-mutant group was 17.5% (P = .01; OR = 3.41; 95% CI = 1.31-8.88) (Table 3).
Comparisons of the rate of the positive immune markers between the patients with and without BRAF V600E gene mutation.

Abbreviations: BRAF, B-type rafkinase; CK19, cytokeratin 19; COX-2, cyclooxygenase 2; HBME-1, Hector Battifora mesothelial 1; RET, rearrangement during transfection.
Chi-square Test.
The relationships between gene mutation and immune markers using a binary logistic regression model were shown in Table 4. The presence of BRAF V600E gene mutation was significantly inversely correlated with the positivity of the HBME-1 marker (P = .037; OR = 0.353; 95% CI = 0.133-0.939) and COX-2 marker (P = .023; OR = 0.325; 95% CI = 0.124-0.856) (Table 4).
Relationships between the BRAF V600E gene mutation and immune markers using a binary logistic regression model.

Abbreviations: CK19, cytokeratin 19; COX-2, cyclooxygenase 2; HBME-1, Hector Battifora mesothelial 1; RET, rearrangement during transfection ; VIF, variance inflation factor.
Table 5 showed the comparisons of the recurrence rate and the mean of the recurrence time between the patients with and without the BRAF V600E mutation. The rate of recurrence in the patients with the BRAF V600E gene mutation was significantly higher than that in the patients without the gene mutation (P = .007). The mean of the recurrence time in patients with the BRAF V600E mutation (12.39 ± 7.44 months) was significantly lower than that in the patients without the gene mutation (18.71 ± 7.44 months) (P = .011) (Table 5).
Comparisons of the recurrence rate and the mean of the recurrence time between the patients with and without the BRAF V600E gene mutation.

Abbreviation: BRAF, B-type rafkinase.
Fisher’s exact test. **Independent-Samples T Test.
Discussion
The BRAF gene mutation is considered to be one of the important mutations related to the invasiveness, advanced stage, recurrence rate, and metastatic degree of thyroid carcinoma. 18 In our study, 60.8% of thyroid carcinoma patients had mutations in BRAF gene at the T1799A (V600E) position. Similarly, Niederer-Wüst et al 26 reported that the rate of BRAF gene mutation was 65% in thyroid carcinoma patients with tumor size ⩾1 cm. Pelizzo et al 27 showed 47.8% of BRAF V600E mutations in 226 patients with papillary thyroid carcinoma, stage T1-T2 without lymph node metastasis (N0). In addition, the results from several studies suggest that the BRAF V600E mutation is common in thyroid carcinomas, particularly papillary thyroid carcinomas, with frequency varying from 18% to 87%.28,29 This difference in the mutation rate may be due to differences in the selection of research patients and geographical locations, especially in histopathological types. In our study, BRAF V600E mutation was present in 61.6% of conventional papillary thyroid cancers (PTCs) and in 56.3% of other histological types; these results are similar to the results of other previous studies when classified according to histological subtypes.30-32
Our study showed no association between the BRAF V600E gene mutation and the disease stage, which is consistent with the study by Peuxeddu et al. 33 In contrast, other studies reported that BRAF V600E mutations are associated with stage III and IV diseases. 34 A multicenter study with a large number of patients showed that patients with stages III and IV had a significantly higher rate of BRAF mutation than the non-mutant group (P = .007). 30 Overall, almost all studies showed that BRAF V600E mutations occur more frequently in patients with the disease in stages III and IV than in stages I and II.12,13,35
Higher rates of positive COX-2 and Ki67 markers were found in the patients with the BRAF mutation than in those without the mutation. There was an inverse correlation between the positive level of marker COX-2 (P = .023) and the occurrence of the BRAF V600E gene mutation, identified using a binary logistic regression. COX is a group of enzymes required for synthesizing prostaglandin E2 (PGE2). 36 Under normal physiological states, COX-2 and PGE2 are upregulated and act as inflammatory factors. 37 COX-2 has been observed to be overexpressed in several human cancers.38,39 Many studies have described the carcinogenesis-promoting mechanisms by COX-2, including inhibition of apoptosis, cell proliferation, stimulation of angiogenesis, and growth of the pathogenic microenvironment inflammation which promotes tumorigenesis.40,41 COX-2 overexpression in different types of thyroid cancer has also been demonstrated.42,43 BRAF V600E mutants have been shown to induce oncogenic cell proliferation by constitutive activation of the MAPK pathway, 44 while BRAF mutations have a role in accelerated production of PGE2 through COX-2. 45 This might explain the association between BRAF mutation status and the appearance of a COX-2 marker through immunohistochemical staining in the current study.
To evaluate postoperative recurrence and supportive treatment with I131, we applied the following diagnostic criteria for recurrence: Patients after surgery and adjuvant I131 therapy were determined to be disease-free at the next follow-up (there was no thyroid tissue, a negative whole-body scintigraphic, and Tg concentration < 10 ng/mL), but at subsequent follow-up visits, there was a recurrence in the thyroid (signs in clinical examination, ultrasound, or scintigraphic), cervical lymph nodes, or distant metastases, and a positive FNA result confirmed the diagnosis. The results of our study showed that the mean of the recurrence time in patients with BRAF V600E gene mutation was earlier than the mean of the recurrence time in the patients without the mutation. Patients with mutations in BRAF V600E gene had a higher recurrence rate than patients without the mutation (P = .007), which is consistent with other studies.12,13,35 Several studies have shown that the prevalence of BRAF mutations was very high at 80% to 85% of recurrent thyroid carcinomas.46,47 Patients with stage I and II diseases usually have a low risk of recurrence, but the risk of recurrence increases markedly in patients with the BRAF gene mutation. 30 Compared with BRAF mutation-negative cases, recurrent thyroid carcinoma patients with the BRAF mutations require more aggressive treatments, such as re-surgery and external beam irradiation, and show a higher rate of treatment failure. 30 The cause of the higher recurrence rate in patients with BRAF mutations is the loss of affinity of the follicular cells with radioactive iodine and the consequent PTC unresponsiveness to therapy with radioactive iodine.30,48 Abnormal activation of the MAPK signaling pathway is responsible for altering the iodine capture mechanism of thyroid follicular cells. 49 The BRAF V600E mutation alters the function of the BRAF protein, thereby inhibiting the expression of thyroid function-specific genes and the loss of the iodine tolerance mechanism of thyroid cells through the activation of the MAPK signaling pathway in thyroid carcinomas. Consequently, iodine is only partially tolerated in thyroid cells carrying the BRAF V600E mutation and sparingly accumulated in the lumen of the thyroid follicle. 49 This is the key to the molecular mechanism of the radioiodide loss induced by BRAF mutations in PTC, leading to the radioiodine therapy failure and increased recurrence rate.
Our study has some limitations. The study design has a limitation of subjects with a small sample size and without a control group. The follow-up time of patients after treatment was not long, so the predictive and prognostic role of immune markers as well as BRAF gene mutations in thyroid carcinoma had not been fully evaluated yet.
Conclusion
The present study found a high prevalence of BRAF gene mutation at the T1799A (V600E) position in thyroid carcinoma patients. The relationships of the positivity rate of HBME-1, COX-2, and Ki67 markers with BRAF V600E gene mutation were identified. Patients with the BRAF V600E gene mutation showed a significantly higher relapse rate and earlier relapse time than patients without the mutation.
Supplemental Material
sj-docx-1-onc-10.1177_11795549231203503 – Supplemental material for Relationships of BRAF V600E Gene Mutation With Some Immunohistochemical Markers and Recurrence Rate in Patients With Thyroid Carcinoma
Supplemental material, sj-docx-1-onc-10.1177_11795549231203503 for Relationships of BRAF V600E Gene Mutation With Some Immunohistochemical Markers and Recurrence Rate in Patients With Thyroid Carcinoma by Tri Bui Dang Minh, Thuan Nghiem Duc, Van Phan Nguyen Thanh, Tuan Dinh Le, Minh Duc Tong, Trung Hoang Nguyen, Anh Le Tuan, Kien Xuan Nguyen, Tien Tran Viet, Thang Ba Ta, Son Tien Nguyen, Hai Anh Vu, Ba Van Nguyen, Dung Nguyen Thi Ngoc, Viet Tran Quoc and Thanh Bui Duc in Clinical Medicine Insights: Oncology
Footnotes
Author contributions
TBDM, TND, VPNT, TDL, and MDT contributed to conceptualization. TBDM, VPNT, MDT, THN, ALT, TBT, STN, and HAV carried out data curation. TBDM, TDL, MDT, THN, KXN, TTV, STN, and DNTN performed formal analysis. TND and DNTN acquired funding. TBDM, TND, VPNT, TDL, and BVN carried out the investigation. VPNT, TTV, TBT, STN, HAV, THN, BVN, and DNTN framed the study methodology. TND, TDL, KXN, TBT, STN, BVN, and DNTN contributed to project administration. TBDM and KXN contributed to resources. TBDM, TDL, MDT, THN, ALT, TTV, HAV, and DNTN contributed to software. TBDM, TND, TDL, KXN and DNTN supervised the study processes. TBDM, TTV, HAV, BVN, and DNTN contributed to validation. TBDM, TDL, and DNTN contributed to visualization. TBDM, THN, TND, KXN, TBT, STN, and DNTN reviewed and edited the article. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The datasets used or analysed during the current study are available from the corresponding author on reasonable request.
Ethical Statement
All participants provided written informed consent and agreed to join our study; and the protocol was approved by the Ethical Review Committee of Vietnam Military Medical University (reference no. 168/2014/IRB-VMMU). The study was also conducted using good clinical practice following the Declaration of Helsinki of 1964, as revised in 2013.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
