Abstract
Background:
Epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) have limited or no response in some certain patients of non-small-cell lung cancer (NSCLC). However, real-world survival analyses comparing clinical data and EGFR-plasma mutation are still lacking.
Methods:
In total, 159 patients with advanced NSCLC resistant to first-generation EGFR-TKIs were included for consecutive blood sampling in this study. Super-amplification refractory mutation system (Super-ARMS) was used to detect EGFR-plasma mutations and correlations between survival and circulating tumor DNA (ctDNA) were analyzed.
Results:
Among 159 eligible patients, the T790M mutation was detected in 27.0% (43/159) of patients. The median progression-free survival (mPFS) was 10.7 months in all patients. Survival analysis revealed that patients with the T790M mutation had shorter progression-free survival (PFS) than those with the T790M wild-type (10.6 months vs 10.8 months, P = .038). Patients who cleared EGFR-plasma mutation had prolonged PFS compared with those with nonclearing EGFR-plasma mutation (11.6 months vs 9.0 months, P = .001). Cox multivariate analysis showed that the nonclearance of EGFR-plasma mutations was an independent risk factor for PFS (RR = 1.745, 95% CI: [1.184, 2.571], P = .005). The T790M mutation was associated with nonclearance of the EGFR-plasma mutation (χ2 = 10.407, P = .001).
Conclusion:
Patients with advanced NSCLC who were resistant to the first-generation EGFR-TKI had a prolonged PFS with clearance of EGFR-plasma mutation. Those nonclearers were more likely to harbor T790M mutations in plasma.
Introduction
Epidermal growth factor receptor-tyrosine kinase inhibitor (EGFR-TKI) therapy is a milestone that prolongs progression-free survival (PFS) and improves the quality of life of non-small-cell lung cancer (NSCLC) patients with EGFR-sensitizing mutations.1-3 However, acquired resistance mutations are unavoidable and mainly occur approximately 1 year after treatment.4,5 Among patients with secondary resistance to EGFR-TKIs, approximately 50% have the T790M mutation.6-8 The presence of the T790M mutation defines a clinical subset with a relatively favorable prognosis and more indolent progression.4,8
Currently, response assessments rely primarily on imaging modalities that may not reflect clonal evolution at the molecular level. Therefore, re-biopsy is recommended for disease progression, if possible. However, feasibility, safety, and tissue adequacy poses challenges for repeat biopsies.9,10 Furthermore, tissue biopsies provide limited information regarding tumor heterogeneity.8,11,12 However, plasma ctDNA predominantly released by apoptosis and necrosis of cancer cells has been shown to closely match that of tumors8,13 and being used to identify molecular targets, predicting response and prognosis, and monitoring resistance in NSCLC.14-16
In the real world, patients have a complex treatment history; therefore, clinical data are limited, and more work is needed to clarify its prognostic and predictive values. Hence, this prospective study of patients with advanced EGFR-mutant NSCLC was conducted to analyze their survival.
Patients and Methods
Study population
A prospective observational study was set up and opened for inclusion from February 2018 to February 2022 in Beijing Chest Hospital affiliated to Capital Medical University. Patients were eligible for inclusion if they were histologically confirmed to have IIIB/IV lung adenocarcinoma, had EGFR gene-sensitive mutations (including exon 19 deletion and exon 21 point mutation L858R), and had received first-generation EGFR-TKI as first-line therapy. Vascular endothelial growth factor (VEGF) inhibitor (anti-VEGF) therapy can be administered to patients with pleural or pericardial effusion. The key exclusion criteria were patients without EGFR gene testing of tissues at diagnosis or without disease evaluation. Clinical staging followed the eighth edition of the American Joint Commission on Cancer (AJCC), 17 and treatment response was assessed using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The last follow-up was performed on February 1, 2023. The Ethics and Scientific Committee of Beijing Chest Hospital, Capital Medical University approved this study (BJXKYY 2018-02-01).
Blood samples and super-ARMS
Blood samples were collected at baseline, radiological progression, and various times during first-line treatment. Next, 10 mL venous blood was collected from each patient using an Ethylene diamine tetraacetic acid anticoagulant tube, placed in a centrifuge tube with serum separation gel, and separated within 2 h. The upper serum was centrifuged at 2000g for 10 min, and the supernatant was centrifuged at 8000g for 10 min. Following the manufacturer’s instructions, peripheral blood cell-free DNA was extracted for immediate detection, or otherwise stored at −20°C. The DNA mass concentration of the samples was then diluted to 1 to 3 ng/µL, and a human EGFR mutation gene detection kit (Amoy Diagnostics Co, Xiamen, China) was used to detect the EGFR driver gene mutation in all samples. A SLAN96S real-time fluorescence PCR instrument (Shanghai Hongshi, China) was used for detection. Briefly, the DNA templates were added to the EGFR Reaction Mix, consisting of novel proprietary primers and probes, dNTPs, buffer, Mg2+, and Taq DNA polymerases. The cycling conditions were as follows: 1 cycle at 95°C for 10 min; 15 cycles at 95°C for 40 s, 64°C for 40 s, and 72°C for 30 s; and 28 cycles at 93°C for 40 s, 60°C for 45 s, and 72°C for 30 s. Positive and negative cutoff points were established for the controls. For positive controls, Ct mutation < 28, and T790MΔCt mutation ⩽ 7 were established.
Definition of clearers and nonclearers
“Clearers” were defined as patients with detectable EGFR mutations in plasma at baseline but without blood mutations during first-line treatment. “Nonclearers” were defined as patients with detectable EGFR mutations in the plasma continuously during the entire course of first-line treatment.
Data analysis
Progression-free survival was defined as the period from the first day of EGFR-TKIs treatment to disease progression, death, and last follow-up. Kaplan-Meier methods were used to evaluate PFS in univariate analysis, and logistic regression was performed to evaluate clinical features and PFS in multivariate analysis. The relationship between the influencing factors and blood T790M mutation was evaluated using a standard chi-squared test. All statistical tests were performed using the SPSS Statistics for Windows or Mac, Version 16.0 (SPSS, Chicago, IL, USA). A 2-sided P < .05 was considered to be statistically significant.
Results
A total of 159 patients ranging in age from 32 to 85 years (median age 61 years) were included in the study. There were 59 males and 100 females, 73.0% (116/159) of whom were nonsmokers. The Eastern Cooperative Oncology Group (ECOG) performance status (PS) was 0 to 1 in 150 patients and 2 to 3 in 1 patient. Twenty patients had stage-IIIB disease, and 139 patients had stage-IV disease. Furthermore, all patients included 82 with 19-Del mutations, 77 with 21-L858R mutations, and no primary T790M mutations.
Among the 159 patients, the mPFS was 10.7 months. As shown in Table 1, PFS was associated with blood T790M mutations and the clearance of EGFR mutations. Figure 1 shows that patients with T790M mutation had shorter PFS than those with the T790M wild type (10.6 months vs 10.8 months, P = .038); As shown in Figure 2, patients with clearance of EGFR mutations at clinical progression had longer PFS compared with patients with non-clearing EGFR-plasma mutation (11.6 months vs 9.0 months, P = .001). Sex, smoking history, initial EGFR mutation status, TKI plus anti-VEGF therapy, and the type of disease progression were not related. As presented in Table 2, the multivariate analysis suggested that PFS was associated only with the clearance of EGFR mutations (RR = 1.745, 95% CI: [1.184, 2.571], P = .005).
Univariate analysis of PFS of patients with advanced NSCLC resistant to first-generation EGFR-TKIs.

Abbreviations: CI, confidence interval; EGFR, epidermal growth factor receptor; NSCLC, non-small cell lung cancer; PFS, progression-free survival; TKI, tyrosine kinase inhibitor; VEGF, vascular endothelial growth factor.

Kaplan-Meier curves for PFS in patients with blood T790M mutation (T790M+) and T790M wild-type (T790M−) during first-generation EGFR-TKI treatment.

Kaplan-Meier curves for PFS in clearers and nonclearers receiving first-generation EGFR-TKI treatment.
Multivariate analysis of PFS of patients with advanced NSCLC resistant to first-generation EGFR-TKIs.

Abbreviations: CI, confidence interval; EGFR, epidermal growth factor receptor; NSCLC, non-small cell lung cancer; PFS, progression-free survival; RR, relative risk; TKI, tyrosine kinase inhibitor.
Among 159 patients, 43 (27%) tested positive for the T790M mutation. Table 3 shows that nonclearers were more likely to have T790M mutation in their blood (χ2 = 15.614, P = 0.001).
Analysis of influencing factors and blood T790M mutation in 159 patients with advanced NSCLC resistant to first-generation EGFR-TKIs.
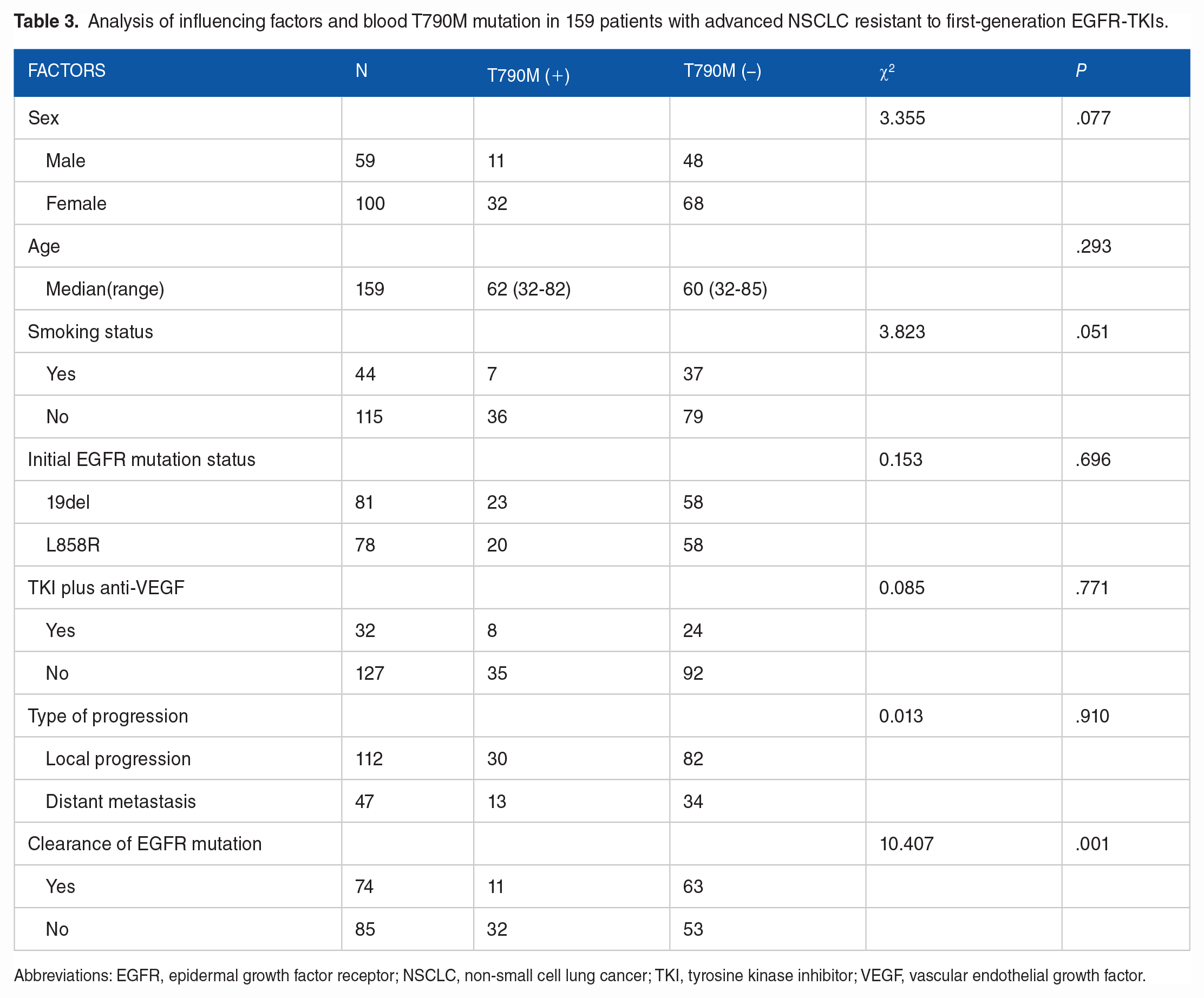
Abbreviations: EGFR, epidermal growth factor receptor; NSCLC, non-small cell lung cancer; TKI, tyrosine kinase inhibitor; VEGF, vascular endothelial growth factor.
Discussion
The blood T790M gene was detected using Super-ARMS. The Super-ARMS is a second-generation ARMS detection technology. In this qualitative detection technology, a ring-forming structure was added to the primer to improve the specific binding ability of the copy template. Notably, the sensor exhibited a high degree of selectivity and sensitivity. Several studies have shown that in patients resistant to first-generation EGFR-TKIs, the rate of blood T790M mutation was lower than that of tissue samples, and the rate of blood T790M mutation was 29.8%-43%.7,11,14 In our study, the rate of blood T790M mutation was lower, possibly because of the difference in the number of patients per study. Currently, tissue biopsy remains the gold standard for T790M mutation detection; however liquid biopsy specimens support this.
Our group showed that patients with blood T790M mutation had a shorter PFS than those with blood T790M wild type after resistance to first-generation EGFR-TKIs. Previous studies7,18 found that the longer the PFS with first-generation TKI, the greater the probability of T790M mutation. Our results contradict these findings, which may be related to the EGFR-TKI plus anti-VEGF therapy. Recently, anti-VEGF therapy has been shown to normalize abnormal blood vessels in tumors and reduced microvascular growth, thereby inhibiting cancer metastasis. Furthermore, VEGF is highly expressed in tumor cells harboring EGFR resistance mutations. Thus, the inhibition of VEGF can overcome the secondary resistance induced by T790M; 19 thereby, prolonging PFS. In our real-world study, 32 patients (32/159, 20.1%) recieved TKI plus anti-VEGF therapy and 24 patients (24/32, 75%) had blood T790M wild type. Therefore, more patients with blood T790M wild type had a longer PFS.
Our results also suggested that a longer PFS was observed in patients with cleared EGFR mutations during first-line treatment. In addition, blood T790M mutation was associated with nonclearance of EGFR-plasma mutation. Recent studies have demonstrated that ctDNA can reliably track molecular dynamics in real time to predict treatment responses in residual tumors.8,16 Song et al 20 conducted a real-world study of 248 patients and found that patients who cleared ctDNA at least once during treatment had significantly longer PFS and overall survival (OS). Mack et al 21 demonstrated that the plasma clearance of mutant EGFR ctDNA at 8 weeks was highly associated with a significantly decreased risk of progression. Furthermore, Verheijen et al 22 found that an increase in T790M mutation copy number could predict disease progression. Thus the induction of T790M mutation may be related to long-term EGFR-TKI therapy and the nonclearance of EGFR mutations, rather than the natural process of NSCLC progression.
Currently, the third-generation EGFR-TKI osimertinib is widely used in advanced NSCLC patients with T790M mutations after resistance to first-generation EGFR-TKIs. It irreversibly binds to the cysteine-797 residue of the adenosine triphosphate (ATP)-binding domain of the EGFR kinase domain. In doing so, it inhibits downstream signaling pathway, thereby inhibiting tumor proliferation.
Conclusion
Patients with advanced NSCLC who were resistant to the first-generation EGFR-TKI had a prolonged PFS with clearance of EGFR-plasma mutation. Those nonclearers were more likely to harbor T790M mutations in plasma. Detection of the blood T790M mutation has great clinical value, as it is conducive to the early detection of drug resistance and guides individualized treatment. However, owing to the small number of cases included in this study, additional samples and prospective studies are needed to demonstrate the reliability of this conclusion.
Footnotes
Acknowledgements
The authors thank Sage Language Services for English language editing services.
Author Contributions
All authors contributed to the conception of the study, participated in the analysis and interpretation of the data, performed a critical review of its content, and approved the final version of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the Helsinki Declaration as revised in 2013. All patients provided written informed consent. Blood sample collection was conducted according to the protocols approved by the Ethics and Scientific Committee of Beijing Chest Hospital (approval no. BJXKYY 2018-02-01).
