Abstract
Background:
Distant organ metastasis is the leading cause of death in pancreatic neuroendocrine tumor (pNET) patients. In the present study, we aimed to develop and validate a nomogram that could accurately identify pNET metastasizing to distant organs.
Methods:
The cases extracted from the Surveillance, Epidemiology, and End Results (SEER) program were assigned to the training cohort and validation cohort. The cases from the Chinese Gastrointestinal Neuroendocrine Tumors program were assigned to the external validation cohort. The strategy was developed with the support of a nomogram, and the predictive value of this strategy was evaluated by the receiver operating characteristic (ROC) curve analysis.
Results:
In total, 2024 American cases were involved in the present study. Besides, 1450 and 574 patients were allocated into training and internal validation cohorts, respectively. In addition, 122 Chinese patients were assigned to the external validation cohort. The results of the univariate logistic regression analysis suggested that tumor grade, tumor size, and the number of metastatic lymph nodes were the risk of metastasis to distant organs, and these 3 clinicopathological characteristics were used to develop the nomogram. We observed that the accuracy of the nomogram for predicting metastasis to distant organs was 0.797, 0.819, and 0.837 in the training cohort, internal validation cohort, and external validation cohort, respectively.
Conclusions:
A predictive nomogram was developed and validated, and it showed an acceptable performance in predicting metastasis to distant organs. The results will enable clinicians to identify pNETs, metastasizing to distant organs, and develop an effective individualized therapeutic strategy for these patients.
Keywords
Introduction
Pancreatic neuroendocrine tumor (pNET) originates from the peptide neurons and neuroendocrine cells in the pancreas. It scarcely occurs, and it accounts for only 0.1% of all pancreatic tumors. 1 Compared with pancreatic ductal adenocarcinoma, pNET is associated with a relatively indolent pathophysiological behavior. 2 Notably, our previous study proved that 40% of pNETs were metastasized to distant organs, such as the liver, lung, bone, and brain at the time of initial diagnosis. 3 In addition, a study reported that 45% to 90% of patients suffered from liver metastasis during the disease course. 4 Similar to other tumors, when a pNET metastasizes to distant organs, it significantly decreases the survival time of patients. 3 Thus, identification of pNETs will reduce the risk of metastasis to distant organs and prolong the survival time of patients, and it is essential to take the advantage of individualized therapies for these patients with early-stage cancer.
To monitor the tumor progression and indicate whether the tumor is metastasized to distant organs, physicians recommended patients undergo computed tomography (CT) every 2 or 6 months. 5 However, this strategy is accompanied by some limitations. For instance, this examination is expensive. Generally, the CT examination of the head, chest, or abdomen costs $1400 to $2500, and patients are suggested to undergo this examination every 2 or 6 months. This increases the financial burden on patients. 6 In addition, this examination delivers a high dose of radiation, and this may impair the functions of different organs. 7 Identifying tumors at low risk of metastasis and increasing the interval between CT scans will reduce the financial burden and the amount of radiation. This might give rise to benefits for these patients.
Nomogram is a strategy that graphically represents mathematical formulas or algorithms to predict outcomes, such as surgical complications, metastasis of tumors, and death. 8 Shariat et al systematically compared nomogram with other methods for predicting outcomes. They found that the performance of the nomogram was superior to other methods, and it assisted physicians to make perfect decisions. 9
The present study aimed to develop and validate a nomogram, which could accurately identify pNETs metastasizing to distant organs. The findings may assist physicians to detect pNETs, metastasizing to distant organs, and developing an efficacious individualized therapeutic strategy for these patients.
Cohorts and Methods
The American and Chinese Cohorts
The American cohort was developed with the support of the Surveillance, Epidemiology, and End Results (SEER) program (1975-2017). The processes of patients’ enrollment have been described in our previous study. 10 The topography codes (C25.0 to C25.9) were used in combination with histology codes (8150, 8151, 8152, 8153, 8154, 8155, 8156, 8157, 8240, 8241, 8242, 8243, 8244, 8245, 8246, and 8249) of the International Classification of Diseases for Oncology (the third edition). To achieve the Chinese cohort, at least one cancer hospital and one general hospital from 7 geographical areas of China were selected. 11 In total, 23 hospitals were involved in the present study, and patients who were pathologically diagnosed with gastroenteropancreatic neuroendocrine neoplasm (GEP-NEN) between 2001 and 2010 were enrolled. This study was approved by the ethics committee of the investigators’ and sub-investigators’ affiliations (Supplementary Table 1).
Training and validation cohorts
As shown in Figure 1, we included cases with pNETs who were pathologically diagnosed in the American cohort and Chinese cohort, and we excluded cases with incomplete clinicopathological data. Subsequently, we followed the suggestions of the Transparent Reporting of a multivariable prediction model for Individual Prognosis Or Diagnosis (TRIPOD) Statement, and 70% or 30% of the American cases were randomly assigned to the training cohort or internal validation cohort. 12 To evaluate the performance of the nomogram, the Chinese cohort was defined as the external validation cohort, and the predictive performance of the nomogram was evaluated.

Flowchart of patient selection.
Variables and statistical analyses
The clinicopathological characteristics including age, gender, race, insurance status, marital status, tumor size, the location of the tumor (pancreatic head, body, tail, or other), tumor grade, the number of positive lymph nodes (PLNs), and the risk of metastasis to distant organs (eg, liver, lung, bone, and brain) are presented in Table 1. The statistical differences of these variables between distinct cohorts were determined by the chi-square test. To develop a nomogram, the relationship between these variables and metastasis was investigated by univariate or multiple logistic regression analysis (Table 2). The results were presented as odds ratio (OR) and 95% confidence interval (CI). Notably, the Ki-67 and mitotic index of World Health Organization (WHO) 2017 classification were not recorded in the SEER program, and the tumor grade in the training and the internal validation cohorts were classified according to the morphological description (well-, moderately, poorly differentiated, and undifferentiated) of ICD-O-3. We, therefore, graded tumors in the external validation cohort based on the morphological description. In addition, the optimal cut-off values of the continuous variables, such as age, tumor size, and PLN, were determined by the Youden index. 13 To measure the performance of these variables for predicting the risk of metastasis to distant organs, the receiver operating characteristic (ROC) curve analysis was undertaken, and the value of the area under the curve (AUC) with 95% CI was calculated (Figure 2). If the AUC value was equal to 0.5, it was revealed that the nomogram had no discrimination ability. 14 These analyses were performed with SPSS 19.0 software (IBM, Armonk, NY, USA). Subsequently, tumor size, tumor grade, and PLN which had predictive valuable were used to develop the nomogram using the “rms” package in the R 4.1.2 software (Figure 3). To evaluate the predictive performance of the nomogram, the total scores of each case in the training, internal validation, and external validation cohorts were calculated, and the AUC value was determined (Figure 4). P value ⩽ .05 was considered statistically significant.
Characteristics of patients.

Abbreviations: AA, African American; AI/AN, American Indian/Alaska Native; A/P Islander, Asian or Pacific Islander; PLN, positive lymph node.
Excluding the data of external validation cohort.
Univariable logistic regression analysis in training cohort.

Abbreviations: AA, African American; AI/AN, American Indian/Alaska Native; A/P Islander, Asian or Pacific Islander; CI, confidence interval; OR, odds ratio; PLN, positive lymph node.
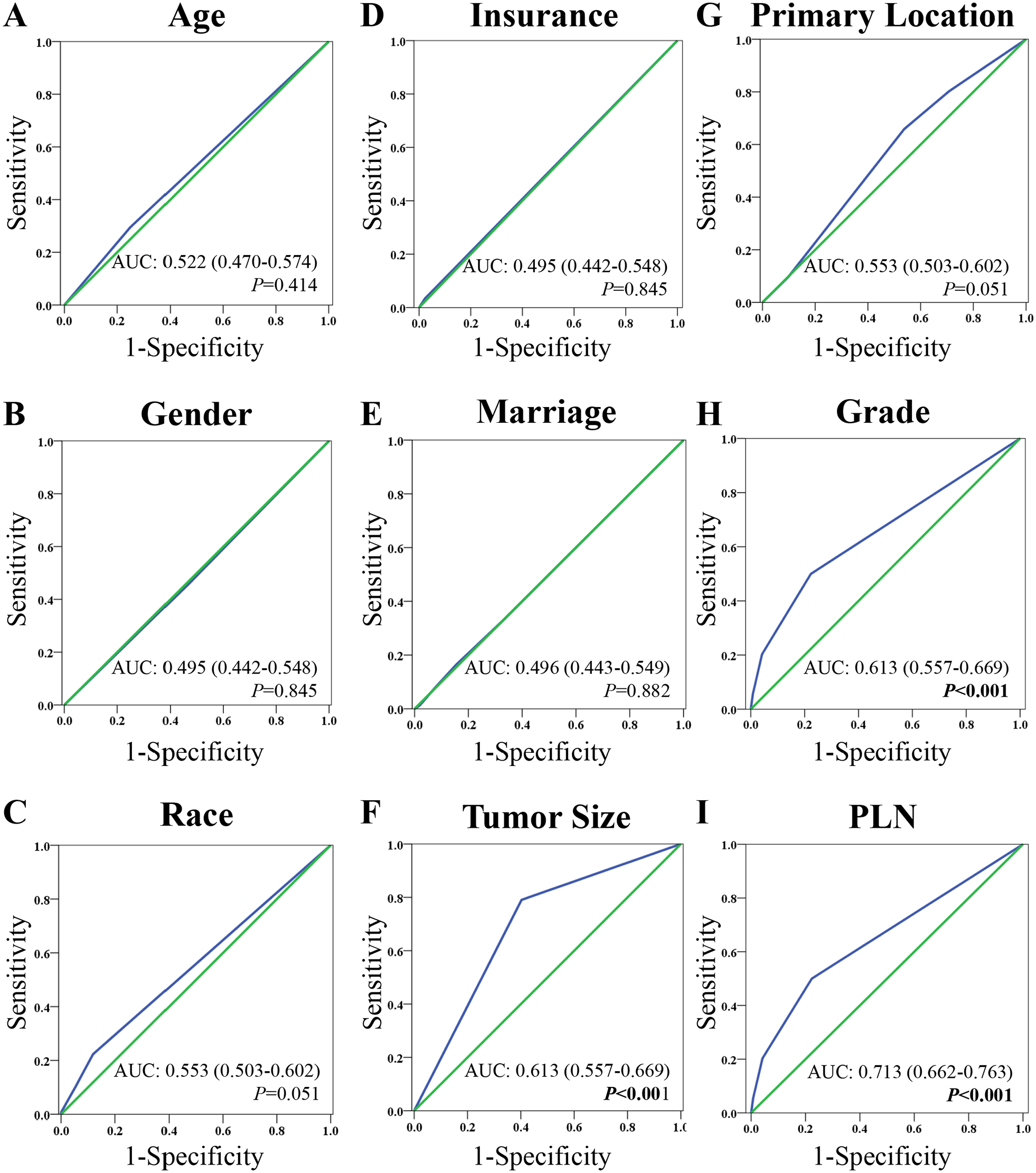
The ROC curves and AUCs of (A) age, (B) gender, (C) race, (D) insurance status, (E) marriage status, (F) tumor size, (G) primary location of tumors, (H) grade, (I) metastatic lymph node.

The nomogram for predicting metastasis in pNETs.

The ROC curves and AUCs of the nomogram in predicting (A) multiple organs metastasis, (B) liver metastasis, (C) lung metastasis, (D) bone metastasis in the training cohort and in the (E) internal validation cohort or in the (F) external validation cohort.
Results
Clinicopathological characteristics of patients in training, internal validation, and external validation cohorts
Following the inclusion and exclusion criteria, 2024 American cases were enrolled in the present study. Subsequently, these cases were split into the training cohort (N = 1450) and the internal validation cohort (N = 574). In addition, 122 Chinese cases were enrolled and this cohort was defined as an external validation cohort (Figure 1). The characteristics of patients are summarized in Table 1. Compared with the training and internal validation cohorts, the number of patients who were younger than 66 years old was greater in the external validation cohort (92.6% in the external validation cohort, 69.5% in the internal cohort, 70.9% in the training cohort). In addition, 18.9% of pNETs were metastasized to distant organs in the external validation cohort, and this rate was higher than that in the internal cohort (10.6%) and the training cohort (9.7%).
The value of clinicopathological characteristics for predicting metastasis
In order to evaluate the value of the clinicopathological characteristics for predicting metastasis, the univariate logistic regression analysis was performed, and it was revealed that compared with whites, African Americans, and Asians (OR: 0.475; 95% CI: 0.227-0.992; P < .048; Table 2) were at a lower risk of tumor metastasis. The tumor size, which was larger than 3 cm, significantly increased the risk of metastasis compared with a tumor size of no more than 3 cm (OR: 5.687; 95% CI: 3.664-8.827; P < .001; Table 2). In addition, compared with well-differentiated tumors, the moderately-differentiated (OR: 2.543; 95% CI: 1.708-3.786; P < .001; Table 2) and the undifferentiated tumors had a higher risk of metastasis (OR: 11.514; 95% CI: 4.412-30.042; P < .001; Table 2); the PLN was also found as a risk factor of the metastasis to distant organs (PLN ⩽ 2 vs PLN = 0, OR: 4.469; 95% CI: 2.826-7.066; P < .001; PLN > 2 vs PLN = 0, OR: 7.303; 95% CI: 4.618-11.551; P < .001; Table 2). The multiple logistic regression proved that tumor size, grade, and PLN were the independent risk factors of metastasis (Table 2).
To evaluate the predictive performance of these clinicopathological characteristics, we calculated the AUC values by the ROC curve analysis. The AUC values of age (Figure 2A), gender (Figure 2B), race (Figure 2C), insurance status (Figure 2D), marital status (Figure 2E), and primary site (Figure 2G) were not significantly higher than 0.500 (Figure 2). This suggested that these predictors could not discriminate between the tumors that were metastasized to distant organs and tumors that could not be metastasized to distant organs. The AUC values suggested that tumor size (Figure 2F), tumor grade (Figure 2H), and PLN (Figure 2I) could significantly discriminate between the tumors with metastasis and tumors without metastasis. We, therefore, used tumor size, tumor grade, and PLN to develop the nomogram.
Development and validation of the nomogram
As mentioned earlier, we used tumor size, tumor grade, and PLN of the training cohort to develop the nomogram. A point was assigned to each predictor. For instance, if the tumor size was not more than 3 cm, zero point was assigned to this predictor; otherwise, 3 points were assigned to the predictor. Subsequently, the total score of these predictors for each case was calculated, and the risk of metastasis was determined (Figure 3).
In order to assess the predictive performance of the nomogram, the ROC curve analysis was carried out, and the AUC value was determined in the training cohort. It was found that the AUC value of the nomogram (AUC: 0.804; 95% CI: 0.768-0.839) was significantly (P < .001) high than 0.500 (Figure 4A). Additionally, the performance of the nomogram was evaluated for predicting metastasis to distant organs. It was revealed that the mentioned strategy could significantly discriminate between patients who suffered from liver metastasis (AUC: 0.807; 95% CI: 0.772-0.842; P < .001) and patients without liver metastasis (Figure 4B). In addition, the nomogram also had an acceptable accuracy in predicting lung metastasis (AUC: 0.730; 95% CI: 0.465-0.996; Figure 4C) and bone metastasis (AUC: 0.834; 95% CI: 0.722-0.946; Figure 4D).
To validate the predictive performance of the nomogram, the ROC curve analysis was conducted in the internal validation and external validation cohorts. It was found that the nomogram could significantly distinct patients with metastasis from patients without metastasis in both cohorts (internal validation: AUC: 0.839, 95% CI: 0.790-0.889, P < .001; external validation: 0.717; 95% CI: 0.597-0.837, P < .001; Figure 4E and F, respectively).
Discussion
Due to the indolent nature of neuroendocrine tumors and patients have a better prognosis than those who had malignant tumors, neuroendocrine tumors have not noticeably attracted clinicians’ attention. 15 For a long time, neuroendocrine tumors were considered as begin carcinoma. Notably, several studies confirmed that similar to other malignant tumors, neuroendocrine tumors can invade and metastasize to distant organs,16 -19 which is consistent with the findings of the present study. In the present study, we observed that 220 (10%) patients in the American cohort and 32 (22.5%) patients in the Chinese cohort were at risk of metastasis to distant organs at the time of initial diagnosis (Table 1). In addition, in contrast to the survival of patients with neuroendocrine tumors that were located in the rectum (24.6 years) and appendix (more than 30.0 years), the survival of patients with pNETs was only 3.6 years. 3 Thus, neuroendocrine tumors, especially pNETs, are malignant tumors, and patients should undergo standardized treatment regimens.
Our previous study showed a poor prognosis for patients with pNETs and metastasis. 4 Tumors that metastasize to distant organs significantly decrease the survival time of patients. Therefore, patients who are at a high risk of metastasis should undergo perioperative screening for metastases, particularly early-stage cancer, to save on the cost of cancer therapy. Notably, Gerber et al 20 suggested that it is not necessary to screen for metastasis in breast cancer patients without clinical signs. However, it is noteworthy that unlike breast tumors, pNETs are located in the abdomen. If a pNET patient develops symptoms such as intestinal obstruction, jaundice, or back pain, it means the pNET has compressed the intestine or metastasized to the liver or nerve.19,21 These patients cannot undergo radical surgery and have a poor prognosis.21,22
In general, if tumors are metastasized to distant organs, surgical resection is not recommended for patients with malignant tumors.23,24 However, the resection of primary tumor in pancreas or the tumor in combination with the metastasized tumor in liver is suggested to pNET patients.25 -27 This is supported by the findings by Bertani et al. 28 They retrospectively analyzed 94 patients, and they observed that compared with patients who underwent peptide receptor radionuclide therapy, the hazard ratio (HR) of death significantly decreased in patients who underwent both surgical resection and peptide receptor radionuclide therapy (HR: 0.18; 95% CI: 0.05-0.66; P = .010). This suggested the importance of primary tumor resection prior to peptide receptor radionuclide therapy in patients with diffuse liver metastasis. In addition, Nigri et al 29 systematically reviewed 116 publications and found that surgical resection is the only curative treatment for pNET patients and surgery may improve the survival of patients.
The clinical value of the present study is that we developed a nomogram that could identify pNETs, metastasizing to distant organs. An individualized follow-up strategy and resection of the metastasized tumors at the early stage may prolong the survival of patients. Notably, even though we validated the diagnostic performance of the nomogram in 2 validation cohorts and both cohorts suggested that the nomogram had acceptable discrimination ability, there are still some limitations in the present study. First, this is a retrospective study and the selective bias might impair the robustness of the findings. Second, in the Chinese cohort, most cases were from cancer hospitals, and most of the hospitalized patients had tumors that were metastasized to distant organs. This might be another source of bias in the present study.
Conclusions
In conclusion, a nomogram was developed that could identify pNETs, metastasizing to distant organs in American and Chinese patients. The results may assist clinicians to develop individualized follow-up strategies.
Supplemental Material
sj-docx-1-onc-10.1177_11795549221099853 – Supplemental material for A Nomogram to Accurately Identify Pancreatic Neuroendocrine Tumors Metastasizing to Distant Organs: A Study Based on Two National Population-Based Cohorts From the United States and China
Supplemental material, sj-docx-1-onc-10.1177_11795549221099853 for A Nomogram to Accurately Identify Pancreatic Neuroendocrine Tumors Metastasizing to Distant Organs: A Study Based on Two National Population-Based Cohorts From the United States and China by Xianbin Zhang, Lili Lu, Jun Liu, Weihan Liu, Li Li, Yushan Wei, Jinhu Fan, Li Ma and Peng Gong in Clinical Medicine Insights: Oncology
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by National Natural Science Foundation of China (grant number: 81973646 and 82104596); Natural Science Foundation of Shenzhen University General Hospital (grant number: SUGH2018QD021); Shenzhen Key Medical Discipline Construction Fund & Sanming Project of Medicine in Shenzhen (SZSM202111002); The Project of Department of Education of Guangdong Province (2018KTSCX193 and 2020KZDZX1170); Shenzhen Science and Technology Innovation Commission (Grant number: RCBS20200714114958333 and JCYJ20190808114203755).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization: PG, LM, JF. Analysis and acquisition of data: XZ, LL, JL, WL, YW. Original draft: XZ and LL. Revised draft: All authors.
Compliance with ethics requirements
We signed the “Surveillance, Epidemiology, and End Results Program Data-Use Agreement” in accordance with the requirement of using SEER database (SEER ID: 21939-NOV 2019). The ethics committee of the Cancer Hospital, Chinese Academy of Medical Sciences; and the First Affiliated Hospital of Dalian Medical University approved the study protocol (LCKY 2012–32).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
