Abstract
Background:
Small bowel adenocarcinoma (SBA) is a rare malignancy that accounts for 3% of all gastrointestinal tumors. We evaluated the clinical characteristics, outcomes, and prognostic factors of primary SBAs.
Methods:
We retrospectively analyzed the clinicopathological features and clinical outcomes of 300 patients with SBA from three institutions in China between January 2003 and July 2020. Overall survival (OS) was analyzed using the Kaplan–Meier method and it was statistically compared using the log-rank test. Single-variable and multivariate analyses were used to identify the significant correlates of OS.
Results:
The primary tumor was on the duodenal papilla in 156 patients (52%), in the duodenum in 60 patients (20%), and in the jejunum–ileum in 84 patients (28%). The median OS of the entire cohort was 32.5 months (range, 0-213 months), with a 1-year OS rate of 78.0%. For jejunoileal adenocarcinoma, advanced age, advanced T stage, advanced N stage, more positive lymph nodes, distant metastasis, high carcinoembryonic antigen (CEA), and lymphocyte-to-monocyte ratio < 2.32 predicted worse survival on single-variable analysis. Multivariate analysis showed that advanced age, advanced tumor node metastases (TNM) stage, high CEA level, high alpha fetoprotein (AFP) level, and low prealbumin level were independent prognostic factors for non-ampullary SBA. The independent prognostic factors for duodenal papilla adenocarcinoma included TNM Stage III, nerve invasion, low platelet/lymphocyte ratio, and high CA19-9.
Conclusion:
We found different independent prognostic factors for tumors at different locations. This finding warrants further investigation to ensure more effective management strategies for SBA.
Introduction
Small bowel tumors are relatively rare malignant neoplasms that account for only 3% (approximately) of all newly identified gastrointestinal tumors and approximately 0.6% of all malignancies each year.1(p9) According to previous reports, the incidence of small intestinal tumors rose to an average annual rate of 2.3% over the past 20 years.2(p64) It is estimated that there will be 11 390 new cases and approximately 2100 deaths from these tumors by 2021. 3
The main histological subtypes of small intestinal tumors are adenocarcinoma, neuroendocrine tumor, gastrointestinal stromal tumor, lymphoma, sarcoma, and melanoma. 4 In recent years, the incidence of neuroendocrine tumors has increased compared with that of small bowel adenocarcinoma (SBA). SBA has become the second most common histological subtype, accounting for 30% to 40% of small bowel cancers. 4 The small intestine is divided into duodenum, jejunum, and ileum. The most common primary site of SBA is the duodenal papilla (papilla of Vater), where the junction of the bile duct and pancreatic duct enters the duodenum.5(p303)
Surgical resection and local lymph node dissection are effective treatments for SBAs.6(p15),7(p1117) If radical surgery is performed at an early stage of the disease, it may improve long-term survival, but early surgical treatment is often difficult for patients with SBA. The unique anatomical structure and nonspecific symptoms of the small intestine limit the likelihood of early diagnosis of small bowel cancer; thus, patients with SBA are often diagnosed at an advanced stage. Surgery combined with chemotherapy may help improve the survival rate; however, the effect of the treatment remains controversial.8 -10 Considering the low incidence of the disease in the population, limited clinical data, and lack of prospective studies, there is no consensus regarding the most effective treatment strategy. Therefore, the prognosis of patients with SBA is poor, and the 5-year overall survival (OS) rate after surgery is very low. Previous studies have shown that the 5-year survival rate is 14% to 35%, and the median survival time is 15.9 to 36 months.2(p66), 4 ,11(p1)
Many prognostic markers for patients with SBA are evaluated in previous studies. Histopathological features, such as positive surgical margins, vascular infiltration, lymph node metastasis rate, T4 tumor staging, and distant metastasis, were independent predictors of survival.12(p218),13(p5), 14 Other studies have also shown that age, tumor location, tumor size, and high CEA levels in the blood may be related to prognosis.15,16 Peripheral blood cell counts are associated with the survival of patients with malignant solid tumors, such as colon and gastric cancers, 17 ,18(p342) but there is insufficient information on SBA.
Due to the low incidence of SBA in the general population and the lack of clinical data, previous studies were mainly retrospective in nature and were conducted in single tertiary care centers. The dates of these studies make them prone to a selection bias. Additionally, research on small intestinal adenocarcinoma is concentrated in the West and Japan and is less concentrated in China. In this study, we collected data from a multicenter cohort of patients with SBA in Anhui, China. We aimed to retrospectively evaluate the clinicopathological features of SBA patients undergoing surgical treatment to identify important prognostic factors.
Method
Participants
This was a multicenter retrospective cohort study of 300 patients diagnosed with SBA at the following three hospitals from January 2003 to July 2020: the First Affiliated Hospital of Anhui Medical University, the Second Affiliated Hospital of Anhui Medical University, and Anhui Provincial Hospital.
Data collection
Study data were obtained from patients with small intestinal adenocarcinomas confirmed by biopsy specimens or mass resection. The exclusion criteria were as follows: (1) suspected invasive tumor of the pancreas; (2) small intestinal metastasis from cancer of other organs; and (3) no surgery. The following clinicopathological parameters were collected: sex, age, site of primary tumor, type of operation, symptoms at diagnosis, tumor node metastases (TNM) stage, lymph node metastasis, nerve invasion, vascular invasion, preoperative blood examination data (carcinoembryonic antigen [CEA], carbohydrateantigen 19-9 [CA19-9], alpha fetoprotein [AFP], hemoglobin [Hb], neutrophil-to-lymphocyte ratio [NLR], lymphocyte/monocyte ratio [LMR], platelet/lymphocyte ratio [PLR], albumin [Alb], and prealbumin [PA]), and treatment. The TNM stage was determined according to the American Joint Committee on Cancer standards (AJCC, 8th edition). Follow-up information was obtained through telephone interviews and outpatient follow-up.
Statistical analyses
All continuous variables are reported as median (range), and all categorical variables are summarized as frequencies (percentages). The optimal cutoff values for NLR, LMR, PLR, and Hb were determined using a receiver operator characteristic (ROC) curve and Youden’s index on the binary outcome of death/survival at the last known follow-up. OS time, defined as the period between surgery and death (or last follow-up), was the dependent variable. OS was estimated using the Kaplan–Meier method, and differences between groups were evaluated using the log-rank test. Single-variable and multivariate predictors of OS were determined using the Cox proportional hazards model. The Cox proportional hazard model with backward stepwise selection was used to identify the factors independently associated with survival time. We adjusted for confounding factors, such as age (25-45, 46-55, 56-65, 66-75, and ⩾76 years) and sex (male and female). Risk ratios and 95% confidence intervals (CIs) were calculated. All tests were two-sided, and a P value less than 0.05 was considered statistically significant. SPSS (26.0) was used for statistical analysis.
Results
Patient characteristics
Overall, from the three institutions, 300 patients, 122 women (40.7%), and 178 men (59.3%) with SBA were included. The patient characteristics are summarized in Table 1. The median age was 58.5 years (range, 26-90 years). Small bowel lesions were located on the duodenal papilla in 156 patients (52.0%), in the duodenum in 60 patients (20%), and in the jejunum–ileum in 84 patients (28%).
Patient characteristics.

Abbreviation: TNM, tumor node metastases.
There is some overlapping.
At the time of diagnosis, 283 (94.3%) patients were symptomatic. Intestinal bleeding-related symptoms were defined as melena and anemia secondary to tumor hemorrhage. The clinical presentations included jaundice in 95 patients, abdominal pain and distension in 173 patients, bleeding-related symptoms in 12 patients, and other symptoms in 31 patients. Among the patients with papilla of Vater adenocarcinoma (PVA), 147 (94.2%) were asymptomatic at diagnosis. In contrast, 58 patients (96.7%) with duodenal adenocarcinoma (DA) and 78 patients (92.9%) with jejunoileal adenocarcinoma were symptomatic at diagnosis. Jaundice is the primary symptom of patients with PVA, whereas it is abdominal pain for those with duodenal and jejunoileal adenocarcinomas.
All patients underwent surgical treatment, and the majority (n = 245; 81.6%) underwent surgery. In total, 300 patients were treated surgically, of whom the majority underwent pancreaticoduodenectomy (n = 208; 69.3%), whereas 77 underwent small bowel segmental resection (25.7%). Resection of the small intestine complicated by viscera was performed in 12 patients (4%) and 3 patients (1.0%) patients underwent palliative surgery. DA is mostly treated by pancreaticoduodenectomy, while jejunal and ileal adenocarcinomas are mostly treated by partial resection of the small intestine.
Based on the clinical tumor, node, and metastasis classification, the number of duodenal papilla adenocarcinoma patients in stages 0/I, II, and III was 34 (21.8%), 77 (49.4%), and 43 (27.6%), respectively. The number of DA patients in stages 0/I, II, III, and IV was 9 (15.0%), 29 (48.3%), 17 (28.3%), and 5 (8.3%), respectively. The number of jejunoileal adenocarcinomas in patients in Stages II, III, and IV was 34 (40.5%), 36 (42.9%), and 11 (13.1%), respectively. Non-ampullary small intestinal adenocarcinoma metastasized to a distant location, while duodenal papillary adenocarcinoma did not.
OS and prognostic factors
The median OS for the entire cohort was 32.5 (range, 0-213) months, with a 1-year OS rate of 78.0%. In total, 161 patients were followed-up until death, and 139 patients survived the entire follow-up period. Figure 1 shows the complement of the Kaplan–Meier curves for TNM staging. The median OS for all stage 0/I disease was 159 months, Stage II disease was 70 months, Stage III disease was 32 months, and Stage IV disease was 8 months. The 1-year survival rates for stages 0/I, II, III, and IV disease were 90.9%, 82.6%, 75.6%, and 33.3%, respectively. As tumor stage advanced, the survival rate progressively decreased. Figure 2 shows the complement of the Kaplan–Meier curves restricted to Stages I to III tumors only for the primary tumor site. The 1-year survival rates for duodenal papillary, duodenal, and jejunoileal adenocarcinomas were 83.3, 75, and 68.4%, respectively.

Kaplan–Meier curves of OS by TNM. Median OS for all Stage I disease was 159 months, Stage II disease 70 months, Stage III disease 34 months, and Stage IV disease was 8 months.
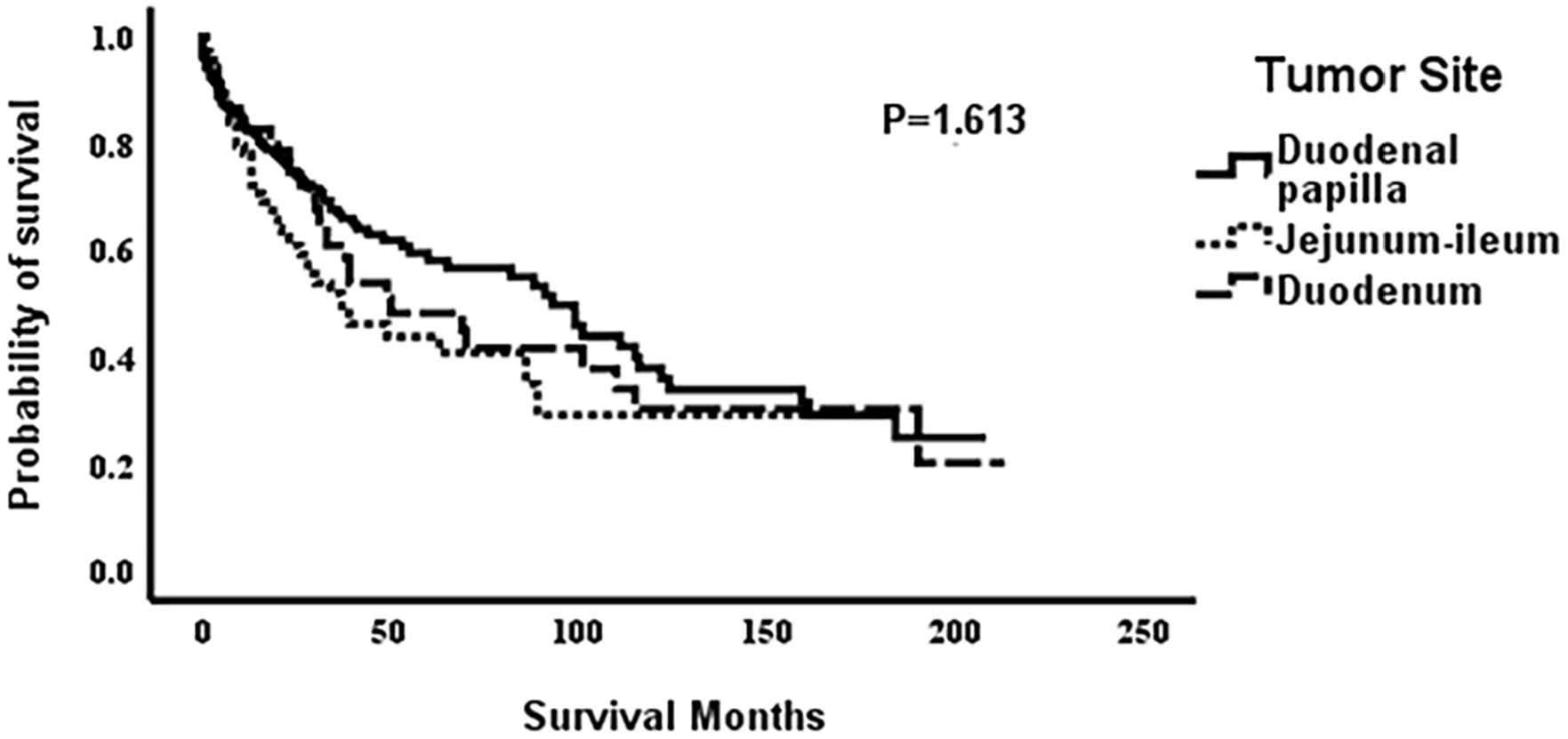
Kaplan–Meier curves of OS by tumor site. Median OS for adenocarcinoma was 93 months, duodenum adenocarcinoma 50 months, and jejunum–ileum adenocarcinoma was 37 months.
Table 2 shows prognostic factors in the primary tumor site (duodenum and jejunum–ileum) determined using single-variable analysis. In DA, distant metastasis, Stage IV disease, and nerve invasion were prognostic factors for OS. In jejunoileal adenocarcinoma, T4, lymph node positivity, distant metastasis, Stage IV, advanced age, and LMR (⩽2.31) were prognostic factors. Although not significant, CEA (⩾5 ng/mL) was associated with a poor prognosis in patients with jejunoileal adenocarcinoma. Multivariate analyses were conducted to identify independent prognostic factors for OS; the results of these analyses are shown in Table 3. Multivariate analysis showed that advanced age (hazard ratio [HR]: 7.33; 95% CI: 2.54-21.18; P = .00), high CEA (⩾5 ng/mL) (HR: 2.04; 95% CI: 1.19-3.49; P = .01), high AFP (⩾10 ng/mL) (HR: 2.28; 95% CI: 1.04-4.97; P = .04), low PA (⩽180 mg/L) (HR: 1.69; 95% CI: 1.00-2.87; P = .05), and advanced TNM stage (HR: 18.98; 95% CI: 4.84-74.37; P = .00) were independent prognostic factors for OS.
Single-variable analysis for OS in primary site of tumor (duodenum, jejunum–ileum).
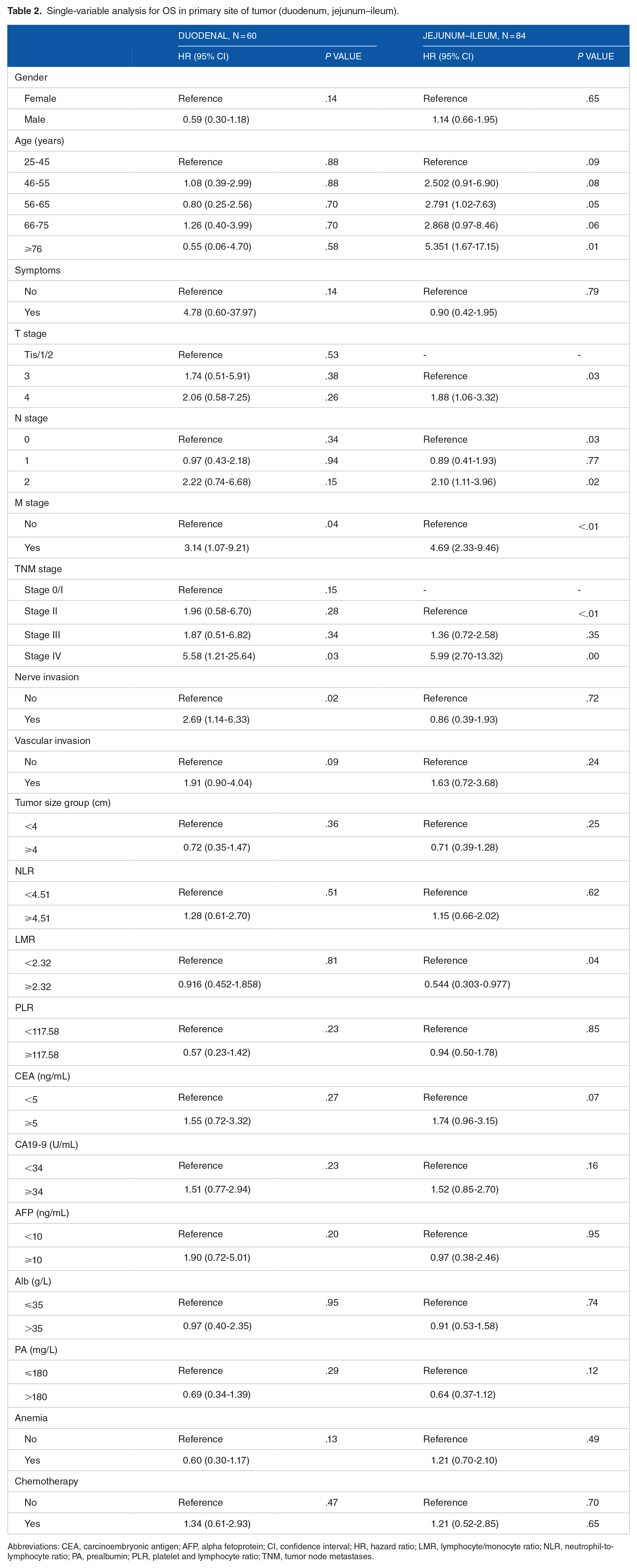
Abbreviations: CEA, carcinoembryonic antigen; AFP, alpha fetoprotein; CI, confidence interval; HR, hazard ratio; LMR, lymphocyte/monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PA, prealbumin; PLR, platelet and lymphocyte ratio; TNM, tumor node metastases.
Multivariate analysis for OS in non-ampullary SBA.

Abbreviations: CEA, carcinoembryonic antigen; AFP, alpha fetoprotein; NLR, neutrophil-to-lymphocyte ratio; PA, prealbumin; TNM, tumor node metastases.
Age (25-45, 46-55, 56-65, 66-75, and ⩾76 years) and gender (male and female) were adjusted.
Single-variable and multivariate analyses were performed to determine the independent prognostic factors for OS in duodenal papilla adenocarcinoma; the results of these analyses are shown in Table 4. TNM Stage III, nerve invasion, and CA19-9 (⩾34 U/mL) were associated with poor prognosis of duodenal papilla adenocarcinoma. Although not significant, NLR (⩾4.51) and PA (⩽180 mg/mL) were associated with a poor prognosis in duodenal papilla adenocarcinoma. Multivariate analysis showed that nerve invasion (HR: 3.12; 95% CI: 1.34-7.28; P = .01), low PLR (⩽117.58) (HR: 2.57; 95% CI: 1.27-5.21; P = .01), TNM Stage III (HR: 3.12; 95% CI: 1.34-7.28; P = .00), and high CA19-9 (⩾34 U/mL) (HR: 2.03; 95% CI: 1.23-3.34; P = .01) were independent prognostic factors for OS.
Single-variable and multivariate analysis for OS in duodenal papilla adenocarcinoma.

Abbreviations: CEA, carcinoembryonic antigen; AFP, alpha fetoprotein; CI, confidence interval; HR, hazard ratio; LMR, lymphocyte/monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; PA, prealbumin; PLR, platelet and lymphocyte ratio; TNM, tumor node metastases.
Age (25-45, 46-55, 56-65, 66-75, and ⩾76 years old) and gender (male and female) were adjusted.
Discussion
In this detailed examination of 300 patients with SBA, we used single-variable and multivariate analyses to identify independent prognostic factors for small intestinal adenocarcinoma and made some new discoveries.
The most common tumor site in primary SBA was the ampullary papilla, and 59.3% of the patients were male. This finding is similar to the results of previous studies.5(p301),19(p257)
Most patients with SBA are diagnosed after the appearance of symptoms; they are often in the stage of cancer progression by the time they are diagnosed. We found that the presence of symptomatic disease at diagnosis does not affect the prognosis of patients with SBA, although symptoms could guide early-stage diagnosis. Some studies reported that patients with jejunoileal adenocarcinoma are more likely to have a high disease stage than those with DA. 16 ,20(p1609) We found that the most common postoperative TNM stage of the PVA and DA patients was Stage II, while it was Stage III for patients with jejunoileal adenocarcinoma. The number of patients with Stage IV jejunoileal adenocarcinoma was much more than those with Stage IV PVA and DA. This could be due to the early clinical manifestations and tumor-related symptoms. The early diagnosis of PVA is owing to early biliary obstruction and subsequent persistent jaundice. Jejunoileal adenocarcinoma is easily confused with other digestive diseases due to abdominal pain, bleeding, and other nonspecific clinical manifestations, which delay the diagnosis. Gastroscopy is widely used in China to detect stomach lesions and to visualize the third part of the intestinal tract. This may also be a reason for the early diagnosis of DAs.
Previous studies showed that old age, tumor markers (high CEA and high CA19-9), tumor stage, pancreatic-positive removal edge, lymphatic metastasis positivity rate, distant metastasis, lymph vascular invasion, and the primary part of the duodenum were poor prognostic factors in patients with small intestinal adenocarcinoma.20(p1608),21(p397), 22 In this study, when analyzed separately, the duodenal papilla, non-ampullary duodena, and jejunum–ileum adenocarcinoma cohorts were associated with different clinical and pathological variables. By adjusted analysis, we determined that the prognostic factors independently correlated with PVA were nerve invasion, Stage III disease, low PLR, and high CA19-9. The independent prognostic factors for non-ampullary SBA included advanced age, advanced TNM stage, high CEA, high AFP, and low PA. Our analysis also showed that other variables, such as sex, tumor size, and chemotherapy, may not independently play a major role in prognosis.
The effect of the primary tumor location on survival is controversial. Several studies reported that the primary tumor location is not an important prognostic factor for small intestinal adenocarcinomas.21(p397),23(p800) Koo et al24(p3) reported that DA patients had shorter survival time and worse prognosis than patients with jejunoileal adenocarcinoma. Two large population-based cohort studies showed that DA is an independent prognostic factor of SBA.19(p257),21(p397) In this study, the median survival time of patients with jejunoileal adenocarcinoma was shorter than that of patients with DA and PVA, which may be influenced by patients with Stage IV adenocarcinoma. We rectified this and found that the primary tumor site in patients with Stages I to III SBA was not a significant prognostic factor. Previous studies also reported that Stage III DAs tend to worsen.20(p1609)
Systemic inflammation based on host–tumor interactions is currently considered a hallmark of cancer and is closely associated with the development and metastasis of various malignancies.25(p584),26(e493) Neutrophils inhibit the host response to cancer and promote tumor proliferation. 27 Tumor-associated macrophages (TAMs) derived from monocytes are involved in cancer progression. 28 Simultaneously, the decrease in lymphocytes is closely related to the poor prognosis of the tumor. Therefore, patients with tumors may show high NLR and LMR. Previous studies verified that the combination of NLR, PLR, LMR, and other inflammatory markers is an important prognostic factor for different cancers.29,30,31(p49) We found that a low LMR was an independent prognostic factor for jejunoileal adenocarcinoma, and high NLR and low PLR were associated with a poor prognosis of PVA.
This study has several limitations. First, this was a retrospective study; thus, there may be some confounding factors in the data. Second, our patients were from the Anhui Province; therefore, the influence of regional factors may affect the generalizability of our findings. Finally, this study had limitations inherent to all observational studies. For instance, the lack of patient comorbidities, low power, and other potential confounders might have influenced the results of several studies. Selection bias could exist because of insufficient follow-up time for some patients.
Conclusion
We found that TNM Stage III, nerve invasion, low PLR, and high CA19-9 were independent prognostic factors for PVA. The independent prognostic factors for non-ampullary SBA included advanced age, advanced TNM stage, high CEA, high AFP, and low PA levels.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Natural Science Foundation of Anhui Province under Grant 2008085MH294.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
X.W. collected and analyzed data and wrote the article. K.C. carried out the idea and design of the study. D-.C.L., H.L., and L.Z. assisted in the collection of data. Z-.G.W. coordinated and approved the final draft. All authors read and approve the final article.
Availability of Data and Materials
All data generated or analyzed during this study are included in this article.
Ethics Approval and Consent to Participate
The current study complied with the principles of the Declaration of Helsinki and was overseen by the Institutional Review Boards of the First Affiliated Hospital of Anhui Medical University, the Second Affiliated Hospital of Anhui Medical University, and Anhui Provincial Hospital (IDs: AYFY2021LL0091, P2021-13-11, and PJ2021-02-19, respectively). Approval date: 25-02-2021. The need for informed patient consent was waived owing to the retrospective nature of the study.
