Abstract
Introduction
The prognosis differences of gastrointestinal stromal tumors (GISTs) between the jejunum and ileum have rarely been studied. This study aimed to evaluate the long-term survival outcomes in patients with jejunal and ileal GISTs.
Methods
This population-based retrospective cohort study utilized data from the Surveillance, Epidemiology, and End Results (SEER) database from 2000 to 2019. Kaplan-Meier analyses and Cox proportional hazards models were employed to assess overall survival (OS) and cancer-specific survival (CSS). A 1:1 propensity score matching (PSM) approach was implemented to address confounding factors, and subgroup analyses were performed for various variables.
Results
A total of 1237 patients were analyzed, with 848 in the jejunum and 389 in the ileum. Ileal GISTs patients were older, exhibited higher T stages, larger tumor sizes, elevated mitotic rates, and were more susceptible to distant metastasis (P<0.05). After PSM, the jejunal GISTs group and the ileal GISTs group demonstrated comparable OS (hazard ratio [HR]: 1.25, 95% confidence interval [CI]: 0.97-1.60, P=0.082) and CSS (HR: 1.13, 95% CI: 0.81-1.59, P=0.461). Additionally, jejunal GISTs and ileal GISTs exhibited similar OS and CSS across various subgroups. Multivariate Cox regression analysis revealed age, TNM stage, and mitotic rate as independent risk factors influencing OS, whereas race, N stage, and M stage were identified as independent risk factors affecting CSS.
Conclusions
Jejunal GISTs and ileal GISTs have comparable OS and CSS, with the occurrence of lymph node metastasis and distant metastasis being important factors affecting their prognosis.
Introduction
Gastrointestinal stromal tumors (GISTs) are the most prevalent mesenchymal tumors of the digestive tract, with gastric stromal tumors accounting for approximately 60%-70% of all cases, followed by small intestinal stromal tumors, which make up about 30%.1,2 GISTs are distinguished by specific mutations in genes like KIT and PDGFRA, which are the driving force behind their growth and progression.3-5 Currently, surgery is the primary treatment method for GISTs.6-8 For patients with high-risk pathological assessments post-surgery, adjuvant chemotherapy with imatinib is necessary, and it has significantly enhanced the prognosis for GISTs patients.9,10 The location of the tumor plays a crucial role in determining the prognosis of GISTs patients.2,11 Previous research has indicated that extra gastrointestinal stromal tumors (EGISTs) have a poorer prognosis compared to GISTs, primarily due to their location outside the gastrointestinal tract, which makes early detection of symptoms like gastrointestinal bleeding less feasible. Consequently, EGISTs are often diagnosed at a later stage when the tumor is larger and has a higher mitotic index.12-14 Furthermore, previous studies have shown that gastric stromal tumors have a better prognosis than small intestinal stromal tumors, possibly because small intestinal stromal tumors have an insidious onset and lack specific clinical symptoms, posing significant challenges for diagnosis and treatment.15-17
However, the small intestine is subdivided into the jejunum and ileum, which exhibit notable differences in anatomical location, structural characteristics, and physiological functions.18,19 Consequently, it remains uncertain whether there are variations in the prognosis between jejunal stromal tumors and ileal stromal tumors. Therefore, in this study, we leveraged the population-based database from the Surveillance, Epidemiology, and End Results (SEER) to analyze and contrast the overall survival (OS) and cancer-specific survival (CSS) between patients with jejunal stromal tumors and ileal stromal tumors in the post-imatinib era. Our aim is to provide valuable insights for the development of treatment strategies and patient management for individuals with jejunal stromal tumors and ileal stromal tumors.
Materials and Methods
Data Sources
This population-based retrospective cohort study utilized data obtained from the SEER database using SEER*Stat 8.4.0, which is accessible at https://seer.cancer.gov/seerstat/. This database covers approximately 28% of the US population and provides extensive information on various cancer types. GISTs were identified by using the histological subtype code 8936/3, aligning with the International Classification of Diseases for Oncology, Third Edition (ICD-O-3), specifically targeting codes C17.1 for Jejunum and C17.2 for Ileum. A series of screening criteria were applied to the initial patient pool, with the specific details outlined in a flowchart presented in Figure 1. Cases lacking a definitive pathological histological diagnosis or survival data were excluded from the study. A wide range of variables were extracted, including age, race, gender, TNM stages, tumor size, surgical status, chemotherapy status (which, in the SEER database, includes both traditional cytotoxic chemotherapy and molecularly targeted therapies such as imatinib, sunitinib, and regorafenib; however, specific details regarding the type, dosage, or duration of these agents are not available), mitotic rate, cancer-specific mortality, overall health condition, and duration of follow-up. Participants were divided into two age groups: those under 60 (classified as the young group) and those over 60 (classified as the elderly group). In terms of race, participants were categorized as white, black, or other races, which encompassed American Indian, Alaska Native, and Asian/Pacific Islander. The cancer’s TNM stages were classified according to the 7th edition of the AJCC Cancer Staging Manual. The study used the “CS site-specific factor 6” variable to evaluate the mitotic rate. OS and CSS were determined from the date of GISTs diagnosis until death, cancer-related death, or the most recent follow-up date. As the SEER database comprises anonymized, publicly accessible information, this research did not directly engage with human subjects, and all patient details have been de-identified. Therefore, institutional review board (IRB) approval and informed consent were not applicable. This study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2024. Clinical trial number: not applicable. The reporting of this study was conducted following the Reporting of studies Conducted using Observational Routinely-collected health Data (RECORD) statement.
20
Patients were identified from the SEER database based on the defined histological and temporal inclusion criteria, representing a population-based sample rather than a consecutive, random, or selectively recruited cohort. Flow chart of the study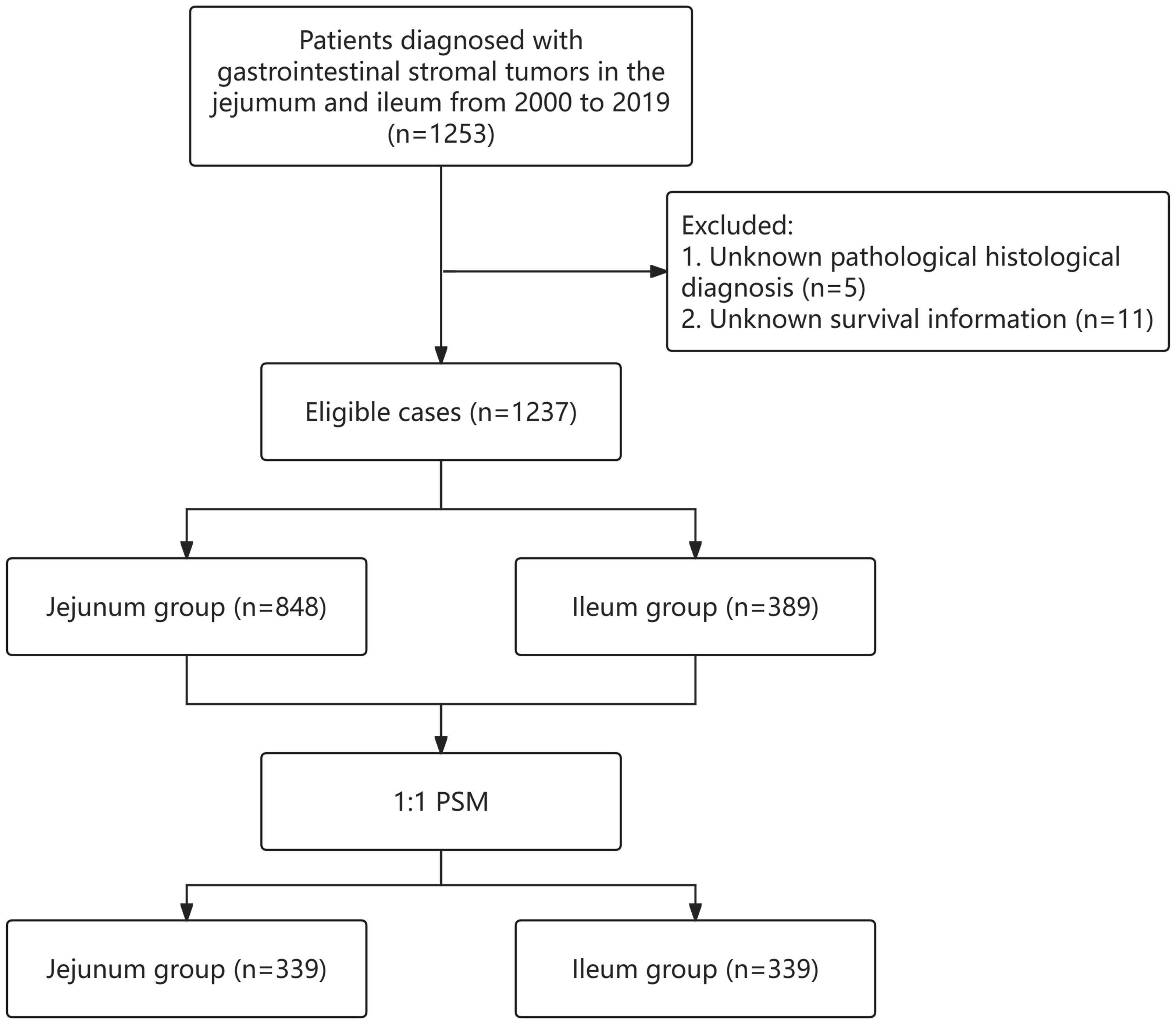
Statistical Analysis
Categorical data were presented using counts and percentages, and chi-square tests were employed for comparisons between groups. For continuous variables that did not follow a normal distribution, medians and interquartile ranges (IQR) were reported, with comparisons made using the Mann-Whitney U test. To ensure comparability between the jejunum and ileum groups, a 1:1 propensity score matching (PSM) technique was applied, incorporating factors such as age, race, sex, TNM stage, surgery status, tumor size, chemotherapy status, and mitotic rate, with a caliper width set at 0.01. The Kaplan-Meier method was utilized to assess OS and CSS, and survival curves were compared using the log-rank test. Cox proportional hazards models were used for both univariate and multivariate analyses, with hazard ratios (HRs) and their corresponding 95% confidence intervals (CIs) being calculated. Variables with a P-value less than 0.05 in the univariate Cox regression analysis were included in the multivariate analysis. To address missing data in the SEER database and enhance the statistical analysis, multiple imputation (MI) combined with the random forest method was employed. A total of 5 imputed datasets were generated to ensure stability and reproducibility of the estimates. Convergence of the imputation process was assessed by visually inspecting trace plots of imputed values across iterations, which demonstrated adequate convergence and stability. All statistical computations were performed using R software (version 4.1.0), with statistical significance set at a P-value of less than 0.05.
Results
Patient Demographics and Clinical Characteristics
Comparison of the Demographic and Clinical Characteristics of Patients Before and After PSM

PSM: propensity score matching; Others: American Indian, Alaska Native, Asian/Pacifc Islander; HPF: high power field; IQR: interquartile range; bold values indicate P < 0.05.
Survival Analysis of Patients With GISTs in the Jejunum and Ileum
Before PSM, Kaplan-Meier curves revealed that the ileum group had poorer OS (HR 1.33, 95% CI 1.10-1.61, P = 0.004) (Figure 2A) and CSS (HR 1.33, 95% CI 1.01-1.74, P = 0.040) (Figure 2B) compared to the jejunum group. However, after PSM, the ileum group and the jejunum group had comparable OS (HR 1.25, 95% CI 0.97-1.60, P = 0.082) (Figure 2C) and CSS (HR 1.13, 95% CI 0.81-1.59, P = 0.461) (Figure 2D). OS and CSS were compared between jejunum group and ileum group before and after PSM A. OS before PSM; B CSS before PSM; C OS after PSM; D CSS after PSM. Shaded areas represent 95% confidence intervals (CIs) for each survival curve. OS: overall survival; CSS: cancer-specific survival; PSM: propensity score matching
Subgroup Analyses of Patients With GISTs in the Jejunum and Ileum
After PSM, subgroup analysis showed that in most cases, the OS between the jejunum group and the ileum group was comparable (P > 0.05). However, when considering subgroups of patients older than 60 years, those at T2 stage, M1 stage, and with tumor sizes between 2.1-5.0 cm, the jejunum group exhibited better OS than the ileum group (P < 0.05). Yet, in terms of the P value for interaction, a statistically significant difference between the jejunum group and the ileum group was only observed in the M stage subgroup (Figure 3). When it came to the subgroup analysis for CSS, the jejunum group demonstrated better prognosis than the ileum group in the N1 stage and M1 stage subgroups (P < 0.05). For the remaining subgroups, the prognosis between the jejunum group and the ileum group was comparable (P > 0.05). As for the P value for interaction, a statistically significant difference between the two groups was only present in the M stage subgroup (Figure 4). Additionally, Supplementary Table 2 shows the demographic and clinical characteristics of jejunal GISTs and ileal GISTs in the M1 stage group. Subgroup analysis of overall survival between jejunum group and ileum group after PSM. PSM: propensity score matching Subgroup analysis of cancer-specific survival between jejunum group and ileum group after PSM. PSM: propensity score matching

Univariate and Multivariate Cox Regression
Univariate and Multivariate Cox Regression Analysis of Overall Survival in Patients With GISTs in the Jejunum and Ileum

GISTs: gastrointestinal stromal tumors; Others: American Indian, Alaska Native, Asian/Pacifc Islander; HPF: high power field; HR: hazard ratios; CI: confidence interval; bold values indicate P < 0.05.
Univariate and Multivariate Cox Regression Analysis of Cancer-Specific Survival in Patients With GISTs in the Jejunum and Ileum

GISTs: gastrointestinal stromal tumors; Others: American Indian, Alaska Native, Asian/Pacifc Islander; HPF: high power field; HR: hazard ratios; CI: confidence interval; bold values indicate P < 0.05.
Discussion
Previous studies have reported on the long-term prognosis of GISTs located in various sites, including the esophagus, stomach, small intestine, duodenum, rectum, and areas outside the gastrointestinal tract.12,21-26 However, it is important to note that the small intestine can be further subdivided into the jejunum and ileum based on anatomical distinctions. Currently, there is a notable lack of research examining the differences in prognosis between jejunal GISTs and ileal GISTs. To address this gap, this study utilized data from the SEER database to analyze the long-term prognosis of jejunal GISTs and ileal GISTs in the post-imatinib era, spanning from 2000 to 2019. To mitigate confounding factors, a 1:1 PSM approach was employed. The findings of this study indicate that jejunal GISTs and ileal GISTs have comparable OS and CSS rates (P > 0.05). Additionally, subgroup analyses based on variables such as age, race, gender, T stage, N stage, tumor size, surgical status, chemotherapy status, and mitotic rate also demonstrated comparable OS and CSS rates between jejunal GISTs and ileal GISTs (P > 0.05).
Our study encompassed a total of 1237 patients, revealing that the incidence of jejunal GISTs surpassed that of ileal GISTs, with a ratio approximating 2.2. Consistent with our findings, Feng et al. 27 conducted a study involving 702 patients and similarly demonstrated that the incidence of jejunal GISTs was higher than that of ileal GISTs, with a ratio of approximately 2.1. Another study, which included 906 patients with GISTs located in the jejunum and ileum, also confirmed that the incidence of jejunal GISTs was higher than that of ileal GISTs. 16 To date, Feng et al. 27 are the only researchers who have utilized data from the SEER database to compare the long-term prognosis of jejunal GISTs and ileal GISTs. However, their study was hindered by a substantial amount of missing data for key variables, including mitotic rate (71.4% missing), histological grade (66.7% missing), and lymph node metastasis (67.0% missing). Analyzing such incomplete data directly could potentially introduce selection bias and compromise statistical power. In contrast, our study had minimal missing data, and we employed MI techniques to preprocess the data, thereby enhancing the reliability of our analysis. Moreover, Feng et al's study was limited to analyzing disease-specific survival for jejunal GISTs and ileal GISTs, while our study conducted a more comprehensive analysis by examining both CSS and OS, providing a fuller understanding of the prognosis of jejunal GISTs and ileal GISTs. Since the introduction of imatinib in clinical practice in 2000, the prognosis of patients with GISTs has significantly improved.28,29 However, Feng et al’s study included data from the SEER database spanning from 1973 to 2013. Combining data from both before and after the imatinib era may compromise the accuracy of the results. Therefore, our study exclusively included data from the post-imatinib era (2000 to 2019), which bolstered the reliability of our findings. Furthermore, in Feng et al’s prognostic analysis, the baseline data were unbalanced, with the tumor size in the ileal GISTs group being significantly larger than that in the jejunal GISTs group (P<0.05), introducing potential bias in the direct comparison between the two groups. Previous studies have also highlighted that tumor size is a crucial factor influencing the prognosis of patients with GISTs.30-32 Consequently, in our study, we employed a 1:1 PSM method to ensure comparability between the jejunal GISTs group and the ileal GISTs group, addressing baseline data imbalances. Although both our study and Feng et al's study concluded that jejunal GISTs and ileal GISTs have comparable prognoses, our study’s conclusions are more comprehensive and reliable.
Furthermore, it is important to contextualize our findings within the evolving landscape of targeted therapy during the study period (2000–2019). While our analysis focused on the post-imatinib era, it is noteworthy that additional targeted agents, such as sunitinib (approved in 2006) and regorafenib (approved in 2013), became available for patients with advanced GISTs who were resistant or intolerant to imatinib.33,34 These subsequent-line therapies have been demonstrated to prolong progression-free survival and may have contributed to improved overall and cancer-specific survival in specific patient subsets. Although the SEER database lacks granular data on the specific sequence or type of targeted therapy administered, the availability of these multi-line treatment options likely influenced the long-term outcomes observed in our study. This broader therapeutic context may partially explain the comparable OS and CSS between jejunal and ileal GISTs after PSM, as patients with disease progression had access to effective subsequent treatments. Future studies incorporating detailed treatment histories are warranted to further elucidate how the timing and selection of targeted therapies impact site-specific prognosis in GISTs.
In this study, a total of 213 patients (17.2%) developed distant metastasis. Following PSM analysis, 57 cases in the jejunal group and 61 cases in the ileal group were identified with distant metastasis. Across various subgroups, jejunal GISTs and ileal GISTs demonstrated similar OS and CSS, except within the M1 subgroup, where jejunal GISTs exhibited a better OS and CSS compared to ileal GISTs. We postulate that the prognosis of ileal GISTs with distant metastasis may be less favorable than that of jejunal GISTs, potentially due to underlying differences in mutation types and expression patterns between jejunal and ileal GISTs. These disparities could result in a relatively weaker adaptability of jejunal GISTs to the metastatic environment, slower growth and diffusion rates, ultimately leading to a more favorable prognosis. Moreover, patients with jejunal GISTs may exhibit a more positive response to treatments such as chemotherapy and targeted therapy, which can more effectively control tumor growth and diffusion, thereby enhancing the prognosis. In addition to these potential biological and treatment-related factors, the anatomical and physiological distinctions between the jejunum and ileum may also contribute to the prognostic differences observed in these high-risk subgroups.18,19 Variations in lymphatic drainage patterns, vascularization, and the local immune microenvironment could influence the patterns of tumor progression and metastatic efficiency. Furthermore, the biological heterogeneity, such as differences in the frequency and type of KIT/PDGFRA mutations between jejunal and ileal GISTs, may lead to variable tumor aggressiveness and differential responses to targeted therapy.4,5 These combined factors could collectively explain why jejunal GISTs exhibit relatively better outcomes in specific subgroups like T2, N1, and M1. Furthermore, our observations revealed that the mitotic rate in the jejunal GISTs group was significantly lower than that in the ileal GISTs group among patients with distant metastasis (P=0.022) (Supplementary Table 2). Notably, the mitotic rate is a pivotal factor influencing the prognosis of GISTs patients, and previous research has indicated that GISTs patients with a high mitotic rate are prone to a poorer prognosis.35-37 It is important to note that the number of cases with distant metastasis in this study was relatively small, and the analysis based on this limited sample size might have influenced the results. Therefore, further validation with a larger sample size is warranted in future studies.
Several limitations of our study ought to be acknowledged. Firstly, our retrospective analysis relies on the SEER database, which inherently bears the risks of data inconsistencies and biases. Secondly, while the SEER database provides a large, population-based sample with racial diversity representative of the United States, it is crucial to recognize that these findings are primarily derived from the American medical context. Therefore, caution should be exercised when extrapolating our study’s conclusions to non-U.S. or non-Western cohorts. We acknowledge that external validation using independent, non-U.S. datasets (such as multicenter registries from Asia or Europe) would significantly strengthen the evidence. We consider this an important direction for future international collaborative research, as such comprehensive validation is beyond the scope of the current study. Thirdly, the identification of tumor location using ICD-O-3 topography codes (C17.1 for jejunum and C17.2 for ileum) may be subject to potential misclassification. This could arise from variations in coding practices, inaccuracies in pathological reports, or ambiguity in determining the exact anatomical origin of tumors located near the junction of the jejunum and ileum. Such misclassification, if present, could introduce bias and affect the robustness of our comparative analysis between jejunal and ileal GISTs. Fourthly, our study lacks numerous patient-specific characteristics, including detailed treatment information such as surgical techniques and chemotherapy regimens, postoperative complications, and comprehensive follow-up data. Specifically, the SEER database does not provide granular data on surgical methods (e.g., laparoscopic versus open resection, or local excision versus radical resection), which are recognized as important variables influencing postoperative outcomes and long-term prognosis in GIST patients.6,8 The absence of this information prevented us from performing subgroup analyses based on surgical approach, which could have further refined the preoperative risk factor system. Future studies incorporating detailed surgical records from institutional databases or prospective cohorts are warranted to address this important clinical question. Additionally, the SEER database does not contain information on KIT/PDGFRA mutation status, which is a known critical molecular marker influencing GIST prognosis and response to targeted therapy.4,5 The absence of this genetic data prevented us from performing subgroup analyses based on mutation type, which could have provided further insights into the prognostic differences between jejunal and ileal GISTs. Future studies incorporating detailed molecular and genetic information are warranted to validate and extend our findings.
Conclusion
Our analysis of the long-term OS and CSS of patients with jejunal and ileal GISTs in the post-imatinib era, utilizing the SEER database, revealed comparable survival outcomes between the two groups. Furthermore, we identified lymph node metastasis and distant metastasis as common risk factors that significantly impact the OS and CSS of patients with jejunal and ileal GISTs. Hence, early detection, accurate diagnosis, and prompt treatment are crucial for improving the prognosis of these patients.
Supplemental Material
Supplemental material -Long-term Prognosis Analysis of Gastrointestinal Stromal Tumors in the Jejunum and Ileum: A SEER-Based Study
Supplemental material for Long-term Prognosis Analysis of Gastrointestinal Stromal Tumors in the Jejunum and Ileum: A SEER-Based Study by Xiaochun Lu, MD, Dong Liu, MD, Shimeng Zhang, Shuaiyang Zhou, Rencheng Cai, Zhenguo Qiao, and Tinghua Cao in Cancer Control.
Footnotes
Ethical Considerations
This study utilized de-identified data from the publicly available SEER database. As the research did not involve direct interaction with human subjects or the use of identifiable private information, ethical approval and informed consent were not required. The study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments.
Author Contributions
Conception and design: Tinghua Cao and Rencheng Cai. Administrative support: Zhenguo Qiao. Provision of study materials or patients: Xiaochun Lu. Collection and assembly of data: Dong Liu. Data analysis and interpretation: Shimeng Zhang, Shuaiyang Zhou. Manuscript writing: All authors. Final approval of manuscript: All authors.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Development Program of Suzhou (SYWD2024077, SYWD2024322) and the Development Fund of Xuzhou Medical University Affiliated Hospital (XYFM202426, XYFZ202404). There was no additional external funding received for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Supplemental Material
Supplemental material for this article is available online.
