Abstract
Background
Antiangiogenic therapy had been tested in urothelial cancer (UC) without reaching the clinic.
Objective
We provide a systematic review and meta-analysis of trials to assess efficacy of immune checkpoint inhibitors (ICI) combined with antiangiogenic agents in UC.
Methods
Following PRISMA guidelines, we searched for trials with at least one arm of patients with UC treated with ICI plus antiangiogenics. Data were analyzed with the “meta” package from R using a one-staged frequentist meta-analysis.
Results
After screening 13,708 titles and abstracts, 9 studies were selected for analysis with 14 identified cohorts comprising 621 patients: 448 were ICI-naïve (ICI-N) and 173 were ICI-exposed (ICI-E). The estimated objective response rate (ORR) in all patients was 27% (21–35). In the ICI-N group, ORR was 34% (28–41). Conversely, the ICI-E group had a lower ORR of 16% (9–28). This difference was mainly driven by a higher partial response rate of 27% (23–31) in ICI-N group compared to 13% (8–20) in the ICI-E group. Disease control rate was 72% (66–77) ICI-N group vs. 71% (64–78) in ICI-E group. Median overall survival ranged from 6.4 to 24.9 months in the ICI-N group, and 8.2 to 10.4 months in ICI-E group. Median progression free survival ranged from 1.9 to 10.1 months and from 3 to 3.9 months in both groups, respectively.
Conclusion
ORR with ICI plus antiangiogenics was lower after prior ICI exposure, with substantial variability estimates among included trials, either due to differences among antiangiogenic agents used or trial-related factors. Future exploration of ICI combined with antiangiogenics in UC, especially in ICI-refractory setting, will benefit from better patient selection.
Keywords
Background
Angiogenesis is a key event in tumoral growth and progression in urothelial carcinoma (UC).1,2 Among many studied pro-angiogenic factors, the vascular endothelial growth factor (VEGF) pathway was strongly incriminated in UC angiogenesis based on both pre-clinical and clinical evidence. 3 Notably, VEGF overexpression in tissue and high serum levels were both associated with disease aggressivity, higher relapse and poor prognosis in UC.4–6
Pivotal research by McConkey and colleagues revealed that gene expression patterns in UC could forecast patient outcomes following treatment with a potent chemotherapy regimen (dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin [MVAC]) combined with the anti-VEGF-A monoclonal antibody, bevacizumab. 7 They identified that patients with the basal subtype of the disease had a higher survival rate, while those with a p53-like subtype were more likely to develop bone metastases and exhibit resistance to chemotherapy. A subsequent phase III study by Rosenberg et al. assessed the benefits of combining gemcitabine, cisplatin, and bevacizumab versus using gemcitabine and cisplatin alone, finding no significant improvement in overall survival with the addition of the antiangiogenic therapy to chemotherapy. 8 While bevacizumab was the first antiangiogenic agent approved by the Food and Drug Administration (FDA) for treatment of solid tumors, several antiangiogenic agents have been subsequently approved in genitourinary malignancies, including tyrosine kinase inhibitors (TKIs), namely: sunitinib, sorafenib, pazopanib, axitinib, lenvatinib and cabozantinib.9,10
Currently, the frontline of treatment of locally advanced or metastatic UC (LA/mUC) has shifted towards the early utilization of immunotherapy combinations, yielding higher response rates, better survival outcomes and more options for clinicians and patients to personalize treatment.11–13 However, primary or acquired resistance still emerge and limit the benefit of these treatments. The relationship between VEGF expression and the effectiveness of immune checkpoint inhibitors (ICIs) has been also evaluated in different other cancer models. VEGF was found to enhance co-expression of inhibitory receptors involved in CD8+ T cell exhaustion in a colon cancer mouse model, and targeting VEGF led to decreased expression of these inhibitory receptors. 14 Moreover, studies in melanoma indicate an association between lower VEGF-A levels and positive responses to anti–Programmed Death 1 (anti-PD-1) therapies, hinting at anti-angiogenesis as a mechanism of therapeutic resistance. 15 Several clinical trials in LA/mUC have hypothesized that targeting VEGF in combination with ICIs may enhance antitumor responses. However, a comprehensive synthesis of their data has not been done yet. Therefore, we conducted a systematic review and meta-analysis to consolidate data on the treatment outcomes of combining ICIs with antiangiogenic agents, aiming to bridge these critical knowledge gaps.
Materials and methods
Data sources and search strategies
We performed a systematic search in Ovid MEDLINE, Ovid Embase, Clarivate Web of Science and Wiley Cochrane Library from the inception of the databases to January 28, 2024. Search structures, subject headings, and keywords were tailored to each database by a medical research librarian (YG). The following concepts were searched using subject headings and keywords as needed, “cancer”, “neoplasm”, “immunotherapy”, “checkpoint inhibitor”, “cytotoxic t- lymphocyte-associated antigen 4”, “programmed cell death 1”, “programmed cell death ligand 1”, “vascular endothelial growth factor”, “VEGF inhibitor”, “vascular endothelial growth factor receptor”, “anti-vascular”, “anti-VEGF”, “anti-angiogenic”, “angiogenesis inhibitor”, “ipilimumab”, “tremelimumab”, “pembrolizumab”, “nivolumab”, “spartalizumab”, “cetrelimab”, “atezolizumab”, “durvalumab”, “avelumab”, “cemiplimab”, “monalizumab”, “aflibercept”, “bevacizumab”, “ranibizumab”, “brolucizumab”, “conbercept”, “pazopanib”, “sunitinib”, “sorafenib”, “regorafenib”, “cabozatinib”, “lenvatinib”, “ponatinib”, “axitinib”, “tivozanib”, “ramucirumab”, “vandetanib”, and “sitravatinib”. The search terms were combined by “or” if they represented the similar concept, and by “and” if they represented different concepts. Database search strategies are detailed in the Supplementary Tables S1–S4.
Eligibility criteria
For our review, we set specific criteria for selecting studies. These included clinical trials ranging from phase 1 to phase 3, published in English, involving adult patients with UC treated with both antiangiogenic agents and ICI. We ruled out studies that were not original or failed to report the outcomes and trials in progress. Additionally, retrospective studies were not considered to maintain the accuracy and dependability of our data. However, abstracts that aligned with our inclusion criteria were included in the analysis even if the full manuscript was not available.
Study selection
Two reviewers (IK and EP) independently conducted the study selection process. They reviewed titles and abstracts to determine eligibility based on pre-established inclusion and exclusion criteria. They also assessed the full texts of pertinent references. In instances of disagreement, a third reviewer, OA, was consulted to reach a consensus.
Data extraction
Two independent reviewers (MJM and IK) utilized Microsoft Excel for data extraction and annotation. If there were any differences in their findings, two additional reviewers, OA and RB, were brought in to reconcile them. The data compiled from each study included study and participant characteristics, details of the intervention, and outcomes of interest. This encompassed the total number of participants and the occurrence of events in each group.
Outcomes of interest
The outcomes of interest included complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD). Overall response rate (ORR) was defined as the proportion of patients with CR or PR, while disease control rate (DCR) covered CR, PR and SD.
Data analysis
A pooled logit transformed proportion of events across the included studies using the DerSimonian-Laird (DL) random-effects model in a one-staged frequentist model of meta-analysis. A random effects model was chosen a priori due to the expectation that treatment effect would vary between trials using different anti-angiogenic agents in combination with ICI. The I2 and Tau2 were used as a measure of total variability due to between-study heterogeneity. The p-value for Cohran's Q was used to test the null hypothesis of no between-study heterogeneity. We conducted prior-specified subgroup analyses based on ICI exposure status and defined two subgroups: ICI-naïve (ICI-N) and ICI-exposed (ICI-E). Statistical analyses were performed using the RStudio graphical interface v.2024.04.2 + 764 for R software environment v.4.4.1 (http://www.r-project.org) with Meta package.
Ethics statement
This study was exempt from Institutional Review Board review as it involved the analysis of existing publicly available data.
PRISMA statement
This study was conducted in accordance with the PRISMA guidelines (Preferred Reporting Items for Systematic Reviews and Meta-Analyses). The PRISMA checklist was used to guide the study and is available upon request (
Results
Study characteristics
A total of 16,267 potential titles and abstracts were identified through the electronic search strategy, with 32 duplicates removed internally and 2527 duplicates removed through the assistance of a medical research librarian (YG). The remaining 13,708 studies underwent primary screening, and 731 full-text articles or abstracts were evaluated for eligibility (Figure 1). After secondary screening,
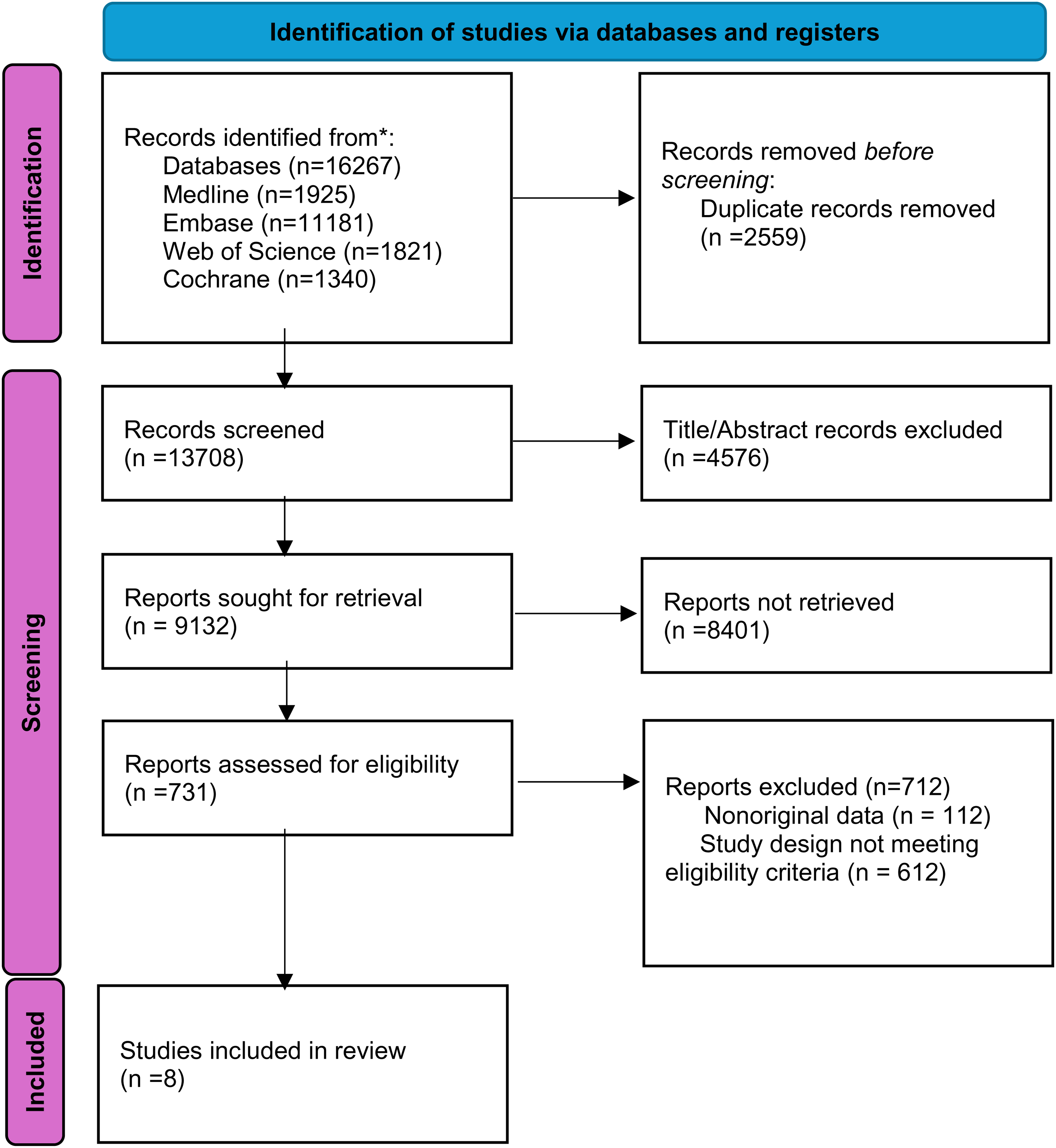
Flowchart demonstrating the process of study selection.
Description of enrolled studies involving antiangiogenic therapy combined with immune checkpoint inhibitors (AA + ICI). Key: N/A: Not assessed; ASCO: American Society of Clinical Oncology; GU: Genitourinary; LA/mUC: Locally advanced or metastatic urothelial cancer; CTx: Chemotherapy; ICI: Immune checkpoint inhibitor; ADC: Antibody drug conjugate; tx: treatment; MIUC: Muscle-invasive urothelial cancer; neoCTx: Neoadjuvant chemotherapy, PD-1: Programmed Death 1; PD-L1: Programmed Death Ligand 1, ICI-E: Immune checkpoint inhibitor-exposed, ICI-N: Immune checkpoint inhibitor-naïve.
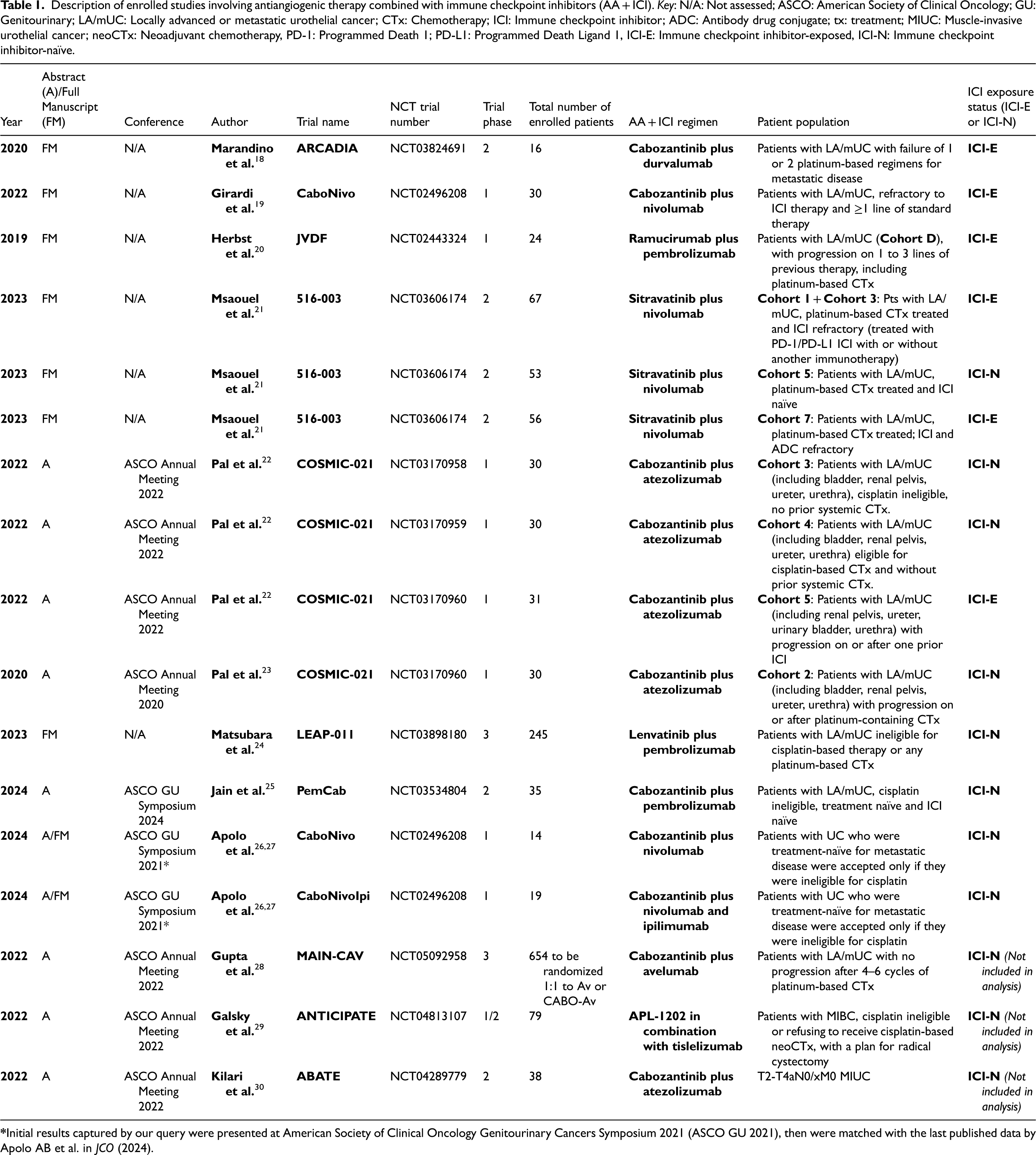
In our analysis, the final
In contrast, the ABATE and ANTICIPATE studies, which focused on the localized setting of muscle-invasive bladder cancer, lacked reportable outcomes suitable for inclusion in our meta-analysis.29,30 Results from MAIN-CAV, a randomized controlled trial (RCT) comparing cabozantinib with avelumab to avelumab alone in the metastatic setting, are pending. 28 Further details on main characteristics of the studies are found in Table S6 in the appendix.
Outcome analysis
Radiographic response analysis
In this one-staged frequentist meta-analysis, the estimated overall response rate (ORR) in all patients was 27% [95% CI: 21–35] (Figure 2). The ORR in ICI-N group was 34% [28–41] with low heterogeneity, while it was lower in ICI-E group with substantial heterogeneity: 16% [9–28]. This difference was mainly driven by a higher PR rate of 27% [23–31] observed in the ICI-N group compared to that of the ICI-E group, 13% [8–20] (Figure 3), with the same patterns of heterogeneity. However, different CR rates were estimated at 9% [6–15] and 5% [2–10] in each group, respectively (Figure 3).
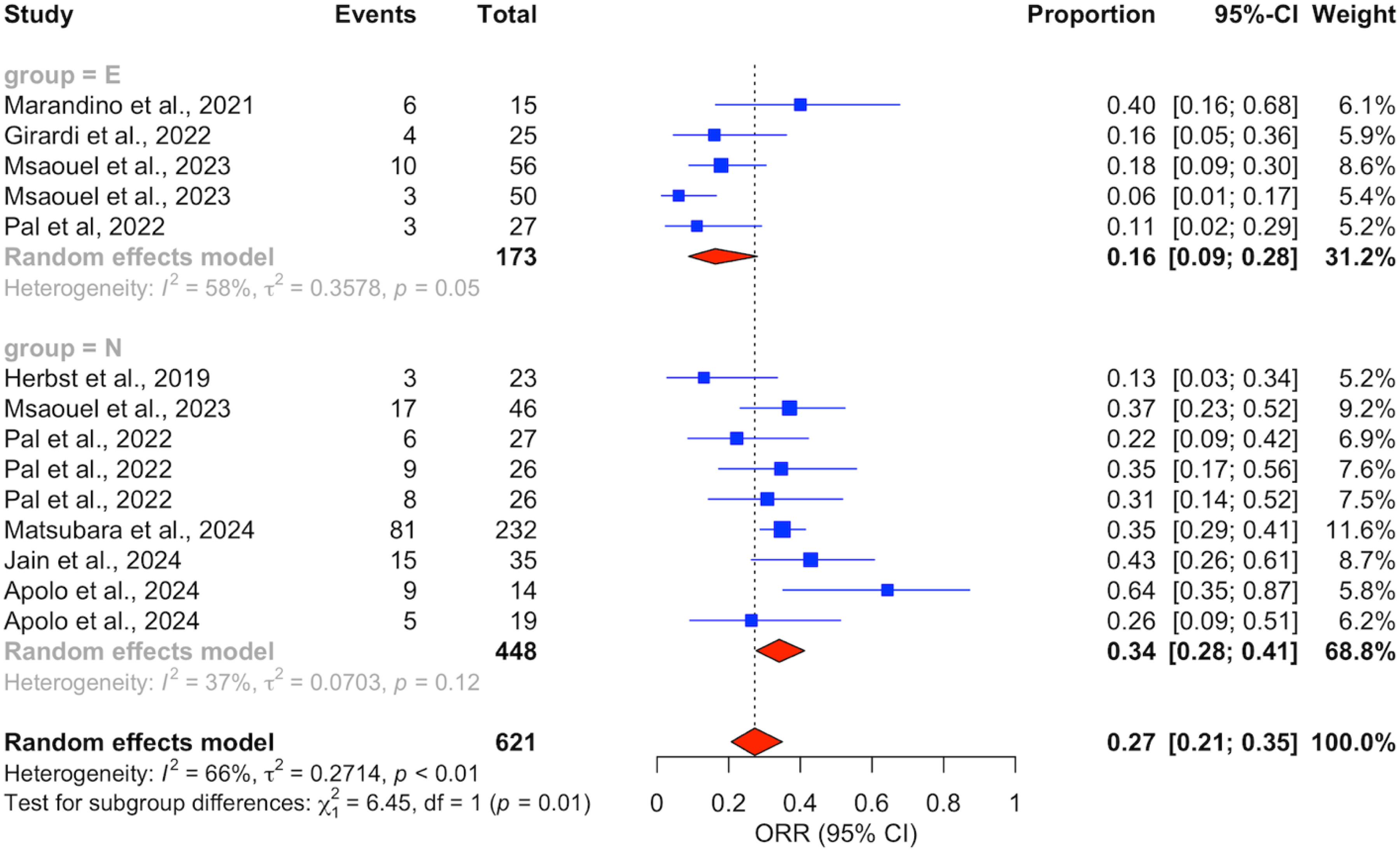
Forest plot comparing overall response rates (ORR) between ICI-exposed (ICI-E, ‘E’ in Figure) and ICI-naïve (ICI-N, ‘N’ in Figure) patients. Key: DL: DerSimonian and Laird.
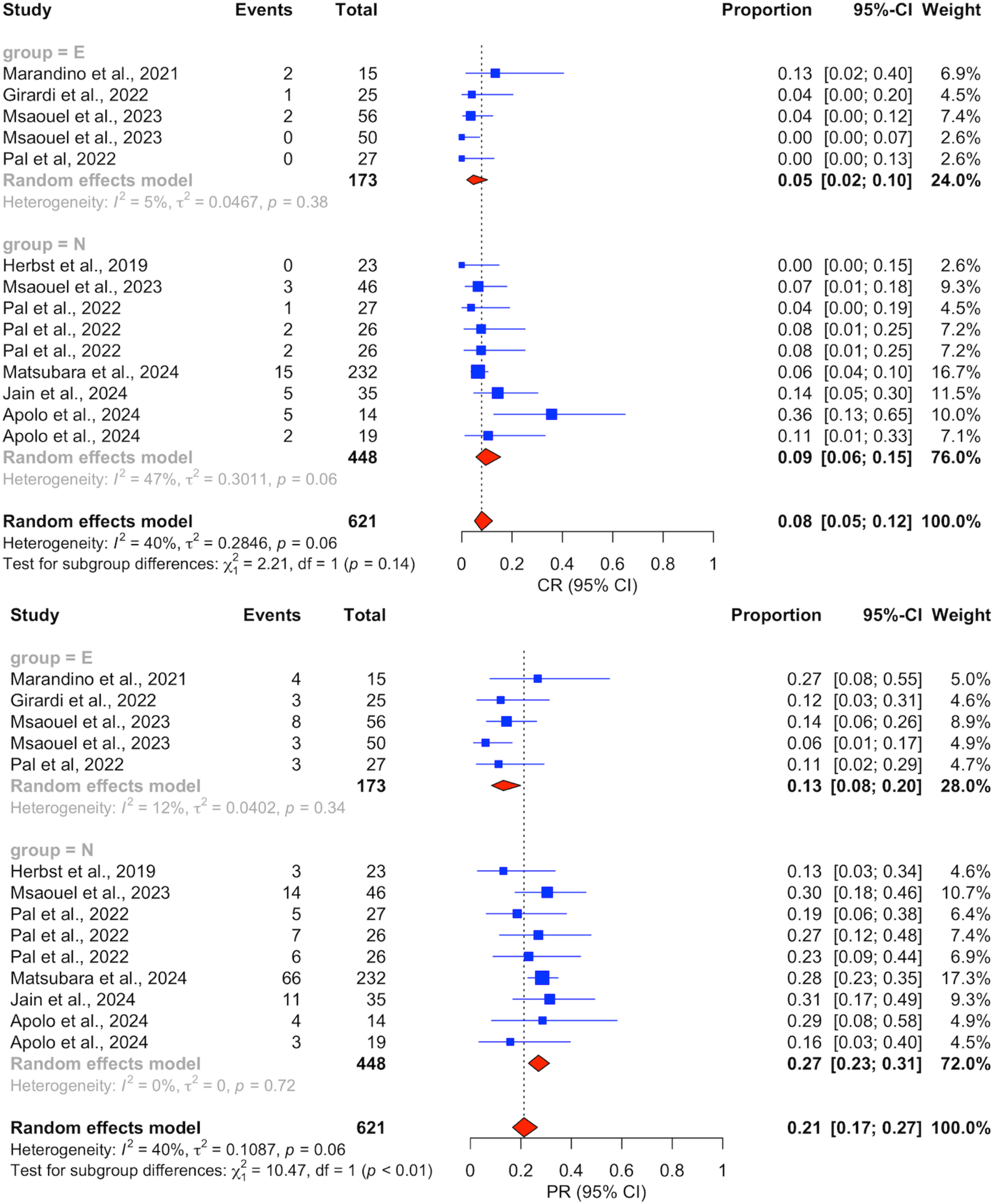
Forest plot comparing complete response (CR) and partial response (PR) rates between ICI-exposed (ICI-E, ‘E’ in figure) and ICI-naïve (ICI-N, ‘N’ in figure) patients. Key: DL: DerSimonian and Laird.
DCR estimates were not much different between both groups: 72% [66–77] in ICI-N vs. 71% [64–78] in ICI-E (Supplementary Figure 1). SD rates were estimated at 39% [32–46] for ICI-N patients versus 56% [47–65] for ICI-E patients. PD rates were 23% [16–33] versus 29% [22–36], respectively for non-exposed and exposed patients. A breakdown of the response outcomes of each study is displayed in Table S7 in the appendix.
Survival outcome analysis
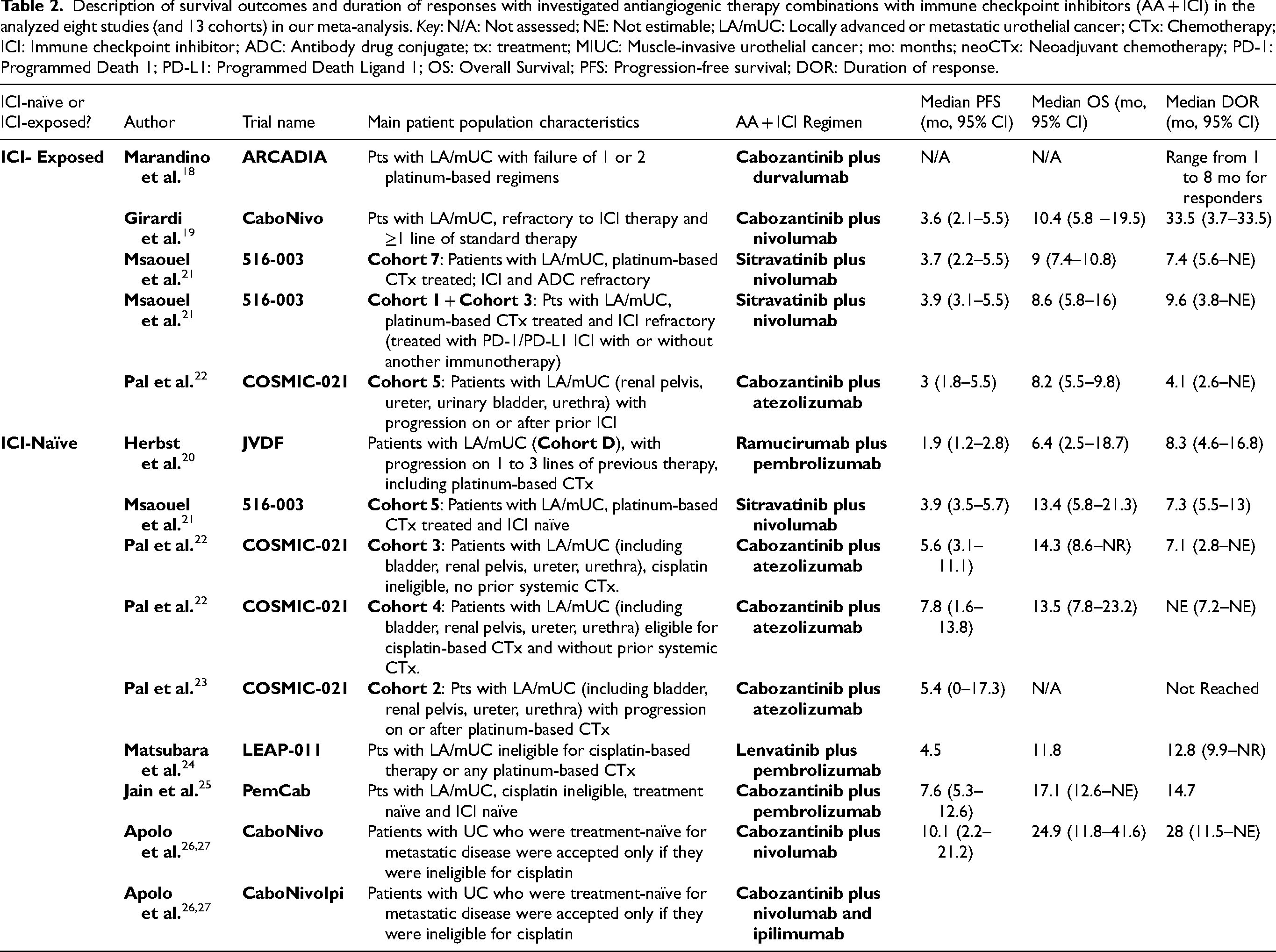
Data on length of outcomes, like overall survival (OS), progression-free survival (PFS), and duration of response (DoR) could not be pooled due to insufficient individual data points from every study and the lack of randomization in most studies. Median OS ranged from 6.4 (JVDF) to 24.9 months (CaboNivo/CaboNivoIpi) in the ICI-N group, and 8.2 (COSMIC-021) to 10.4 months (CaboNivo by Girardi et al.) in ICI-E group. Median PFS ranged from 1.9 (JVDF study) to 10.1 months (CaboNivo/CaboNivoIpi by Apolo et al. [2024]) in the ICI-N group and from 3 (COSMIC-021) to 3.9 months (516-003) in ICI-E group. A detailed information about survival outcomes of each study is displayed in Table 2.
Description of survival outcomes and duration of responses with investigated antiangiogenic therapy combinations with immune checkpoint inhibitors (AA + ICI) in the analyzed eight studies (and 13 cohorts) in our meta-analysis. Key: N/A: Not assessed; NE: Not estimable; LA/mUC: Locally advanced or metastatic urothelial cancer; CTx: Chemotherapy; ICI: Immune checkpoint inhibitor; ADC: Antibody drug conjugate; tx: treatment; MIUC: Muscle-invasive urothelial cancer; mo: months; neoCTx: Neoadjuvant chemotherapy; PD-1: Programmed Death 1; PD-L1: Programmed Death Ligand 1; OS: Overall Survival; PFS: Progression-free survival; DOR: Duration of response.
Studies without reported results
Two studies (ABATE and ANTICIPATE) are evaluating antiangiogenic/ICI combinations in the localized disease setting.29,30 The forthcoming results of the Phase 1/2 ANTICIPATE trial will shed light on the effectiveness of a novel oral antiangiogenic agent, APL-1202 (nitroxoline), a methionine aminopeptidase 2 (MetAP2) inhibitor, in combination with tislelizumab, a more recently developed anti-PD-1 agent, compared to tislelizumab alone. 29 The target population consists of cisplatin-ineligible patients with muscle-invasive bladder cancer or those refusing standard neoadjuvant chemotherapy. Primary endpoints include overall survival and PFS, while secondary endpoints include response rates, safety and Quality of Life (QoL) metrics. Moreover, ABATE is a single-arm study assessing efficacy and safety of cabozantinib with atezolizumab as neoadjuvant therapy, with a primary endpoint of evaluating pathological downstaging defined as absence of residual muscle invasion in the surgical specimen (<pT2). 30
Important knowledge in the metastatic setting will also be generated from the Phase 3 RCT, MAIN-CAV, interested in the possible synergistic effect of the combination of cabozantinib with avelumab [anti-PD-1] compared to maintenance avelumab alone, after disease stability on 4–6 cycles of platinum-based chemotherapy. 28 Its original insights consist of few cross-resistance mechanisms and few overlapping toxicity profiles between the two agents, aiming to better contextualize the contribution of this multityrosine kinase inhibitor to a commonly used ICI in mUC. Other comparative details about the characteristics of these trials figure in Table S6.
Discussion
This systematic review synthesizes findings from 9 studies, encompassing 621 patients, to evaluate the effectiveness of combining ICIs with antiangiogenic agents in mUC treatment. It represents the inaugural systematic review and meta-analysis to probe objectively defined radiographic efficacy of this combination, with a keen emphasis on the comparative outcomes between patients newly introduced to ICI treatment and those with prior ICI exposure. Historically, studies of earlier antiangiogenic agents, up to 2014, studying single-agent sunitinib and pazopanib (PLUTO trial), in first or second-line therapies in mUC, were challenged by suboptimal clinical outcomes, low accrual and toxicity (sunitinib: ORR 3–6%, mPFS of 2.3 months; pazopanib: mPFS of 3.1 mo and mOS of 4.7 mo with pazopanib).31,32 However, the interest was sparked again in 2016 after the first introduction of cabozantinib, a multi-TKI (VEGFR, AXL, MET and RET inhibitor), which showed promising single agent activity with 8 mUC patients showing response out of 49 enrolled. 33 Nonetheless, erdafitinib, a fibroblast growth factor receptor (FGFR) inhibitor, remains the only TKI reaching clinical use in mUC, underscoring the need to explore additional targeted therapies in the field. 13
In the ICI-E group, we noted considerable variability in response outcomes, including ORR and PR. This variation could be attributed to mixed responses to subsequent immunotherapy observed in the ICI exposure setting. 34 Preclinical studies in an ICI-resistant small cell lung cancer mouse model have shown that the subsequent administration of antiangiogenic agents in conjunction with ICI can counteract T-cell exhaustion, a pivotal factor in ICI resistance. 35 Furthermore, the observed variability in response may also be influenced by the inherent varying immunomodulatory properties of different antiangiogenic agents. 36
The response rates among ICI-N patients displayed less variability. Interestingly, the modest improvement in ORR and, most importantly, PR rates, among ICI-N patients hints at the potential benefits of initiating this combination therapy earlier in the treatment course.37,38 This approach leverages the immunomodulatory effect of antiangiogenics to counteract the immunosuppressive impact of VEGF on both innate and adaptive antineoplastic immune responses. 33 Moreover, the involvement of hepatocyte growth factor (HGF)/cMET pathway in epithelial-mesenchymal transition and progression in bladder cancer gives a rationale for targeting this pathway with the potent multi-TKI, cabozantinib. 39 However, it remains unknown whether these responses rates are mainly driven by the ICI component. Previous clinical data from KEYNOTE-045 and KEYNOTE-052 suggest that single-agent pembrolizumab can confer good anti-tumor activity.40,41 The ORRs with pembrolizumab in both studies, respectively, were 21% for patients with mUC treated with platinum-based chemotherapy and 29% for platinum-ineligible, treatment-naïve patients. RCTs in the setting are useful to dissect the individual contribution of each of the two components in the antiangiogenic/ICI combination.
Due to the lack of data availability and randomization in treatment arms in most studies, as well as the absence of studies directly comparing survival outcomes between ICI-E and ICI-N, we were unable to conduct a formal analysis of survival rates (OS, PFS, and DoR). This limitation is further compounded by the extensive pre-treatment in the ICI-E group, significantly affecting outcomes compared to the ICI-N group, especially considering the more resistant disease state and compromised overall health status of ICI-E group. Future analyses should stratify outcomes based on the number and type of prior therapies received for a more balanced comparison. Finally, sub-analyses of key survival and oncological outcomes, such as primary UC location (upper vs. lower urinary tract), metastatic burden and patient performance status, were not possible due to paucity of data.
The particular interest in cabozantinib can be traced back to its earliest sign of clinical activity as a single agent in heavily pretreated, platinum-refractory cohort with mUC (n = 49) (including 1 CR and 7 PR). 33 Later, combinations of cabozantinib with single agent and dual agent ICIs were thoroughly studied in the expansion cohorts by Apolo AB et al., presented first at the American Society of Clinical Oncology Genitourinary Cancers (ASCO GU) Symposium 2021, and published more recently in the Journal of Clinical Oncology (2024).26,27 These expansion cohorts were triggered by considerable ORRs observed with these combinations in the phase 1 trial (n = 54). 42 In the expansion cohorts, the ORR was 42.1% in platinum-exposed LA/mUC, compared to 16% in the ICI-refractory setting (Girardi et al.) (n = 30). 19
Limitations of our analysis include the fact that it involved only 9 peer-reviewed publications, with most of these entries available only as abstracts. This limited availability of full texts complicates the assessment of bias risk. Furthermore, the ICI-E group had heterogeneity in the number of prior lines of therapy. Additionally, our analysis did not find trials comparing the efficacy of ICI + antiangiogenic combination therapy to single-agent antiangiogenic therapy, which limits the attribution of benefit. This suggests the need for higher-evidence level trials to fully elucidate the individual contribution of the two components (ICIs and antiangiogenics agents) to the therapeutic value in LA/mUC.
Our findings support for higher-evidence level trials to fully elucidate the individual contribution of the two components (ICI and antiangiogenic agents) in the treatment of patients with UC.37,38 The forthcoming results of the phase 3 MAIN-CAV clinical trial, examining the efficacy and safety of cabozantinib with avelumab versus avelumab alone, will be essential to understand the true contribution of a multi-targeted TKI antiangiogenic agent like cabozantinib in mUC after disease stability or response to platinum-based chemotherapy. 28 The study's primary endpoint is OS, with key secondary endpoints including PFS, safety, tolerability, and efficacy of cabozantinib plus avelumab compared to avelumab alone, assessed using RECIST 1.1, iRECIST criteria, and PD-L1 status. Its target completion date is estimated to be in December 2024. 43 Looking ahead, there is a need for more precise patient selection when exploring antiangiogenics combined with ICIs in UC. Implementing an immunohistochemical workflow during the pathological analysis of UC tissues may be feasible compared to the complex and less practical approach of utilizing UC transcriptome subtypes. 44 For example, McConkey and colleagues’ work suggested that patients with the basal subtype exhibit the best survival rates, potentially indicating a more targeted application for VEGF-based therapies. 7
Conclusion
While the combination of antiangiogenic agents with ICIs shows promise, particularly for ICI-N patients, the overall complexity of response patterns necessitates further investigation. Response rates with these combinations were inferior among ICI-E patients, despite substantial variability among included trials. Limitations include differences in immune modulation mechanisms among different antiangiogenic agents, study designs or patient characteristics. The need to develop therapies for patients with ICI-refractory mUC is rising especially with recent FDA approvals of frontline ICI combinations. Elucidating the role of angiogenesis in ICI resistance in a subset of patients may allow for better selection in future trials.
Supplemental Material
sj-docx-1-blc-10.1177_23523735241296763 - Supplemental material for Antiangiogenic therapy combined with immune checkpoint blockade in urothelial cancer: Systematic review and meta-analysis
Supplemental material, sj-docx-1-blc-10.1177_23523735241296763 for Antiangiogenic therapy combined with immune checkpoint blockade in urothelial cancer: Systematic review and meta-analysis by Mohammad Jad Moussa, Iuliia Kovalenko, Emanuele Crupi, Ekaterina Proskuriakova, Yimin Geng, Giuseppe Fallara, Raed Benkhadra, Daniele Raggi, Matthew T. Campbell, Pavlos Msaouel and Omar Alhalabi in Bladder Cancer
Footnotes
ORCID iDs
Author contributions
All authors contributed to the article, jointly reviewed it and approved the submitted version.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Moussa MJ, Kovalenko I, Crupi E, Geng Y, Proskuriakova E, Fallara G, Benkhadra R and Raggi D report no disclosures.
Msaouel P reports honoraria for service on scientific advisory boards for Mirati Therapeutics, Bristol Myers Squibb, and Exelixis; consulting for Axiom Healthcare Strategies; nonbranded educational programs supported by DAVA Oncology, Exelixis, and Pfizer; and research funding for clinical trials from Takeda, Bristol Myers Squibb, Mirati Therapeutics, Gateway for Cancer Research, and the University of Texas MD Anderson Cancer Center.
Campbell MT reports consultancy or advisory role for Astellas, AstraZeneca, AXDev, Eisai, EMD Serono, Exelixis, Genentech, Pfizer, and SeaGen; research funding from ApricityHealth, Aravive, AstraZeneca, Exelixis, Janssen, and Pfizer/EMD Serono; nonbranded educational programs from Bristol Myers Squibb, Merck, Pfizer/EMD Serono, and Roche.
Alhalabi O reports scientific advisory board fees from Seagen, Silverback Therapeutics, and Cardinal Health; educational program speaker from Curio Science and Aptitude Health; and research funding to the institution from AstraZeneca, Ikena Oncology, Genentech, and Arcus Biosciences.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
