Abstract
Immuno-oncology drugs (IODs) have revolutionized the treatment of some cancers. Although IODs are enabling some patients with cancer to become long-time survivors, only 30% to 40% respond to these drugs. There is experimental and clinical evidence that the gut microbiome may play a role in IOD response, leading to speculation that manipulation of the gut microenvironment might improve the response rate to IODs. We review the evidence relating to how gut microorganisms may affect response to IODs and discuss the implications of targeting the microbiome to improve IOD response, including the challenges to refine and translate the findings to practical clinical use.
Keywords
Introduction
Immuno-oncology drugs (IODs) have revolutionized the management of several cancers, including melanoma, lung, pancreatic, kidney, bladder, and more. Ipilimumab, nivolumab, pembrolizumab, atezolizumab, avelumab, durvalumab, and other immune checkpoint inhibitors have vastly altered the therapeutic and prognostic landscape of these previously difficult-to-treat cancers. However, although IODs have enabled some cancer patients to become long-term survivors, only 30% to 40% of patients’ tumors demonstrate clinical responses. Although multiple factors influence response to IODs in individual patients (Table 1), most of these factors cannot currently be targeted for modification. However, there is experimental and clinical evidence that the gut microbiome may play a role in IOD response, leading to speculation that manipulation of the gut microenvironment might improve the response rate to IODs.
Factors associated with response to IOD. 1
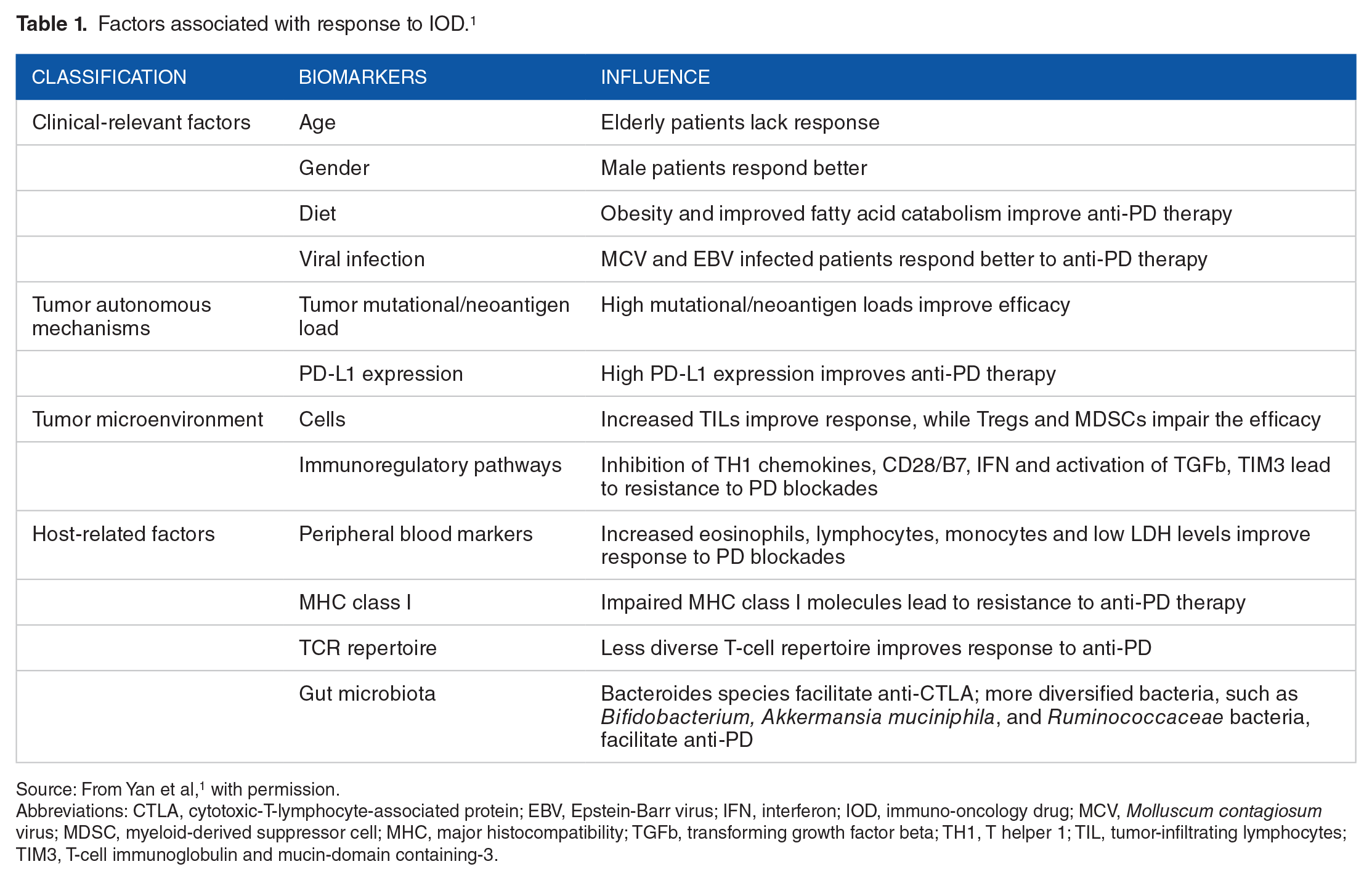
Source: From Yan et al, 1 with permission.
Abbreviations: CTLA, cytotoxic-T-lymphocyte-associated protein; EBV, Epstein-Barr virus; IFN, interferon; IOD, immuno-oncology drug; MCV, Molluscum contagiosum virus; MDSC, myeloid-derived suppressor cell; MHC, major histocompatibility; TGFb, transforming growth factor beta; TH1, T helper 1; TIL, tumor-infiltrating lymphocytes; TIM3, T-cell immunoglobulin and mucin-domain containing-3.
We review the evidence relating to how gut microorganisms may affect response to IODs and discuss the implications of targeting the microbiome to improve IOD response.
Microbiota and IOD
The gut microbiome’s influence on the host immune system is well known. Particularly relevant to natural antitumor defenses is its effect on T-cell function. 2 A number of clinical and experimental observations support a role for the gut microbiome in IOD responsiveness (Table 2):
Sivan et al 4 modeled the influence of commensal microbiota on spontaneous antitumor immune responses and their effect on anti-PD-1/PD-L1 therapy by examining the growth of B16.SIY melanoma tumors implanted subcutaneously into genetically similar mice raised in 2 separate locations. Tumor growth did not show a good response in the first group that had lower intratumoral CD8 + T cell infiltrates, while the second group (raised in a different location) had a good response to the treatment. When fecal material from responding mice was administered to the non-responding group, not only was tumor growth inhibited, but PD-L1 responses were markedly enhanced. The investigators concluded that fecal material from the mice sourced from the second location impeded tumor growth in the first group, and that components of this fecal material were sufficient in and of themselves to enhance the IOD response. They identified anaerobic Bifidobacterium spp, specifically Bifidobacterium breve, Bifidobacterium longum, and Bifidobacterium adolescentis, as candidate species responsible.
Matson et al 5 examined stool samples from metastatic melanoma patients obtained prior to administration of anti-PD-1 therapy and found that B longum, Collinsella aerofaciens, and Enterococcus faecium were more abundant in stools from patients who responded to these IODs.
Frankel et al 6 evaluated the effects of human gut microbiota and its metabolites on immune checkpoint inhibitor response in 39 metastatic melanoma patients treated with ipilimumab, nivolumab, ipilimumab plus nivolumab, or pembrolizumab. Responders for all therapies were enriched for Bacteroides caccae. Among those whose tumors responded to ipilimumab + nivolumab, the gut microbiome was enriched for Faecalibacterium prausnitzii, Bacteroides thetaiotaomicron, and Holdemania filiformis. Among pembrolizumab responders, the microbiome was enriched for Dorea formicogenerans.
Gopalakrishnan et al 7 examined the oral and gut microbiomes of 112 melanoma patients undergoing anti-PD-1 treatment. Those patients whose tumors responded to the treatment had an abundance of bacteria of the Ruminococcaceae family. In contrast, patients with abundance of Bacteroidales had impaired tumor responses. In addition, these researchers observed that germ-free mice, when transplanted with stool samples from the responding patients, had significantly reduced tumor growth and improved responses to anti-PD-1 and anti-PD-L1 therapy.
Routy et al 8 examined data from 140 patients with advanced non-small-cell-lung cancer, 67 patients with renal cell carcinoma, and 42 patients with urothelial carcinoma. Sixty-nine patients who took antibiotics before or soon after initiating anti-PD-1 therapy had shorter progression-free survival and overall survival. The authors observed that stools from patients whose cancers responded to anti-PD-1 therapy were enriched for Akkermansia muciniphila. Using a fecal microbiota transplant (FMT) process, feces from these patients then were transplanted to germ-free or antibiotic-treated mice. The antitumor effects of PD-1 blockade were ameliorated, whereas FMT from nonresponding patients failed to do so.
Summary of experiments providing evidence of microbiota’s influence on IOD response. 3
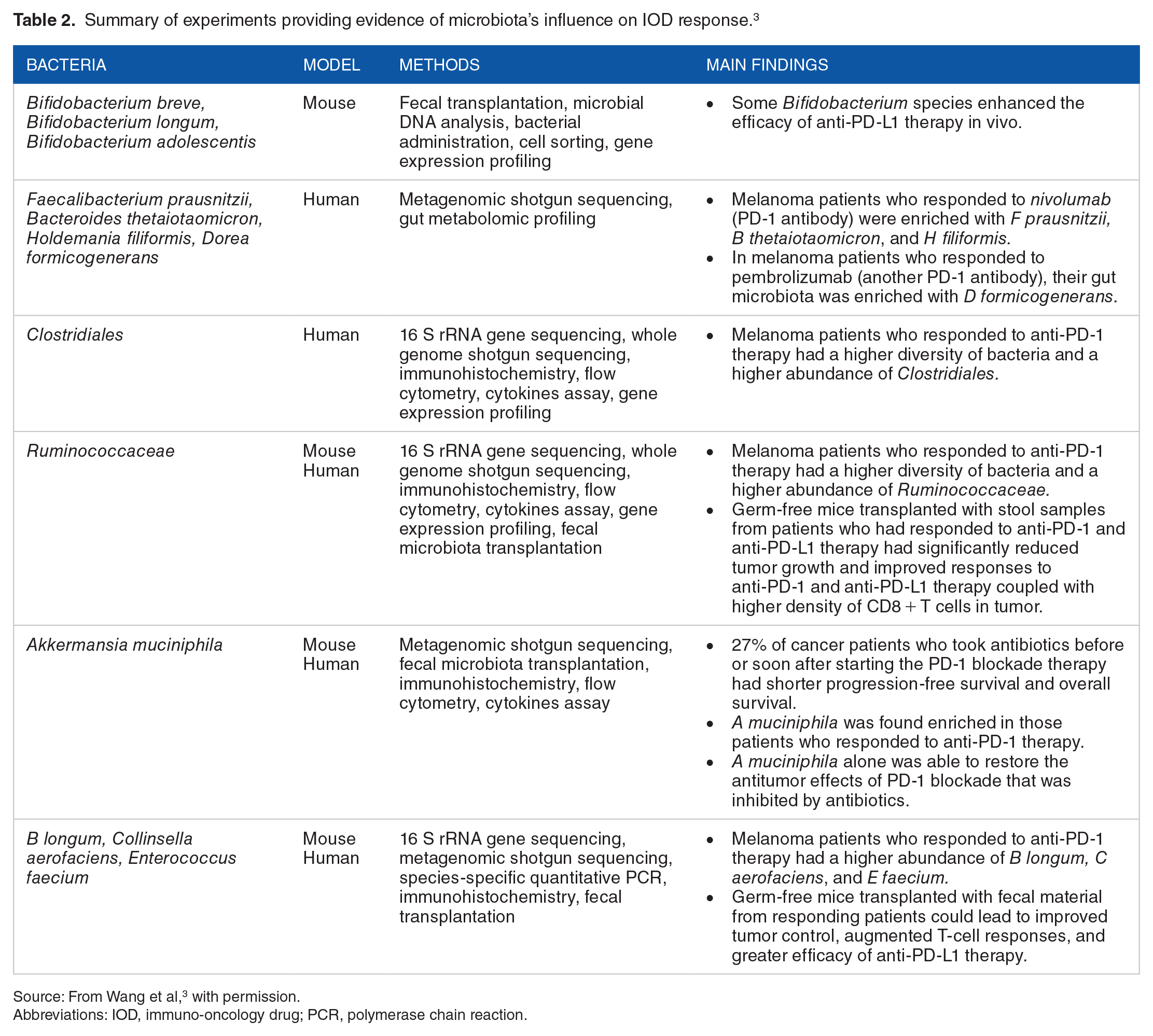
Source: From Wang et al, 3 with permission.
Abbreviations: IOD, immuno-oncology drug; PCR, polymerase chain reaction.
In addition, Pinato et al 9 showed in a prospective trial that prior broad-spectrum antibiotic therapy was associated with a worse treatment response to IOD in patients with non-small cell lung cancer, melanoma and other tumor types.
While these and other studies strongly suggest that gut microbiota can influence IOD response, the mechanisms by which this may occur remain undefined. Furthermore, the gut microenvironment is complex; apart from the bacteria, it is possible that nutrients released from specific microorganisms, or other components influenced by these microbes, may be playing a role. Because cancer is a systemic disease, it is important to know whether interaction of drugs with bacteria or bacterial products themselves, or immune-associated cells in the gut affected by bacteria, or nutrients released from bacteria is responsible alone or in combination.
Challenges to Translation of Observations to Therapeutics
Despite encouraging data from preclinical and clinical studies, there are multiple challenges to the refinement and translation of these findings to practical clinical use.
Precision identification of microbiome components associated with IOD responsiveness
Through metagenomic sequencing, it is estimated that healthy adults harbor >1000 bacterial species. 10 While bacteria at the species or phylum level have been associated with responses in cancer patients to IOD treatments, few bacteria have been common across studies. Further studies applying more sophisticated genomic/metagenomic and sequencing technologies, as well as a deeper understanding of mechanism(s) of action, will be required.
Formulation of therapeutics
Many questions remain unanswered regarding the nature and source of potential therapeutics. If specific bacterial species or phyla are shown to correlate with response to IODs, will fecal material containing these bacteria, together with other components to preserve the microenvironment, be more effective than purified bacterial preparations? Would laboratory-grown/Good Manufacturing Practice (GMP)-manufactured bacteria retain the same degree of effectiveness? Given individual variability of the microbiome, will a personalized approach to composition, dose level, or dose frequency be required? Should bacteria be sourced from healthy donors or from responding patients? These and other issues relating to mechanism of action should be addressed before choosing a specific approach and initiating clinical trials.
The only proven approach to altering the gut microbiome, FMT, has been safely and successfully administered for Clostridium difficile colitis, ulcerative colitis, and functional bowel disorders. 11 However, donor screening and selection, standardization, regulation, long-term safety, and other issues are unresolved. Commercial fecal donor banks are attempting to standardize screening, collection, and processing of material for FMT; yet, one company’s experience suggests that as few as 3% of healthy individuals screened meet preset criteria for donation. 12 Recent reports of multidrug-resistant bacteria transmission via FMT have raised alarm bells regarding safety of FMT and will no doubt spur additional regulatory oversight. 13 If FMT is sourced from responding patients, then what would appropriate screening criteria be and what would be the screen failure rate? We do not yet know the answers to these important questions.
Use of bacteria as a therapy in the cancer patient
Treatment of patients with cancer poses risks beyond those typically encountered in people with intestinal disorders. Cancer patients, especially those undergoing chemotherapy, are known to have altered immunity and disrupted natural barriers. IODs themselves also are known to alter host immune responses. This is important to consider because IODs are increasingly used in combination with cytotoxic drugs, which carry side effects, such as neutropenia, mucositis, diarrhea, and vomiting, that will likely affect the outcome of microbiome-targeted therapies. Furthermore, administering a live bacterial preparation (eg, FMT or purified live bacterial preparations) could be problematic in the setting of natural or therapeutically induced immunosuppression, and may also increase the risk of infection if an invasive procedure such as colonoscopy or endoscopy is involved.
Regulatory challenges
The US Food and Drug Administration (FDA) has defined FMT as a biological product and as a drug, which makes it subject to investigational new drug (IND) application regulations. However, the agency has stated its intent to use discretion regarding the IND requirement for FMT when used to treat refractory C difficile infection, if (1) adequate informed consent is obtained from the patient; (2) the FMT product is not obtained from a stool bank; and (3) the stool donor and stool are qualified by screening and testing performed under the direction of the licensed health care provider for the purpose of providing the FMT product for treatment of the patient. 14
The implications of these policies for process development and standardization, as well as controlled preclinical and clinical evaluation, are significant. Moreover, as noted earlier, safety concerns around FMT preparations have become increasingly relevant.
In Europe, many countries have developed country-specific guidelines. Belgium authorities have outlined conditions that will enable fecal microbiota to evolve toward the status of a medicine. 15 In France, fecal microbiota is considered a pharmaceutical drug, while in Austria, fecal microbiota transplantation is considered a therapeutic intervention. 16
Intellectual property–related challenges
Ownership and proprietary interest pertaining to bacteria and other products derived from the human microbiome are complex issues and in some ways ill-defined. It is our understanding that the bacteria (or microbial product) itself cannot be patented while the specific formulation intended for use as a specific therapy and its development process (along with the method of use) can be patented. In the absence of legal intellectual property protection, the commercial opportunities surrounding microbiome-derived products remain obscure.
Conclusion
Immune checkpoint inhibitors and other IODs have transformed the practice of clinical oncology but have helped only 30% to 40% of the patients treated. While research suggests that components of the gut microenvironment, and specifically the gut microbiome, affect the response to these drugs, few bacteria or their byproducts have been identified as common across studies, and researchers have yet to understand the mechanisms of action. Furthermore, issues relating to the quality and the method of administering the bacteria remain unresolved, and intellectual property protections and regulations must be considered. Clearly, more questions than answers exist now, but the promising results from multiple research studies demonstrate the importance of continuing this research with the hope of improving health outcomes for more oncology patients in the future.
Supplemental Material
Permission_doc_1 – Supplemental material for Gut Microbiota and Response to Immunotherapeutic Drugs in Oncology: More Questions Than Answers
Supplemental material, Permission_doc_1 for Gut Microbiota and Response to Immunotherapeutic Drugs in Oncology: More Questions Than Answers by Dhiraj Abhyankar, Kelly T McKee and Pavle Vukojevic in Clinical Medicine Insights: Oncology
Supplemental Material
Permission_doc_2_1 – Supplemental material for Gut Microbiota and Response to Immunotherapeutic Drugs in Oncology: More Questions Than Answers
Supplemental material, Permission_doc_2_1 for Gut Microbiota and Response to Immunotherapeutic Drugs in Oncology: More Questions Than Answers by Dhiraj Abhyankar, Kelly T McKee and Pavle Vukojevic in Clinical Medicine Insights: Oncology
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
DA and KTMK contributed to writing the article, PV reviewed it.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
