Abstract
Although immune checkpoint inhibitors (ICIs) have greatly improved the prognosis of some cancer patients, the majority still fail to respond adequately, and the available biomarkers cannot reliably predict drug efficacy. The gut microbiota has received widespread attention among the various intrinsic and extrinsic factors contributing to drug resistance. As an essential regulator of physiological function, the impact of gut microbiota on host immunity and response to cancer therapy is increasingly recognized. Several studies have demonstrated significant differences in gut microbiota between responders and nonresponders. The gut microbiota associated with better clinical outcomes is called ‘favorable gut microbiota’. Significantly, interventions can alter the gut microbiota. By shifting the gut microbiota to the ‘favorable’ one through various modifications, preclinical and clinical studies have yielded more pronounced responses and better clinical outcomes when combined with ICIs treatment, providing novel approaches to improve the efficacy of cancer immunotherapy. These findings may be attributed to the effects of gut microbiota and its metabolites on the immune microenvironment and the systemic immune system, but the underlying mechanisms remain to be discovered. In this review, we summarize the clinical evidence that the gut microbiota is strongly associated with the outcomes of ICI treatment and describe the gut microbiota characteristics associated with better clinical outcomes. We then expand on the current prevalent modalities of gut microbiota regulation, provide a comprehensive overview of preclinical and clinical research advances in improving the therapeutic efficacy and prognosis of ICIs by modulating gut microbiota, and suggest fundamental questions we need to address and potential directions for future research expansion.
Introduction
Immune checkpoint inhibitors (ICIs) have revolutionized the field of cancer therapy. Although chemotherapy and radiotherapy remain the primary treatments for most cancers, ICIs are now effective treatments for many unresectable solid or nonsolid tumors, with response rates between 20% and 40% for most tumor indications. 1 ICIs also have excellent performance in neoadjuvant and adjuvant therapy. Neoadjuvant nivolumab did not delay surgery and caused major pathologic responses in nearly half (45%) of resectable non-small-cell lung cancer (NSCLC) with minimal side effects. 2 Pembrolizumab as adjuvant therapy for stage IIB or IIC melanoma significantly reduces the risk of disease recurrence or death compared to placebo while having a manageable safety profile. 3 As the most commonly used immunotherapy, ICIs work by relieving the functional suppression of T cells, inducing potent activation of the immune system and an effective antitumor response. To date, the US Food and Drug Administration has approved ICIs against four different targets for use in humans: antibodies against cytotoxic T lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1) and its ligand, PD-L1, and lymphocyte-activation gene 3 protein.4,5 Over the years, the clinical use of anti-PD-1 and PD-L1 antibodies has significantly surpassed that of anti-CTLA-4 antibodies because of greater clinical efficacy and better tolerability.
Although some patients experienced dramatic tumor regression after treatment with ICIs, most did not respond. Among the melanoma patients with the highest response rates to ICIs, 40–50% showed no objective response to the combination of nivolumab and ipilimumab.6,7 In patients with mismatch repair proficient/microsatellite stable metastatic colorectal cancer, no efficacy was observed with ICIs alone, 8 and the objective response rate in a phase II clinical trial of durvalumab combined with tremelimumab was only 1%. 9 Why most patients do not respond to ICIs deserves to be studied. A complex and interconnected set of factors, such as host, tumor, and environmental variables, 10 can contribute to drug resistance, including gut microbiota. The gut microbiota plays a vital role as a regulator of physiological functions, including obtaining nutrients from the diet, maintaining the integrity of the mucosal barrier, and promoting the development and homeostasis of the immune system. 11 It affects the threshold of the anticancer immune response.12,13 Increasing numbers of preclinical and clinical studies have demonstrated that the efficacy and prognosis of ICI treatment are closely related to gut microbiota diversity and variety. 14 Therefore, strategies to modulate gut microbiota [e.g. fecal microbiota transplantation (FMT), probiotics, and dietary interventions] are being developed and used to improve clinical outcomes in patients using ICIs.
In this review, we present evidence for the role of gut microbiota in the therapeutic response to ICIs, outline the potential mechanisms, summarize the advances in research modulating the efficacy of ICIs by intervening with gut microbiota, and provide an outlook on future directions.
The gut microbiota is closely related to the efficacy of ICIs
The gut microbiota regulates cancer at multiple levels of initiation, genetic instability, susceptibility to host immune response, progression, and response to treatment. 15 There is evidence that the gut microbiota may play a role in the response to chemotherapy. 16 The immune system is an essential pathway by which the microbiome influences the chemotherapeutic response.16,17 As a treatment that is most closely associated with the immune response, the relationship between ICIs and the gut microbiota has attracted attention.
Two landmark studies published in 2015 uncovered a link between gut microbiota and ICIs in preclinical murine models. They indicated that CTLA-4 and PD-1 blockade only reduced tumor growth in mice carrying specific commensal bacteria and that modulation of gut microbiota could enhance treatment response.18,19 Three subsequent studies in 2018 further explored the gut microbiota characteristics of responders (Rs) and nonresponders (NRs) to ICIs and successfully reduced tumor volumes in preclinical murine models by FMT from responders.20–22 Moreover, two independent cohorts showed that antibiotic-induced dysbiosis adversely affected the response to ICI therapy.22,23 The importance of specific bacterial composition has been supported by several clinical studies investigating the relationship between different gut microbiota characteristics and clinical benefits (Supplemental Table 1).20–22,24–43 Most of these studies focused on NSCLC, melanoma, and hepatocellular carcinoma, where alpha diversity and some specific gut microbiome constituents have been associated with successful treatment with ICIs, such as
Due to the limited sample size, differences in the characteristic microbiome among cancers were not detected. Even in patients with the same cancers responding to the same ICIs, a low overlap of gut microbiota was observed, which may be related to different reference databases, diverse sample analysis techniques, and individual differences. It may also suggest that, rather than individual strains, the integral gut ecosystem is more likely to be a key determinant of the response to ICIs. The human gut microbiome includes multiple discrete ecologically balanced communities.49,50 Most current studies link individual taxa to medication outcomes, but little is known about the gut ecosystem. McCulloch
In addition, some studies use gut microbiota as a biomarker of therapeutic response. Patients were classified into clusters based on the intrinsic gut microbiota composition at baseline by unsupervised hierarchical clustering, these clusters had different response ratios to immunotherapy.19,20,25,26,42 Peng
Modulating the efficacy of ICIs by regulating gut microbiota
There is growing evidence that the gut microbiota has a role in influencing the efficacy of ICIs in preclinical models and cancer patients. In particular, antibiotic treatment prior to ICIs inhibits clinical efficacy, further suggesting that disruption of the balanced gut microbiota (i.e. switch from eubiosis to dysbiosis) and loss of specific bacteria may impede the success of anticancer therapy.23,51 A lack of immunostimulatory bacteria or a dysregulated gut ecosystem containing excessive immunosuppressive bacteria can lead to therapeutic failure. Therefore, modulating gut microbiota composition may be an effective strategy to avert ICI resistance. The gut microbiota composition is determined by host genetics, colonization at birth, type of delivery, individual lifestyle, disease, and drug exposure.52–55 It remains relatively stable in adulthood 56 but can be altered by external interventions or disturbances in the internal environment. Clinical studies preliminarily evaluated the effectiveness of improving immunotherapy outcomes through gut microbiota modulation (Table 1).24,29,57–63 Additional clinical trials have been initiated, which may provide practical and accessible tools for future adjuvant cancer therapy (Table 2).
Clinical studies on modulating gut microbiota composition to improve the efficacy of ICIs.
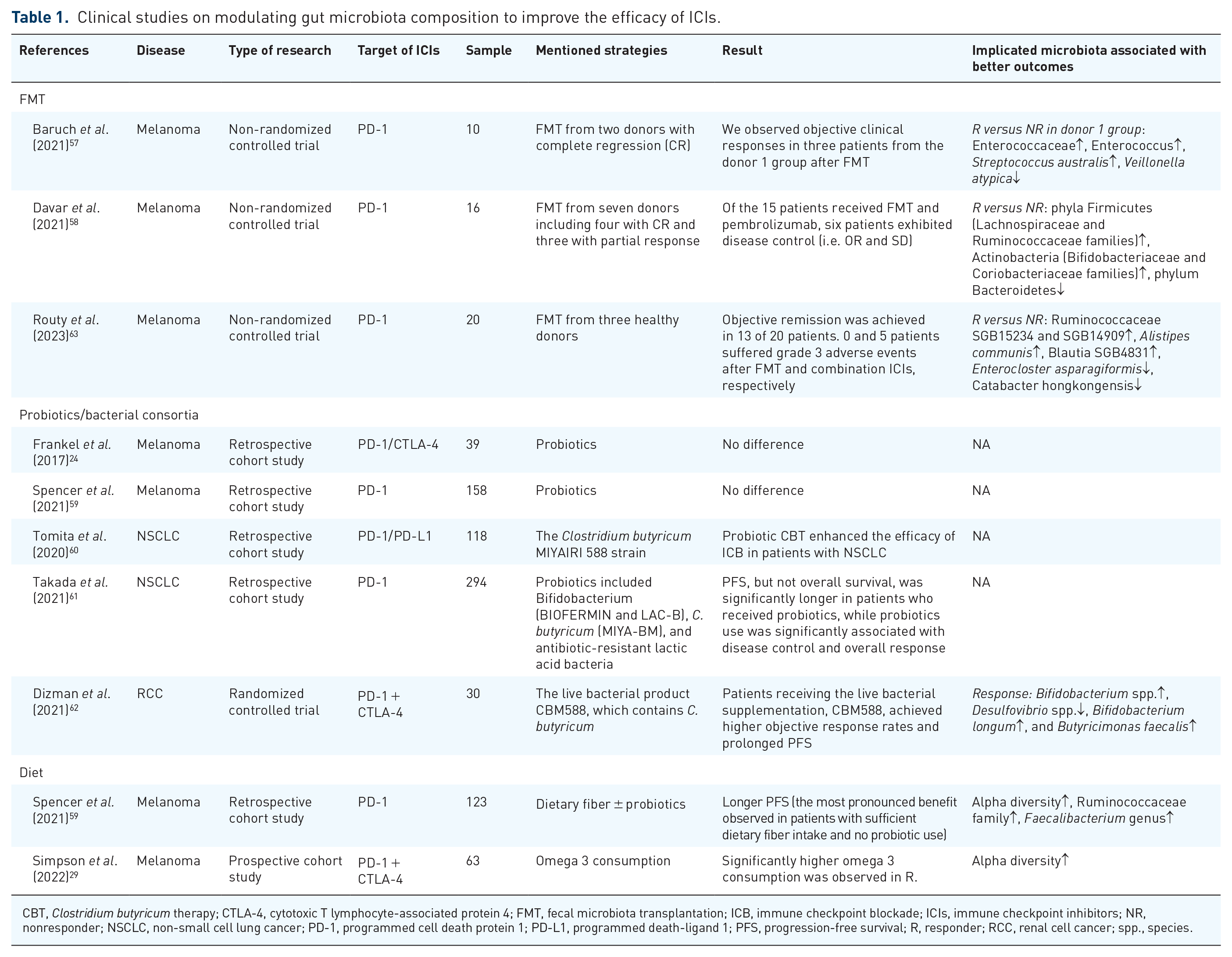
CBT,
Ongoing trials of gut microbiome modulation (FMT/probiotics/bacterial consortia) in cancer immunotherapy.

dMMR, mismatch repair deficiency; FMT, fecal microbiota transplantation; ICIs, immune checkpoint inhibitors; MSS, microsatellite stable; NSCLC, non-small-cell lung cancer; PD-1, programmed cell death protein 1; PD-L1, programmed death-ligand 1; pMMR, mismatch repair proficient.
Fecal microbiota transplantation
FMT is the administration of fecal matter from a donor into the intestinal tract of a recipient to directly change the recipient’s microbial composition and confer a health benefit. 64 It is effective in numerous diseases, such as irritable bowel syndrome (IBS), inflammatory bowel disease (IBD), metabolic disorders, autoimmune diseases, and cancer.65,66 FMT transplants the entire gut microbial ecosystem from the donor containing beneficial bacteria and overall diversity, which enables the introduced bacteria to be more stable and less subject to competitive rejection by the recipient microbiota.
Due to imperfect quality control assessment, pathogenic bacteria, especially multidrug-resistant bacteria, and other pathogens may be unknowingly transferred in fecal material, resulting in severe infections and complications. 67 In addition, complicated operating procedures make the products expensive and challenging to access.
Nevertheless, several studies have demonstrated that FMT effectively improved the efficacy and prognosis of ICIs in preclinical murine models (Supplemental Table 2).18–22,34,39,59,63,68–74 Transplantation of fecal matter from Jackson Laboratory (JAX) mice with better responses into Taconic Farms (TAC) mice significantly improved the efficacy of anti-PD-L1 treatment. It increased the abundance of
The combined application of FMT from responders and anti-PD-1 treatment was well tolerated in patients with anti-PD-1 refractory melanoma, resulting in clinical benefit in 6 of 15 patients. FMT combination therapy induced microbiota fluctuations, with posttransplant responders exhibiting an increase in the abundance of taxa previously shown to be associated with anti-PD-1 treatment response.
58
In phase I clinical trial by Baruch
The results of the above clinical trials support the fact that FMT from responders could overcome resistance to ICIs in melanoma. Recently, a phase I clinical trial of FMT in combination with immunotherapy (NCT03772899) enrolled 20 ICI-naïve patients with advanced melanoma who underwent FMT
FMT was delivered mainly by two modalities, endoscopy or (and) oral capsules.57,58,63 Due to insufficient clinical experience, comparing the effects of different delivery modalities in immunotherapy is difficult. Each modality can improve the composition of the gut microbiota effectively and has been widely used in digestive diseases other than cancer.82,83 Endoscopic transplantation is the most direct way to accurately transplant larger amounts of intestinal bacteria into the implicated bowel segment with potentially greater clinical efficacy, especially in recurrent
More clinical trials are currently underway (Table 2). Most trials use fecal matter from responders (e.g. NCT03341143), and a few use healthy individuals with specific favorable strains or with the same microbiota profile as responders as donors (e.g. NCT04163289). Identifying and functionalizing critical bacterial species that drive favorable clinical outcomes in ICIs are crucial and can help select suitable donors. Meanwhile, the optimal route for FMT has yet to be determined. Factors such as patient compliance and cost-effectiveness are important in selecting the appropriate transplantation modality. Whether there is heterogeneity in the favorable strains associated with the efficacy of ICIs in different cancers and whether the modulation of gut microbiota by FMT to improve the efficacy of ICIs is always sensitive and feasible are questions that need further exploration.
Bacterial consortia and probiotics
An alternative approach to modulating the gut microbiota is the oral administration of bacterial consortia or beneficial bacteria (i.e. probiotics). Concentrating on identified bacteria prevents the transfer of other pathogens and may provide stable therapeutic products that can be obtained and applied easily. However, there are substantial problems and discrepancies in bioavailability and standardization quality control. In addition, administration may cause a reduction in the overall diversity of gut microbiota. 89
As early as 2015, Sivan
Apart from conventional bacteria, Griffin
Probiotics are one part of the bacterial consortia. They are living bacteria that provide health benefits to the host and show varying degrees of clinical efficacy in many gastrointestinal disorders.93–95 The efficacy of combination therapy with ICIs and probiotics varies (Table 1).24,59–62 Two retrospective studies in NSCLC patients showed that probiotic use improved immunotherapy efficacy and was associated with favorable prognostic outcomes. These probiotics included the
Preclinical studies are warranted based on promising early data.18,47,60 Oral probiotics combined with antitumor immunotherapy demonstrated superior efficacy in murine models. The commercial cocktail of Bifidobacterium, including
Only one interventional clinical trial has presented preliminary results. The open-label, single-center, randomized, controlled study showed that 19 treatment-naïve patients with metastatic renal cell carcinoma receiving nivolumab–ipilimumab plus probiotics (CBM588) achieved higher objective response rates (58%
Although the clinical trial presented favorable results, the instability of probiotics or other bacterial products in the intestine caused by the resistance of the original bacteria prevented follow-up research and the clinical application of this technology. Previous studies have primarily used antibiotic pretreatment to prepare the gut ecosystem. Han
Clinical trials of probiotics in combination with immunotherapy are being conducted gradually (Table 2). Several hospitals in China have expanded the research direction to other cancers, such as NCT05220124 and NCT05032014, focusing on bladder and liver cancer, respectively. Indeed, further research is needed to determine how effectively probiotics can improve immunotherapy efficacy. We should take a very cautious position until the safety of commercially available probiotics is thoroughly evaluated in preclinical and clinical studies.
Diet and prebiotics
Dietary intake can contribute to different microbiome compositions, changes in dietary regimen significantly alter the gut microbiota within 5 days.
105
Although responders to ICIs have different bacterial compositions, these taxa have something in common regarding their upstream dietary substrates.106,107 For example,
With excellent safety, accessibility, and low cost, dietary interventions are simple ways to regulate the gut microbiota in patients using ICIs. However, behavioral changes, particularly dietary patterns, are notoriously tricky, resulting in a paucity of prospective interventional studies and evidence-based dietary guidelines for patients to follow. As a widely known healthy diet, a high-fiber diet can be helpful for immunotherapy.59,69 It could enhance the response to ICIs in preclinical mouse models, which is closely related to increased fiber-fermenting bacteria.
Dietary fiber is the main subject of current diet-related research. Indeed, consuming specific dietary components, such as dietary fiber and prebiotics, is a common way to regulate the microbiota. Intervention clinical trials have shown that dietary fiber intake increases gut bacterial diversity.
108
Dietary fiber intake may affect clinical outcomes in patients treated with ICIs. A retrospective cohort study showed that patients treated with ICIs taking sufficient dietary fiber intake had significantly longer PFS than those taking insufficient dietary fiber intake (median PFS not reached
Bindels
In addition, Simpson
Modulation of gut microbiota affects irAEs of immune checkpoint inhibitors
Research has demonstrated that irAE occurrence is connected to better ICI response and long-term benefits,117,118 making early and appropriate management a critical task. Nevertheless, the relationship between specific genera and irAEs has yielded controversial results. Certain bacteria have been identified to facilitate ICI responses and reduce ICI toxicities, including Akkermansia, Bifidobacterium, etc.
119
Chaput
While improving the efficacy of immunotherapy through modulating gut bacteria, the diversity and abundance of microbes might also influence ICI therapy’s immunotoxicity. In a currently published clinical trial, the efficacy of ICIs can be safely modulated by FMT. 63 However, it is difficult to draw a definite conclusion regarding highly diverse and multifaceted microbial-immune system interactions. We still need to monitor the possible induction or attenuation of irAEs in immunotherapy and find the balance between ICI response and irAEs step by step.
Gut microbiota modulates the function of immune checkpoint inhibitors by several potential mechanisms
Although studies suggest that the gut microbiota may be an important factor in the therapeutic response to ICIs, the mode of action remains to be elucidated. We summarize the potential mechanisms as follows (Figure 1).

Gut microbiota modulates the antitumor immunity by several potential mechanisms in immunotherapy. (a) Microbial proteins cause T-cell cross-reactivity by mimicking tumor antigens. (b) Gut microbiota creates a relatively “hot” tumor microenvironment by regulating the expression and distribution of immune checkpoint molecules. (c) Microbes or microbial products activate PRRs, promoting tumor cell death by directly inducing or indirectly activating adaptive immune cells
Regulate the expression and distribution of immune checkpoint molecules
The TME consists of many different cell types that play an essential role in tumor tolerance and evasion of immune surveillance 120 and largely influence the efficacy of ICIs,121,122 including T cells, fibroblasts, natural killer (NK) cells, and DCs.123–125
Chaput
Programmed death ligand 2 (PD-L2) is a member of the same B7 family proteins as PD-L1 and exerts immunomodulatory effects by binding to PD-1 and repulsive guidance molecule b (RGMb) as a ligand.
130
In 2017, Yearley
Activate antitumor immunity
One pathway is that microbial proteins cause T-cell cross-reactivity by mimicking tumor antigens. The theory of ‘molecular mimicry’ suggests that specific CD4+ or CD8+ T lymphocytes induced by major histocompatibility complex class I and class II binding epitopes encoded by bacterial or viral genomes may unexpectedly recognize autoantigens and attack tumor tissues.133,134
Microbial-specific T lymphocytes can activate an effective antitumor immune response. Deficits in the efficacy of ICIs were overcome by the adoptive transfer of
Until 2020, Bessell
The other pathway is that gut microbes or microbial products activate pattern recognition receptors (PRRs), leading to the activation of immune responses. Several PRR agonists have been proven to overcome resistance to ICI therapy.140–142 PRR stimulation can promote tumor cell death by directly inducing or indirectly activating adaptive immune cells
Release metabolites
Metabolites of the gut microbiota suppress inflammation by maintaining the epithelial barrier and intestinal integrity and interact with host cells to influence the immune response.144,145 SCFAs are the most crucial metabolites, including acetate, propionate, and butyrate, which can modulate effector T-cell function by regulating G protein-coupled receptor signaling through a receptor-dependent mechanism.146,147 A cohort study of 52 patients with solid tumors revealed that high fecal acetate, propionate, butyrate, and valerate concentrations were significantly associated with prolonged PFS and higher response rates to anti-PD-1 therapy.20,148 NSCLC patients receiving anti-PD-1 therapy showed similar gut metabolomics, in which fecal SCFAs, particularly propionate, were associated with a better long-term response to ICIs.
31
The supplement of a probiotic strain,
Other metabolites also contribute to modulating immunotherapy. Trimethylamine
By specifying the connection between gut microbiota metabolites and immunotherapy, we may pioneer a new approach to adjuvant cancer treatment.
Affect intratumoral microbiome
The intratumoral microbiome constitutes a significant part of TME, influencing tumorigenesis, progression, metastasis, and antitumor immunity,
152
especially the extent of immune infiltration and the degree of activation of CD8+ T cells,153–156 which are important factors affecting ICI efficacy. Intratumor microbiota can promote CD8+ T-cell activation and TH1 differentiation of CD4+ T cells by increasing differentiation of classical activated macrophages (M1) and decreasing myeloid-derived suppressor cells, ultimately increasing PD-1 expression and responsiveness to immunotherapy.
156
Nejman
By comparing the microbiomes of stool samples, tumor specimens, and non-tumor adjacent normal tissues from PDAC patients, approximately 25% of the intratumoral microbiome was derived from the gut microbiome. When fecal microbiota from patients with advanced PDAC was transferred to the murine intestine, the bacterial composition of the intratumoral microbiome was significantly altered, and a small proportion of fecal bacteria from human donors could be detected. 153 In the Lewis lung cancer mouse model, exogenous Akk increases the abundance of Akk in the circulation or tumor tissue, affecting the commensal microbiome and reprogramming the tumor metabolic network. 158 These results demonstrate that the intestinal microbiome could modulate the intratumoral microbiome.
Targeting the gut microbiome may reverse the intratumoral immune tolerance environment. Mackenzie
T-cell cross-reactivity has been mentioned above as a possible mechanism of ICI enhancement by gut microbiota. Whereas a study in melanoma showed that intratumoral bacteria can enter melanoma cells, and that their peptides can be presented by the human leukocyte antigen class I and class II molecules of melanoma tumor cells and elicit immune reactivity, again providing the basis for T-cell cross-reactivity. 160
With the above studies, the authors demonstrated a new way for gut microbiota to modulate immunotherapy through bacteria translocation to gut-distal tumors, providing a rational basis for designing new combination therapeutic strategies.
Regulate inflammatory pathways
The cyclic GMP-AMP synthase (cGAS)/STING signaling pathway has become a key mediator of inflammation, with the capacity to sense and regulate cellular responses to microbes. Gut microbiota associated with a high-fiber diet improves ICI efficacy
The inflammatory pathway is an important link in the activation of antitumor immunity. Except for the above, it can be widely detected among the pathways by which the gut microbiota modulates immunotherapy, especially the T-cell receptor signaling pathway. There are far more inflammatory pathways involved, pending further exploration.
Others
MYC, a pivotal signal molecule regulating colorectal cancer proliferation, migration, stemness, and growth, induces PD-L1 expression in tumors,
161
leading to immunosurveillance escape and nonresponse or resistance to ICIs.
Intestinal
Some components of intestinal bacteria or their metabolites can drive tumorigenesis,15,164,165 induce an immunosuppressive microenvironment,
156
and cause therapeutic tolerance,
20
known as ‘unfavorable gut microbiota’, such as
Future perspectives
The evidence above highlights the potential effect of the gut microbiota on the immunotherapeutic response and supports future efforts to improve treatment efficacy through its modulation.
However, each of these strategies has important variables to be considered. For FMT, it is essential to find expedient preparation and effective delivery methods, and strict policies must guide donor selection. Standardizing commercially available commodities is the most urgent requirement for probiotic or bacterial consortium application, and self-administration of off-the-shelf probiotics for cancer treatment is not recommended. We also have to decide on the type, dose, and formulation based on bacterial colonization to achieve optimal outcomes while avoiding potentially reduced microbiome diversity. Maintaining patients’ dietary patterns over time is the main challenge for dietary interventions. Whether the diet is sufficient to modulate therapeutic efficacy needs to be further demonstrated.
Limitations of this review include the following points. Gut microbiota modulates immunotherapy through multiple mechanisms. We mainly focus on the relevant mechanisms with well-defined evidence and only briefly describe the possible mechanisms that have yet to be rigorously confirmed. Besides, the regulation of gut microbiota is a new approach to enhancing ICIs. Although many clinical studies are underway, the published data are scarce and of limited value for clinical guidance. Research on the biological characteristics, pharmacokinetics, and optimal delivery of the potential interventions could further guide clinical medication. In conclusion, future challenges and directions are as follows:
➢ We need to further explore the relationship between gut microbiota and tumorigenesis, immune function, and antitumor immune response and uncover the biological effects and modes of action of commensal bacteria to lay the foundation for clinical research.
➢ The discovery of reliable microbiome-related biomarkers for predicting outcomes and stratifying patients accordingly could help guide the application of coregulatory interventions for the gut microbiota. The characterization of favorable and unfavorable gut microbiota is crucial, and the identification of microbial characteristics associated with different tumors or different drugs can contribute to the evolution of precision medicine.
➢ We need to determine the optimal modalities to effectively induce and maintain the favorable gut microbiota that is beneficial for immunotherapy over time, as well as factors that disrupt the beneficial bacterial component, and implement appropriate preventive measures.
➢ The evaluation of bacterial metabolites that enhance antitumor immunity can help facilitate the invention of novel products and target new molecular pathways.
➢ Other microbiomes in the gut, such as fungi and viruses, together with bacteria, form an integral part of the microecosystem. However, their roles have been underestimated in current studies. We need to refine relevant studies to confirm their value.
By addressing the above, we expect to establish a convenient and cost-effective approach to clarify the distribution of the gut microbiota and create a reproducible pathway to manipulate the gut ecosystem in combination with immunotherapy to improve patient survival.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231204854 – Supplemental material for Modulation of gut microbiota: a novel approach to enhancing the effects of immune checkpoint inhibitors
Supplemental material, sj-docx-1-tam-10.1177_17588359231204854 for Modulation of gut microbiota: a novel approach to enhancing the effects of immune checkpoint inhibitors by Jinglong Huang, Caifeng Gong and Aiping Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
