Abstract
Euthyroid sick syndrome (ESS) is characterized by abnormal thyroid function tests, most notably a low triiodothyronine (T3) level, occurring in the absence of intrinsic thyroid disease. A 54-year-old woman presented to the endocrinology clinic with a 4-month history of progressive fatigue, lethargy, and new-onset cold intolerance after initiation of semaglutide for weight management. The dose was titrated monthly over 4 months, during which she experienced significant weight loss of 22 kg. Laboratory evaluation revealed a thyroid function profile classic for ESS, with low free T3, low-normal free T4, and a normal thyroid-stimulating hormone (TSH) level that was inappropriately low relative to the reduced T3. After exclusion of primary thyroid and pituitary disorders, a diagnosis of ESS secondary to the catabolic state induced by rapid weight loss was made. The patient was counseled that this represented a physiological adaptation rather than intrinsic thyroid disease. Semaglutide was continued given its metabolic benefits, and nutritional optimization with adequate caloric and protein intake was advised.
Plain Language Summary
This article describes a temporary change in thyroid blood tests that can happen during rapid weight loss. Thyroid tests are commonly used to assess how the body regulates energy and metabolism, but these tests can sometimes appear abnormal even when the thyroid gland itself is healthy. This condition is known as euthyroid sick syndrome (ESS) and is usually seen during illness or physical stress. With the increasing use of weight-loss medications such as semaglutide, understanding this phenomenon has become more important. We report the case of a 54-year-old woman who was treated with semaglutide for obesity. Over 4 months, she experienced substantial and intentional weight loss of 22 kg, which improved her metabolic health. During this period, she developed symptoms including fatigue and increased sensitivity to cold. Blood tests showed a low level of one thyroid hormone (T3), while other thyroid hormones were within or near the normal range. There was no evidence of underlying thyroid or pituitary disease. These findings were explained by ESS, a reversible condition caused by the body adapting to reduced calorie intake and a catabolic state during rapid weight loss. In this situation, the body temporarily lowers energy expenditure as a protective mechanism. Importantly, this does not indicate permanent thyroid disease or failure. As the patient’s weight stabilized and nutritional intake improved, her symptoms resolved and her thyroid tests began to normalize without the need for thyroid hormone treatment. This case highlights that abnormal thyroid test results during rapid weight loss particularly in people using medications like semaglutide do not always require treatment. Recognizing ESS can help avoid unnecessary investigations, prevent inappropriate thyroid hormone use, and reassure patients that these changes are often temporary and reversible.
Introduction
Euthyroid sick syndrome, also known as nonthyroidal illness syndrome or low T3 syndrome, is characterized by abnormal thyroid function tests in patients experiencing acute illness without intrinsic defects of the thyroid gland. Chronic illness is seldom associated with euthyroid sick syndrome unless additional endocrine pathology exists. Current evidence suggests that alterations in thyroid hormone levels in euthyroid sick syndrome are the result of both physiological adaptation and pathological mechanisms. Laboratory abnormalities typically include low total and free T3 levels, increased reverse T3 (rT3), and low or normal thyroxine (T4) and thyroid-stimulating hormone (TSH) levels. 1
Iodothyronine deiodinases are enzymes responsible for the conversion of T4 to T3. Three types have been identified: type 1 (D1) and type 2 (D2), which mediate peripheral activation of thyroid hormones, and type 3 (D3), which inactivates thyroid hormones by converting T3 to rT3. Numerous factors inhibit D1 activity, including acute illness, caloric deprivation, malnutrition, medications such as glucocorticoids, β-adrenergic antagonists (eg, propranolol), amiodarone, and propylthiouracil, as well as fatty acids, the fetal or neonatal period, selenium deficiency, and hepatic disease. In euthyroid sick syndrome, decreased D1 and increased D3 activity result in reduced T3 and increased rT3 levels, respectively. 2
During the early phase of illness, suppression of the hypothalamic–pituitary–thyroid (HPT) axis also contributes to these changes. Thyroid-stimulating hormone levels may initially remain normal; however, as illness progresses, TSH levels may decrease due to the inhibitory effects of steroids and dopamine. Additionally, increased D2 activity in the pituitary gland further suppresses TSH secretion. During fasting, reduced leptin levels also influence TSH secretion. Leptin, a hormone produced by adipose tissue, normally stimulates TSH through regulation of thyrotropin-releasing hormone (TRH); therefore, decreased leptin levels lead to lower TSH secretion. 3 High catabolic states and malnutrition may also alter thyroid-binding globulin (TBG) synthesis. 2
Semaglutide is a glucagon-like peptide-1 (GLP-1) receptor agonist. GLP-1 is released from intestinal L cells and stimulates insulin secretion, suppresses glucagon release, delays gastric emptying, and reduces appetite, resulting in significant weight loss. 4 GLP-1 receptor agonists are approved for the treatment of obesity in patients with a body mass index (BMI) ⩾ 30 kg/m2 or ⩾27 kg/m2 in the presence of comorbidities such as hypertension, dyslipidemia, or diabetes mellitus, particularly when lifestyle modifications fail to achieve at least 5% weight loss within 3 to 6 months. 5 individuals with obesity may achieve an average body weight reduction of 15% to 25% after approximately 1 year of therapy. 6
A novel absorption enhancer, sodium N-[(2-hydroxybenzyl) amino caprylate] (SNAC), enables effective oral absorption of semaglutide by protecting it from enzymatic degradation and facilitating gastric mucosal uptake. Studies have demonstrated comparable glycemic control and weight management outcomes between oral and subcutaneous semaglutide formulations. 7 While studies have shown that liraglutide can induce C-cell hyperplasia and increase the risk of C-cell tumors in animal models, with an associated rise in calcitonin levels, 8 but recent observational studies have demonstrated the absence of this hypothesized association between GLP-1 Receptor Agonists and thyroid cancers. 9 However, there remains limited evidence regarding the effects of GLP-1 receptor agonists on T3, T4, and rT3 levels in humans.
Case Presentation
A 54-year-old woman presented to the endocrinology clinic with a 4-month history of progressive fatigue, lethargy, and new-onset cold intolerance.
Her medical history was significant for obesity (baseline BMI 38.2 kg/m2), prediabetes with HbA1c of 5.9%, and stage 1 hypertension that was well controlled with lifestyle measures and required no pharmacologic therapy. Four months prior to presentation, she was initiated on semaglutide for weight management, with the dose titrated monthly from 0.25 to 1.0 mg weekly. During this period, she reported marked appetite suppression and achieved significant weight loss of 22 kg, decreasing from 105 to 83 kg, representing an 18% reduction in body weight. She reported no personal or family history of thyroid disease, and semaglutide was her only medication.
On physical examination, the patient was alert but appeared fatigued with decrease activity. Vital signs were stable, including blood pressure of 118/74 mmHg, heart rate of 64 beats per minute, and she was afebrile. Her BMI at presentation was 30.8 kg/m2. Skin examination revealed cool, dry skin without pallor or jaundice, and there were no palpable thyroid enlargement or masses. Neurological examination demonstrated normoactive deep tendon reflexes with a normal relaxation phase. Review of systems was positive only for fatigue and cold intolerance; she denied palpitations, heat intolerance, or changes in bowel habits.
Laboratory evaluation revealed a thyroid function profile consistent with euthyroid sick syndrome (Table 1). Free T3 was low, free T4 was low-normal, and TSH was within the normal range. This pattern excluded primary hypothyroidism, which would be characterized by elevated TSH levels. Further evaluations showed negative thyroid autoantibodies and a normal thyroid ultrasound, effectively excluding autoimmune thyroiditis and structural thyroid disease. Other pituitary axes were intact, ruling out panhypopituitarism. A comprehensive metabolic panel was unremarkable, and inflammatory markers were within normal limits, excluding an occult systemic inflammatory or infectious process.
Serial Thyroid Function Tests, Weight, and Key Laboratory Parameters.
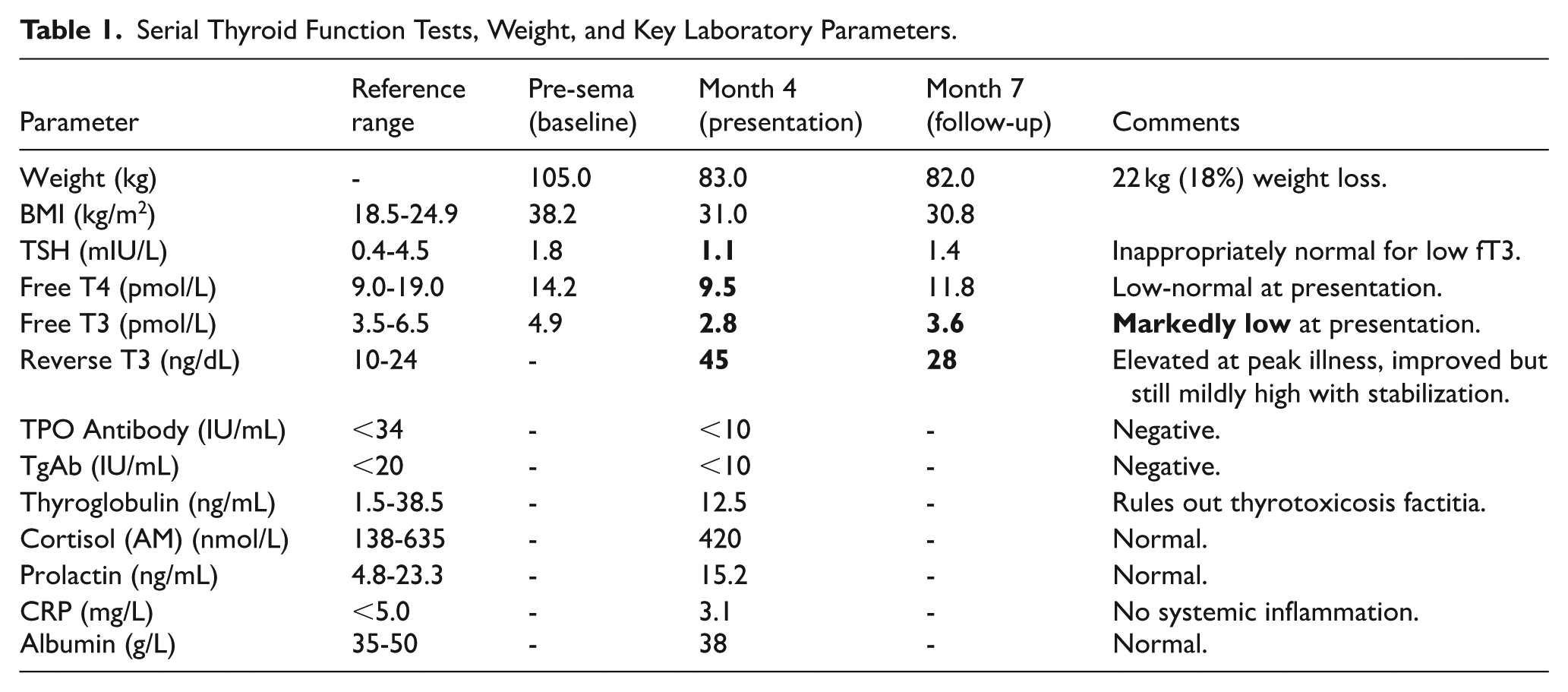
A diagnosis of euthyroid sick syndrome secondary to a catabolic state resulting from rapid weight loss was established. The patient was counseled extensively that this condition represented physiological adaptation rather than intrinsic thyroid dysfunction. Semaglutide therapy was continued due to its favorable impact on her metabolic comorbidities. She was advised to maintain adequate caloric and protein intake to mitigate ongoing catabolism.
At 3-month follow-up, her weight stabilized at 82 kg, and her symptoms of fatigue and cold intolerance had markedly improved. Repeat thyroid function testing demonstrated a trend toward normalization, with free T3 levels rising into the low-normal range and stable TSH levels (Tables 1 and 2). Thyroid hormone replacement therapy was not initiated.
Clinical Course Timeline of Semaglutide-Induced Euthyroid Sick Syndrome.

Discussion
GLP-1 receptor agonists such as Semaglutide have transformed obesity management through their potent effects on appetite suppression, gastric emptying, and central modulation of satiety. 10 Despite their widespread use, the endocrine adaptations accompanying pharmacologically induced weight loss remain incompletely understood, particularly in relation to thyroid hormone homeostasis.
This report presents an unusual instance of Euthyroid Sick Syndrome (ESS) following rapid, semaglutide-induced weight loss. ESS, or non-thyroidal illness syndrome (NTIS), represents a transient alteration in thyroid hormone metabolism that develops in the absence of an intrinsic thyroid pathology. It is characterized biochemically by reduced triiodothyronine (T3), low or low-normal thyroxine (T4), and inappropriately normal or low thyroid-stimulating hormone (TSH) concentrations.11,12 These alterations are regarded as adaptive mechanisms that minimize energy expenditure and protein catabolism during systemic illness, starvation, or caloric restriction. 13
As euthyroid sick syndrome (ESS) refers to alterations in thyroid hormone levels observed during acute or chronic systemic illness in the absence of intrinsic thyroid disease. ESS pathophysiology is multifactorial and involves disturbances in both peripheral thyroid hormone metabolism and central hypothalamic–pituitary regulation. A key mechanism is the altered activity of iodothyronine deiodinases. In critical illness, type-1 deiodinase activity is reduced, leading to decreased peripheral conversion of thyroxine (T4) to the biologically active triiodothyronine (T3). Concurrently, increased type-3 deiodinase activity promotes the conversion of T4 to reverse triiodothyronine (rT3), an inactive metabolite, resulting in reduced circulating T3 levels and accumulation of rT3. 11
Furthermore, inflammatory cytokines such as interleukin-1 (IL-1), interleukin-6 (IL-6), and tumor necrosis factor-α (TNF-α) suppress hypothalamic thyrotropin-releasing hormone (TRH) and pituitary thyroid-stimulating hormone (TSH) secretion and inhibit peripheral T4-to-T3 conversion.11,14,15 These mechanisms collectively result in the characteristic biochemical pattern of low serum T3 and elevated rT3 seen in ESS.
In the present case, an 18% body weight reduction within 4 months of semaglutide initiation, resulting in a catabolic metabolic state. Such rapid weight loss is known to suppress hepatic type-1 deiodinase activity, which converts T4 to T3, while increasing type-3 deiodinase activity, responsible for generating reverse T3 (rT3). The outcome is a characteristic decline in circulating T3 with an elevation in rT3 levels, reflecting an energy-conserving physiological adaptation.10,13
The temporal association between Semaglutide initiation, marked weight reduction, and subsequent thyroid profile suggests a causal link mediated by the catabolic consequences of sustained caloric restriction. Previous studies investigating thyroid function during rapid weight loss, have documented a similar pattern of decline in circulating T3 without compensatory TSH elevation. 16
Although current evidence indicates no direct thyrotoxic or goitrogenic effect of semaglutide, the metabolic consequences of rapid caloric restriction can transiently influence thyroid hormone dynamics.17,18 A recent systematic review confirmed that GLP-1 receptor agonists may be associated with mild, reversible alterations in thyroid function tests, without inducing clinical thyroid disease. 19 These findings support the interpretation that the abnormalities observed in this patient are secondary to metabolic adaptation rather than drug-induced thyroid dysfunction.
Clinically, the differentiation between ESS and true hypothyroidism carries critical therapeutic implications. The absence of TSH elevation and the spontaneous normalization of thyroid parameters upon weight stabilization in this case reinforce the appropriateness of observation rather than thyroid hormone replacement. Initiating levothyroxine in such settings could interfere with endogenous adaptive mechanisms and may expose patients to iatrogenic thyrotoxicosis. Thus, thyroid function tests in individuals undergoing rapid weight loss should be interpreted in the context of metabolic state and overall clinical stability, rather than as isolated laboratory abnormalities.
An additional clinical consideration relates to the overlap between symptoms associated with euthyroid sick syndrome and the gastrointestinal adverse effects commonly reported with GLP-1 receptor agonists. Agents such as semaglutide frequently induce gastrointestinal symptoms including nausea, delayed gastric emptying, and constipation due to their effects on gastrointestinal motility and appetite regulation, and these manifestations may complicate the clinical assessment of patients undergoing rapid pharmacologically induced weight loss, as symptoms such as fatigue, reduced appetite, or altered bowel habits may be attributed either to medication-related effects or to metabolic adaptations accompanying caloric restriction. Constipation has increasingly been recognized in individuals treated with GLP-1 receptor agonists, likely reflecting delayed gastrointestinal transit and altered colonic motility. Awareness of this overlap is important to avoid misinterpretation of symptoms and to ensure appropriate clinical monitoring during therapy. 20
Another key implication highlighted by this case is the importance of maintaining adequate caloric and protein intake during rapid weight loss induced by GLP-1 receptor agonists. These agents significantly suppress appetite and may reduce total energy intake by up to 40%, which can predispose individuals to insufficient macronutrient and micronutrient intake if dietary counseling is not provided. Emerging evidence suggests that patients receiving GLP-1 receptor agonists may fail to meet recommended protein requirements during hypocaloric diets, potentially increasing the risk of lean body mass loss and metabolic adaptations associated with rapid weight reduction. Therefore, clinicians should emphasize on balanced nutritional intake, with adequate protein consumption and dietary monitoring.21,22
This case emphasizes the need for clinician awareness of transient thyroid adaptations during Semaglutide-induced weight loss. Future prospective studies examining thyroid hormone kinetics in GLP-1 receptor agonist users could further clarify the prevalence, duration, and clinical significance of such biochemical changes.
Conclusion
The ESS in this case was considered due to the highly catabolic effect on the body through rapid weight loss period after initiation GLP-1 agonist drug (Semaglutide), as a physiological adaptation action in the body in response to the temporary alteration of the thyroid enzymes activity, our recommendation that thyroid function test should be considered for following up in symptomatic patients experiencing a rapid weight loss using GLP-1 agonists.
Footnotes
Ethical Considerations
IRB: Not applicable.
Consent to Participate
Consent for participation was taken by signed consent form
Consent for Publication
Signed informed consent obtained directly from the patient.
Author Contributions
Ziad W. Elmezayen: Project administration; Resources. Farah Qrareya: Methodology; Writing – original draft. Abdallah Abdallah: Data curation; Writing – original draft. Hossam Salameh: Investigation; Writing – original draft. Waheed Qaisi: Conceptualization; Supervision; Validation; Writing – review & editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Original data generated and analyzed for this case report are included in this published article.
