Abstract
Background:
Semaglutide, a Glucagon-like peptide 1 (GLP-1) is highly effective as an anti-diabetic medication along with additional benefits of significant reduction in cardiovascular events and weight.
Objectives:
There is a lack of reliable evidence confirming the benefits of Semaglutide in the Pakistani cohort. Our study aims to ascertain the efficacy of Semaglutide in the Pakistani cohort and patient satisfaction.
Methods:
An ambi-directional cohort study was conducted from August 2022 to January 2023 at The Aga Khan University Hospital, Karachi, Pakistan. A chart review was done for all patients with Type 2 diabetes who were prescribed Semaglutide, and baseline data was recorded. Patients were followed prospectively in the endocrinology clinics after at least 3 months for the change in HbA1c, BMI, adverse effects profile, treatment satisfaction questionnaire for medications (TSQM-9) and medication effect score. Mean + STD or median with IQR were computed for continuous data, while categorical variables and percentages were measured. The association between variables was assessed by applying the chi-square test.
Results:
A total of 112 patients were recruited who took the medication for at least 3 months. The mean age of the patients was 50.9 ± 10.5 years with 51.2% females. The mean difference in weight, body mass index, fasting blood glucose, HbA1c, and medication effect score were decreasing from baseline to follow-up (−4.0 kg, −1.5 kg/m2, −23.1 mg/dl, −1.2%, −0.4) respectively. A decrease in appetite was reported in 72.7% of the participants. The major adverse effects observed were dyspepsia (21.4%) followed by nausea (20.5%) and constipation (19.6%). Medication was discontinued in 9.8% of patients due to gastrointestinal side effects. The majority (72.3%) of patients were satisfied with their medication.
Conclusion:
Semaglutide is effective in reducing HbA1c and weight in Pakistani population with measurable tolerability and patient satisfaction.
Introduction
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder with multifactorial pathogenesis that can cause numerous complications that, in addition to affecting the quality of life, also reduce life expectancy. Diabetes is the ninth leading cause of mortality worldwide and >one-third of deaths from diabetes are occurring in individuals below the age of 60 years. 1 The risk of T2DM rises exponentially as the body mass index (BMI) increases above 25 kg/m2 and it is expected that the prevalence of obesity-related diabetes will double to 300 million by 2025. 2 Hence, treatment strategies targeting both weight and glycemic control are the need of the hour while managing patients with T2DM. 3
Glucagon-like peptide 1 receptor agonists (GLP-1 RAs), the synthetic version of the body’s naturally occurring incretin hormone that is, GLP-1, in addition to their role in managing diabetes, have various beneficial nonglycemic clinical effects notably weight reduction and cardiovascular risk reduction. These agents augment insulin release with concomitant suppression of glucagon, hence improving glycemic control. GLP-1 RAs promote weight loss by various receptor-dependent and receptor-independent mechanisms including delaying gastric secretion and motility, promoting the sensation of satiety, and decreasing food intake4,5.
Semaglutide, the once-weekly GLP-1 RA, is highly effective as a medication for diabetes with additional benefits in terms of reduction in weight and cardiovascular adverse events. 6 This is also proven through the SUSTAIN-6 trial which has shown a reduction in body weight (2.9-4.4 kg), systolic blood pressure (SBP) (3.4-5.4 mmHg), and glycated hemoglobin (HbA1c; 1.1%-1.4% [11.9-15.4 mmol/mol]), along with reductions in total cholesterol and serum triglycerides with the use of Semaglutide as compared to placebo. 7 Post hoc analysis of sustain trials has shown that 0.5 and 1.0 mg of Semaglutide lead to an HbA1c reduction of 1% to 1.5% (10.7-16.4 mmol/mol) and 1.3% to 2% (14.2-22.3 mmol/mol) from baseline along with weight reduction by 2.3 to 4.7 kg and 3.6 to 6.1 kg respectively. 8 A retrospective evaluation of 189 patients with T2DM initiated on Semaglutide has shown mean HbA1c reductions of 1.2% (13.3 mmol/mol) and 1.5% (16.4 mmol/mol) at 6 and 12 months respectively and mean weight loss of 3 kg at 6 months. 9
Semaglutide, like other GLP-1 RA, has predominantly gastrointestinal adverse effects; however, the drug is usually well-tolerated. 10 Among gastrointestinal symptoms, nausea is the most commonly reported (⩾10%), whereas vomiting, constipation, diarrhea, abdominal pain, and dyspepsia are relatively common (1-10%). 11
Although GLP-1 RA is frequently used in the management of T2DM in the Pakistani population, there is a lack of reliable evidence confirming the benefits of Semaglutide in the Pakistani cohort.12,13 Moreover, South Asian population was not included in Sustain trials.7,8 Our study aimed to examine the real-world impact of Semaglutide on glycemic control and weight reduction in South Asian population of Pakistani origin as well as to assess the tolerability of medication in terms of side effects experienced by the patients, the convenience of use, and overall satisfaction of patients with the medication.
Methods
Study population
An ambi- directional cohort study was conducted from August 2022 to January 2023 at The Aga Khan University Hospital, Karachi, Pakistan (AKUH). A thorough ethical review was supervised by the Ethical Review Committee of AKUH after which the study was conducted (2022-7520-21896). Informed consent was taken from the patients for conducting an interview at the end of the study visit. Written informed consent was taken from the patients who visited endocrinology clinic. For those patients who could not visit the clinic, verbal informed consent was taken through telephonic call. The process of verbal as well as written consent was approved by the Ethical Review Committee of AKUH prior to initiation of study. A total of 135 patients with T2DM were prescribed Semaglutide at the endocrinology clinic of AKUH during the study period. Baseline data of all these patients was collected by reviewing their medical records. However, 9 patients were lost to follow up and 14 patients took Semaglutide for <3 months. Hence, 112 patients were included in the final analysis.
Inclusion and exclusion criteria
All the patients with T2DM aged 18 to 65 years who were prescribed Semaglutide and took it for at least 3 months were included in the study.
Patients who were prescribed Semaglutide for an indication other than T2DM were excluded.
Study variables and measurements
Baseline and follow-up data were extracted by reviewing the medical records of the patients who took the medication for at least 3 months. The mean duration of follow up was 150.3 ± 54 days. Data collected at baseline included demographic factors (age, gender), clinical characteristics (duration of diagnosis of T2DM, drug regimen, comorbidities, complications of T2DM), lifestyle factors, weight, and BMI. Fasting blood glucose, mean HbA1c and creatinine were measured. At the follow-up visit, fasting blood sugar, HbA1c, and BMI were recorded. An interview was conducted by either face to face or telephonic conversation at the end of the study. Tolerability was assessed by questioning about side effects at the end of the study visit. Any change in the symptoms with a change in dose was noted. Patients were questioned about the effect of Semaglutide on appetite if any. In those patients who had discontinued the medication without medical consultation, the cause was ascertained. Any change in the dosage of other anti-diabetic medications with concomitant use of Semaglutide was noted. The medication effect score reflects overall intensity of a diabetes regimen by consolidating dosage and potency of agents used. The medication effect score was calculated at baseline and follow-up to assess the change in intensity of the treatment regimen after the introduction of Semaglutide. 14 Semaglutide was not included in the medication effect score at the follow up as the aim was to observe the change in doses and number of other antidiabetic medications. A treatment satisfaction questionnaire for medication version 9 (TSQM-9) was used to assess the satisfaction of patients taking Semaglutide. 15 Diabetes Treatment Satisfaction Questionnaire (DTSQ) is currently not licensed for use in Pakistan and could not be obtained despite multiple attempts. 16
Statistical Analysis
STATA version 14 was used to analyze the data. Mean with standard deviation or median with Inter Quartile Range (IQR) were computed for continuous data after assessing the normality assumption, while for categorical variables, frequencies and percentages were measured. Chi-squared test and student t-test were used to compare the variables from baseline to follow-up. A P-value of <.05 was considered significant with a confidence level of 95%.
Results
Demographics and baseline characteristics
This study included 112 patients having a mean age of 50.9 (±10.5) years. Among them, 51.8% were females (n = 58). The mean diabetes duration of 8 years. Most of the patients had dyslipidemia and hypertension at the time of initiation of Semaglutide while coronary artery disease was documented in 17% (n = 19).
Before initiating Semaglutide, metformin and SGLT-2 inhibitors were the most used medications. Baseline medications and key characteristics are provided in Table 1.
Baseline characteristics of patients (n = 112).

Abbreviations: CABG, coronary artery bypass graft; SGLT2, sodium-glucose co-transporter-2; TZD, thiazolidinedione; DPP4, dipeptidyl peptidase IV; HbA1c, glycated hemoglobin; MES, medication effect score.
The MES is calculated for each diabetes medication in a regimen using the following equation: (actual drug dose/maximum drug dose) ×drug-specific adjustment factor. Subgrouping according to control based on HbA1c.* Group 1: well controlled diabetes (HbA1c < 7%). Group 2: partially controlled diabetes (HbA1c 7-9%). Group 3: poorly controlled diabetes (HbA1c > 9%).
Comparison of baseline and follow-up investigations
The comparison of baseline and follow-up fasting blood glucose, HbA1c, weight, Body mass index and medication effect score is reported in Table 2. In 86 patients, Semaglutide was initiated at a dose of 0.25 mg (76.85%) whereas 20 patients were given 0.5 mg at the start (17.9%) and only 6 patients (5.4%) were given a dose of 1 mg. The mean dose of Semaglutide at baseline was 0.33 mg. An increase in the dose of Semaglutide was observed at follow-up with 64 patients taking 0.5 mg (57.1%) and 31 patients taking 1 mg (27.7%). The dosage of 0.25 mg was continued in only 17 patients (15.2%). The mean dose of Semaglutide at follow up was 0.60 mg. The mean HbA1c at follow up was 7.0 ± 0.8, 7.0 ± 1.1 and 6.7 ± 1.1 in patients taking 0.25, 0.5 and 1 mg of Semaglutide, respectively.
Comparison of variables from baseline to follow up.
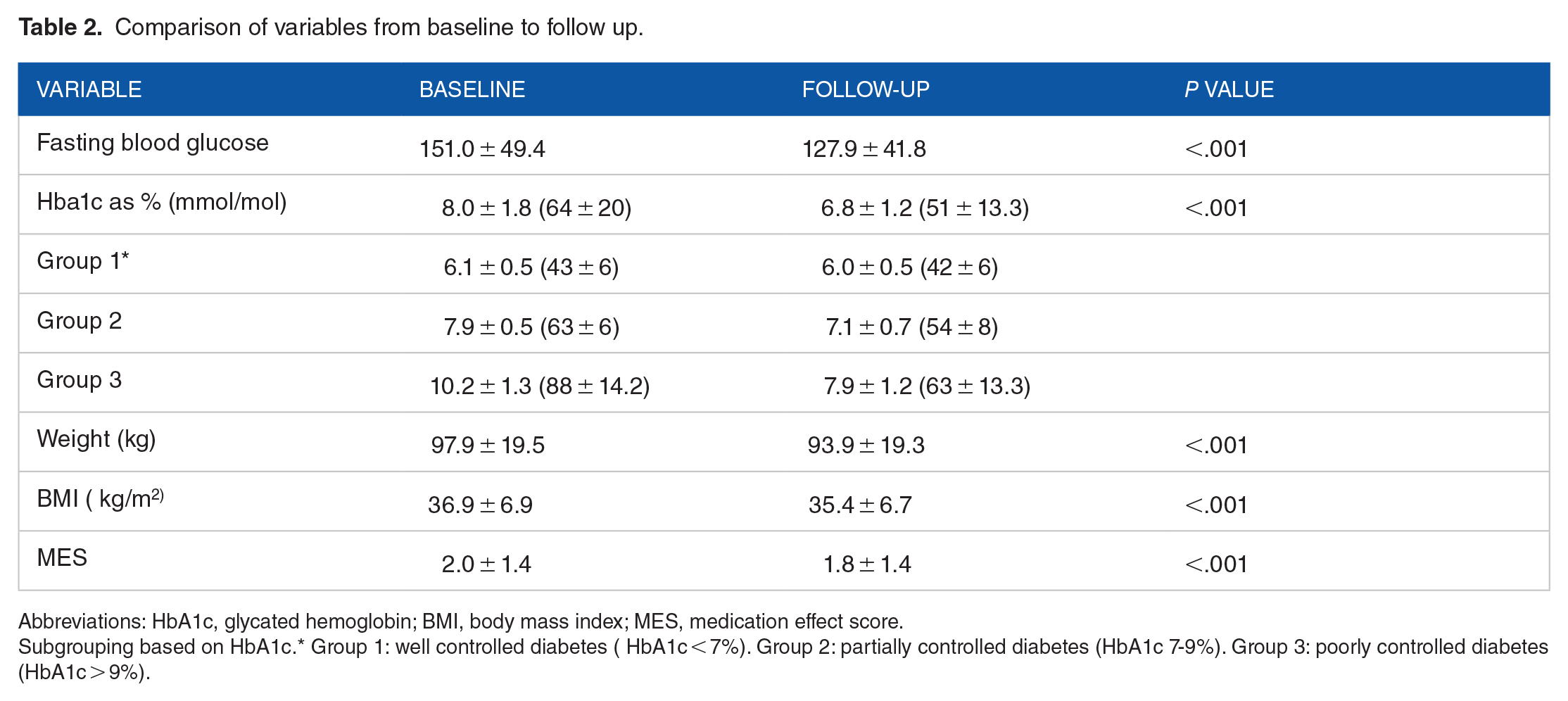
Abbreviations: HbA1c, glycated hemoglobin; BMI, body mass index; MES, medication effect score.
Subgrouping based on HbA1c.* Group 1: well controlled diabetes ( HbA1c < 7%). Group 2: partially controlled diabetes (HbA1c 7-9%). Group 3: poorly controlled diabetes (HbA1c > 9%).
Tolerability analysis
Semaglutide was discontinued by 11 patients at follow-up (9.8%). The reasons for discontinuation were gastrointestinal adverse effects in 10 patient (90.9%) and cost in 5 patients (45.4%).; dyspepsia was reported in 20 patients (17.8%) while 15 (13.3%) patients had nausea at the starting dose of Semaglutide. At the end of study visit, dyspepsia was still the major adverse effect reported in 24 (21.4%) patients whereas 23 (20.5%) patients had complaint of nausea. Many patients reported a decrease in their appetite while taking the medicine (80, 72.7%). Tolerability analysis is further reported in Table 3, Figure 1a and b.
Adverse effects experienced with Semaglutide.
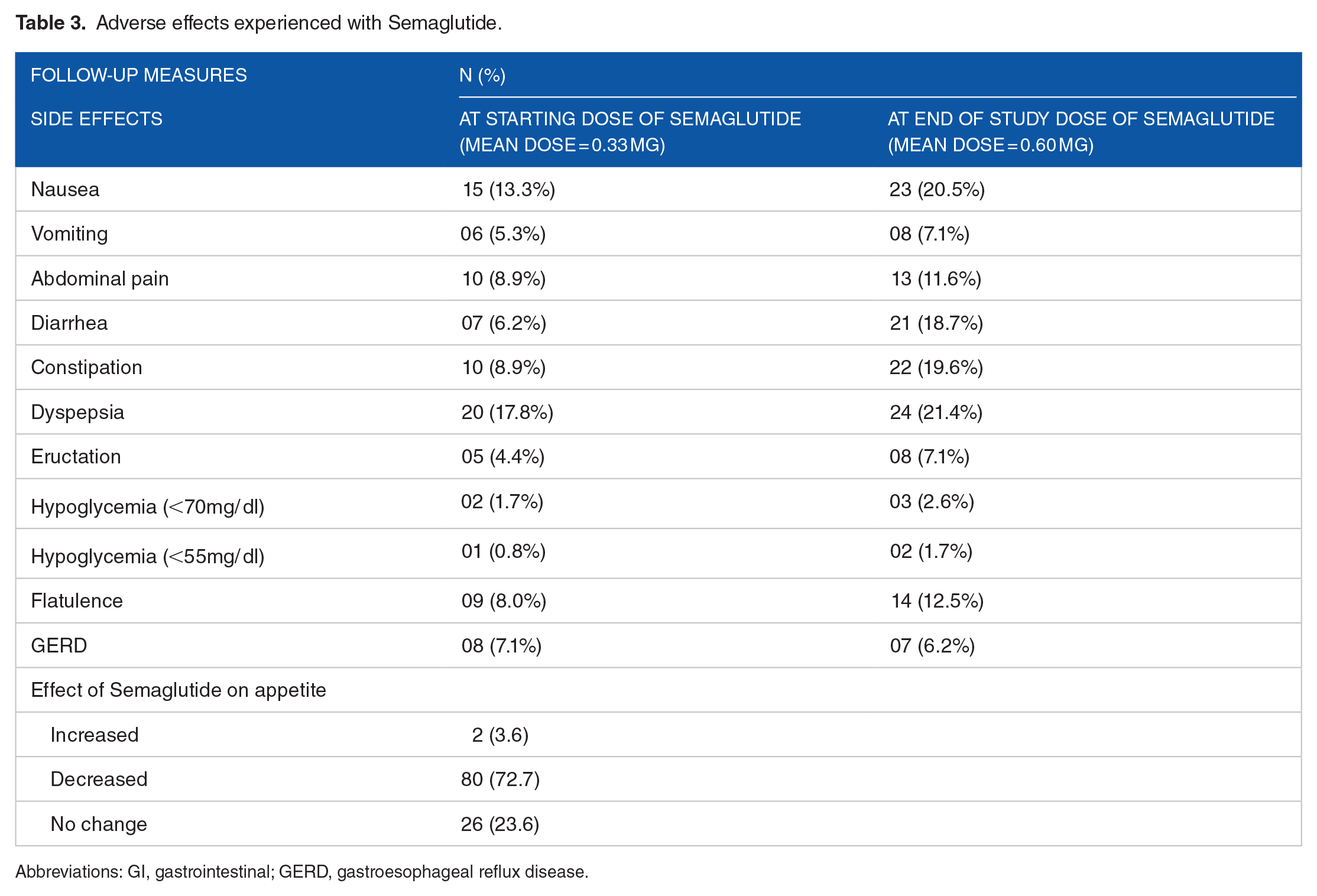
Abbreviations: GI, gastrointestinal; GERD, gastroesophageal reflux disease.

Adverse effects are various doses are plotted across x-axis. Percentages showing frequency of adverse effects are plotted across Y axis. (a) shows the frequency of major adverse effects at various starting doses of Semaglutide and (b) shows the frequency of major adverse effects at end of study visit at various doses of Semaglutide.
When questioned about their satisfaction regarding the treatment of their condition with Semaglutide, 81 patients reported satisfaction with the drug (72.3%) and 106 patients found it easy to use Semaglutide in its current form (94.7%).
Comparison in doses of other medication at baseline and follow-up
DPP4 inhibitors were stopped in all patients after initiating Semaglutide. Metformin was prescribed at a mean dose of 1388.1 at baseline which decreased to 1304.3 at follow-up. The mean dose of insulin was 69 units at baseline while it decreased to 55 units at follow-up; however, no difference in the mean number of injections was noted at follow-up. The comparison in doses of other medication at baseline and follow-up are reported in Table 4.
Comparison in doses of other medications from baseline to follow-up.

Abbreviations: SGLT2, sodium-glucose co-transporter-2; TZD, thiazolidinedione; DPP4, dipeptidyl peptidase IV.
Discussion
This study shows that Semaglutide is beneficial in reducing fasting blood glucose levels, HbA1c, weight, body mass index, and medication effect score in Pakistani patients with T2DM. The results are comparable to those observed in SUSTAIN 5 trial which showed a mean reduction in HbA1c of 1.4% (15.8 mmol/mol) and 1.85% (20.2 mmol/mol) along with weight loss of 3.7 kg and 6.4 kg at 0.5 mg and 1 mg of Semaglutide, respectively at 30 weeks of treatment. 17 In our study, at follow-up, 57.1% of the patients were receiving 0.5 mg of Semaglutide and 27.7% were taking 1 mg with a mean reduction of HbA1c, weight, and BMI of 1.2% (13.3 mmol/mol), 4 kg, and 1.5 kg/m2, respectively. Study results are also consistent with real-world studies conducted in other countries. In Canada, a study based on the retrospective analysis of a diabetes registry (SPARE study) on 937 naïve patients documented a statistically significant mean reduction in HbA1c of 1.03% (11.3 mmol/mol) and in weight of 3.9 kg, with no significant change in the self-reported incidence of hypoglycemia. 18 Similarly, in the SURE Denmark/Sweden cohort (n = 331), the use of Semaglutide was associated with HbA1c reduction of 1.2% (13.3 mmol/mol) and weight loss of 5.4 kg. 19 Finally, in the clinic-based analysis of 189 patients in Wales, HbA1c was reduced by 1.5% (16.4 mmol/mol) and weight by 3 kg after 6 months. 9
Global Burden Disease Study (2019), reported that there is a rapid increase in the incidence of cardiovascular diseases (CVDs), cancer, and diabetes in Pakistan and these may be the 3 leading causes of death until 2040. 20 Hypertension and coronary artery disease were 2 commonly reported comorbidities in our study with 13 (11.6%) patients being previously treated with percutaneous coronary intervention (PCI) and 3 with coronary artery bypass graft (CABG). Semaglutide is reported as a safe drug in cardiovascular patients. This is also proved in the SUSTAIN-6 trial where the patients of T2DM with a high risk of CVDs had a lower chance of non-fatal MI, non-fatal stroke, and death. 7
Subsequently, the association of obesity with T2DM has been an established notion through various studies. T2DM and obesity both are considered a major peril for CVD. 21 However, after the use of Semaglutide, the mean weight, and BMI reduced to 4 kg and 1.5 respectively, showing a positive change in the data. Thus, the use of Semaglutide in T2DM patients can not only prevent cardiovascular diseases but also keep obesity at bay.7,17,22
In our study, a decrease in medication effect score of 0.4 was observed at follow up showing that Semaglutide therapy could lead to dose reduction of other anti-diabetic medications as well as insulin. The mean dose of insulin decreased from 69 units at baseline to 55 units at follow-up. In SUSTAIN 5 trial, the end-of-treatment to baseline ratio for insulin dose with Semaglutide 0.5 mg and Semaglutide 1.0 mg was 0.90 and 0.85 at 30 weeks which in our study was 0.79. 17
Semaglutide was generally well tolerated, with GI symptoms reported as the most common adverse effects, as it delays gastric emptying. 23 The GI symptoms were responsible for the discontinuation of treatment prematurely in 11 patients (9.8%). This is lower than the discontinuation rate of 13.1% observed in the SUSTAIN-6 trial; the period of drug exposure in SUSTAIN-6 was, however, longer at 2 years. 6 Dyspepsia was the most reported adverse effect (27.7%) in our study followed by nausea and constipation. One possible explanation for high rate of dyspepsia at various doses of Semaglutide could be high prevalence of functional dyspepsia in Pakistani population. 24 Despite the adverse effects, 72.3% of the patients were satisfied with the medication and 78.6% reported it to convenient to use considering the injectable nature of medication. Our study also documented that Semaglutide can be used in patients with newly diagnosed as well as more advanced stages of diabetes, supporting its benefits in patients with a wide range of baseline characteristics that is, age, duration of diabetes, and background lowering glucose-lowering treatments including insulin. However, in this study, no subgroups were defined based on prior GLP-1 analog, baseline glycemic control, or baseline metabolic profile. This subgrouping would have been further beneficial in identifying the target population who would achieve maximum benefits from Semaglutide. Furthermore, in our study, the lipid profile, serum ALT and blood pressure of the patients were not considered. Given the cardiovascular superiority of Semaglutide in SUSTAIN 6 trial, the observation of these parameters would be important in future studies.6,7
Strengths and Limitations
The main strength was that this is the first Pakistani study documenting the real-world impact of Semaglutide on glycemic control and weight as well as looking at the adverse effect profile and treatment satisfaction of the medication. However, being an observational study, it is subjected to various limitations. Firstly, the interval between baseline and follow-up collection data was variable. Secondly, the effects of several confounding variables for example, diet and physical activity were not monitored at follow-up. Thirdly, due to the short duration of the study, the persistence of therapeutic response could not be predicted. Fourthly, recall bias could not be ruled out while questioning the adverse effects. Another limitation of our study is that power analysis for sample size calculation was not done. Finally, real-world assessment of the impact of Semaglutide on major adverse cardiovascular events, as well as assessment of the impact on microvascular outcomes was limited by the short duration of follow-up and would be an important observation in future studies with longer follow-up.
Conclusion
In conclusion, Semaglutide was associated with significant glycemic and weight-loss benefits in adults with T2D, supporting its real-world use in all stages of diabetes disease. Gastrointestinal adverse effects, although frequently reported, were generally well tolerated as reflected by adequate satisfaction of the patients as well as good adherence to the medication at the end of the study. Hence, we support the use of Semaglutide in heterogenous population with T2DM.
Supplemental Material
sj-docx-1-end-10.1177_11795514231213568 – Supplemental material for Efficacy and Tolerability of Semaglutide in Patients With Type-2 Diabetes Mellitus: Experience of a Tertiary Care Hospital, Pakistan
Supplemental material, sj-docx-1-end-10.1177_11795514231213568 for Efficacy and Tolerability of Semaglutide in Patients With Type-2 Diabetes Mellitus: Experience of a Tertiary Care Hospital, Pakistan by Maria Wajid, Saadia Sattar, Farah Khalid and Muhammad Qamar Masood in Clinical Medicine Insights: Endocrinology and Diabetes
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
