Abstract
The global rise in diabetes mellitus presents a major healthcare challenge due to its associated complications. Effective glycemic control, crucial for reducing diabetes-related morbidity and mortality, encompasses 3 key components: fasting plasma glucose (FPG), postprandial plasma glucose (PPG), and glycated hemoglobin (HbA1c). While FPG and HbA1c are commonly used for evaluating glycemic control, PPG also significantly influences overall glucose management. Postprandial hyperglycemia, the early deviation in type 2 diabetes mellitus (T2DM), plays a dominant role in individuals with near-target HbA1c levels. Advances in continuous glucose monitoring (CGM) provide a more comprehensive understanding of PPG fluctuations, offering real-time data and reducing the limitations of traditional monitoring methods. CGM technology revolutionizes glycemic monitoring, enhancing the management of PPG and supporting better diabetes care. This review emphasizes the importance of monitoring and managing PPG throughout the postprandial state in individuals with diabetes. It further consolidates evidence highlighting the importance of viewing PPG as a continuum and the potential of CGM in improving PPG management.
Plain language summary
The growing number of people with diabetes around the world is a major heath challenge due to complications associated with it. Hence, effective blood sugar level control is essential to reduce risk of adverse conditions associated with diabetes. Blood sugar level before meal, blood sugar level after meal, and glycated hemoglobin are the three important key factors which identify blood sugar characteristics. High blood sugar level after meal is an early sign of type 2 diabetes mellitus (T2DM). Continuous blood sugar monitoring (CGM) is a technique that gives an understanding of blood sugar levels throughout the day, which may potentially result in better care for people with diabetes mellitus. This article explores the recognition of a continuum for blood sugar level after meals. This recognition has been possible due to increasing use of CGM in diabetes management. Further, this article also emphasizes on the need for monitoring and managing blood sugar levels after meal as a continuum rather than at specific time.
Keywords
Introduction
The escalating prevalence of diabetes mellitus on both the global and national scale poses a significant challenge for healthcare systems. This is primarily attributed to the development of various complications associated with diabetes, leading to increased morbidity and mortality. As per International Diabetes Federation (IDF) report, 10.5% of the adult population (20-79 years) has diabetes and an estimated 240 million people are living with undiagnosed diabetes worldwide, translating to almost one-in-two adults with diabetes being unaware about their condition. The IDF projections show that by 2045, 1 in 8 adults, approximately 784 million, will be living with diabetes, an increase of 46% over the next 25 years. 1 More than 80% of people with diabetes reside in low and middle income countries. A recent report from the Indian Council of Medical Research–India Diabetes (ICMR-INDIAB) study estimated that over 100 million adult individuals in India have diabetes, representing a prevalence of 11.4%. 2 Effective management of blood glucose levels in diabetes mellitus is a fundamental aspect of reducing the negative health impact of the disease. Attaining glycemic control is associated with lowering the risks of both microvascular and macrovascular complications linked with diabetes. Recent evidences from real-world studies provide compelling evidence highlighting the challenge of inadequate glycemic control despite availability of various treatment options for diabetes management.3-6 Therefore, achieving and maintaining glycemic control are crucial aspects in the management of diabetes.
Applying principles of clinical pharmacology plays a pivotal role in optimizing diabetes care, particularly in managing complex medication regimens and addressing treatment non-adherence. Incorporating clinical pharmacological reconciliation review and feedback (CPRRF) into the diabetes management team ensures that therapeutic interventions are tailored to each patient’s unique clinical and pharmacokinetic profiles. 7 This co-professional support facilitates better understanding of drug-drug interactions, adverse drug reactions (ADRs), and the importance of medication reconciliation. A recent cross-sectional observational study demonstrated that patients receiving clinical pharmacological consultation exhibited significantly higher awareness of hypoglycemia management, self-monitoring of blood glucose (SMBG), insulin injection techniques, and ADR awareness. These patients also demonstrated improved treatment adherence and lower rates of drug-drug interactions, underscoring the crucial role of clinical pharmacologists in promoting patient safety and optimizing glycemic control. 8 As diabetes management becomes more patient-centric, the inclusion of applied clinical pharmacology care, CPRRF can further aid in achieving better and safer patient outcomes, ultimately improving both glycemic and non-glycemic targets.
It is widely acknowledged that effective glycemic control is contingent upon 3 key components that form the glucose triad: fasting plasma glucose (FPG), postprandial plasma glucose (PPG), and glycated hemoglobin (HbA1c). Although the most commonly practiced methods for evaluating glycemic control involve HbA1c and fasting glucose measurements, it is crucial to recognize that PPG also plays a significant role in the overall management of blood glucose levels. 9 In individuals with previously undiagnosed diabetes, when they underwent various tests for diabetes diagnosis, it was observed that HbA1c identified 30% of cases and 2-hour PPG identified 90% of cases. 10 The PPG have has also shown stronger correlation with HbA1c compared to FPG, which significantly contributes to overall glycemic control. 10 Hence, it is essential to give special attention to PPG both for identifying new cases and for monitoring in those with existing diabetes. 11 Postprandial hyperglycemia, characterized by high post-meal blood glucose levels, represents one of the initial deviations of glucose homeostasis associated with type 2 diabetes mellitus (T2DM). The balance of fasting and postprandial hyperglycemia undergoes a progressive change as hyperglycemia severity increases. In individuals with diabetes with blood glucose levels closer to target (HbA1c < 7.3%), postprandial glucose excursions play a predominant role, while the contribution of fasting hyperglycemia becomes more pronounced as glycemic level worsens. 12 Current treatment guidelines for T2DM recommend a 1 to 2 hour test post meal to monitor PPG.13-15 Though the 1 and 2 hour time frames are often used for monitoring PPG, studies have suggested that assessing postprandial peaks is complex and these values may not adequately reflect glycemic excursions because each value reflects only a single time point in PPG dynamics. With the advent of continuous glucose monitoring (CGM), PPG is being monitored longitudinally providing more valuable information about the situation. The purpose of this review is to gather evidence supporting the idea that PPG is a continuum in diabetes and therefore, it is crucial to monitor and manage PPG throughout this entire phase in individuals with T2DM.
Significance of PPG in Diabetes
PPG is primarily controlled by 3 separate mechanisms: gastric emptying, the release of insulin by β-cells, and the inhibition of glucagon secretion by α-cells. 16 Furthermore, the emergence of postprandial hyperglycemia occurs simultaneously with a decline in or absence of the first-phase insulin response, reduced insulin sensitivity in peripheral tissues, and impaired suppression of hepatic glucose production following meals attributable to insulin deficiency.17,18
A cross-sectional study including 605 T2DM patients was conducted with the aim of understanding the correlation of HbA1c with FPG and PPG. The results revealed that both FPG and PPG showed a very good correlation with HbA1c; however, correlation of HbA1c with PPG was higher when compared with that of FPG. 19 Furthermore, a systematic review and meta-analysis assessing the significance of FPG and PPG, and their correlation with HbA1c demonstrated that PPG is better correlated with HbA1c as compared to FPG and thus can be preferred to predict overall glucose control in the absence of HbA1c. 11 It has been reported that ethnicity is an important factor to be considered while deliberating the importance of contribution of PPG to HbA1c. Studies have revealed that in Caucasian populations contribution of FPG became significantly higher than PPG in T2DM patients as the HbA1c worsens; whereas, in Asian patients with T2DM, PPG was a predominant contributor to excess hyperglycemia in well-controlled patients and was equally important as FPG in moderately to poorly controlled patients with mean HbA1c up to 10%.20,21 STARCH study done in Indian patients with T2DM revealed that the diet of these patients had high carbohydrate content (64.1% of total energy from diet), which has a direct effect on postprandial blood glucose and insulin response. 22
The cholesterol rich apolipoprotein B, which contains lipoproteins is the most known cause of atherosclerotic cardiovascular disease (CVD). Atherosclerosis requires apoB containing lipoproteins and hyperglycemia increases their atherogenicity. 23 Hyperglycemia is associated with endothelial dysfunction that may promote CVD. 24 It is important to note that chronic hyperglycemia as well as periodic hyperglycemia can induce endothelial dysfunction. This dysfunction is characterized by an increase in local reactive oxygen species which accelerates a decrease in the vasodilatory nitric oxide. The resulting damage causes the expression of adhesion molecules resulting in an inflammatory response recruiting macrophages which engulf the damaged tissue forming foam cells. The influx of platelets into these foam cell populations cause the synthesis and secretion of various growth factors (platelet-derived growth factor (PDGF), epidermal growth factor (EGF), transforming growth factor-alpha (TGF-alpha)) that stimulate smooth muscle cell proliferation synthesizing collagen and extracellular matrix contributing to the formation of the nidus of an atherosclerotic plaque. The continued glycemic insult builds on the nidus to cause accelerated atherosclerosis.24,25 The complex nature of atherosclerotic plaques, including foam cells, lipid cores, fibrotic caps, necrosis, and calcification are caused due to retention as well as due to accumulation of low-density lipoprotein in subendothelial matrix. 26 An increasing volume of research has revealed a significant connection between PPG and cardiovascular risk and outcomes, as well as with factors such as oxidative stress, carotid intimal thickness, and endothelial dysfunction. 27 Various studies28-31 have observed postprandial hyperglycemia to be an independent risk factor of CVD and of mortality for patients with diabetes. The DECODE study 32 revealed high blood glucose levels 2h after an oral glucose overload was associated with a greater risk of death, regardless of blood glucose levels under fasting conditions. The San Luigi Gonzaga Diabetes study 33 concluded that blood glucose levels 2h after meals was an independent risk factor of cardiovascular disease.
Current Scenario of PPG Monitoring
In a healthy individual, a 24-hour day involving the intake of 3 meals is characterized by 3 distinct phases: fasting, postprandial, and postabsorptive states. Tentatively, the postprandial and postabsorptive states span 4 and 6 hours, respectively (Figure 1). Generally, the postprandial state, encompassing the time frame after a meal, constitutes an intricate physiological process pivotal for metabolizing nutrients and furnishing vital metabolic fuels to various tissues (Figure 2). During this period, the rise in plasma glucose levels, coupled with higher insulin secretion from the pancreatic β-cells, regulates postprandial glucose homeostasis by prohibiting endogenous hepatic glucose production and promoting the hepatic glucose disposal.34,35 The 6-hour period following the postprandial period is termed as the postabsorptive state. In this interval the glucose concentrations remain within a normal range in individuals without diabetes through glycogenolysis, that is, breakdown of the glycogen stored during the postprandial period. 36 The hepatic and extrahepatic insulin resistance are one of the responsible factors for exacerbation of postprandial glucose intolerance in impaired fasting glucose (IFG)/impaired glucose tolerance (IGT). 37 A study by Basu et al indicates that excessive glucose production is the cause of fasting hyperglycemia in people with either isolated IFG or combined IFG/IGT. Increased rates of gluconeogenesis are the cause of the excessive fasting glucose production in people with IFG/normal glucose tolerance (NGT) and, along with increased rates of glycogenolysis, probably contribute to increased fasting glucose production in people with IFG/IGT. 37 Another study by Varghese et al shows that subjects with isolated normal fasting glucose (NFG)/IGT have defects in peripheral, but not hepatic, insulin action. Although insulin-induced stimulation of glucose disappearance is impaired in people with NFG/IGT, insulin-induced suppression of endogenous glucose production, gluconeogenesis, and glycogenolysis is normal. This defect in extrahepatic insulin action is accompanied by an inability of β-cells to secrete sufficient insulin to overcome this defect, thereby causing glucose intolerance. 38
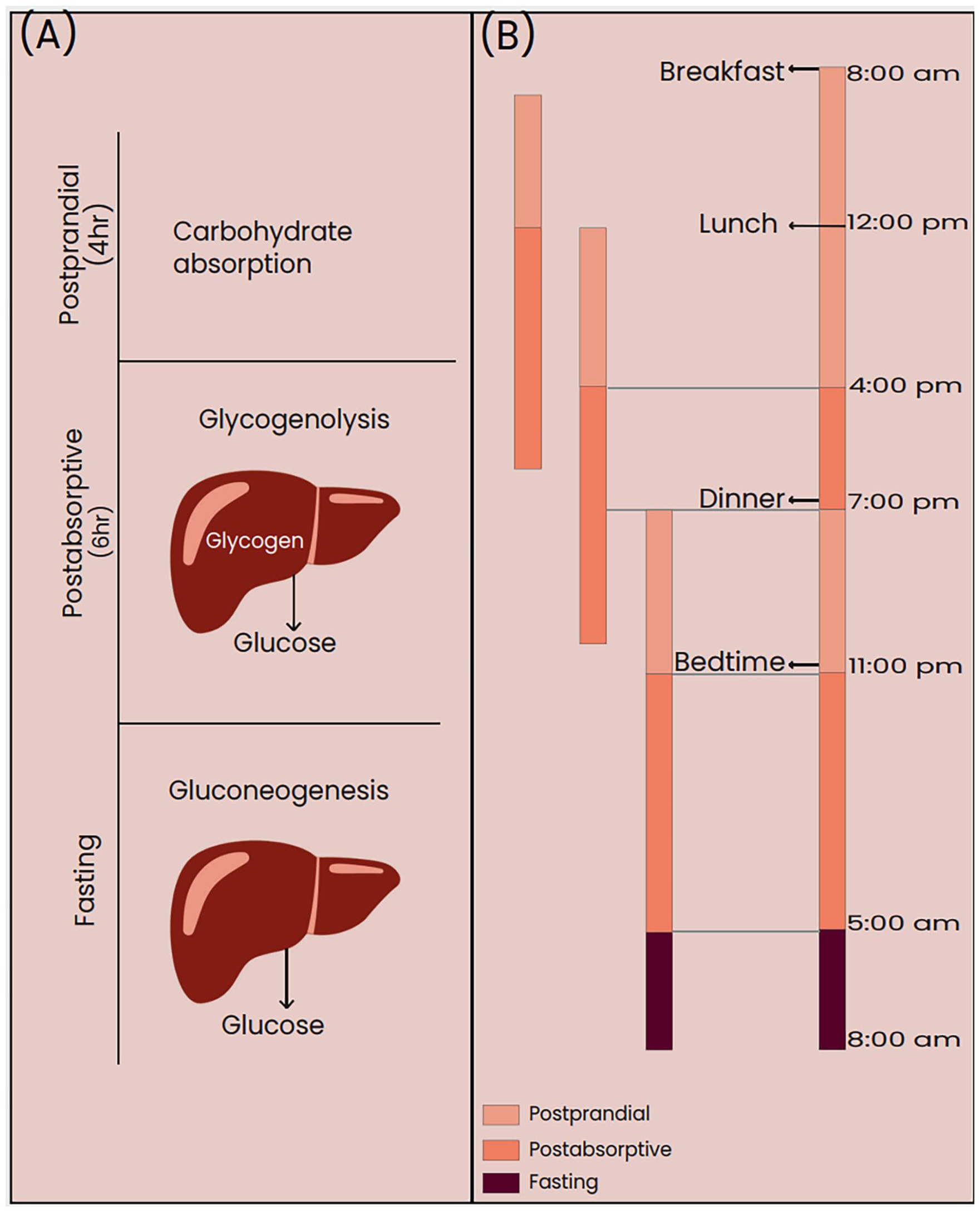
(A) Glucose metabolism and (B) duration of the postprandial, postabsorptive, and fasting states.

Glucose metabolism in the postprandial state.
While FPG and HbA1c are prioritized for diabetes management, evidence from observational studies suggest that testing glucose levels at postprandial and postabsorptive time points could also play a crucial role.20,39 The available guidelines suggest that PPG measurements should be taken 1 to 2 hours after the start of the meal. Additionally, these guidelines mention the importance of CGM in different scenarios among individuals with diabetes. SMBG is a method that involves intermittent fingerstick testing with capillary blood glucose meters, offering timely information to guide management. It is recommended that SMBG should be considered for monitoring PPG. The guidelines note that CGM systems could be a valuable tool for identifying postprandial hyperglycemia and enhancing therapeutic management in patients with T2DM. 15 They also mention the importance of CGM in the management of glycemia in different special populations with diabetes (such as elderly, pregnant women, insulin-dependent patients with diabetes and presence of comorbid conditions). 40
Preference of FPG Monitoring over PPG Monitoring
Although multiple guidelines for diabetes care recommend use of FPG and/or 2 hour-PPG monitoring, in resource-limited settings where HbA1c measurements are not available, there is no unanimous agreement among healthcare professionals on whether FPG or PPG serves as a more reliable indicator of glycemic control. 11
There are several reasons for the preference of FPG monitoring over PPG monitoring in routine clinical practice. PPG monitoring involves taking measurements at specific time points after eating, which can be more cumbersome. The PPG levels are influenced by an intricate interaction of biological, chemical, and physical elements. Additionally, various factors including carbohydrate intake, glycemic index and nutritional content of foods consumed, water intake habits, meal timings, physical activity, and stress41-47 also impact PPG levels in individuals with diabetes. FPG levels are less affected by day-to-day variations in meal composition and timing, which can make PPG measurements more variable. Furthermore, FPG measurements are typically taken after an overnight fast, ensuring consistency and eliminating the influence of recent meals, making the results more reproducible. FPG testing is less complex and more convenient for patients since it doesn’t require them to time their tests after meals. The preference for FPG testing also stems from the traditional training that emphasizes FPG as the initial focus, leading physicians to perceive PPG management as less crucial. 48
Common Timepoints to Monitor PPG Levels
The postprandial period is a finely orchestrated process that ensures the efficient utilization of nutrients from the meal to meet the body’s energy needs and maintain metabolic balance. Postprandial hyperglycemia is defined by the IDF as blood glucose levels exceeding 140 mg/dL within 1 to 2 hours after a meal. Similarly, the American Association of Clinical Endocrinologists and the American College of Endocrinology define it as blood glucose levels greater than 140 mg/dL specifically at 2 hours post-meal.40,49 Individuals with diabetes experience greater and more prolonged PPG excursions, with greater glycemic variability compared to those without diabetes.50-52
Usually, optimal timing considered for measuring PPG levels to assess glucose excursions is 2 hours after a meal. 53 The rationale for selecting the 2-hour time point for PPG measurement within the broader postprandial period is grounded in the distinct glucose response patterns observed between individuals with normoglycemia and those IGT/diabetes. In a healthy individual exhibiting normoglycemia, blood glucose levels typically peak approximately 30 minutes after eating. Conversely, in individuals with IGT, the peak in glucose levels is often delayed, occurring within the 1 to 2-hour timeframe post meal.52-54 Evidence, including recent IDF position statement, indicates that a 1 hour-PPG level ⩾ 155 mg/dL can serve as a valuable indicator for identifying individuals who are at an elevated risk for both T2DM and cardiovascular disease.55-58
In addition to the physiological reasons for choosing the 1 to 2-hour post meal window for PPG measurements, practical and technical limitations also play a significant role. Traditionally, monitoring PPG excursions throughout the entire postprandial period would require frequent blood sampling, which necessitates repeated needle pricks. The need for frequent sampling at shorter intervals would not only increase patient discomfort but also pose challenges in terms of time, resource allocation, and compliance. Therefore, the traditional practice of measuring PPG at a 2-hour interval, is the standard of care which has been in place for many years. However, these values should be evaluated with care as they actually reflect a complex combination of underlying insulin resistance, β-cell dysfunction, meal composition/timing, composition/timing of previous meals and daily activities, and not solely glycemic levels. Thus 1 and 2-hour PPG values should always be evaluated with care as they reflect only a single time point in postprandial phase and may not adequately reflect glycemic excursions.
PPG Monitoring Techniques
In recent years, developments in PPG monitoring techniques have greatly enhanced the ability to understand and manage blood glucose levels after meals. These advancements have had a significant impact on diabetes care and overall health. Currently, PPG is measured at particular time points using traditional methods of blood glucose monitoring in routine clinical practice and in various clinical studies. These include SMBG and laboratory tests using venous blood samples at different time points of the day. SMBG offers only snapshots of blood glucose concentration.59,60 and the constraints of SMBG encompass discomfort, inconvenience arising from the need for frequent testing throughout the day, and associated costs. 61 Similarly, blood glucose monitoring using venous blood in a commercial-grade laboratory with appropriate sophisticated quality control checks also has limitations. These include the potential for a painful procedure, the risk of local tissue damage, and its impracticality for frequent specimen collection.62,63 Even though these conventional methods remain the primary choice in routine clinical practice, it is crucial to recognize that these approaches do not enable continuous monitoring of glucose levels, thereby missing the glycemic excursions occurring during this period. The CGM helps to identify glycemic response of an individual to the particular diet that is consumed. This information is crucial and helps the individual make informed dietary choices in order to reduce the postprandial glucose. The CGM combined with dietary counseling has demonstrated improved glycemic reduction in T2DM patients. 64
Importance of Viewing PPG as a Continuum
Typically, PPG in healthy individuals refers to the concentration of glucose in the bloodstream after consuming a meal. In individuals with diabetes, the postprandial state, defined by the hydrolysis and absorption period of dietary carbohydrates, generally mirrors that observed in individuals without diabetes, unless there is delayed gastric emptying. Post meal a gradual decline in glucose absorption occurs over time and the total duration of absorption extends to approximately 4 hours, corresponding to the postprandial state. The subsequent postabsorptive state encompasses a 6-hour period, during which plasma glucose concentrations consistently remain within the normal range. Therefore, the postprandial state with respect to glucose is a state that represents the rise in blood glucose levels within a 4-hour period immediately following the ingestion of a meal (Figure 1). 36 Consequently, the combined duration of the postprandial states approximates 12 hours (assuming 3 main meals in a day), equivalent to a full half-day period and generally, the fasting state is confined to a 3-hour interval at the end of the night. Consequently, to enhance long-term metabolic control and reduce to the risk of chronic diabetic complications, monitoring glucose during the entire postprandial state becomes crucial.
The postprandial exercise has proven to be safe and effective among patients with T2DM. Studies have reported that moderate intensity exercise after meal will be beneficial for acutely lowering post meal glucose levels. Added benefits of exercise were observed, when started soon after the meal and around 10 to 20 minutes before the expected glucose peak in healthy individuals and exercise within 15 to 30 minutes of the onset of meal in diabetes population.65,66 Combining CGM with regular exercise may be important to find the appropriate time to exercise to blunt the postprandial glucose rise. Combination of the 2 has demonstrated greater efficacy in reducing 2 h postprandial glucose compared to CGM alone. 67
Stress is known to affect blood glucose levels. The use of CGM has further given an additional lens on this issue. Glycemic variability has been shown to increase when there is a change in mood. 68 It has been observed that in individuals with T2DM, acute stress is associated with altered glucose concentrations in the postprandial period but not in the fasting state. 69 It has also been reported that adult individuals with T2DM who are using CGM, perceive an improved quality of life. 70
Technological Advances Enabling PPG State Monitoring
A diverse range of technologies are now accessible for continuous glucose measurement. These include flash glucose monitoring (FGM) and CGM. 71 FGM devices exclusively offer an instantaneous glucose reading when the sensor is scanned using a handset or a smartphone with a suitable software application, displaying data trends.72,73 CGM devices offer continuous glucose measurement and automatically transmit data to a monitor, smart device, or insulin pump. This technology enables both users and healthcare providers to establish alerts for high or low glucose levels, as well as the speed at which these levels are changing. CGM devices offer continuous and real-time information about glucose levels, as well as insights into trends and fluctuations. CGM measures interstitial fluid every 10 seconds and an average glucose value is recorded every 5 minutes for 24 hours a day offering comprehensive data that unveils temporal trends and patterns in glucose control. This gives a more accurate pattern of daily glucose fluctuations allowing identification of the glycemic effect of food, physical activity and different medication types and doses aiding in better self-management and avoiding unrecognized hypoglycemia. 74 Integration of CGM data in the electronic health record has enormous potential to assess actionable trends on a continuous basis. With this approach patients/caregivers actively participate in their health management and have direct engagement with healthcare providers from a smartphone or computer via the secure patient portal, and their data is stored and easily retrieved. This methodology also holds the potential to facilitate telehealth diabetes care in regions with insufficient access to pediatric diabetes providers, and for those patients who do not benefit from quarterly visits. 75 These systems enable healthcare professionals to measure the various key parameters including mean glucose (mg/dL), glycemic variability using coefficient of variation (⩽36%: stable, >36%: unstable), time in range (TIR: % of readings and time in the range of 70-180 mg/dL), time above range (TAR: % of readings and time in the range of 180-250 mg/dL [Level 1] and % of readings and time in the range of > 250 mg/dL [Level 2]), and time below range (TBR: % of readings and time in the range of 54-69 mg/dL [Level 1] and % of readings and time in the range of <54 mg/dL [Level 2]). 76 Numerous CGM systems have been seamlessly integrated with smartphone applications and wearable devices. The CGM reports can be quickly delivered to the treating physician (via email or directly via the platform with the user’s permission) and then discussed during a telephone call between healthcare professionals and the person with diabetes. Insulin pumps and CGM systems from the same manufacturer or systems from different manufacturers that communicate with each other allow combined documentation of insulin delivery and glucose values. This is also possible when using specific software solution. Using these tools, CGM data, insulin doses, and food intake can easily be visualized and discussed between patients with diabetes and healthcare professionals. 77 Hence, CGM offers a more holistic picture of glucose fluctuations that goes beyond the scope of SMBG, providing a broader understanding of glycemic trends rather than just isolated data points, enabling better monitoring of glycemic states including postprandial state. Furthermore, these techniques not only provide essential information but also serve to inform, educate, motivate, and alert individuals with diabetes about their glucose levels and trends.78,79
One of the significant advantages of using CGM in individuals with insulin-treated T2DM is the capability to set alerts for hypoglycemia. CGM can be used to evaluate glucose control in patients with insulin-treated T2DM or those who experience frequent, severe, or nocturnal hypoglycemia, especially when accompanied by hypoglycemia unawareness.79-81 Similarly, it also helps in monitoring and managing blood glucose levels to prevent dangerous high blood glucose episodes. The Endocrine Society suggests using professional CGM as it could be advantageous for adults with diabetes in identifying dawn phenomenon (early morning blood glucose excursions) and postprandial hyperglycemia.82,83 A study demonstrated the ability of CGM to detect a higher number of postprandial hyperglycemic episodes than finger-prick blood glucose levels.84-86 Particularly, CGM devices are capable of showing the current glucose levels at regular intervals of few minutes and can notify the individuals of potential (projected alert) or actual (threshold alert) instances of hyperglycemia or hypoglycemia, as well as rate of change in glucose levels.83,87 Observations from another study evaluating the potential benefit of CGM in individuals without diabetes (n = 448) and in individuals with diabetes (n = 192) suggested a positive effect of CGM in decreasing the episodes of increased blood glucose spikes among insulin-naïve individuals. This benefit can also be extended to individuals with early stages of dysglycemia. 88 TIR is a CGM parameter that is often used to analyze patient’s glycemic condition; however, it may not provide adequate information about hypoglycemia. Hence, as an alternative, various composite scores have been proposed to combine measures of glycemic control. Glycemia Risk Index (GRI) is such a composite score which describes the quality of glycemia in a CGM tracing and can be a useful metric for clinicians and researchers for assessing the glycemic effects of prescribed treatments. 89
Beyond its technical advantages, CGM profoundly influences patients’ psychological well-being and behavioral patterns. By providing real-time visual feedback on the impact of lifestyle choices on glucose levels, CGM fosters a heightened sense of personal responsibility and awareness. This direct connection between actions and consequences fosters stronger internal motivation to adhere to recommended lifestyle modifications, including dietary adjustments and increased physical activity. A study in type 1 diabetes patients has shown that CGM use significantly enhances quality of life, boosting confidence in blood sugar management and alleviating diabetes-related distress. 90 Despite the many pros that CGM has, it has some cons such as higher cost, relatively complex operation and more time required for training patients when compared with SMBG. These factors impede the widespread use of CGM, especially in resource-constrained settings. Hence, SMBG remains an irreplaceable glycemic monitoring technique. In conditions where application of CGM is limited, blood glucose control can also be assessed by using SMBG calculations to obtain indicators such as TIR and TAR, that are recommended by diabetes clinical guidelines, which can help compensate for the inability of HbA1c to reflect daily blood glucose fluctuations. 91 Currently, CGM use does not obviate the need for fingerstick SMBG as fingerstick blood glucose monitoring (BGM) confirmation will be necessary whenever extremes of glucose are reported (low glucose < 40 mg/dL and high glucose > 400 mg/dL) even for those who use latest CGM devices. Structured 5- to 7-point BGM profiles can empower patients with diabetes to play a more active role in the management of their disease, as well as improve the efficacy and safety of glucose-lowering therapeutics.
To recapitulate, CGM devices aid in comprehending glucose levels and fluctuations over time, providing a comprehensive view of glycemic patterns. They offer real-time displays of glucose levels and rate of change, alerts for current or impending hypoglycemia and hyperglycemia, continuous 24-hour coverage, and the ability to assess glycemic variability.79,86 As postprandial state is a dynamic state wherein glucose levels fluctuate depending on multiple factors, continuous monitoring of glucose levels would be particularly useful to understand glycemic patterns in this state as compared to the single time point measurement techniques.88,92 Thus, CGM technology has revolutionized glycemic monitoring, offering a potent tool for tracking and thus managing PPG fluctuations throughout the entire postprandial state. The structured SMBG remains a very important tool, especially in conditions that preclude the use of CGM. Hence, adhering to the principle of ‘the right tool for the right patient at the right time’, decisions about use of CGM and SMBG should be made through a shared decision-making process between clinicians and patients with diabetes. 93
Limitations
The present review does not report novel findings on PPG. Demographic factors (like age, pregnancy, etc.) impact on PPG are not explored here. The review does not explore the impact of PPG in other diabetes related complications beyond cardiovascular disease.
Conclusion
Achieving and maintaining glycemic control emerge as crucial aspects in the management of diabetes. The glucose triad consists of 3 key components: HbA1c, FPG and PPG. Postprandial hyperglycemia, characterized by high post-meal blood glucose levels, represent one of the initial deviations in regulating glucose levels linked to type 2 diabetes mellitus. Various studies have demonstrated that PPG is better correlated with HbA1c as compared to FPG. Moreover, literature reveals a significant connection between PPG and cardiovascular risk and outcomes. Despite recognition of PPG’s significance, PPG management is not pursued rigorously in practice. This can be attributed to cumbersome methods needed for PPG monitoring and various factors that affect PPG leading to variations in PPG measurements. Furthermore, the traditional practice of measuring PPG 1- and 2-hour post-meal may not adequately reflect glycemic excursions as they reflect only single time points, whereas the postprandial state, encompassing the 4 hour time frame after a meal, constitutes an intricate physiological process pivotal for metabolizing nutrients. Hence, to enhance long-term metabolic control and to reduce the risk of chronic diabetes complications, monitoring glucose during the entire postprandial state becomes crucial. Traditionally, such monitoring would require SMBG and laboratory tests with constraints of associated costs and inconvenience to patients. However, diverse range of technologies such as CGM are now accessible which enable continuous glucose measurement and identification of the glycemic effect of food, physical activity and medications, aiding in better self-management and avoiding unrecognized hypoglycemia. These technological advancements need to be implemented in routine practice for better diabetes management; especially to effectively monitor and manage the PPG state continuum.
Footnotes
Acknowledgements
Authors would like to thank Tejal Vedak and Rupika Pawar from Sqarona Medical Communications LLP, Pune.
Ethical Considerations
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Shashank Joshi: Conceptualization; Methodology; Project administration; Supervision; Validation; Writing - review & editing. Jothydev Kesavadev: Conceptualization; Resources; Supervision; Validation; Writing - original draft. K M Prasanna Kumar: Conceptualization; Methodology; Resources; Resources; Supervision; Supervision; Writing - original draft; Writing - original draft. Banshi Saboo: Conceptualization; Methodology; Resources; Validation; Writing - review & editing. Adi Mehta: Conceptualization; Data curation; Methodology; Writing - review & editing. Arpandev Bhattacharyya: Conceptualization; Data curation; Methodology; Resources; Writing - review & editing. S R Aravind: Conceptualization; Data curation; Methodology; Resources; Supervision; Writing - review & editing. P K Jabbar: Data curation; Investigation; Methodology; Resources; Writing - original draft. R Santosh: Conceptualization; Methodology; Project administration; Resources; Validation; Writing - review & editing. Vaishali Deshmukh: Conceptualization; Methodology; Resources; Supervision; Visualization; Writing - original draft. Nilakshi Deka: Data curation; Methodology; Resources; Validation; Writing - review & editing. Shambo Samrat Samajdar: Data curation; Methodology; Supervision; Validation; Writing - original draft.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This initiative was funded by Cipla Ltd. Mumbai.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
NA.
