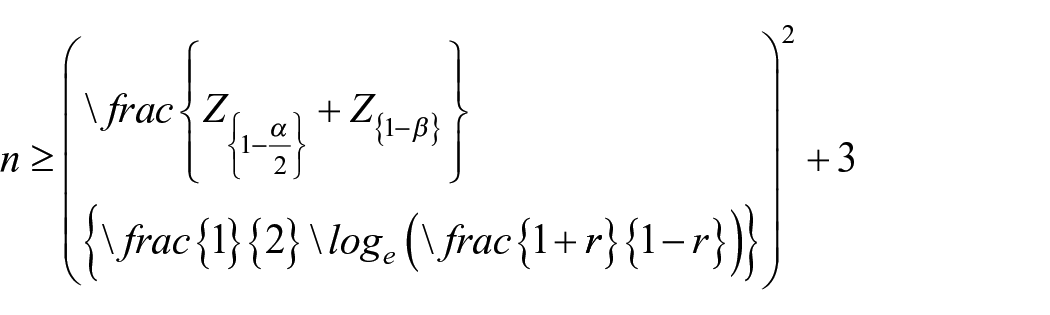Abstract
Background:
Serum thyrotropin levels rise above normal while thyroid hormone levels remain within normal ranges, indicating subclinical hypothyroidism. PLT and MPV levels significantly increase in AITD patients; PLT levels are higher in hypothyroidism and Hashimoto’s disease, whereas MPV levels are higher in hyperthyroidism and Graeve’s disease. An increased MPV and other abnormal platelet values lead to a higher incidence of cardiovascular events. The aim was to evaluate RDW and platelet indices in Egyptian patients with subclinical Hashimo’s thyroiditis using high-sensitivity CRP Hs-CRP as an indicator of inflammation.
Methods:
Data for patients (60 patients) with subclinical Hashimoto’s thyroiditis who visited the Menoufia University Hospital, Internal Medicine outpatient clinic included. We measured RDW, MPV, PDW, PC, LPCR, (FT3), (FT4), (TSH), lipid profile, Hs-CRP, and Antithyroid antibodies. According to Antithyroid antibodies positivity the 60 patients subdivided into 3 groups 1 (negative for Abs), 2 (positive to 1 Ab), and 3 (positive for both Abs).
Results:
Among our patients 40 (66.7%) tested positive for anti-thyroglobulin Abs while 36 (60.0%) tested positive for antiperoxidase Abs. RDW, MPV, and LPCR demonstrated positive significant correlations with serum Hs-CRP levels. The study revealed negative significant correlations between TSH levels and PDW and between T4 levels and MPV.
Conclusion:
Early diagnosis of Hashimoto’s thyroiditis can be supported by platelet indices and RDW as diagnostic tools and these markers can also offer prognostic insights into disease severity and complications to help initiate timely treatment for hypothyroidism and prevent atherosclerotic cardiovascular disease.
Plain Language Summary
A frequent autoimmune disease that affects the thyroid gland is called Hashimoto’s thyroiditis. Early on, it might not exhibit obvious symptoms; this is referred to as subclinical Hashimoto’s thyroiditis. Even in this early stage, though, the body may exhibit certain changes that point to inflammation, which is the body’s reaction to injury or stress.
In this study, we examined a few basic blood test indicators that may indicate inflammation. Measurements of red blood cells and platelets, which are microscopic cells involved in clotting, are among them. In particular, we looked at platelet indices such mean platelet volume, as well as red cell distribution width RDW, a measure of variation in red blood cell size. An established indicator of inflammation, a blood plasma protein called hs-CRP, was also tested.
Sixty patients with subclinical Hashimoto’s thyroiditis were studied. According to our findings, greater levels of the inflammatory plasma protein hs-CRP were substantially correlated with RBCs anisocytosis RDW, and platelet indices MPV, and LPCR. This implies that the body may already be experiencing low-grade inflammation even when thyroid hormone levels are only slightly off. These blood indicators have the potential to be helpful for early identification and monitoring because they are simple to measure and already included in standard blood tests.
Our findings suggest that people with early thyroid problems might already be at risk for inflammation-related complications, and that these simple markers could help doctors assess that risk. However, the study had some limitations, such as the small number of participants and the lack of a healthy control group. More research on larger populations is needed to confirm our results.
In summary, our study highlights the potential value of RDW and platelet indices as early indicators of inflammation in subclinical Hashimoto’s thyroiditis.
Introduction
Serum thyroid hormone levels below the reference range, which indicates thyroid insufficiency, are indicative of overt hypothyroidism. Thyrotropin (also known as thyroid-stimulating hormone, or TSH) levels are suitably increased in overt hypothyroidism caused by thyroid malfunction (primary hypothyroidism). When blood thyrotropin levels are raised outside of the standard range but thyroid hormone levels are within the reference range, this is known as subclinical hypothyroidism. 1
Thyroid function tests alone are used to make the biochemical diagnosis of subclinical hypothyroidism. Up to 10% of people in iodine-sufficient populations suffer from subclinical hypothyroidism, with women and the elderly having the highest frequency. 2
Subclinical hypothyroidism, however, often returns to euthyroidism, 3 and the prevalence of subclinical hypothyroidism has probably been exaggerated since thyrotropin levels increase with aging in those without thyroid illness. 4
Individuals have a range of values for serum thyrotropin and free thyroxine (FT4) that are maintained within a narrower range than the broader population reference range. 5 Due to the highly sensitive correlation between pituitary thyrotropin secretion and serum FT4 levels, if a person’s serum FT4 level falls below their FT4 reference range, even though their FT4 level is still within the population-based FT4 reference range, their serum thyrotropin level may be higher than the upper limit of the population-based reference range. The intraindividual thyroid axis set point is mostly influenced by genetics. 6
RDW is a marker of anisocytosis and considered as an inflammation marker in various inflammatory conditions, autoimmune diseases such in Hashimoto’s thyroiditis. 7
Indeed, it was found that the Platelet to lymphocytes ratio PLR, neutrophil to lymphocytes ratio NLR, and systemic inflammatory index SII indices were higher in Hashimoto’s thyroiditis and non-immunogenic hypothyroidism. 8
Patients with Autoimmune thyroiditis (AITD) have considerably higher PLT and MPV, with HT and hypothyroidism showing a greater increase in PLT and Graves’ disease (GD0 and hyperthyroidism showing a greater increase in MPV. When abnormal platelet indices are discovered, patients’ thyroid function can receive the proper clinical attention; on the other hand, the consequences of abnormal platelet parameters, like elevated MPV, increase the risk of cardiovascular events, which should also be addressed in the AITD population. 9
Aim of the Study
Analyze the relationship between platelet indices and RDW with high-sensitivity CRP (Hs-CRP) as an inflammation marker in Egyptian patients with subclinical Hashimoto’s thyroiditis.
Patients and Methods
Study Design
A cross-sectional observational study. Data for 60 patients with subclinical Hashimoto’s thyroiditis (Variable disease durations not accurately determined from the history) who visited the Menoufia University Hospital, Internal Medicine outpatient clinic included. Also, data from 60 healthy controls were collected for comparison.
Inclusion and exclusion criteria; The eligibility criteria were: an age of 18 to 70 years and the absence of other chronic diseases. Patients with a history of liver or kidney dysfunction, diabetes, cancers including thyroid cancer, infection, and any chronic inflammatory or autoimmune disease such as rheumatoid, lupus, etc. were excluded. The use of acetylsalicylic acid and other drugs that affect platelet function was not permitted during the study.
Patients and Procedures
We included a 60 Individuals who had not previously been diagnosed with Subclinical Hashimoto’s thyroiditis and whose parenchyma was homogeneous on ultrasound and we contrasted them against 60 healthy volunteers as a control group. A diagnosis of subclinical Hashimoto’s thyroiditis was based on positivity for thyroid peroxidase antibody (TPOAb) and thyroglobulin antibody (TgAb) and disturbed thyroid functions. Platelet indices measured using an XN-1000 hematology analyzer (Sysmex, Kobe, Japan). The normal values for the analyzed parameters are as follows: PDW, 9.7 to 15.1 fL; PC, 173 × 109/L to 360 × 109/L; free triiodothyronine (FT3), 2.3 to 4.2 ng/L; free thyroxine (FT4), 0.84 to 1.76 ng/L; thyroid-stimulating hormone (TSH), 0.35 to 5.5 mIU/L; TPOAb, 0 to 60 U/mL; and TgAb, 0 to 60 U/mL. The aforementioned parameters measured using an ADVIA Centaur® XP Immunoassay System (Siemens, Munich, Germany). Biochemical parameters were measured using a Cobas 8000 system (Roche, Basel, Switzerland).
According to the results of serum Antithyroid antibodies positivity the 60 patients subdivided into 3 groups 1 (negative for Abs), 2 (positive to 1 Ab), and 3 (positive for both Abs).
Study Duration
From March 2024 to December 2024.
Ethics
Informed written consent was provided by all patients authorizing the use of their data for research purposes after explanation of benefits and risks of the study. Blood samples was obtained under complete a septic technique prior to the beginning of anticoagulant therapy. All precautions will be taken to avoid any infections or hazards during sampling or imaging. The study was approved by the local ethical and scientific committee of Faculty of Medicine, Menoufia University, Egypt. Ethics approval code number (INTM13/date-6-2023).
Sample Size Estimation
An analytical cross-sectional study proposing primarily to detect the assess the platelet indices and RDW in Subclinical Hashimoto’s thyroiditis. A previous study showed that the correlation coefficient between TSH and PDW was .269 (Avni Findikli and Tutak). 10 So, the sample size to study the results of the current study with a significant P < .05 and a power of 80%, is calculated according to this formula:
So, n = 107, then at least 107 subjects should be included in this study.
Statistical Analyses
The differences between the groups determined using Student’s t-test for continuous variables and the Mann–Whitney U test for non-continuous variables. Significance was indicated by P < .05. Statistical analysis was performed using Graph Pad Prism version 6.00 (Graph Pad Software, San Diego, CA, USA, www.graphpad.com).
Results
We had a 60 patients diagnosed as subclinical thyroiditis and we contrasted them against 60 healthy volunteers as a control group, the age range in our patients was from 22 years to 54 years with a mean was 35.9 ± 8.5, 52 of them were females, according to their lab investigations the mean of hemoglobin level was 12.1 ± 0.81, and the mean for white blood cells, platelets, TSH, high sensitive CRP, serum cholesterol, triglycerides, LDL were 6.3 ± 1.5, 294.0 ± 61.4, 7.9 ± 1.9, 4.68 ± 3.99, 234.5 ± 41.7, 170.0 ± 28.7, 167.3 ± 28.7, respectively. CBC indices were 13.9 ± 1.4 for RDW mean and 10.1 ± 1.4 for MPV and 16.2 ± 2.9 for PDW and 0.26 ± 0.03 for PCT. We had 40 (66.7%) patients which were tested positive for anti-thyroglobulin Abs, and 36 (60.0%) patients were positive for antiperoxidase Abs (Table 1).
Socio-demographic and clinical data of the studied cases (60).

Abbreviations: HDL, high density lipoprotein; HG, hemoglobin; HsCRP, high sensitive CPR; LDL, low density lipoprotein; LPCR, large platelet concentration ratio; MCV, mean corpuscular volume; MPV, mean platelet volume; PCT, plateletcrite; PDW, platelet distribution width; Plt, platelet; RDW, red cell diameter distribution width; TgAb, thyroglobulin antibody; TGDs, triglycerides; TPOAb, thyroid peroxidase antibody; TSH, thyroid stimulating hormone; WBCs, white blood cells.
We compared cases with 60 healthy controls for demographic and lab investigations then the significant differences were for Hs-CRP and Platelet counts, RDW, PDW, MPV, and PCT (Table 2).
Comparison between cases and control regarding their laboratory data.

Abbreviations: HsCRP, high sensitive CPR; LPCR, large platelet concentration ratio; MPV, mean platelet volume; PCT, plateletcrite; PDW, platelet distribution width; RDW, red cell diameter distribution width; TgAb, thyroglobulin antibody; TPOAb, thyroid peroxidase antibody.
U (Mann-Whitney test). **T-test. #Chi-square test.
We subdivided the 60 thyroiditis patients into 3 groups in Table 3 according to positivity to antibodies group 1 (20) was the negative patients to both antibodies, group 2 (4) was the positive for 1 ab only and group 3 (36) was the positive for both. The mean of Hs CRP was significant in-between group 1 and 3 (group 1, 2, 3 were 0.63 ± 0.37, 1.92 ± 0.9, 7.23 ± 3.13) respectively. The mean of RDW in group 1, 2, 3 were 10.6 ± 1.1, 12.6 ± 0.40, 14.9 ± 1.08 respectively with a significant difference between the 3 groups. Platelet counts were 302.3 ± 37, 176.5 ± 0.57, and 302.4 ± 61.8 in groups 1, 2, 3 respectively. According to platelets indices there were a statistically significant differences in MPV, PDW, LPCR, and PCT between the negative group 1 and the positive group 3 for Abs (the means for MPV were 8.0 ± 0.51, 9.8 ± 0.01, 11.1 ± 0.76 and for PDW 15.06 + 2.6, 15 ± 0.01, 16.9 ± 3.3 and for LPCR 29.08 ± 12.2, 22 ± 0, 39.3 ± 9.8 and for PCT were 0.26 ± 0.46, 0.24 ± 0.10, 0.26 ± 0.03 in groups 1, 2, 3 respectively.
Comparison between positivity of antibodies and their laboratory data.

Abbreviations: HsCRP, high sensitive CPR; LPCR, large platelet concentration ratio; MPV, mean platelet volume; P1, Negative versus positive peroxidase or thyroglobin; P2, Negative versus Positive both peroxidase and thyroglobin; P3, positive peroxidase or thyroglobin versus Positive both peroxidase and thyroglobin; PCT, plateletcrite; PDW, platelet distribution width; RDW, red cell diameter distribution width; TgAb, thyroglobulin antibody; TPOAb, thyroid peroxidase antibody.
Kruskal Wallis test.
From Table 4 there were a positive significant correlation between RDW, MPV, and LPCR and serum level of Hs-CRP. However, from Table 5 there was a negative significant correlation between TSH level and PDW, also a negative significant correlation between T4 level and MPV
Correlation between Platelet indices and RDW with high sensitive CRP in subclinical Hashimoto’s thyroiditis.

Abbreviations: HsCRP, high sensitive CPR; LPCR, large platelet concentration ratio; MPV, mean platelet volume; PCT, plateletcrite; PDW, platelet distribution width; RDW, red cell diameter distribution width.
P < 0.05.
Correlation between platelets count, MPV, PDW, and thyroid function subclinical Hashimoto’s thyroiditis.

Abbreviations: MPV, mean platelet volume; PCT, plateletcrite; PDW, platelet distribution width; RDW, red cell diameter distribution width.
P < 0.05.
Discussion
Dividing patients according to thyroid antibody status (TPOAb and TgAb) further reveals that patients positive for both antibodies exhibit the highest levels of inflammatory markers and platelet activation indices. This stratification strongly suggests that the serological profile could reflect disease severity or higher autoimmune activity, corroborating the concept that thyroid autoimmunity is a continuum, where the inflammatory burden escalates with higher autoantibody titers. This may explain why some subclinical HT patients eventually progress to overt hypothyroidism or develop cardiovascular complications.
The mean age of the patients in this study was 35.9 ± 8.5 years, in Aygun et al 11 study, the mean age of patients with Hashimoto thyroiditis was 34.45 ± 8.43 years, and in Erge et al 12 study, the median age was 45 years, and the post hoc analysis revealed that the age of the euthyroid Hashimoto’s thyroiditis group was not significantly different from the age of the hypothyroid-thyrotoxic Hashimoto’s thyroiditis group.
About 86.7% of the patients in this study were females, this is in concordance with Aygun et al (86.1%) and Erge et al (87%) study results.12,13
Hs CRP was significantly higher in the patients group compared to the control group. The same significant difference was found in Aygun et al and Erge et al studies.12,13
In contrast to the control group, patients with Hashimoto’s thyroiditis in this study had higher platelet count, RDW, PDW, and PCT, and lower MPV levels. The same results were found in Aygun et al study, 11 Erge et al study, 12 and Bilge et al study. 13
The higher platelet counts and Hs CRP levels in Hashimoto’s thyroiditis patients were reported by many studies in the literature.11 -13
As an autoimmune disorder, Hashimoto’s thyroiditis can lead to a state of systemic inflammation. 14 The pathophysiology of Hashimoto’s thyroiditis is mostly related to the activation of T-cells, HLA, DR3, DR4, DR5, and multiple genetic factors. These data suggest that Hashimoto’s thyroiditis causes a state of chronic inflammation. 15
High platelet count has been reported in systemic infections, 16 chronic inflammatory disorders and autoimmune diseases as Behcet’s disease, 17 , and rheumatoid arthritis. 18 It is clear that Hashimoto’s thyroiditis is an inflammatory condition. Therefore, the same inflammatory pathways may induce platelet production.
Postulated reasons why RDW is elevated in chronic inflammatory disorders include: First, the impairment of red blood cell maturation caused by the desensitization of the inflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β) and interleukin-6 (IL-6) against red cell progenitors, second, the entry of juvenile red blood cells into the circulation, and third, anisocytosis of the red blood cells. 19
Interestingly, the positive correlation between RDW and hs-CRP suggests that erythropoiesis is altered in these patients, likely as a result of chronic inflammatory cytokine activity (eg, IL-6, TNF-α), which disrupts normal red blood cell maturation and increases anisocytosis. This mechanism is consistent with observations in other autoimmune and inflammatory diseases, indicating that RDW may serve as a surrogate marker for systemic inflammation. Moreover, elevated RDW has been linked to cardiovascular morbidity and mortality in various populations, implying that subclinical HT patients may share similar risks even without overt clinical signs.
RDW, CRP, and ESR were observed to be significantly higher in the patient group compared to controls in another retrospective study that included 165 patients with inflammatory bowel disease. 20
In Chang et al ’s meta-analysis of 22 studies involving 80 216 coronary artery disease patients, a positive correlation was found between increased RDW and mortality due to increased coronary artery disease. Also, RDW levels were found to be higher among heart failure patients with diabetes mellitus than those without diabetes. 21
Thyroid stimulating hormone (TSH) is also known as an accelerating factor of atherosclerosis. In addition, Hashimoto’s thyroiditis is a well-established risk factor for atherosclerotic cardiovascular disease independent of thyroid hormones. 22 In our study, we found that RDW increased in accordance with the literature.
By comparing different patients’ groups with subclinical hypothyroidism according to serology of Hashimoto’s thyroiditis (negative antibodies, 1 positive antibody test, and 2 positive antibody test), there was a significant difference between the 3 groups regarding Hs CRP, RDW, MPV, PDW, LPCR, and PCT.
Also, there was a positive significant correlation between RDW, MPV, and LPCR with serum hs-CRP level, And a negative significant correlation between TSH level and PDW, T4 level and MPV.
Bremner et al reported a negative correlation of serum T4 levels with RDW, 23 and Montagnana et al showed a positive correlation between serum TSH levels and RDW. 24
Carlioglu et al performed a study on 51 patients with subclinical Hashimoto’s thyroiditis, and 51 age-matched people. MPV level was found to be higher in the patient group, and in the analysis, there was a positive correlation between both thyroid autoantibodies, and MPV. 22
Many studies showed similar results. In Jabeen et al’s study, MPV and hs-CRP were significantly higher in diabetics than non-diabetics. 25 Higher MPV levels have been shown in many diseases such as DM, acute coronary syndrome, and cerebrovascular diseases. 26 It was also found that MPV may have an acute phase reactant like function that correlates with disease activity in some rheumatologic diseases, as rheumatoid arthritis and ankylosing spondylitis. 27
The negative correlation between TSH and PDW, and between T4 and MPV, provides a new perspective on how thyroid function intricately modulates platelet dynamics, even in subclinical states. Thyroid hormones influence hematopoiesis and platelet production, and deviations in their levels—even within “normal” limits—could significantly affect platelet morphology and reactivity. This finding suggests a possible compensatory response to maintain hemostatic balance in the face of evolving autoimmune thyroiditis, although the exact mechanism warrants further investigation.
Autoimmune thyroiditis and subclinical hypothyroidism have been considered as risk factors for atherosclerotic cardiovascular diseases. 28
Using some specific and cheap markers for early detection of Hashimoto’s thyroiditis is important for early diagnosis and prevention of possible complications, such as heart diseases, 29 birth defects, 30 dyslipidemia, 31 and Hashimoto encephalopathy. 32
However, some limitations need to be acknowledged:
Cross-sectional design limits causal inference—whether these hematologic changes contribute to, or merely reflect, disease activity remains undetermined.
Lack of longitudinal follow-up prevents understanding of whether these markers predict progression to overt hypothyroidism or cardiovascular events.
We did not evaluate other inflammatory markers such as ESR, IL-6, or TNF-α, which could further clarify the inflammatory milieu.
Possible confounders such as smoking status, BMI, or subclinical infections were not controlled.
the small number of the cohort included.
Future studies should consider:
- Prospective cohort designs to establish the prognostic value of these markers.
- Larger multi-center studies to validate findings across different populations.
- Exploration of cytokine profiles and other immune markers to deepen understanding of the pathophysiology.
- Investigation of whether intervention strategies (eg, early levothyroxine therapy, anti-inflammatory treatments) could modulate these hematologic indices and improve patient outcomes.
Conclusion
The present study highlights significant alterations in hematologic indices, specifically RDW, MPV, PDW, and LPCR, in patients with subclinical Hashimoto’s thyroiditis (HT). The observation that RDW and MPV are significantly elevated and positively correlated with hs-CRP underscores the existence of a low-grade inflammatory state even in the absence of overt hypothyroidism. These findings align with the growing body of evidence suggesting that subclinical HT is not a benign condition, but rather a dynamic state characterized by ongoing immune-mediated thyroid injury and systemic inflammation. Our results are consistent with prior studies (Aygun et al., Erge et al., Carlioglu et al.), but also add novel insights by focusing on the Egyptian population, providing region-specific data that could help tailor preventive strategies. Notably, this is one of the few studies that correlate platelet indices and RDW with hs-CRP and autoantibody titers in subclinical HT, reinforcing the relevance of these markers beyond mere hematologic curiosities—they are potential tools for early risk stratification.
Footnotes
Ethical Considerations
The study was approved by the local ethical and scientific committee of Faculty of Medicine, Menoufia University, Egypt. Ethics approval code number (INTM13/date-6-2023).
Consent to Participate
After obtaining a written informed consent from all patient/participant/guardian to be included in this study and the consequence of publication.
Author Contributions
Ahmed Salah: Investigation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available upon request by contacting the corresponding author (Dr. Alaa Efat).