Abstract
Diabetes mellitus is one of the most debilitating diseases, diabetic neuropathy happens to be the most common and perhaps the most serious complication of diabetes mellitus, often leading to morbidity and mortality. A 60 year old female presented with disorientation, history of vomiting, shortness of breath, respiratory failure initially. Blood reports revealed that she was positive for ketone bodies with elevated HbA1c and general random blood sugar. Chest radiogram revealed atelectasis of the right lung with prominent involvement of right middle and lower lobes. High-resolution computed tomography of chest confirmed the findings and unilateral diaphragmatic paralysis due to phrenic nerve neuropathy due to undetected type 2 diabetes was diagnosed. Although phrenic nerve paralysis is a rare occurrence with diabetes, the possibility shouldn’t be overlooked as a presentation of diabetes mellitus.
Keywords
Introduction
Diabetes mellitus (DM) has become a rapidly increasing disease over the past 3 decades, raising concerns due to its significant morbidity and mortality rates associated with complications. 1 The evaluation and treatment options for diabetic peripheral neuropathy are complex and involve multiple factors. It is crucial to screen patients with diabetes for autonomic neuropathy as well as sensorimotor neuropathy. 2 Different classification systems have been developed to categorize the various manifestations of neuropathy, with distal symmetrical polyneuropathy (DSPN) being the most common form. Autonomic neuropathy, particularly cardiovascular autonomic neuropathy (CAN), has received significant attention in research. However, the incidence and prevalence estimates for CAN, similar to DSPN, vary. 3 While phrenic nerve neuropathy is a rare complication of diabetic neuropathy, it is noteworthy, and other potential causes of diaphragmatic palsy should be ruled out before diagnosing diabetic phrenic nerve neuropathy. Unilateral involvement of the phrenic nerve appears to be more prevalent than bilateral involvement, but both can lead to severe respiratory failure.4,5 Our case highlights the significance of considering diabetic neuropathies beyond the commonly observed sensorimotor and autonomic neuropathies. The diagnosis of unilateral diabetic phrenic nerve neuropathy presents notable challenges due to its rarity and the need to rule out alternative causes. Additionally, our case underscores the importance of implementing comprehensive management strategies, encompassing glycemic control, respiratory support, and targeted antibiotic therapy.
Case Presentation
A 60-year-old female was presented to the emergency department in a state of disorientation with history of vomiting since 2 days, shortness of breath since 4 months which is insidious in onset, gradually progressing. Physical examination revealed hypotensive state with systolic blood pressure of 60, with tachycardia, tachypnea and fall in oxygen saturation. Patient was stabilized. Patient was conscious and cooperative, she was afebrile. Cardiovascular examination was normal except for tachycardia; patient had decreased breath sounds over the right lung field. Use of accessory respiratory muscles was noted. Abdominal examination revealed no significant findings. Peripheral pulses especially in the lower limbs were difficult to palpate. Pin sensation was decreased in distal limbs. Patient had a history of burning sensation and tingling numbness in lower limbs and upper limbs since few weeks.
On admission, general random blood sugar (GRBS) was found to be 400 mmol/L. She was not previously diagnosed with diabetes mellitus or not on any hypoglycemic medication. She complained of having difficulty in performing strenuous tasks which resulted in breathlessness, she was used to sleeping with pillows propped up rather than supine. Sleeping supine would often lead to an episode of breathlessness and disturbed sleep. She had no history of smoking or substance abuse. She was not a known case of thyroid/ hypertension/ bronchial asthma/epilepsy/coronary artery disease. She was not affected by COVID-19 neither vaccinated with booster dose. No major surgeries except for total abdominal hysterectomy 10 years ago. The rest of the examination remained insignificant. Over the next few days, patient complained of severe dyspnea, with falling oxygen saturation. Her arterial blood gas analysis revealed pH of 7.23, PO2 of 89 mmHg, HCO3 of 15 mEq/L and PCO2 of 54 mmHg, developing hypercarbic respiratory failure. Patient was initially supported with continuous positive airway pressure (CPAP) and 4 L of O2 to maintain patient’s vitals stable.
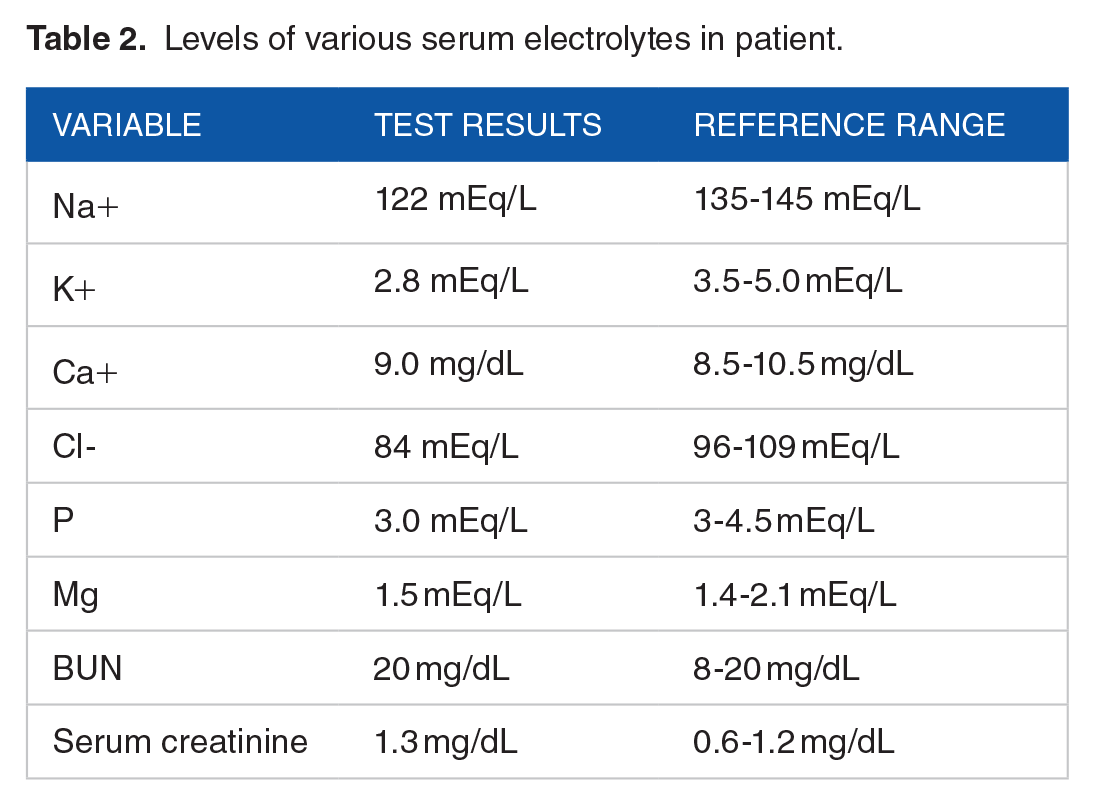
Patient’s body mass index (BMI) was noted to be 32. Her red blood count, thyroid function tests, B-natriuretic peptide, cardiac enzymes, serum protein electrophoresis, anti-acetylcholine receptor antibodies, procalcitonin and proBNP levels were normal. Patient was positive for urine ketones. Complete blood count (CBC) revealed an elevated total white blood count of13 000 cells/cumm as shown in the Table 1. Table 2 shows the serum electrolytes levels of sodium and potassium as 122 and 2.8 mEq/L respectively suggestive of hyponatremia and hypokalemia. HbA1C was revealed to be 10%. Her lipid profile was deranged with high total cholesterol and triglyceride levels as shown in Table 3. Electrocardiogram (ECG) was suggestive of tachycardia, 2D ECHO revealed normal findings.
Complete blood count of the patient.

Levels of various serum electrolytes in patient.
Lipid profile of patient.

Chest radiogram revealed blunting of bilateral pleural angles, mild haziness involving both lungs, collapse/consolidation of right middle and lower lobes as shown in the Figure 1. Her RT-PCR for COVID-19 was negative. Patient was advised for High-resolution computed tomography (HRCT) for further work up, which revealed consolidation with sub segmental atelectasis involving anterior segments of right upper lobe, right lower lobe segments and right middle lobe segments as shown in the Figure 2. Possible eventration of right hemi diaphragm was disclosed as shown in Figure 3. Pulmonary function tests were consistent with severe restrictive lung disease. Any blunt/surgical trauma history to phrenic nerve, viral infections, cervical spondylosis, tumors of neck, myasthenia gravis were excluded before making a provisional diagnosis of unilateral diabetic phrenic nerve neuropathy with diabetic ketosis.

Blunting of bilateral pleural angles, mild haziness involving both lungs, collapse/consolidation of right middle and lower lobes.

Consolidation with sub segmental atelectasis involving anterior segments of right upper lobe, right lower lobe segments and right middle lobe segments.

Possible eventration of right hemi diaphragm was disclosed.
Main stay of management remained fluid resuscitation and to control the patient’s GRBS. Patient was put on CPAP ventilation with 100% FiO2, Intravenous fluids were given at 100 ml/h, regular insulin and glargine were given subcutaneously according to GRBS. Patient was also given potassium in normal saline depending upon need. Patient was put on empirical antibiotic therapy that included intravenous piperacillin + tazobactum (4.5 g, TID in 100 mL NS), clindamycin (600 mg, TID). Intravenous deriphyllin and nebulization with salbutamol and budesonide was given for pulmonary complaints. Patient was closely monitored, with every 3 hourly monitoring of GRBS, sixth hourly monitoring of arterial blood gases and fourth hourly monitoring of urine ketones. Chest physiotherapy was advised to clear any pleural collections and improve total lung capacity. When the patient’s oxygen saturations improved, she was weaned off CPAP and O2 support. With her GRBS under control, patient negative for ketone bodies and oxygen saturations maintaining above 95%, patient was discharged with the advice to continue chest physiotherapy regularly and follow up for her blood sugars. Patient was advised to continue Injection glargine OD subcutaneously and was put on Tab dapaglifazolin 10 mg OD, Tab sitagliptin 50 mg BD, Tab metformin 500 mg BD to maintain her GRBS. Patient was also advised to take Tab gabapentin NT once a day along with methylcobalamin supplements to prevent any neuropathies in the future. After 15 days, the patient demonstrated gradual improvement and was advised to continue the prescribed medication. The patient was under follow-up for a month and no further complications were reported.
Discussion
Diabetes mellitus is one of the growing problems in developed and developing countries. Because of the expensive treatment modalities, medications are beyond the reach of an average individual, which further complicates the problem.6,7 Undetected diabetes can present with common signs such as unexplained weight loss, fatigue, restlessness, and body pain. It is important to note that symptoms may be mild or have a gradual onset, potentially going unnoticed. 8 High HbA1c levels are often associated with severity of neuropathy as assessed by electrophysiological testing in studies. 9 Peripheral and autonomic neuropathies do not invariably coexist in diabetes. 10
Diabetes is the most common cause of neuropathy, although distal symmetrical polyneuropathy is the most common type, autonomic neuropathy is a troubling complication. 3 Phrenic nerve palsy in particular is a rare complication of diabetes, most common causes include injury during cardiac surgery, surgeries involving spine, shoulder, head and neck, viral infections, autoimmune disorders, tumors involving neck and mediastinum, but most often the cause remains obscure.
Diabetic phrenic neuropathy was the only identifiable etiology in our patient; patient history, decreased breath sounds over the right thorax, chest radiograph with confirmatory findings from HRCT and exclusion of all other possible causes support the diagnosis of diabetic phrenic neuropathy. 2D ECHO, proBNP, troponin were evaluated to rule out any cardiac causes resulting in respiratory failure. Patient’s blood counts, procalcitonin were used to exclude any infective causes. Most diabetic phrenic nerve palsies especially unilateral resolve spontaneously and do not need any other treatment other than proper management of blood sugars. 11 Diaphragm resection and video assisted minimally invasive surgeries are alternative management modalities. 12 For respiratory failure management, patient is put on continuous positive airway pressure or in bilateral phrenic nerve palsy, mechanical ventilation is suggested. 13
The diagnostic challenges in our case were multifaceted due to the rarity of unilateral diabetic phrenic nerve neuropathy as a manifestation of diabetes mellitus. The initial presentation of shortness of breath and decreased breath sounds over the right lung field raised concerns of a respiratory pathology, necessitating the exclusion of other potential causes such as pulmonary infections or tumors. Patient had to be evaluated for all cardiac pathologies. Further, the presence of peripheral neuropathy symptoms, including burning sensation and tingling numbness in the limbs, posed a diagnostic challenge due to the overlapping clinical features with other neuropathies. Additionally, the absence of a known history of diabetes further complicated the diagnosis. To establish a definitive diagnosis, it was crucial to conduct a comprehensive evaluation ruling out alternative etiologies. Through meticulous clinical assessment, utilization of imaging studies, and consideration of the patient’s unique clinical presentation, the diagnosis of unilateral diabetic phrenic nerve neuropathy could be established.
The limitations of our case includes, we could not confirm our findings with nerve conduction tests for phrenic nerve palsy. The patient was not evaluated for other subclinical DN that required electrodiagnostic testing and quantitative sensory and autonomic testing.
Conclusions
In conclusion, we suggest that in elderly patients with phrenic nerve palsy, diabetes mellitus be considered a prominent cause. GRBS stands as an easy enough indicator for hyperglycemia. Although phrenic nerve palsy is a very rare presentation of diabetes mellitus, it shouldn’t be overlooked.
