Abstract
This multicenter, longitudinal, descriptive, observational study of T2DM adults in Thailand aimed to assess real-world outcomes of basal insulin (BI) dose titration on glycemic control. Three-hundred and twenty-four patients were recruited and followed up over 24 weeks. Basal insulin titration was physician-driven in 58.2% of patients and patient-driven in the rest. During the 24-week study period, the total daily BI dose moved from 20.9 to 25.6 in the physician-driven group, while in the patient-driven group, it increased from 25.3 to 29.7. Thirty-five patients (11.2%) achieved their individualized HbA1c targets, with 18 patients (5.8%) achieving HbA1c ⩽ 7% without documented hypoglycemia. In summary, this study highlights that BI titration is suboptimal in the real world, and patients are unable to achieve their glycemic targets.
Introduction
Type 2 diabetes mellitus (T2DM) is recognized as a global pandemic and has an increasing prevalence in low- and middle-income countries (LMICSs), especially in Asia. 1 Rapid changes in environmental, social, and lifestyle factors are some of the factors contributing substantially to this increased prevalence. 2
Clinical management of T2DM depends largely on disease characteristics and progression at the time of diagnosis. Traditionally, lifestyle and dietary modifications with metformin as the first line of treatment are recommended following early diagnosis of diabetes. However, given the progressive nature of T2DM, patients with diabetes eventually require exogenously administered insulin to correct the loss of β-cell function. In Thailand, the guidelines on insulin initiation in patients with T2DM are derived from International Diabetes Federation (IDF) recommendations 3 among several others. While certain glycemic indices such as glycosylated hemoglobin (HbA1c) and fasting plasma glucose (FPG) have been assigned fixed targets in the past to ensure glycemic control in T2DM, at present there is no consensus on these in the various guidelines and instead a patient-centric approach is advocated. In real-world practice, basal insulin is initiated when lifestyle modifications and a combination of various oral antidiabetic (OAD) agents fail to achieve a patient’s target HbA1c or FPG. Basal insulin such as neutral protamine Hagedorn (NPH) or insulin Gla-100 and insulin detemir are highly effective in maintaining normoglycemic conditions and can be combined with OADs or short-acting insulins to maintain glycemic control. However, to achieve target glycemic control, the dose of insulin has to be regularly titrated. Current recommendations from the IDF suggest initiating insulin at 10 U or 0.2 U/kg and titrating the dose once or twice weekly at 1 to 2 U each time till FPG is 70 to 130 mg/dL. 3
Despite these recommendations, glycemic control in Asia is generally found to be suboptimal.4,5 One of the first studies to evaluate glycemic control in the Asian diabetic population following insulin initiation revealed that only one-third of the study patients achieved HbA1c < 7.0% after 6 months of basal insulin therapy 6 and the estimate for Thailand was 15.2%. 7 Inadequate titration of insulin was one of the factors that has been correlated with this lack of glycemic control.6-8 Besides dose titration, various other factors have also been postulated to influence the effectiveness of insulinization. 4 These factors need to be investigated in detail to draft guidelines and policies that are suitable for local management of T2DM in LMICs. In the Thai setting, patients with T2DM are generally attended to by general practitioners or specialties besides endocrinologists. While the Diabetes Association of Thailand conducts routine training on insulinization with the primary care physicians, the outcome of this exercise on the real-world management of T2DM is largely unknown. In this study, we have evaluated the consequence of optimizing the dose of basal insulin in achieving glycemic control in patients with T2DM in real-life practice in Thailand.
Methods
Study design
This 24-week, national, multicenter, noninterventional, observational, prospective study was conducted in Thailand between April 2015 and September 2016. The primary objective was to evaluate and describe the glycemic control (individualized glycosylated hemoglobin [HbA1c] and FPG) in real-world setup, following titration of the basal insulin dose to achieve optimal results in patients with T2DM. Secondary objectives were determination of the number of patients achieving HbA1c < 7.0% without symptomatic hypoglycemia, identification of insulin titration pattern and evaluation of hypoglycemic events.
Study sites were hospitals with laboratory testing facilities for estimation of HbA1c and study investigators were nonendocrinologists who managed patients with T2DM and had adequate knowledge of optimal insulin therapy. Diabetes management, including optimization of insulin dose was the investigators’ prerogative and based on their independent clinical judgment. The study comprised 3 visits—an inclusion visit (V1) and 2 follow-up visits (V2 and V3), 12 and 24 weeks after enrolment.
The study was performed according to the principles laid down by the 18th World Medical Assembly 9 and in compliance with the guidelines for Good Epidemiological Practice and approval from all pertinent regulatory authorities, steering committee and IRBs/IECs. Ethical approval for this study has been obtained from all ethics committees with the following ethical approval reference number: CR 0032.102/9275 (Chiangrai Prachanukroh Hospital), P015h/58 (Fort Mengraimaharaj Hospital), 0032.202/098 (Nakornping Hospital), 34/2015 (Chumphonkhetudomsakdi Hospital), 05022558 (Songkhla Hospital), 26/02/58 (Hat Yai Hospital), 001/58 (Nakhoncity Hospital), CTIREC 004 (Prapokklao Hospital), 0032.202.03/413 (Buddhasothorn Hospital), 42/2558 (Chonburi Hospital), 016/2558 (Sanpasitthiprasong Hospital), HE581012 (Srinagarind Hospital), 010/2015 (Maharat Nakhon Ratchasima Hospital), 1/2558 (Chaiyaphum Hospital), 04/2558 (Phra Chom Klao Hospital), R2015-002 (Banpong Hospital), 1/2558 (Uthai Thani Hospital), 20150201 (Anandhamahidol Hospital), Si072/2015 (Siriraj Hospital & Gloden Jubilee Med Center), 4/2558 (Surin Hospital), and 070/58 (Buddhachinaraj Hospital). All study patients provided informed consent to participate in the study.
Patient recruitment and data collection
All patients in the registry were recruited after obtaining written informed consent, during a 12-month period. Inclusion and exclusion criteria are presented in detail in Supplemental Table 1.
Data collected at baseline included demographics, physical examination, history of diabetes, current treatment for diabetes, prior and current laboratory reports on serum creatinine (SCR), HbA1C, and FPG. At week 12 and 24, data were collected on physical examination, current diabetes treatment, current laboratory reports on HbA1c and FPG (and SCR, if available), hypoglycemic events, patient diary, unplanned hospitalizations, and final assessment (patient status and achievement of glycemic control at week 24). Documented (symptomatic) hypoglycemia was defined as an event in which typical symptoms of hypoglycemia were accompanied by a measured plasma glucose concentration ⩽70 mg/dL (⩽3.9 mmol/L).
Insulin dose titration was classified into 2 categories—physician-driven method, where insulin dose was adjusted at each visit by the physician; and patient-driven method, where patient self-adjusted the insulin dose (based on the physician suggested algorithm).
Statistical analysis
Sample size calculation was made using a formula given by Riddle et al. 10 Assuming 60.0% of patients achieving HbA1c ⩽ 7.0% with each insulin type (NPH and Gla-100) with a 10.0% patient drop-out. A total of 300 patients were planned to be enrolled in the study, with margin of error (e) of 0.06.
For purpose of analysis, the study patients were categorized as eligible population comprising all patients enrolled, and evaluable population which included only those patients who had HbA1c and FPG values for the 24-week follow-up visit (V3).
Descriptive statistics using mean (with 95% confidence interval [CI]), standard deviation (SD), median, and min-max for continuous parameters and counts and percentages (with 95% CI) for categorical parameters were used for data analysis.
Statistical testing of primary endpoints: change from baseline to the end of titration period for HbA1c was done using ANCOVA and change from baseline in FPG was done using rank ANCOVA. Statistical testing of secondary endpoints: rates of hypoglycemia was done using rank ANOVA, and other categorical variables were tested using either Pearson’s Chi-square or Fisher’s exact test. All statistical analyses were conducted using SAS, version 9.2 (SAS Institute, Cary, NC, USA).
Results
Patient disposition and characteristics
Our study enrolled a total of 327 patients at 24 sites across Thailand and 3 patients were excluded due to eligibility criteria during source data verification. A total number of 324 patients comprised the eligible population. Of these, 318 (98.2%) patients completed the study. Three (0.9%) patients were lost to follow-up and 3 (0.9%) patients withdrew consent. Outcome data from 313 patients were available at week 24 and were considered for the evaluable population analysis. Participants were further categorized according to – (1) type of basal insulin received and (2) dose-titration method employed and is given in Figure 1. Briefly, most of the participants in both the eligible as well as the evaluable populations were received NPH as basal insulin (eligible, n = 197; evaluable, n = 190) followed by insulin Gla-100 (eligible, n = 123; evaluable, n = 119) and insulin detemir (eligible and evaluable, n = 4). In addition, physician-driven insulin dose optimization was reported in a larger proportion of patients (eligible, n = 190; evaluable, n = 182) than patient-driven dose optimization (eligible, n = 134; evaluable, n = 131).
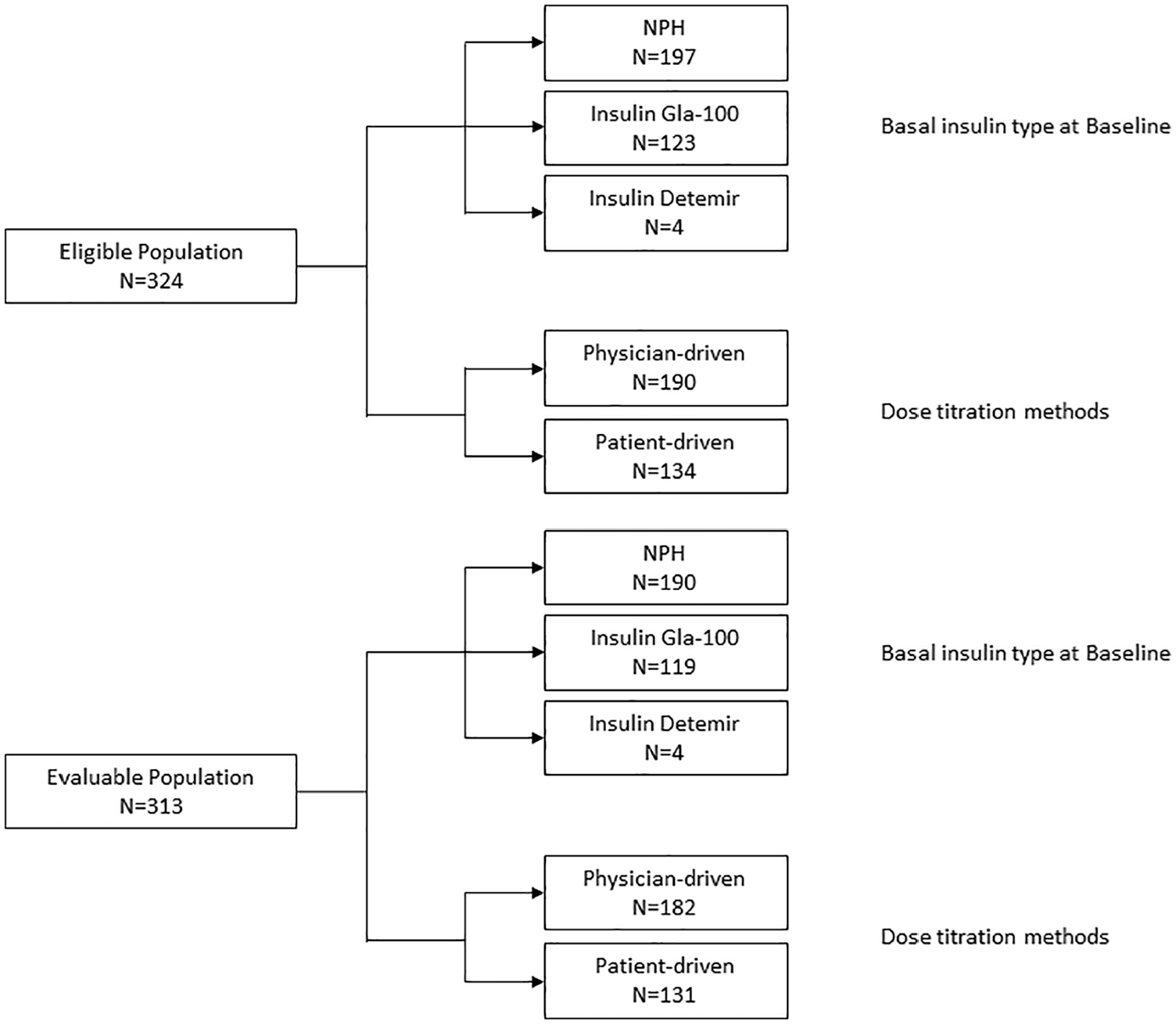
Summary of analysis populations in the study. NPH indicates neutral protamine Hagedorn.
Patient characteristics, including diabetes history, are summarized in Table 1. Almost two-thirds (n = 213, 65.7%) of the patients were female. The median age of the eligible population was 58.0 (interquartile range [IQR]: 51.0–63.0) years and 258 (79.6%) patients were aged ⩾50 years. Average (±SD) body mass index (BMI) of study patients was 26.97 (±4.64) and over half (62.7%) of the patients had BMI ⩾ 25.0 kg/m3. Average (±SD) systolic and diastolic blood pressure were 133.26 (±14.40) mm Hg and 74.16 (±10.20) mm Hg, respectively. Commonly reported comorbidities in the eligible population at the time of enrolment were hypertension (n = 270, 83.3%) and dyslipidemia (n = 250, 77.2%). Most of study patients had either universal coverage (48.5%) or civil servant medical benefit scheme (40.1%).
Demographic and baseline characteristics in eligible participants.

Abbreviations: BMI, body mass index; DBP, diastolic blood pressure; DPP, dipeptidyl peptidasel; FPG, fasting plasma glucose; HbA1c, glycated hemoglobin; NPH, neutral protamine Hagedorn; OAD, oral antidiabetic; SD, standard deviation; SBP, systolic blood pressure.
The average (±SD) duration of diabetes in the eligible population was 10.91 (±5.22) years, and 287 (88.6%) patients had had diabetes for ⩾5.0 years. A total of 287 (88.6%) patients had HbA1c ⩾ 8.0% and over half of the patients (n = 183; 56.5%) had HbA1c in between 8.0% and 9.9%. Moreover, 220 (68.1%) patients had FPG ⩾ 130.0 mg/dL and 30.3% of the patients (n = 98; 30.3%) had FPG values in between 80.0 and 129.0 mg/dL. The eligible population had been receiving OAD therapy for an average (±SD) duration of 10.64 (±5.28) years and 280 (86.4%) patients had received OADs for ⩾5.0 years. The most commonly prescribed OADs were metformin (88.9%) and sulfonylureas (74.1%).
Glycemic control following titration of basal insulin dose
At 24 weeks following study inclusion, a total of 35 (11.2%) and 154 (49.2%) patients in the evaluable population had achieved their individual HbA1c and FPG targets, respectively (Table 2). Mean (±SD) HbA1c reduced from 9.41% (±1.23%) at baseline to 8.96% (±1.57%) at week 24 (P < .0001). Concurrently, mean (±SD) FPG decreased from 170.09 (±69.51) mg/dL at baseline to 150.71 (±64.63) mg/dL at week 24 (P < .0001; Supplemental Table 2). Overall HbA1c and FPG target achievement rates with physician-driven titration were 14.84% and 56.59%, respectively; while for patient-driven titration, the rates were 6.11% and 38.93%, respectively (Table 2).
Summary of HbA1c and FPG target achievement in evaluable patients at Week 24 and by dose-titration method applied.

Abbreviations: FPG, fasting plasma glucose; HbA1c, glycated hemoglobin.
Individualized HbA1c and FPG targets were set by physicians at baseline.
In patients whose insulin dose titration was physician-driven, mean (±SD) HbA1c reduced from 9.22 (±1.15) at baseline to 8.62 (±1.43) at week 24 (P < 0.0001). Similarly, mean (±SD) FPG in these patients decreased from 163.85 (±60.44) at baseline to 140.33 (±57.54) at Week 24. In patients who performed self-titration of their insulin dose, mean (±SD) HbA1c reduced from 9.67 (±1.29) at baseline to 9.44 (±1.64) at week 24 (P = 0.0730); and mean (±) FPG decreased from 178.82 (±79.95) at baseline to 165.14 (±71.09) at Week 24 (P = 0.0618; Supplemental Table 2).
The proportion of patients achieving HbA1c target at Week 24 without any symptomatic hypoglycemia was 5.8%. The proportion of patients achieving HbA1c target at Week 24 without any symptomatic hypoglycemia in the physician-driven titration group was 8.2% and 2.3% in the patient-driven titration group (Table 3).
Summary of HbA1c ⩽ 7.0% target achievement without documented hypoglycemia in evaluable patients at Week 24 by dose-titration method.

Abbreviations: HbA1c, glycated hemoglobin; SD, standard deviation.
Hypoglycemic events
Overall, a total of 64 (20.45%) patients experienced at least one hypoglycemic event during the course of the study. The average event rate at Week 12 and Week 24 were 3.39 (95% CI: 3.00-3.84) events per person-year and 1.71 (95% CI: 1.51-1.94) events per person-year, respectively (Table 4).
Summary of hypoglycemic events in evaluable patients at Week 24 by titration method.

Abbreviation: CI, confidence interval.
Titration patterns
In the evaluable population, between baseline to Week 12, the insulin regimen in 6 patients (5 patients receiving NPH and one patient receiving detemir) was shifted to Gla-100. In addition, one patient on NPH stopped insulin treatment. In between Week 12 and Week 24, 3 patients who were previously on NPH were switched to Gla-100 while 8 patients previously on NPH were switched to non-basal insulin. Moreover, 1 Gla-100 user was switched to NPH and 6 Gla-100 users were switched to non-basal insulin.
In 182 patients (58.2%), the basal insulin titration was physician-driven, while in the remaining 131 (41.8%), patients self-titrated their basal insulin. Over the course of the study, the total daily basal insulin dose moved from 20.9 U/day to 25.6 U/day in the physician-driven group (+4.7 U), while in the patient-driven group, it increased from 25.3 U/day to 29.7 U/day (+4.4U) (Table 5). Overall, the average (±SD) total daily dose at Week 24 increased by 4.48 U (±6.93) in comparison to baseline (P < .0001). The dose increase tended to be the highest at Week 24, irrespective of basal insulin type or dose-titration method. Following insulin dose titration, no increase in body weight was reported at Week 12 and Week 24 compared to baseline, irrespective of type of basal insulin or dose-titration strategy.
Comparison of total daily dose of basal insulin in evaluable patients at baseline and Week 24, by titration method.

Abbreviations: SD, standard deviation; NA, not available.
Significant P value using Wilcoxon sign-rank test.
Discussion
This study aimed to evaluate and describe the effect of a 24-week titration of the dose of basal insulin on target achievement rate of glycemic indices, HbA1c and FPG, in real-world settings in Thailand. NPH and insulin glargine are the preferred basal insulins and used in most of the patients. Data showed that basal insulin titration is physician-driven in most of the patients, indicating a scope for more patient education and empowerment for patient-driven titration, as has been evidenced by the recent studies11-13 and recommended by the current American Diabetes Association (ADA) guidelines. 14 Overall target achievement was poor in this real-world study, with almost 90% of the patients failing to achieve their individualized HbA1c targets. This may be, in part, due to the suboptimal dose titration of basal insulins. Overall, 20.45% of the patients experienced at least 1 hypoglycemic event during the study period, and the proportions were matched irrespective of the type of basal insulin or titration method.
Various studies report a wide range in HbA1c target achievement rate following initiation of basal insulin treatment.15-17 However, various treat-to-target (TTT) studies have shown that titration of the insulin dose is vital for attaining glycemic targets.10,11,18-20 However, in our study, the target achievement rate for glycemic indices was far from desirable even after a 24-week period. Certain factors regarding the study design, especially the investigators and patients, need to be considered to put these findings in context. The study investigators were nonendocrinologists, but physicians most routinely responsible for providing care to patients with T2DM. There could have been a possibility of variation in the standard of care/ titration algorithms being used by these physicians. From the currently available treatment options, basal insulin analogues have been shown to be better than NPH in multiple trials, especially in terms of reduction of the risk of hypoglycemia and weight gain. 21 Despite this fact, we found that NPH was being used in more patients. This perhaps is largely out of economic consideration and insurance policies. Moreover, patients in this study had an average duration of T2DM >10 years, almost 90.0% of them had HbA1c >8.0%, and all of them had failed to achieve HbA1c <7.0% while being on basal insulin. Hence, it could also be possible that most of our patients were refractory to moderate dose titration and required further treatment intensification. Also, it is important to put into perspective that this study was not aimed or powered to compare and contrast between different insulins or different titration regimens or methods, but to describe the current status and overview of basal insulin use pattern in the real-world. There was no randomization and patients were not entirely comparable on baseline characteristics or on the management followed. Therefore, no comparative analysis between groups could be attempted. This study provides a snapshot of current basal insulin use and provides insights into likely gaps and ways to improve glycemic control in T2DM patients.
Patient awareness and education is a critical part of long-term management of chronic diseases. In insulinized T2DM patients, the responsibility of daily administration of the recommended insulin dose as well as routine monitoring for blood glucose levels and hypoglycemic events is largely borne by the patients themselves. Various TTT trials such as the Canadian INSIGHT trial 11 and the AT.LANTUS trial, 12 have shown that the patient-driven titration process results in improved glycemic control with a lower incidence of hypoglycemia and higher treatment satisfaction. These findings have been reproduced in the Asian context in the ATLAS trial. 13 In this study, the incidence of hypoglycemia was lower in patients who self-titrated, in accordance with the TTT trials.
Taking findings from this study into consideration, especially in the context of studies that have been previously published, it becomes obvious that newer strategies and options are required for improving glycemic control with basal insulin in Thailand. Various titration algorithms for optimizing basal insulin dose 22 have been validated and these need to be tested for appropriateness in the Thai settings. Being a multimodal disease, the management of T2DM requires participation from the patient and their family, as well as the physician and other medical personnel. Certain initiatives like family-oriented programs for diabetes control 23 or the use of current Internet- and gadget-based technologies 5 could also be very helpful in spreading education and awareness. Early insulinization, especially in combination with existing treatments 24 or with newer agents such as glucagon-like peptide 1 receptor antagonists25-27 or the introduction of newer basal insulins such as glargine-30028-30 are also clinical interventions that could improve glycemic control in Thai patients with diabetes.
Conclusion
This study provides important insights in the real-world scenario with regard to basal insulin use in routine clinical settings in Thailand. It shows that a substantially low proportion of patients achieve their glycemic targets, with minimal dose titration over time. This may underlie the suboptimal basal insulin titration in the real-world scenario. These findings could be useful in creating more relevant strategies such as physician education and patient empowerment to safely and effectively titrate insulin for successful management of T2DM in Thailand.
Limitation
While this study highlights a couple of key issues regarding glycemic control in the Thailand context, certain others were beyond the scope of the study and hence limit the interpretability of the results. Given the choice of patients and investigators, we could not assess target achievement in the entire spectrum of diabetic patients in Thailand. In addition, given the noninterventional and observational nature of this study, as well as differences in key baseline and treatment characteristics, we cannot statistically compare and interpret the target achievement rates in between the groups as these factors may have an additional impact on glycemic control, thereby confounding the results. Moreover, we could not follow-up patients to ascertain the further course of action taken by the investigators. This study also did not ascertain factors that affect the outcome of insulin dose titration such as patient adherence, investigators’ knowledge and practices, and time since first insulinization; therefore, we can only hypothesize on possible reasons for low target achievement.
Supplemental Material
supplementary_table_1-2 – Supplemental material for Basal Insulin Dose Titration for Glycemic Control in Patients With Type 2 Diabetes Mellitus in Thailand: Results of the REWARDS Real-World Study
Supplemental material, supplementary_table_1-2 for Basal Insulin Dose Titration for Glycemic Control in Patients With Type 2 Diabetes Mellitus in Thailand: Results of the REWARDS Real-World Study by Chaicharn Deerochanawong, Rattana Leelawattana, Natapong Kosachunhanun and Puntip Tantiwong in Clinical Medicine Insights: Endocrinology and Diabetes
Footnotes
Acknowledgements
The authors acknowledge the REWARDS investigators and study nurses; Anandamahidol Hospital: Dan Tanphaichitra, Pathomporn Bunjongparu; Banpong Hospital: Chatchai Chulapornsiri, Rattanaporn Hongwatthanakul; Buddhachinaraj Hospital: Viruch Sirigulsatien, Rachadda Pipatsat; Buddhasothorn Hospital: Nuttha Leowchavalit; Chaiyaphum Hospital: Thanitsak Thawikhote, Sopapan Nganchaturas; Chiang Rai Prachanukroh Hospital: Samroeng Seekaew, Pornprapa Sompan; Chonburi Hospital: Rachada Kwanjaipanich, Wipawon Homsiang; Chumphonketudomsakdi Hospital: Kanokrat Petsrijun, Jongrak Romnukul;
Fort Meng Rai Maharaj Hospital: Natthapon Kantawee, Saisunee Netsuwan; Golden Jubilee Medical Center: Suphawan Siriwattanakul, Oradee Charitkuan, Sununta Tangpanithadee; Hat Yai Hospital: Tarapat Auppatham, Ulaiwan Kaew-ied; Maharat Nakhon Ratchasima Hospital: Watanyu Parapiboon, Prajamchat Watthanapanich;
Nakhoncity Hospital: Jakkrit Pompet; Nakornping Hospital: Puntapong Taruangsri, Jutapron Chunrithai; Phrachomklao Hospital: Anchana Panich, Wasana Bussaboakaew; Prapokklao Hospital: Nawarat Pengpong, Kunika Treekham; Sanpasitthiprasong Hospital: Amornrat Tepakorn, Issaree Taesongkroh, Supatra Chaikaew; Siriraj Hospital: Chaiwat Washirasaksiri, Manop Pithukpakorn, Wayuda Muangkaew; Songkhla Hospital: Prapol Aengchuan; Srinagarind Hospital: Praew Kotruchin, Chatlert Pongchaiyakul, Sasiporn Yongyuengpan; Surin Hospital: Passorn Sueyunyongsiri, Sumalee Boonmee; and Uthai Thani Hospital: Jira Pattanaphongsak, Phanumas Dech-in, for their contributions toward this study
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Sanofi. Editorial support for the preparation of this publication was provided by Satyendra Shenoy of Describe Scientific Writing & Communications and paid for by Sanofi. Editorial support was also provided by Anahita Gouri and Rohan Mitra of Sanofi India. The authors, individually and collectively, are responsible for all content and editorial decisions and received no payment from Sanofi directly or indirectly (through a third party) related to the development of this publication.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Study conception and design: CD,RL; Acquistion of data: CD, RL, NK, PT; Analysis and interpretation of data: CD, RL,NK, PT; Manuscript drafting: CD, RL,NK,PT; Manuscript revision: CD.
Data Sharing Statement
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and data set specifications. Patient-level data will be anonymized and study documents will be redacted to protect the privacy of trial subjects. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at: ![]() .
.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
