Abstract
Introduction:
My Dose Coach™ (MDC) is a U.S. Food and Drug Administration-approved digital smartphone application designed to help users with type 2 diabetes mellitus (T2DM) to titrate their basal insulin (BI) according to a clinician-prescribed individualized titration plan. The present analysis aimed to compare the efficacy of the MDC app in achieving the fasting plasma glucose (FPG) target, glycemic goals (glycated hemoglobin [HbA1c] <7%), hypoglycemic events, and economic costs with conventional titration strategies in people with T2DM in Mexico.
Methods:
This randomized, controlled, single-center, two-arm, 12-week clinical trial included insulin-naïve participants with uncontrolled T2DM (HbA1c 8%–10%). The study endpoints included the number of participants achieving a weekly mean FPG of 90–130 mg/dL, time to reach the FPG target, reduction in HbA1c, the mean number of insulin applications and FPG measurements per month, hypoglycemia incidence, and cost-effectiveness.
Results:
Study participants (N = 80) were divided equally into control and MDC arms. The percentage of participants achieving the target FPG range was higher (92.5% vs. 75.0%; P = 0.034) in the MDC arm with a greater reduction in fasting glucose (69.0 vs. 62.0 mg/dL; P = 0.028) compared with the control arm. The MDC group achieved the fasting glucose target faster than the control group (18 vs. 30 days; P = 0.001). After 12 weeks, both groups had a reduction in HbA1c levels. The MDC group had a fewer hypoglycemia events than the control group. No severe or nocturnal hypoglycemia was reported in either study arm. The cost of care was lower in the MDC group than in the control group (Mex$4890.00 vs. Mex$7582.00; P = 0.001).
Conclusions:
The MDC app is a viable option which can be used for titrating BI dose in a safe, effective, and cost-efficient manner for individuals with T2DM in Mexico.
Keywords
Introduction
The prevalence of diabetes has increased rapidly in low- and middle-income countries such as Mexico over the past few decades and is expected to rise further. According to the International Diabetes Federation, in 2024, 13.6 million people were living with diabetes in Mexico. This number is expected to increase to 19.9 million by 2050. 1 In 2024, Mexico was among the top 10 nations with substantial total diabetes-related health expenditure in adults, amounting to approximately USD 19.9 billion. 1 The increase in the burden of type 2 diabetes mellitus (T2DM) in Mexico is the result of the confluence of factors such as genetic predisposition, a high prevalence of risk factors and comorbidities, and insufficient coverage of health services, which are serious limitations to early detection and proper control of the disease. 2 Several key factors, such as the fear of hypoglycemia, clinical and therapeutic inertia, patient education, availability and affordability of diabetic treatment, and physical conditions such as obesity and overweight, can impact glycemic outcomes. This emphasizes the multifaceted nature of diabetes management and the various elements that can influence the achievement of glycemic targets.3,4
T2DM is a progressive metabolic condition that often requires complex pharmacotherapeutic treatments along with lifestyle modifications.5,6 Studies showed that in Mexico, despite the availability of many efficacious therapy options, glycemic control remains low (<50%) among people living with diabetes. 4 Many people with T2DM need insulin therapy to achieve optimal glycemic control. It is recommended to initiate insulin therapy with a long-acting basal insulin (BI) analog such as insulin glargine.7–9 However, the initiation of insulin therapy is often delayed and, even when initiated, may not be optimally titrated.3,10
Digital health is transforming the landscape of medicine through sensor data, software, and wireless communication tools, which can help initiate and titrate BI in people with T2DM and reduce the diabetes management burden on physicians. Telehealth, qualitative hypoglycemia alarms, artificial intelligence, flash/continuous glucose monitoring (CGM) devices, closed-loop systems, insulin pumps, “smart” insulin pens, and digital/mobile-based applications are some of the technologies that can be used along with informational brochures and educational programs to overcome barriers to BI initiation and titration.11–13 Digital technologies such as CGM devices have been efficient in diabetes care during an unprecedented health crisis such as coronavirus disease 2019 (COVID-19). 14 BI titration applications such as MyStar DoseCoach™, 15 MyStar WebCoach®, 16 INSULIA®, 17 mobile health applications, 18 TeleDiab-2, 19 and My DoseCoach™ KSA 20 have helped in T2DM management.
The United States Food and Drug Administration (U.S. FDA) has approved a digital smartphone application, My Dose Coach (MDC), to help people with T2DM titrate their BI according to individualized titration plans prescribed by their physicians. 3 The MDC app combines a web portal where healthcare professionals (HCPs) define a BI titration plan and a smartphone application that provides dose and titration recommendations to people based on a predefined glycemic target, fasting plasma glucose (FPG), and hypoglycemia data. 3 People with T2DM can use the MDC app to titrate their BI and achieve their FPG target levels, as reported by previously published study data from India, Mexico, Colombia, and Germany.3,21,22 The objective of the current analysis was to assess the efficacy of the MDC app in achieving the glycemic target and its cost-effectiveness compared with conventional titration strategies in the Mexican population with T2DM.
Methods
Study design and population
In this randomized-controlled, single-center, two-arm, 12-week clinical trial, insulin-naïve participants with uncontrolled T2DM (HbA1c 8%–10%) who required the initiation of BI therapy, as per the treating physician’s discretion, were enrolled. The study was conducted during the COVID-19 pandemic in 2020 at an outpatient clinic at Endocrinology Department, Hospital Civil de Guadalajara Fray Antonio Alcalde, Guadalajara, JA, Mexico. The study included insulin-naïve adults aged >18 years with T2DM who did not achieve their glycemic goals after receiving ≥2 oral antihyperglycemic drugs (OADs) for at least 3 months and required BI therapy. Participants were randomly assigned to use the MDC app for BI titration (MDC group) or follow a physician-guided BI titration plan (control group). Participants assigned to the MDC group were required to have access to a smartphone and internet and complete training in the proper use of the MDC app (Fig. 1).

Study design. HbA1c, hemoglobin A1c; T2DM, type 2 diabetes mellitus.
This study was conducted in compliance with all international guidelines and Mexican national laws and regulations, and any applicable guidelines. All necessary regulatory submissions (e.g., Institutional Review Board/Independent Ethics Committee) were performed in compliance with local regulations, including local data protection guidelines.
Treatments and basal insulin titration
All participants initiated BI therapy with insulin glargine at a starting dose of 0.2 U/kg body weight, based on the American Diabetes Association (ADA) guidelines. The MDC group participants titrated their insulin dose using the MDC app and recorded their FPG and insulin dose data in the application. The control group participants received information on the BI dose based on FPG and a written titration plan from the treating physician and were required to record their FPG and the applied insulin dose in a patient diary.
Outcomes
The study outcomes included the number and percentage of participants achieving a weekly mean FPG level within the target range of 90–130 mg/dL, the time to first reach this FPG target, changes in the weekly mean FPG levels over time, and changes in HbA1c levels and the BI dose from baseline to 12 weeks. Safety outcomes included the incidence of any (by the time of the day
Other outcomes evaluated were adherence to therapy, measured by the average number of insulin applications and FPG measurements per month, and cost-effectiveness, evaluated by analyzing direct costs, including the costs of medications (insulin and OADs), glucometer, test strips, laboratory tests, travel cost (to hospital), and job loss due to attending the evaluations.
Statistical analysis
The statistical analysis for all endpoints was conducted on the intent-to-treat population, which consisted of all participants who were enrolled in the study, regardless of whether they received the intervention (MDC app) or not. The participants were randomized to the intervention and control groups. A statistical summary of continuous variables, with the counts of variable for analysis (n), arithmetic mean (mean), median, and interquartile range, with the required number of decimals, was reported. The categorical variables with the counts of variable for analysis (n) and the percentages based on the level of summarization were reported. The baseline and clinical characteristics of all continuous variables applicable were compared between the intervention and control groups using an independent sample t-test. The actual and changes in anthropometric and blood parameters from the baseline up to Week 12 were compared between the treatments using an independent t-test. Fasting capillary blood glucose differences between the intervention and control groups were evaluated using the Mann–Whitney U test. The intersubject variability in monthly measurements of capillary glucose and insulin administration was analyzed using an independent t-test. The association of hypoglycemia within the treatment group was analyzed using the Fisher’s exact test. A 5% level of significance was used for statistical comparison between the treatments for all variables. All summaries and statistical analyses were generated using SAS Version 9.4.
Results
Study participants and baseline characteristics
A total of 80 people with T2DM were enrolled and randomized in the study (n = 40 in each group) (Supplementary Fig. S1). The demographic and disease-specific baseline characteristics of the study population were comparable between the two groups, except for gender: the proportion of women was slightly lower in the MDC group (26 [65%]) than that in the control group (30 [75%]) (Table 1). The mean age and body mass index (BMI) were 51.92 years and 28.56 kg/m2 in the MDC group and 53.67 years and 27.52 kg/m2 in the control group, respectively. The mean FPG (172.02 mg/dL vs. 167.97 mg/dL) and HbA1c (9.0% vs. 8.92%) levels were similar in the two groups. In both groups, a majority of the participants had completed primary or secondary education, and ≤20% had an undergraduate or prevocational degree. All participants were on two OADs, predominantly glimepiride/metformin or linagliptin/metformin. A few were on vildagliptin/metformin or glibenclamide/metformin (Supplementary Fig. S2).
Demographic Characteristics

Significance P < 0.05; HbA1c, hemoglobin A1c; IU, international unit; DM, diabetes mellitus; MDC, My Dose Coach™; SD, standard deviation.
Fasting plasma glucose
Overall, at the end of the 12-week intervention period, a greater proportion of participants in the MDC group (37 [92.5%]) achieved the FPG target of 90–130 mg/dL, compared with the control group (30 [75%]; P = 0.034) (Fig. 2A). For those who achieved the FPG target, the mean time to first reach the target was significantly shorter in the MDC group than that in the control group (18 vs. 30 days; P = 0.001) (Fig. 2B). The change in the mean weekly FPG levels was observed from Week 2 onward, and both groups achieved stable mean weekly FPG levels by Week 7 (Fig. 2C).

HbA1c
In both groups, a reduction in the mean HbA1c levels was observed from baseline to Week 12 (MDC: 6.90%; control: 7.15%, Table 2). This reduction in HbA1c was greater in the MDC group than that in the control group (−2.1% vs. −1.8%; P = 0.029) (Fig. 3).
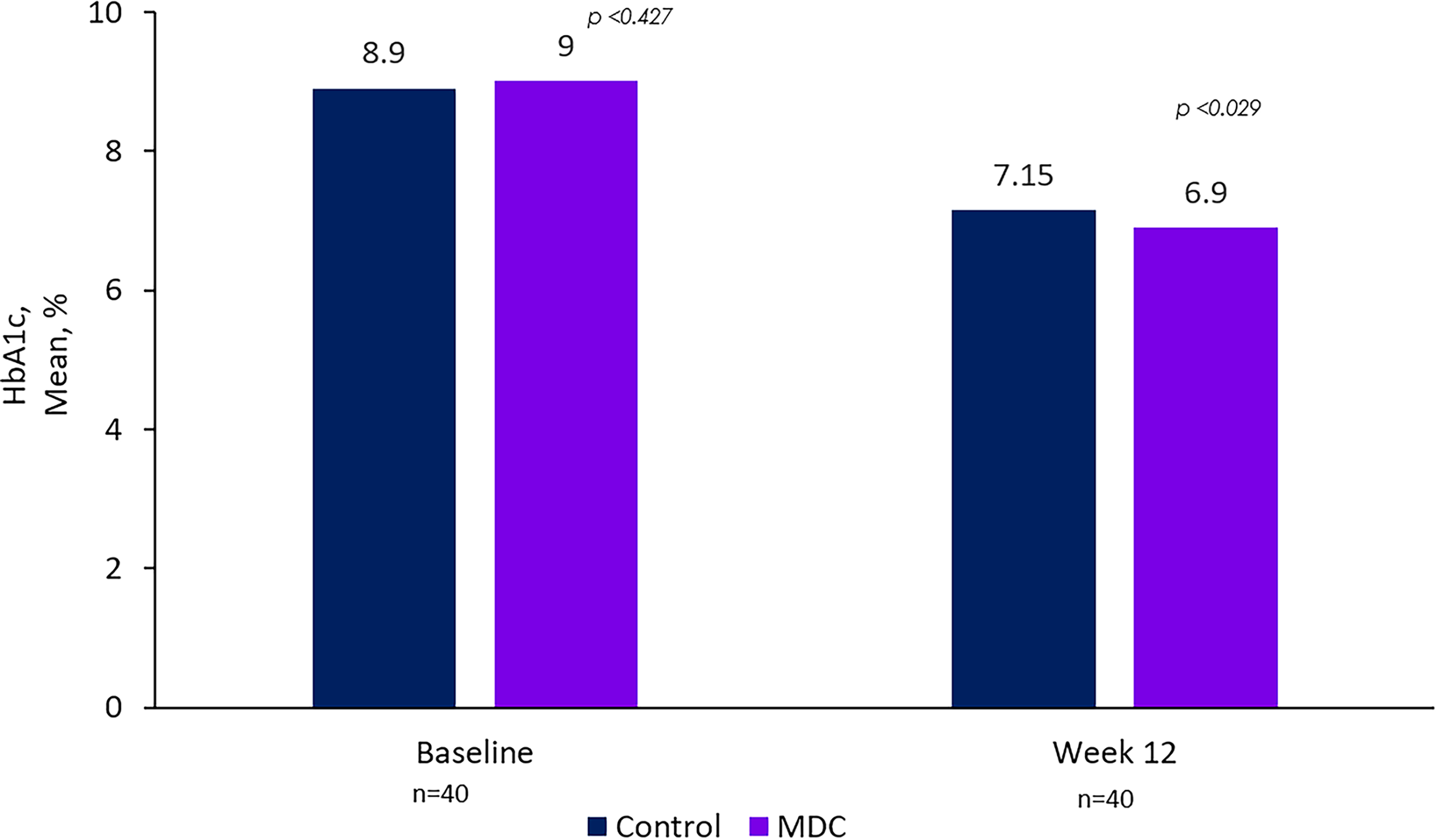
HbA1c (%) comparison between groups: initial and final values. significance P < 0.05. HbA1c, hemoglobin A1c; MDC, My Dose Coach™.
Change in Baseline Characteristics at Week 12

Significance P < 0.05; HbA1c, hemoglobin A1c; IU, international unit; SD, standard deviation.
Insulin dose
The mean initial BI dose was 16.9 U and 17.6 U in the MDC and control groups, respectively (Table 1). At Week 12, the mean BI dose increased in both groups, with a lower mean dose in the MDC group (26.02 U) required to reach FPG target range than the control group (34.08 U; P = 0.001) (Table 2).
Adherence to therapy
In both groups, the number of insulin applications and capillary glucose measurements decreased slightly from month 1 to month 3. At month 3, both the mean number of insulin applications (29.0 vs. 28.4; P = 0.003) and capillary glucose measurements (28.9 vs. 28.42; P = 0.017) were slightly higher in the MDC group than those in the control group (Fig. 4A and B).
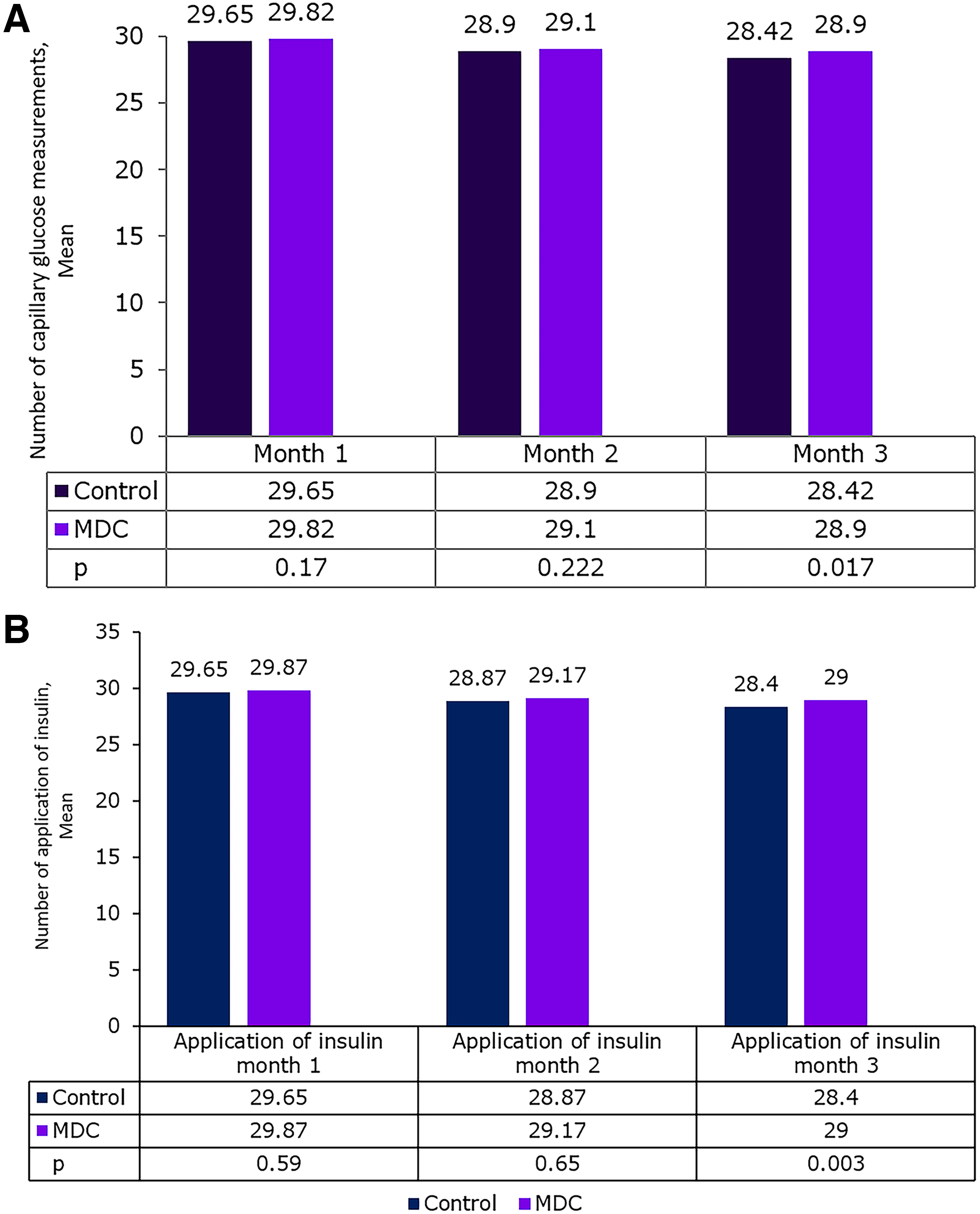
Hypoglycemia
In the MDC group, a total of eight ADA level 1 hypoglycemia events (plasma glucose <70 mg/dL) were reported during the study (during FPG measurement, n = 4; at any time of the day, n = 4). In the control group, a total of 14 ADA level 1 hypoglycemia events were reported (during FPG measurement, n = 8; at any time of the day, n = 6). An ADA level 2 hypoglycemia event (plasma glucose <54 mg/dL) was recorded in one participant in the control group during FPG measurement. Overall, the MDC group had a fewer events than the control group. No severe or nocturnal hypoglycemia events were reported in either study arm (Table 3).
Hypoglycemia

MDC, My Dose Coach™.
Body weight
A comparable weight gain was observed in both groups over the course of the study, with the mean change from baseline of 1.1 kg in the MDC group and 1.8 kg in the control group. The mean BMI at Week 12 was 29.1 kg/m2 and 29.0 kg/m2, respectively, was also comparable.
Cost-effectiveness
Overall, the costs of care and basic supplies were significantly lower in the MDC group (Mex$4890.00) than those in the control group (Mex$7582.00; P = 0.001) (Fig. 5). Direct costs of care and basic supplies over a 12-week period were Mex$4890 for the MDC group and Mex$7582 for the control group (P < 0.001). The direct cost analysis included insulin glargine (Gla-100, Gla-300), OAD, a glucometer, test strips, laboratory tests, travel expenses to the hospital, and income loss due to attending evaluations.

Direct costs of care and basic supplies (expense in Mexican pesos in 12 weeks). significance P < 0.05. MDC, My Dose Coach™.
Discussion
In the present randomized, controlled, open-label parallel group study, 80 participants were randomized to use either the MDC app or the conventional physician-guided method for BI titration. Higher proportion of the MDC app users reported progress in achieving their FPG targets in a shorter period than those in the control group. In addition, the MDC users achieved a greater decrease in FPG and HbA1c levels, with comparable incidence of hypoglycemia events, than those in the control group.
The data from this study are consistent with the results of the MDC study, an open-label, randomized controlled trial (RCT) conducted in 36 diabetes practices in Germany that compared the change in HbA1c after using a smartphone application for 12 weeks versus the standard of care for BI titration. 21 The findings from this study suggest that using the MDC app for BI titration might have resulted in a greater reduction in HbA1c levels, with no negative effects on safety outcomes, among participants with T2DM than BI titration guided by a written titration schedule. The safety and effectiveness of the MDC app have been evaluated in a real-world setting in a multinational observational MDC study. In this study, the overall analysis of real-world data from Algeria, Colombia, India, and Mexico revealed that more than 50% of participants using the MDC app for BI titration were able to achieve their individualized FPG target with a mean time to reach the target of 15.6 days. 23 Further, a sub-analysis of this multinational MDC study showed that a higher number of participants from Mexico (65.7%) achieved the FPG target than the overall study participants (55.6%). 23 The mean time to reach the FPG target was also shorter among participants from Mexico (14.6 days) than those in the overall study. 23 In a prospective open-label pilot study conducted in participants with T2DM (n = 157) from Northern Mexico, the use of the MDC app was associated with reductions in self-monitored plasma glucose (SMPG), FPG, and HbA1c. The proportion of participants reaching the FPG target (90–130 mg/dL) was 55.7%, and the mean reduction in HbA1c was 1.78%, with no events of severe hypoglycemia. 22 The results of the present study are in line with the aforementioned studies. More than 90% of the participants using the MDC app achieved the FPG target with a mean time to reach the target of 18 days. When the patients are involved in treatment decision-making, their understanding of diabetes care increases and positively impacts their self-management. Patient-personalized management, the increase in diabetes education, learning to use a glucometer, empowerment, and confrontation could have helped in achieving the desired results. 22
In the current study, the MDC app users reported a reduction in HbA1c (2.1%), which was in line with a previous study where the mean HbA1c was reduced by approximately 1.0% from a baseline value of 8.2%. A higher number of participants in the intervention group (MDC users) had a reduction in their HbA1c values than those in the control group. 21
In the present study, the BI dose increased in the MDC users from 16.9 U to 26.0 U, which is in line with a previous global study wherein an increase of 4 U in the BI dose was reported in the overall study population. 23 The frequency of hypoglycemia was lower in the MDC app users than in the control group. There was no reported severe hypoglycemia, which was comparable with another randomized multicenter study wherein no case of severe hypoglycemia was reported in the MDC app users. 23
Further, in this study, although both control and MDC groups were frequently measuring capillary glucose (28.4 vs. 28.9 readings per month), the MDC group demonstrated improvement in glycemic parameters, including a reduction in HbA1c and earlier attainment of the FPG target, without any notable difference in hypoglycemia. This suggests that the use of the MDC app may have benefits beyond BI titration. The application has features such as dosing reminders that could help improve treatment adherence. The present study is the first to report the cost-effectiveness of the MDC app. The results highlight the cost-effectiveness of MDC in terms of costs of care and basic supplies compared with the control group.
The users with a high FPG level at baseline might take a longer time to achieve their FPG target, which indicates that FPG baseline readings could have an impact on the time to reach the glycemic target. In general, HCPs set the individualized FPG target for conventional users. Therefore, the success was dependent on the availability of physicians (medical appointments) and how stringently physicians set the personalized goals for non-users. Using the MDC app reduces the dependency on a physician’s availability, and the user-friendliness of the application helps in the self-management of diabetes.
Overall, the findings suggest that the use of the MDC app could allow users to optimize their BI titration, thereby helping them in managing their treatment with the ongoing support of their HCPs. MDC can therefore equip users to overcome known challenges in achieving glycemic control, particularly since it is associated with low risk of hypoglycemia.
The strengths of this study include the randomization of participants into two groups (the control and MDC groups). This is the first study to evaluate the cost associated with BI titration with and without MDC support. Using the MDC app reduces the cost burden on people with T2DM. As very limited RCT data on the MDC app were available, this regional data could be generalized.
The limitations of the present study include a small number of study participants. The follow-up period was relatively short (12 weeks) but sufficient for BI titration. Therefore, it was unclear whether the intervention effect persisted beyond the 12-week period. Larger and long-term studies are required to assess impact on glycemic control, sustainability, and cost of MDC app. Based on results from the current study, future research should also consider variations in treatment duration and participants’ sociodemographic and clinical characteristics. A key limitation of this study is its open-label design, which may have introduced bias, particularly in the assessment of subjective outcomes such as self-reported adherence and hypoglycemia events. Participants’ awareness of their treatment allocation could have influenced their reporting behavior or adherence patterns, potentially leading to overestimation or underestimation of the treatment effects. In addition, the study lacked detailed app usage metrics, such as frequency of use, duration of interactions, and compliance with titration prompts. Future research should aim to integrate detailed app usage analytics to better understand user engagement patterns and how they may influence clinical outcomes.
Nonetheless, we recognize that gender-related biological and social factors can influence study outcomes, and this should be considered when interpreting the results. Future studies with balanced group characteristics or stratified analyses may help further clarify the role of gender in influencing outcomes.
Conclusions
The MDC app users showed a significant improvement in glycemic control with no risk of hypoglycemia at a lower economic cost compared with the conventional strategy. The findings from this RCT can provide further evidence supporting the efficacy and safety of the MDC app to achieve optimal glycemic control with the ease of BI titration in people with T2DM. However, long-term studies are suggested to identify potential factors affecting long-term adherence and to assess the impact of MDC app use on glycemic control, sustainability, and cost-effectiveness.
Authors’ Contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published. Dr. M.Á.P.-P. was involved in the conception, design, and conduct of the study. J.R.B.-A., S.P.-P., L.A.M., and M.E.S.-M. reviewed and approved the article.
Footnotes
Acknowledgments
The authors would like to thank the MDC users and healthcare professionals who participated in the trial. All authors take complete responsibility for the interpretation of the data in this review. The authors acknowledge the medical writing assistance provided by Sasikala Maadwar, PhD; Umakant Bahirat, PhD; and Anuja Vaidya, M. Pharm, from Sanofi.
Author Disclosure Statement
L.A.M. is an employee of Sanofi and may hold stocks in the company. M.E.S.-M. was an employee of Sanofi at the time of the study’s conduct and may hold shares in the company. J.R.B.-A. and S.P.-P. have no competing interests to declare.
Funding Information
Luis Anguiano Medrano (Sanofi) and Dr. Maria Elena Sanudo (formerly Sanofi; currently affiliated with a private office in Tlalpan, Mexico City) reviewed and approved the article. The My Dose Coach™ application was provided by Sanofi, while all other resources and materials were supplied by the investigators. The Mexican division of Sanofi only participated in the data analysis and the writing of the article.
Supplemental Material
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
