Abstract
Background:
Real-world studies of people with type 2 diabetes (T2D) have shown insufficient dose adjustment during basal insulin titration in clinical practice leading to suboptimal treatment. Thus, 60% of people with T2D treated with insulin do not reach glycemic targets. This emphasizes a need for methods supporting efficient and individualized basal insulin titration of people with T2D. However, no systematic review of basal insulin dose guidance for people with T2D has been found.
Objective:
To provide an overview of basal insulin dose guidance methods that support titration of people with T2D and categorize these methods by characteristics, effect, and user experience.
Methods:
The review was conducted according to the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines. Studies about basal insulin dose guidance, including adults with T2D on basal insulin analogs published before September 7, 2022, were included. Joanna Briggs Institute critical appraisal checklists were applied to assess risk of bias.
Results:
In total, 35 studies were included, and three categories of dose guidance were identified: paper-based titration algorithms, telehealth solutions, and mathematical models. Heterogeneous reporting of glycemic outcomes challenged comparison of effect between the three categories. Few studies assessed user experience.
Conclusions:
Studies mainly used titration algorithms to titrate basal insulin as telehealth or in paper format, except for studies using mathematical models. A numerically larger proportion of participants seemed to reach target using telehealth solutions compared to paper-based titration algorithms. Exploring capabilities of machine learning may provide insights that could pioneer future research while focusing on holistic development.
Introduction
Initiation of basal insulin is a complex and time-consuming task associated with clinical inertia.1 -5 Thus, approximately 60% of people with T2D treated with insulin do not reach glycemic targets.4,6 -8 Insulin titration is used when determining the optimal dose for an individual.2,4,9 This is necessary since people with T2D vary in pancreatic insulin production and insulin resistance.9,10 Hence, the optimal dose of basal insulin differs among people with T2D and may change over time due to, for example, stress levels, lifestyle changes, and sickness.
Suboptimal treatment is partly caused by non-adherence to treatment and failure to initiate or intensify treatment promptly.9,11 Lack of adjustment to insulin treatment is mainly caused by the complexity of the titration process. 5 This causes people with T2D to remain on suboptimal insulin doses, leading to less improvement in glycemic control than what could have been accomplished with an optimal dose.5,12,13 In addition, studies based on real-world data have shown both a delay in the initiation of basal insulin and insufficient dose adjustment during titration.1,14,15 Suboptimal insulin titration has been shown in the range of 3 to 12 months after initiation of active titration in clinical practice.3,6,16 -19 This elucidates that people with T2D, in some cases, have not reached glycemic target after 3+ months of active titration. Failure to achieve glycemic targets during the initial 3 months of titration is associated with a higher risk of failure to reach glycemic targets two years after the initiation. 15 This emphasizes the need for dose guidance supporting efficient and individualized basal insulin titration of people with T2D to provide optimal and timely treatment.
In recent years, basal insulin dose guidance has been of rapidly growing interest within international research, emphasized by an increase of publications on the subject. Despite this interest and the fact that it has been a research field for several decades, a preliminary search of the Cochrane Database of Systematic Reviews and Reviews, the International Prospective Register of Systematic Reviews (PROSPERO), and Joanna Briggs Institute (JBI) Evidence Synthesis revealed no systematic review of basal insulin dose guidance for people with T2D. Therefore, this systematic review aims to provide an overview of methods used for basal insulin dose guidance supporting titration of people with T2D and categorize these methods by characteristics, effect, and user experience.
Methods
Study Design
The systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) guidelines. 20 Therefore, a protocol was registered in PROSPERO on December 19, 2021 (CRD42021289364), forming the review’s basis. 21
Eligibility Criteria
Studies evaluating dose guidance methods supporting titration of people with T2D treated with basal insulin analogs in any setting, including adult participants (≤18 years) diagnosed with T2D, were considered. Studies investigating populations of mixed diabetes types without a transparent subgroup analysis or without a clear statement of diabetes type were excluded. Studies including participants on basal-bolus regimens, human or intermediate insulin, or other injectable antidiabetic treatment were excluded.
Primary studies reporting any glycemic outcome published in English, Danish, Norwegian, or Swedish before September 7, 2022, as peer-reviewed full-text, were included. All study designs except study protocols, animal research, expert opinions, and case studies were considered.
Information Sources and Search Strategy
A comprehensive systematic search was performed in PubMed, Embase, and IEEE by one author (C.H.N.T) with assistance from a research librarian. Citation and reference searches were conducted in Google Scholar. Authors of relevant studies were contacted if additional information was needed.
Unstructured searches in PubMed and Google Scholar were performed to identify relevant search terms. The search was adjusted to each database. Search terms included different synonyms and spellings. Search functions were applied, including thesaurus, Boolean operators, phrase, truncation, free text, and advanced search (Supplementary material).
Selection Process
First, studies identified through the systematic search were uploaded to RefWorks (version 2.1.0.1). Second, duplicates were removed using the functions
Data Extraction and Synthesis
One author (C.H.N.T.) extracted data using a sheet in Microsoft Excel (2016). Extracted data included study characteristics (title, authors, publication year, study design, country, sample size, and duration of study), participant characteristics (age, sex, body mass index [BMI], insulin-naïve, and initial HbA1c), characteristics of the dose guidance method (setting, description of the method, and type of insulin used), and glycemic outcomes.
A narrative synthesis of extracted data was conducted, and characteristics of studies and populations were described. The narrative synthesis focused on categorizing dose guidance methods and assessing effect of the interventions and user experience according to the categorization.
Risk of Bias Assessment
Critical appraisal tools from JBI were applied by study design of the studies to assess risk of bias. 22 Study design was determined using Andrews and Likis, 2015. 23 One author (C.H.N.T.) assessed included studies with support from co-authors.
Before critical appraisal was performed, authors agreed on a scoring system and cut-off points per the JBI reviewers manual. 24 Studies were judged as described in Melo et al. 25 A suitable tool for simulation studies was not found from JBI; therefore, the critical appraisal tool from Fone et al 26 was used.
Results
Study Selection
A total of 4,363 papers were found. After removing duplicates, 3,327 papers were included in title and abstract screening. Of those, 280 papers were found eligible for full-text screening. Thirty-one papers met the inclusion criteria and were included in the review. Four additional papers were identified through reference and citation searches. Thus, 35 articles were included in this review. The selection process is presented in Figure 1. The supplementary material contains a tabular overview of data extracted from the included studies.
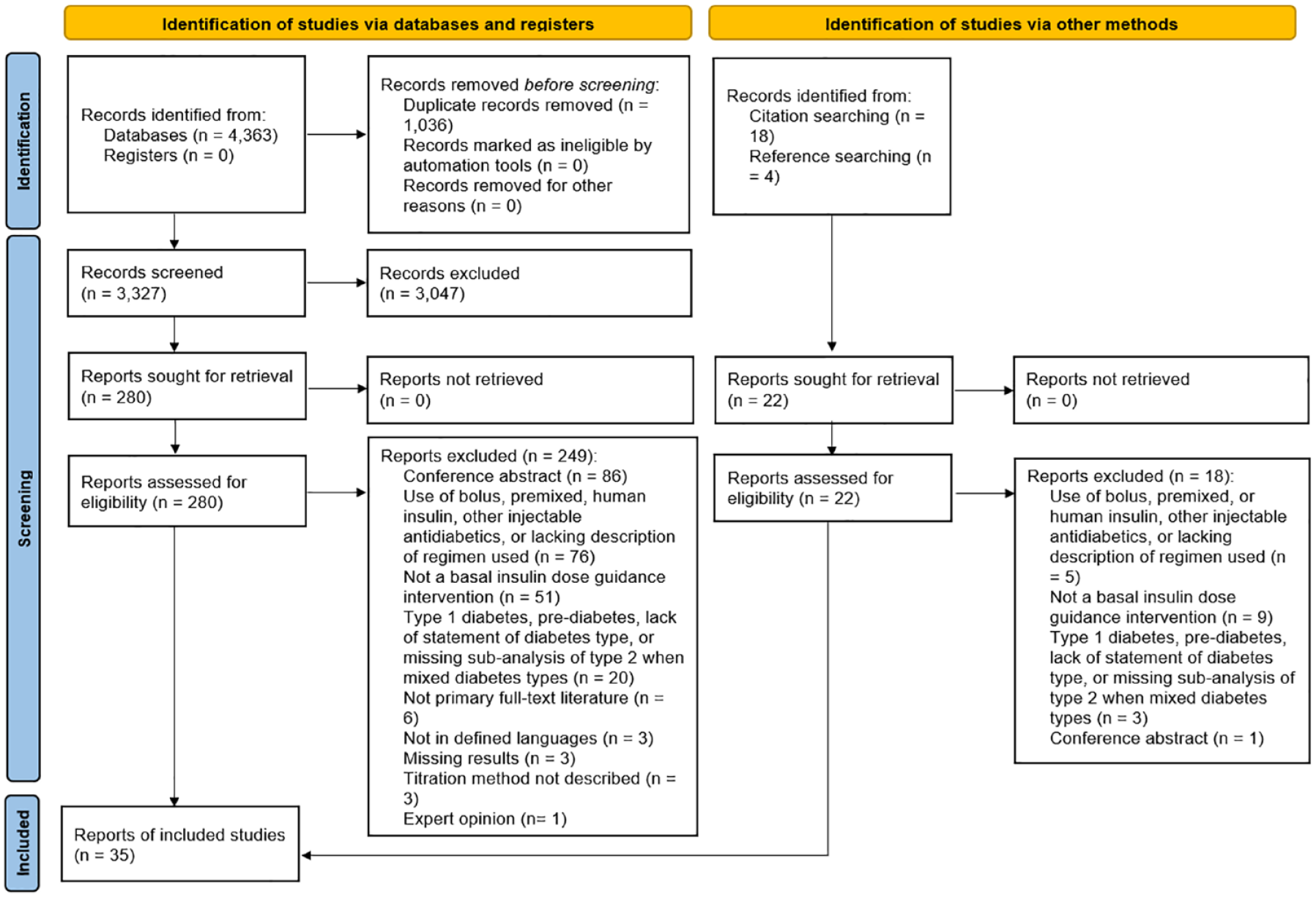
The selection process is illustrated in a preferred reporting items for systematic reviews and meta-analyses flowchart. 20
Some studies seemed eligible but were excluded due to use of human insulin or basal-bolus regime in a subgroup of participants without a transparent subgroup analysis of participants treated only with basal insulin analogs, or the use of bolus insulin as rescue medication.13,27 -29
Study Characteristics
Seven studies were quasi-experimental design,30 -36 20 studies were randomized controlled trials (RCT),37 -56 three studies were mixed method,57 -59 one study was of qualitative design, 60 one study was a cohort, 61 and three studies were simulation design.8,10,62 Mixed method studies were a mix of quasi-experimental and qualitative design. The studies were published from 2006 to 2022 and enrolled 19,432 people with T2D. The length of the studies ranged from 28 days to 12 months.
The studies were conducted in 31 countries across Europa, Asia, North and South America, the Middle East, and Africa. Seven studies did not specify in which country it was conducted.8,10,32,48,55,61,62
Participant Characteristics
Characteristics of participants were similar regarding initial BMI, age, and sex distribution. The most significant difference was whether participants were insulin-naïve at start-of-trial. The study population in 60% of the studies were insulin-naïve.8,10,31,34,35,37,39 -41,45,46,48 -51,53,56 -58,61,62 In 14% of studies, the population continued basal insulin treatment initiated before the study,30,32,33,36,43 and 26% of studies included a study population of both insulin-naïve and people contuneing previously initiated treatment.38,42,44,47,52,54,55,59,60 Initial HbA1c, duration of diabetes, and whether the study population was insulin-naïve are essential factors to consider when comparing the impact on glycemic control from dose guidance interventions.15,63 -67 All study populations had initial HbA1c above 7%, and diabetes duration ranged from 2.9 to 15.9 years.
Characteristics of the Dose Guidance Methods
Twenty-one of identified dose guidance methods were developed for titration of glargine,30,32,34,36 -39,41,42,44,45,47,49,51,53 -56,58,61,62 three for detemir,40,48,52 five for degludec,8,10,31,43,46 one for icodec 50 , and one for glargine and detemir. 59 Four studies did not specify insulin further than it was basal insulin analogs.33,35,57,60
Approximately 70% of the studies were in an outpatient clinic. The remaining studies were in primary care34,35,42,51,52,61 or did not specify the setting.8,10,36,50,62
Categorization of the dose guidance methods
Identified dose guidance methods were divided into three categories: paper-based titration algorithms, telehealth solutions, and mathematical models (Figure 2).
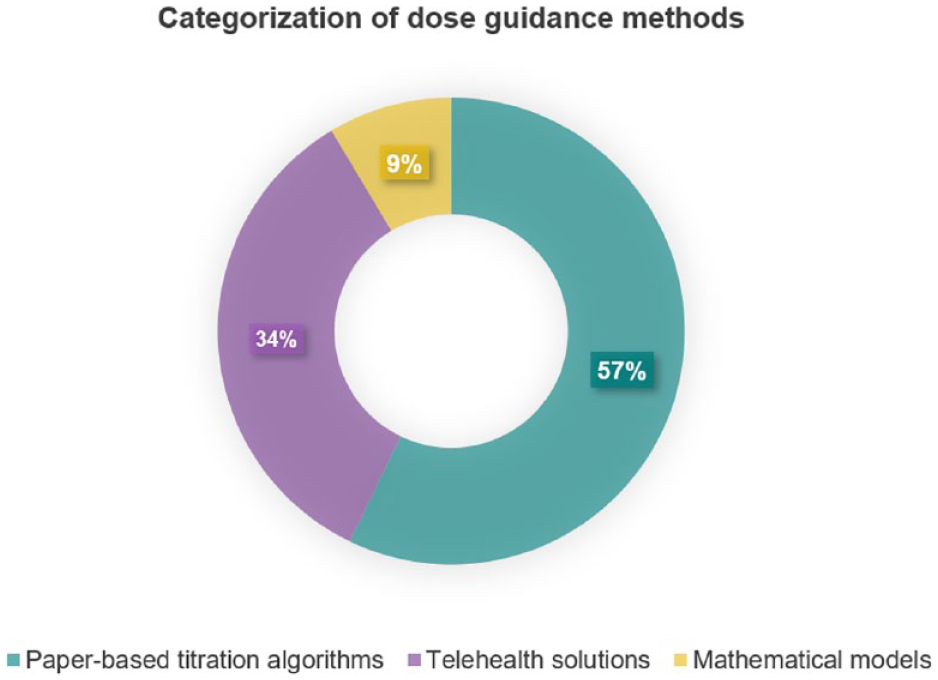
Overview of type of dose guidance methods used in the included studies.
Paper-based titration algorithms reflect standard practice at the time of writing. The studies investigated algorithms with varying targets and sizes of dose adjustment carried out during in-person visits. In total, 20 studies investigated paper-based titration algorithms.32,34,36 -38,40 -43,46,48 -53,56,58,61,62
Telehealth solutions covered telemonitoring solutions with titration across a digital platform30,45,54,57,59,60 and combined with home visits, 35 or self-titration decision support.33,39,44,47,55 In contrast to studies addressing paper-based algorithms, the organizational setup was altered in these studies. Interactions between participants and healthcare professionals (HCP) were primarily handled over distance via phone. In total, 12 studies investigated telehealth solutions.30,33,35,39,44,45,47,54,55,57,59,60
Mathematical models were investigated by three studies using compartment modeling and control theory.8,10,31 Most of these studies did not specify the use case of the method.
Dose guidance methods covered both physician- and patient-led methods. The distribution was similar for paper-based titration algorithms and telehealth solutions, where most approaches based on mathematical models did not specify the intended user (Figure 3).
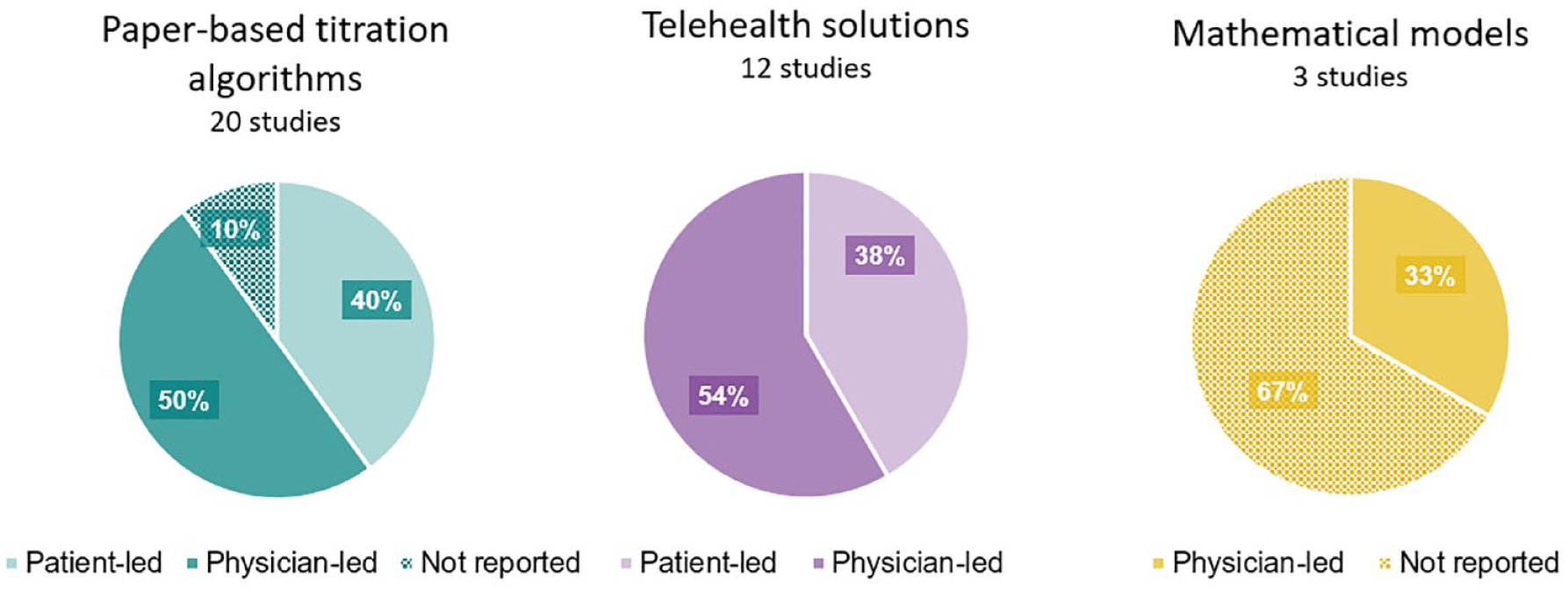
Distribution of the intended user of the identified dose guidance methods according to the three main categories: paper-based titration algorithms, telehealth solutions, and mathematical models.
Description of the dose guidance method is presented in Table 1.
Overview of How Basal Insulin Was Titrated in the Included Studies Grouped by the Titration Algorithm Used.
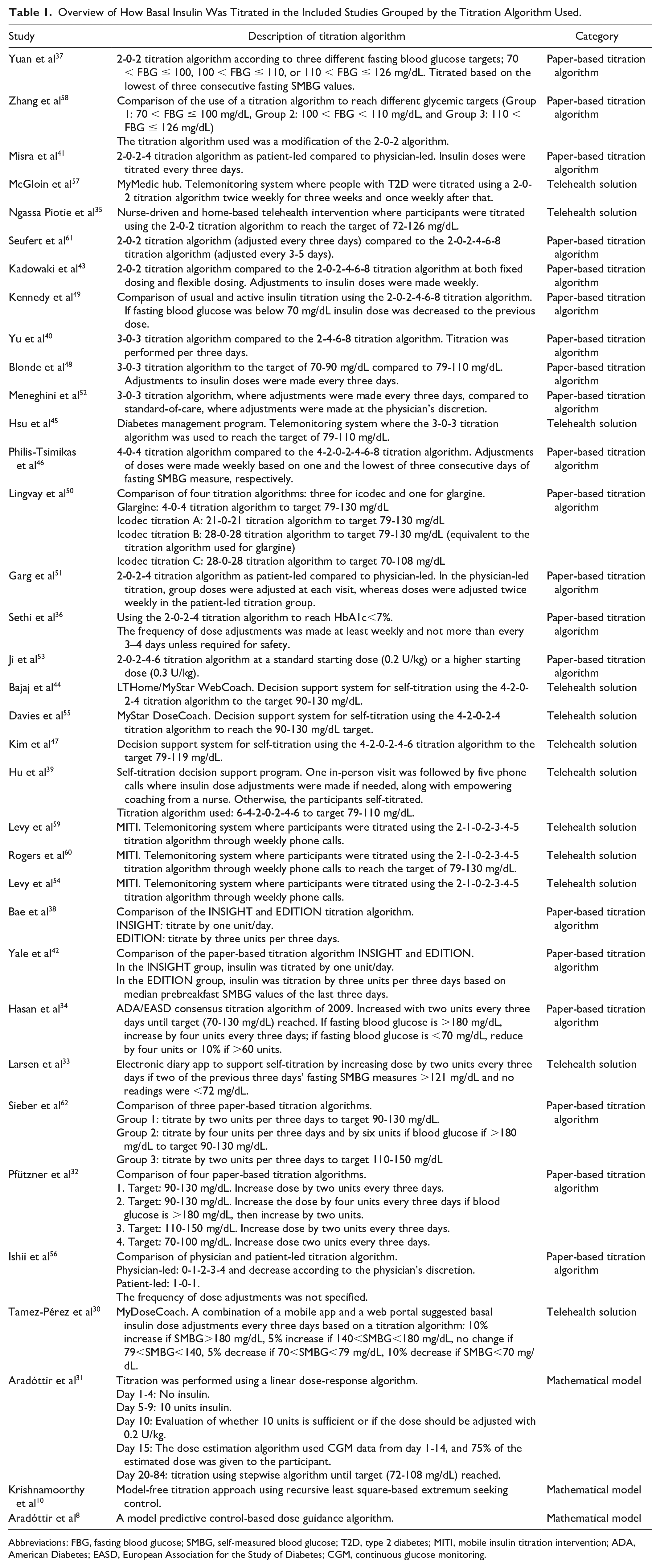
Abbreviations: FBG, fasting blood glucose; SMBG, self-measured blood glucose; T2D, type 2 diabetes; MITI, mobile insulin titration intervention; ADA, American Diabetes; EASD, European Association for the Study of Diabetes; CGM, continuous glucose monitoring.
Table 1 elucidates that all identified dose guidance methods, except in Krishnamoorthy et al 10 and Aradóttir et al, 8 used titration algorithms to titrate basal insulin either in a digital tool or paper-based format. Aradóttir et al 31 mixed the use of a mathematical model with use of a paper-based titration algorithm. Titration algorithms varied considerably among included studies, as approximately 18 algorithms were used. However, similar titration algorithms were found in studies investigating paper-based titration algorithms and telehealth solutions, for example, the 2-0-2 titration algorithm.
Effect of the dose guidance methods
Studies reported very heterogeneous glycemic outcomes (Supplementary material). The most frequently reported outcome was proportion of participants reaching glycemic target. However, this target differed among studies. Some studies used HbA1c < 7% as target, while others used fasting blood glucose within a specific range. The difference in how target was defined made it challenging to compare effect across studies. To enable a comparison to some degree to elucidate tendencies in effect across different dose guidance methods, an overview of the proportion of participants reaching target is presented in Figure 4. Approximately 23% of studies did not report proportion of participants reaching target at end-of-trial.8,10,33,40,45,57,58,62
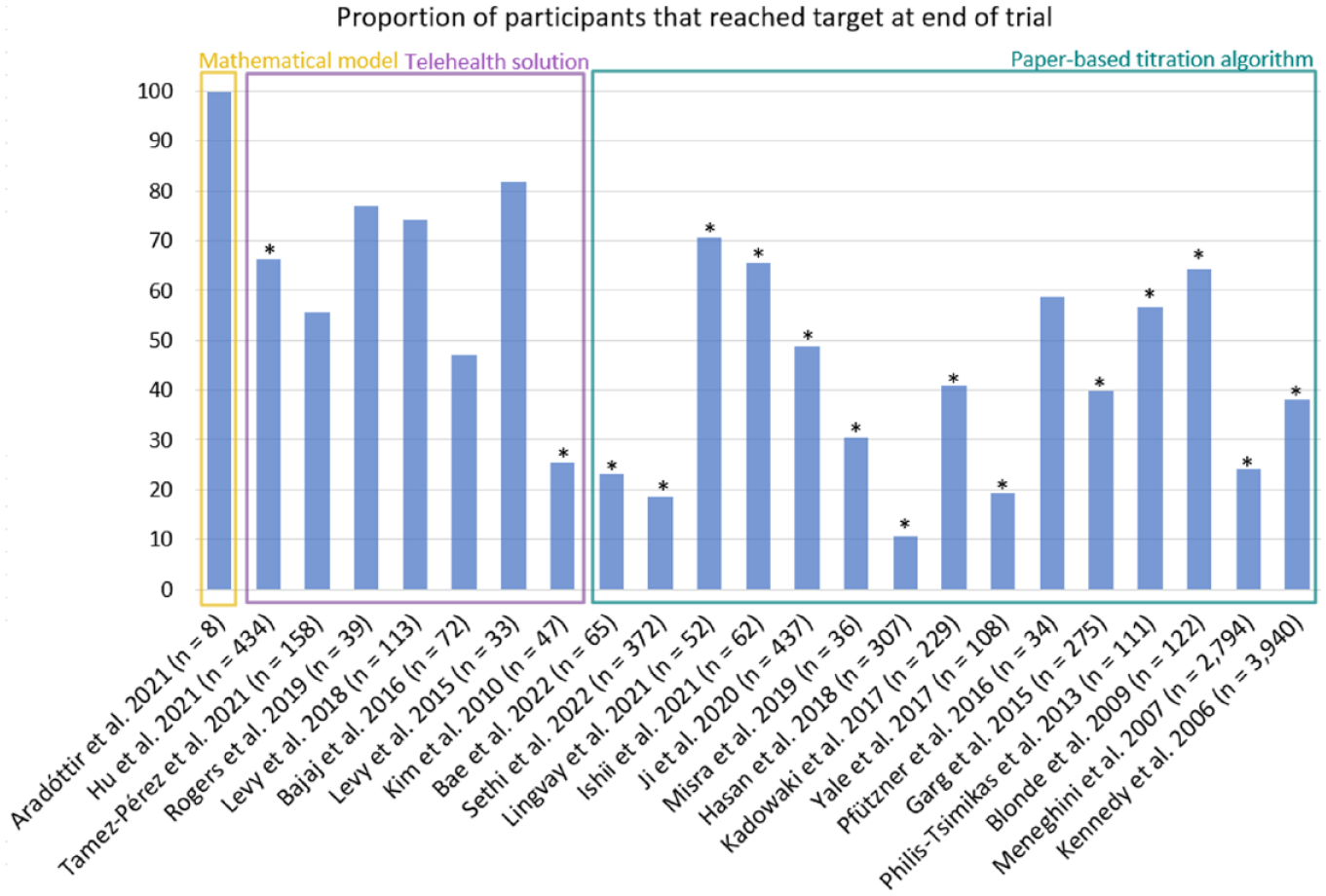
Summary of the proportion of participants that reached a predefined glycemic target. Only studies that reported target as either fasting blood glucose within the target of 79-130 mg/dL, 90-130 mg/dL, or 72-108 mg/dL or HbA1c<7% (marked with *) is included in this figure.
As the only study investigating mathematical models, Aradóttir et al 31 reported that all participants reached target with a mean time to target of 44 days (n = 8). The mean proportion of participants reaching target in studies investigating telehealth solutions was 61 ± 20% when considering both targets and 46 ± 29% when only considering HbA1c targets. The mean for paper-based titration algorithms was 41 ± 19% in both cases. This may indicate a tendency for a numerically larger proportion of participants titrated using telehealth solutions to reach target compared to paper-based titration algorithms.
Among these studies, few reported time-to-target. None of the studies about paper-based titration algorithms reported time-to-target. Three studies about telehealth solutions reported mean time-to-target, which ranged from 20 to 66 days.30,54,59 It should be noted that two of these studies investigated the same telehealth solution.54,59 Since few studies reported time-to-target, it is relevant to consider the mean study duration within the three categories to get an indication of time used to reach target. The mean duration was 22 ± 9 weeks for studies addressing paper-based titration algorithms, 16 ± 6 weeks for studies addressing telehealth solutions, and 11 ± 2 weeks for studies addressing mathematical models, of which most were simulations. On average, study duration of studies investigating paper-based titration algorithms was twice as long as for mathematical models and 6 weeks longer than telehealth studies.
User experience of the dose guidance methods
User experience was investigated by 14 studies, of which 11 studies36,38,41,42,44,45,51,54 -57 reported outcomes from standardized questionnaires (eg, Diabetes Treatment Satisfaction Questionnaire [DTSQ]), three studies35,57,60 reported outcomes from interviews, and three studies35,58,59 reported outcomes from non-standardized questionnaires. Studies addressing mathematical models did not investigate user experience.
The studies reporting baseline changes in the DTSQ scores showed varying results (Supplementary material). For telehealth solutions, the change ranged from 0.8 to 10.1 and from 0.1 to 11.7 for paper-based titration algorithms. This revealed no apparent difference in the change of DTSQ score between the two methods.
From non-standardized questionnaires and interviews, HCPs and people with T2D found telehealth solutions convenient and appropriate for titration of basal insulin.35,57,59,60 Two of these studies investigated the same telehealth intervention.59,60 People with T2D found it convenient to have fewer in-person interactions while maintaining contact with HCP via phone. In Rogers et al, 60 HCPs found telehealth intervention could reduce the burden of titration. In contrast, McGloin et al 57 elucidated an increased workload among HCPs caused by a large amount of generated data.
Only the study by Zhang et al 58 reported qualitative findings on the use of paper-based titration algorithms. The study found a gap between preferences of people with T2D and HCPs when choosing a titration algorithm. People with T2D preferred simple and easy-to-use algorithms. In contrast, HCPs preferred algorithms recommended by guidelines with higher perceived efficacy in lowering blood glucose levels and which were known to the HCP.
Critical Appraisal of the Studies
Table 2-6 show the results of critical appraisal of the included studies.
Summary of Critical Appraisal Assessed by JBI Critical Appraisal Checklist for Randomized Controlled Trials.
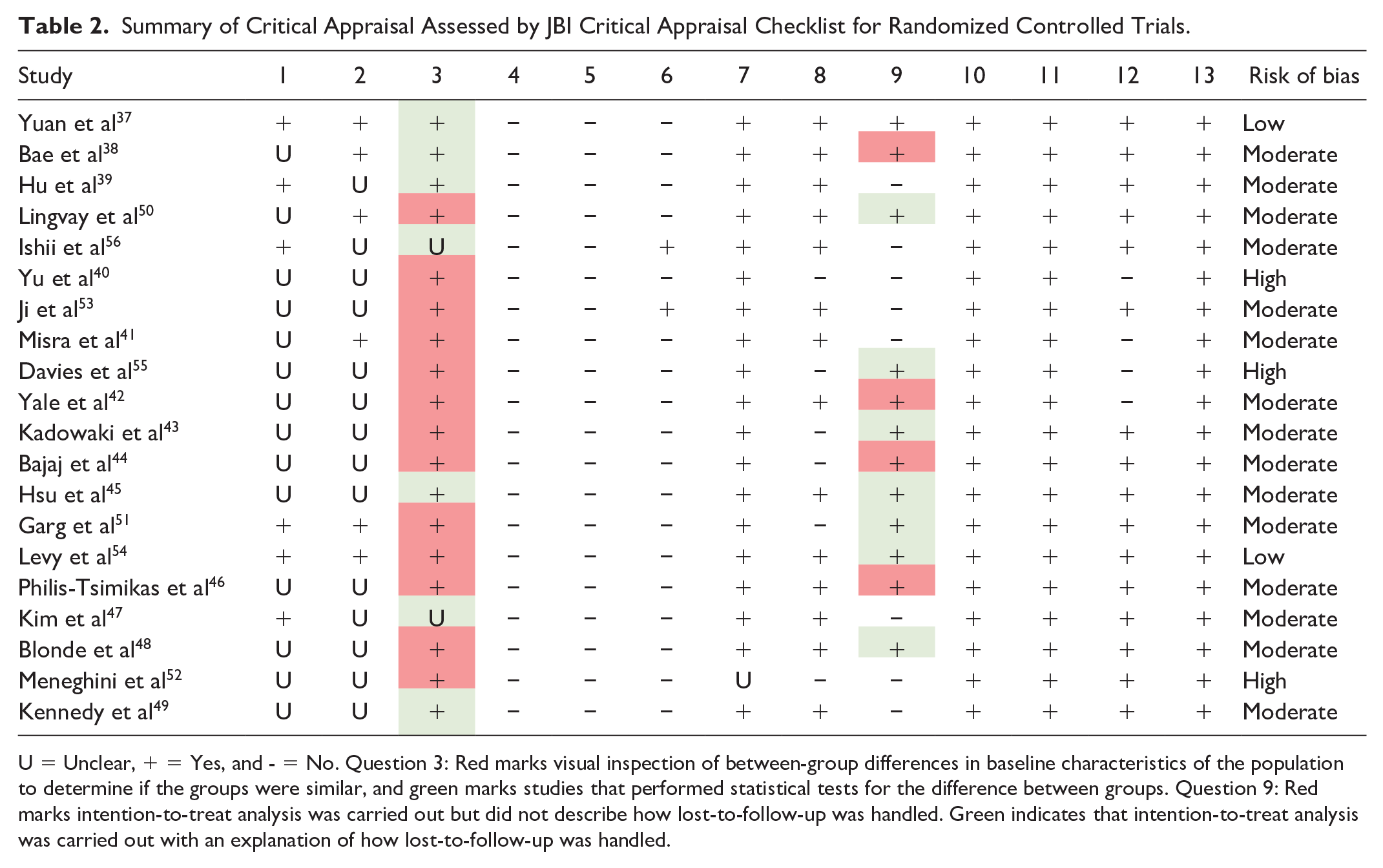
U = Unclear, + = Yes, and - = No. Question 3: Red marks visual inspection of between-group differences in baseline characteristics of the population to determine if the groups were similar, and green marks studies that performed statistical tests for the difference between groups. Question 9: Red marks intention-to-treat analysis was carried out but did not describe how lost-to-follow-up was handled. Green indicates that intention-to-treat analysis was carried out with an explanation of how lost-to-follow-up was handled.
Summary of Critical Appraisal Assessed by JBI Critical Appraisal Checklist for Quasi-Experimental Studies, Including Assessment of the Qualitative Part of Mixed Method Studies.
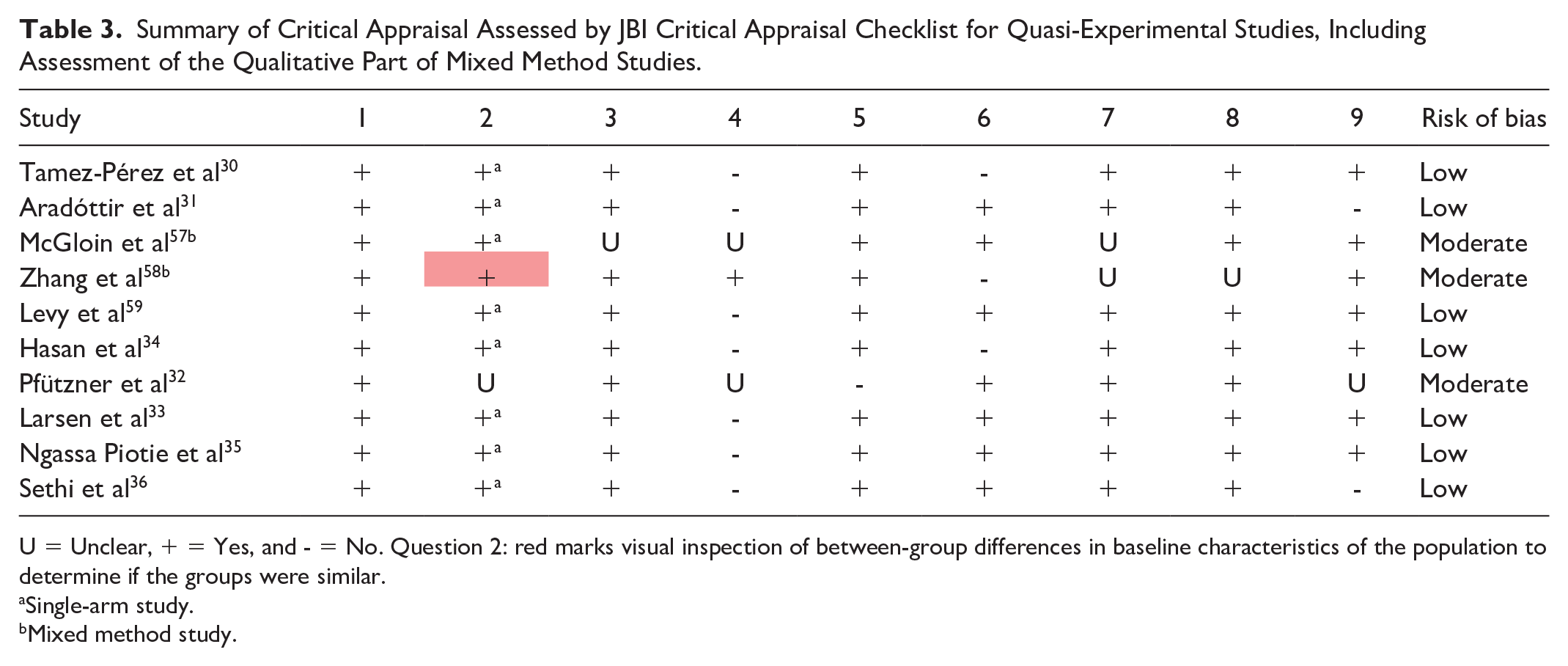
U = Unclear, + = Yes, and - = No. Question 2: red marks visual inspection of between-group differences in baseline characteristics of the population to determine if the groups were similar.
Single-arm study.
Mixed method study.
Summary of Critical Appraisal Assessed by JBI Critical Appraisal Checklist for Qualitative Studies, Including Assessment of the Qualitative Part of Mixed Method Studies.
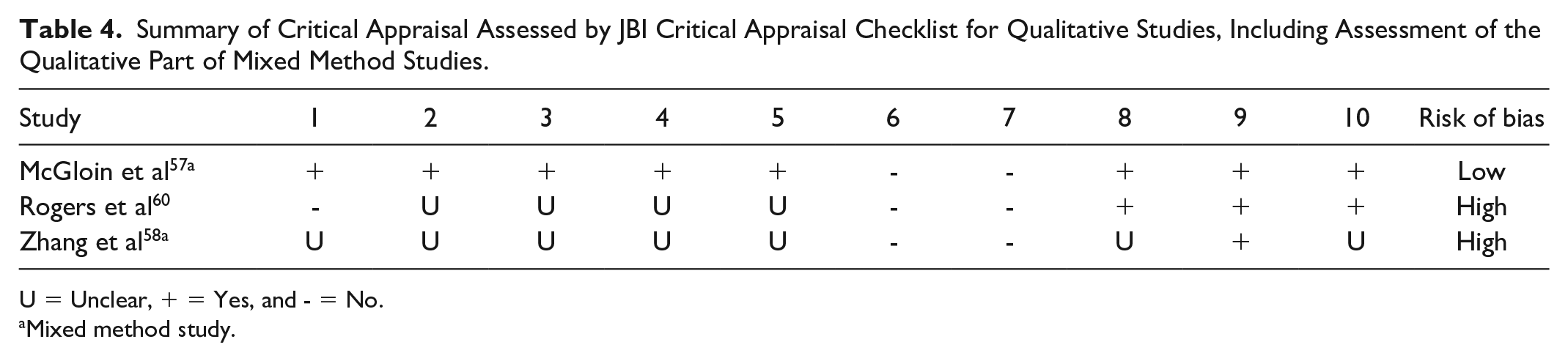
U = Unclear, + = Yes, and - = No.
Mixed method study.
Summary of Critical Appraisal Assessed by JBI Critical Appraisal Checklist for Cohorts.

U = Unclear, + = Yes, and - = No.
Summary of Critical Appraisal Assessed by the Checklist in Fone 26 for Simulation Studies.
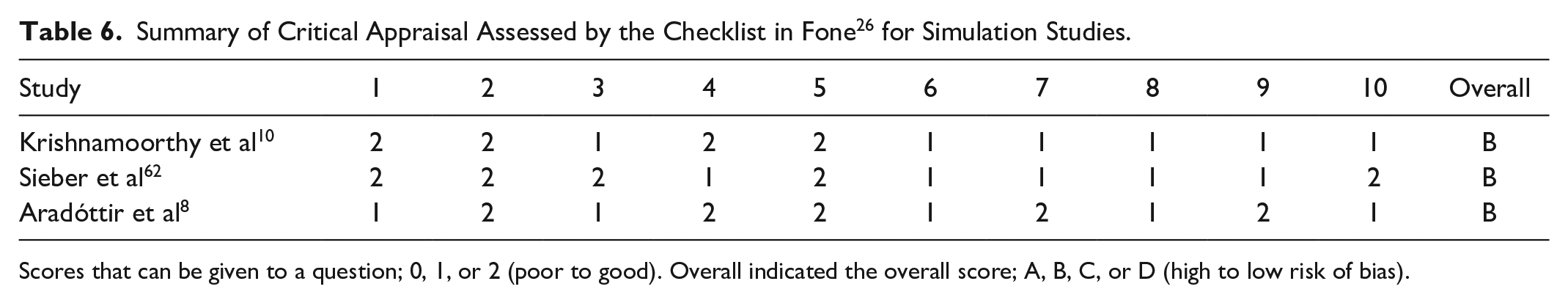
Scores that can be given to a question; 0, 1, or 2 (poor to good). Overall indicated the overall score; A, B, C, or D (high to low risk of bias).
Discussion
Summary of Evidence
The review aimed to provide an overview of dose guidance methods supporting basal insulin titration of people with T2D and categorize these according to characteristics, effects, and user experience. Overall results showed three categories of methods: paper-based titration algorithms, telehealth solutions, and mathematical models. Most studies investigated implementations of paper-based titration algorithms. Studies investigating digital solutions for basal insulin titration for people with T2D were limited to simple telehealth solutions and, in one case, a mathematical model embedded into a decision support system. In summary, all studies used titration algorithms either in paper form or digital, except for the mathematical models.
Similar findings are seen in Deerochanawong et al, 19 which highlighted use of paper-based titration algorithms and telehealth solutions when investigating titration of insulin glargine 100 U/mL in an Asian population. However, use of mathematical models was not reported. Furthermore, Kerr et al 68 found indications for improved glycemic control when using digital solutions to manage T2D treatment compared to standard of care. This is further supported by Hangaard et al, 69 which found a significant improvement in HbA1c when using telemedicine among people with T2D. These studies did not focus on basal insulin titration but overall treatment of people with T2D. However, it is feasible to assume that a similar effect may be seen when using telemedicine for titrating basal, which aligns with the tendency observed in this review.
User experience was not investigated thoroughly by included studies. Yet, common characteristics were the wish of people with T2D for simple and easy-to-use solutions and HCPs’ attention to effect on workload. Concerning telehealth solutions, HCPs, in some cases, uttered concern about increased data being generated compared to standard practice affecting workload. 57 None of the studies investigating mathematical models looked at user experience. Consideration of user experience when developing methods for basal insulin dose guidance is essential to ensure a holistic solution aimed at the intended end-user and thereby to secure effect in a real-world setting. 70 Especially considering solutions aimed at people with T2D due to known issues of non-adherence to treatment.71,72
Strengths and Limitations
The broad scope and comprehensive literature strengthen the present systematic review. However, relevant studies may have been overlooked since the search was limited to use of basal insulin analogs and English, Danish, Norwegian, and Swedish language.
The heterogeneity of reported glycemic outcomes and differences in study design complicated comparison of effect. Validity of the review is weakened since mainly one reviewer screened the search results. To minimize this effect, co-authors were continuously consulted to clarify doubts about inclusion of studies and during critical appraisal. Furthermore, the review was strengthened since the structured search was performed with assistance from a research librarian, ensuring a thorough search.
Implications for Future Research
Mathematical models were limited to three studies which were mainly evaluated through simulation. Expect a study by Aradóttir et al, 31 where the solution was tested on eight participants showing promising results. The limited use of mathematical models may be due to the complex nature of T2D and heterogeneity of the population caused by varying insulin sensitivity and production. This complicates modeling of insulin’s effect on blood glucose. The modeling task is further complicated by the limited available information about people with T2D. Glucose measures are typically performed using glucometers, and frequency of these measures varies depending on the individual in question.
In contrast, people with type 1 diabetes more often use continuous glucose monitoring to measure blood glucose, enabling more thorough insight into blood glucose levels throughout the day.73 -75 Similar challenges have been recognized by studies addressing mathematical models.8,10
New technologies enabling improved data collection might ease some of the challenges in modeling insulin’s effect on blood glucose levels for people with T2D using mathematical models. Kerr et al 68 highlight that new technology that supports improved data capturing may facilitate better treatment support when combined with dose recommendation software. Furthermore, addition of automated data-driven dose guidance might help rectify the increased workload for HCP that, in some cases, has been reported when introducing new technology. 76
At the time of writing, machine learning methods, which are frenquently used in other areas of diabetes reasearch, have focused on detection or prediction of hypoglycemic events, blood glucose levels, and optimal bolus insulin dosing in relation to people with T2D. 77 In the future, exploring the capability of machine learning methods for basal insulin dose guidance for people with T2D may provide insight into the field that could pioneer future research.
Conclusions
Three basal insulin dose guidance categories aimed at people with T2D were identified: paper-based titration algorithms, telehealth solutions, and mathematical models. Compared to paper-based titration algorithms, a numerically larger proportion of participants reached a predefined target using telehealth solutions.
Few studies investigated user experience. Some studies underlined a possible increase in workload when using telehealth solutions due to increased data. Futhermore, it was found that people with T2D preferred simple and easy-to-use solutions and fewer in-person visits.
Future work might benefit from exploring the capabilities of machine learning methods for basal insulin dose guidance for people with T2D, focusing on a simple and easy-to-use solution that does not increase the workload for HCPs.
Supplemental Material
sj-xlsx-1-dst-10.1177_19322968221145964 – Supplemental material for Time for Using Machine Learning for Dose Guidance in Titration of People With Type 2 Diabetes? A Systematic Review of Basal Insulin Dose Guidance
Supplemental material, sj-xlsx-1-dst-10.1177_19322968221145964 for Time for Using Machine Learning for Dose Guidance in Titration of People With Type 2 Diabetes? A Systematic Review of Basal Insulin Dose Guidance by Camilla Heisel Nyholm Thomsen, Stine Hangaard, Thomas Kronborg, Peter Vestergaard, Ole Hejlesen and Morten Hasselstrøm Jensen in Journal of Diabetes Science and Technology
Supplemental Material
sj-xlsx-2-dst-10.1177_19322968221145964 – Supplemental material for Time for Using Machine Learning for Dose Guidance in Titration of People With Type 2 Diabetes? A Systematic Review of Basal Insulin Dose Guidance
Supplemental material, sj-xlsx-2-dst-10.1177_19322968221145964 for Time for Using Machine Learning for Dose Guidance in Titration of People With Type 2 Diabetes? A Systematic Review of Basal Insulin Dose Guidance by Camilla Heisel Nyholm Thomsen, Stine Hangaard, Thomas Kronborg, Peter Vestergaard, Ole Hejlesen and Morten Hasselstrøm Jensen in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors thank research librarian Connie Skrubbeltrang for competent assistance in the literature search.
Abbreviations
DTSQ, Diabetes Treatment Satisfaction Questionnaire; HCP, healthcare professional; JBI, Joanna Briggs Institute; PRISMA, preferred reporting items for systematic reviews and meta-analyses; PROSPERO, International Prospective Register of Systematic Reviews; RCT, randomized controlled trial; T2D, type 2 diabetes.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Author P.V. is head of research at the Steno Diabetes Center North Denmark, funded by the Novo Nordisk Foundation. Author M.H.J is a former Novo Nordisk employee and holds Novo Nordisk shares.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
