Abstract
Objectives:
We aim to increase awareness of pleomorphic mantle cell lymphoma as a rare, but aggressive form of lymphoma with propensity for recurrence in secondary locations.
Methods:
We report the case of a 70-year-old man who presented with chronic post-nasal drainage, dysphagia, and voice changes caused by a tongue base mass.
Results:
Partial excision and pathology showed a pleomorphic mantle cell lymphoma, and radiation treatment was completed. A regional recurrence was detected 3 years later and treated with radiation. He had no evidence of disease 17 months after treatment of the recurrence and is under close surveillance.
Conclusions:
Pleomorphic mantle cell lymphoma is an aggressive subtype of non-Hodgkin lymphoma that can affect the head and neck. Confirming the diagnosis with immunotyping and genotyping from fresh specimens can guide appropriate treatment and then close clinical follow-up.
Keywords
Introduction
Oropharyngeal non-Hodgkin lymphoma (NHL) is rare, accounting for about 1% of oropharyngeal malignancies. 1 Although the most common type of NHL is large B-cell lymphomas, it is necessary to distinguish between the types due to differences in prognosis and treatment. 2 Mantle cell lymphoma (MCL) is a rare B-cell malignancy, accounting for only 5% of malignant lymphomas. 3 It typically occurs in men over the age of 60 years. 4 Extranodal sites can include Waldeyer ring, bone marrow, peripheral blood, and the gastrointestinal tract.5-7
The World Health Organization (WHO) classifies MCL into variant, blastoid, and pleomorphic subtypes. 8 Determining the specific type of MCL using immunohistochemistry, flow cytometry, and cytogenetics is important to determine the prognosis as well as the proper clinical treatment.3,9 There has only been 1 previous published case report of tongue base pleomorphic variant MCL, but the case did not detail treatment or follow-up. 2 There are a few reported cases of other subtypes of MCL in the tongue base, but there was no reported long-term follow-up, because all the cases died of the disease within 18 months.5,7,10 Pleomorphic MCL, in particular, needs to be recognized and treated early, because it compounds the low cure rate of low-grade with the aggressive nature of high-grade lymphoma. 11
Case Report
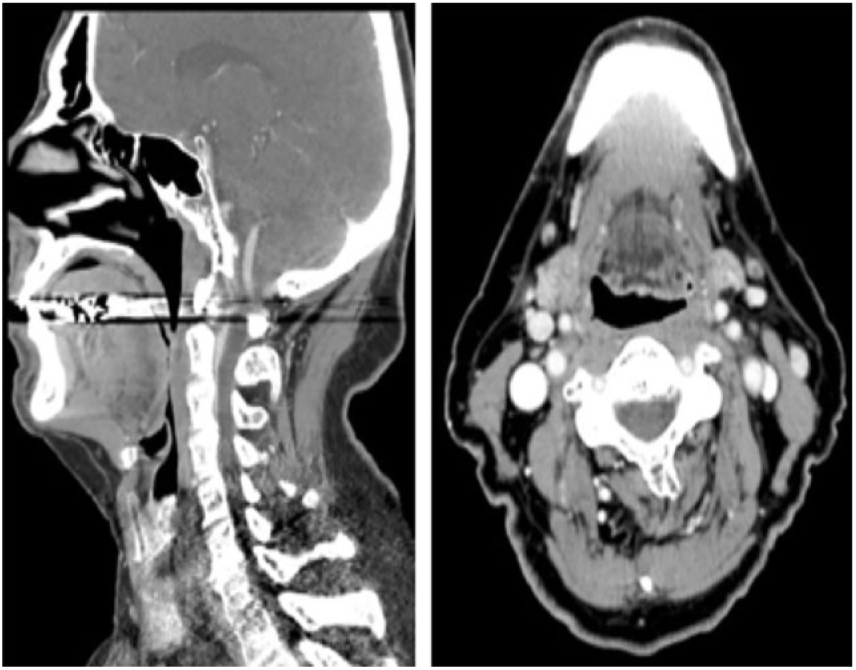
A 70-year-old man with a distant 10-year history of smokeless tobacco use presented with 1 month of progressive dysphagia, difficulty with secretions, throat discomfort, and a muffled voice. He had nasal congestion and postnasal drainage and therefore attributed his throat symptoms to his sinuses. Further workup revealed a tongue base mass, and he was referred to our institution for evaluation. Flexible fiberoptic laryngoscopy showed a partially obstructive, smooth, multi-lobulated mass taking up two-thirds of the tongue base. A computed tomography scan with contrast showed a 4 cm × 2.8 cm × 4.6 cm mass displacing the epiglottis posteriorly and narrowing the airway. There was no cervical lymphadenopathy (Figure 1). A positron emission tomography (PET) scan showed a fluorodeoxyglucose (FDG) avid tongue base mass with a standardized uptake value (SUV) of 10.5 and likely inflammatory mediastinal nodes with moderate avidity and a SUV of 5.1, but no other sites of disease.
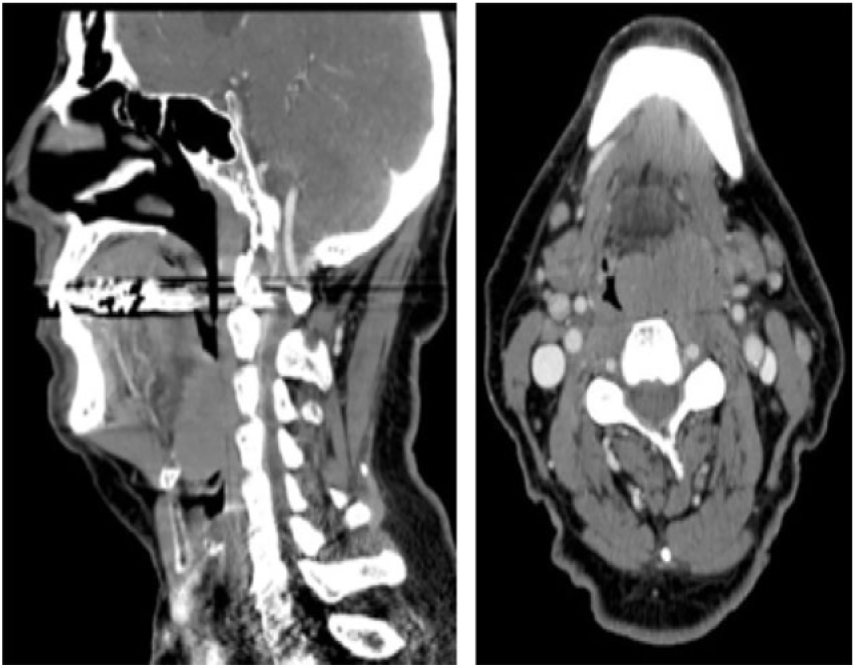
Sagittal and axial computed tomography of the neck with contrast showing the obstructing tongue base mass at initial presentation.
He underwent direct laryngoscopy, esophagoscopy, and biopsy. No secondary lesions were identified. Because of the obstruction caused by the mass, it was partially debulked, including part of the posterolateral pharyngeal wall to which it was attached. He recovered well postoperatively, without any significant aerodigestive compromise.
Histology and flow cytometry were consistent with pleomorphic MCL (Figures 2 to 4). The B-cell population was positive for cyclin D1 (Figure 5), CD20, CD5, Bcl-6, and MUM-1 and negative for CD23 and CD10. Ki-67 staining averaged 30%. Fluorescent in situ hybridization (FISH) for CCND1/IGH (11;14) translocation was positive and further supported the diagnosis of stage 1 pleomorphic MCL (Figure 6).2,5,12
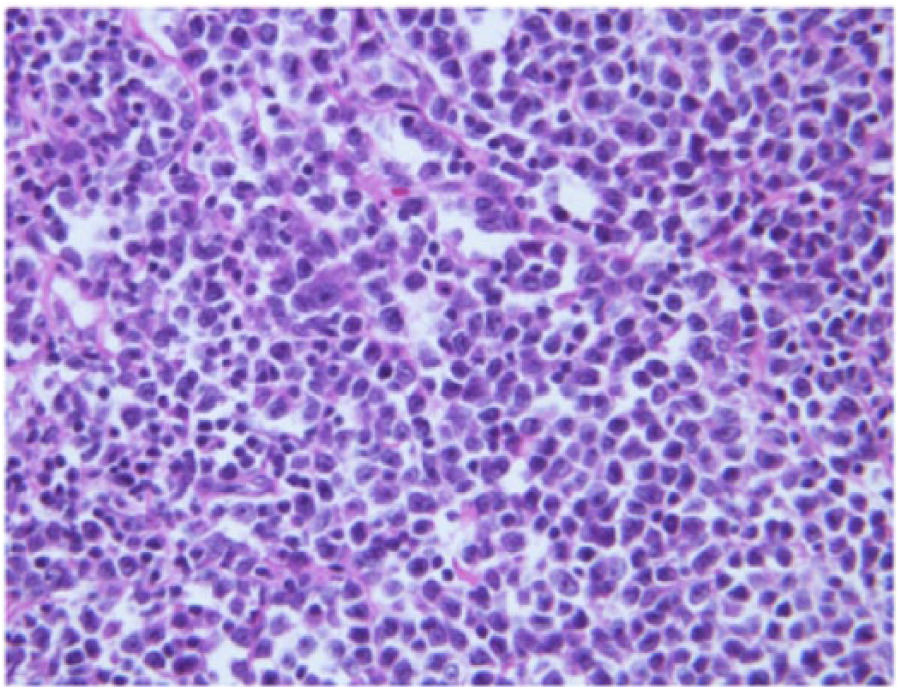
Hematoxylin and eosin stain of the mass, 400×.
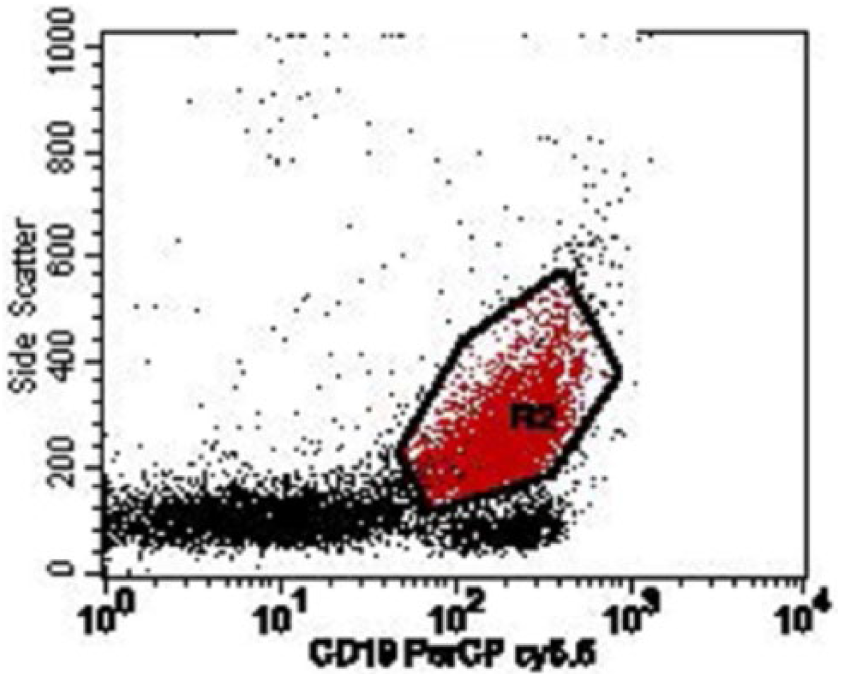
CD19 flow cytometry.
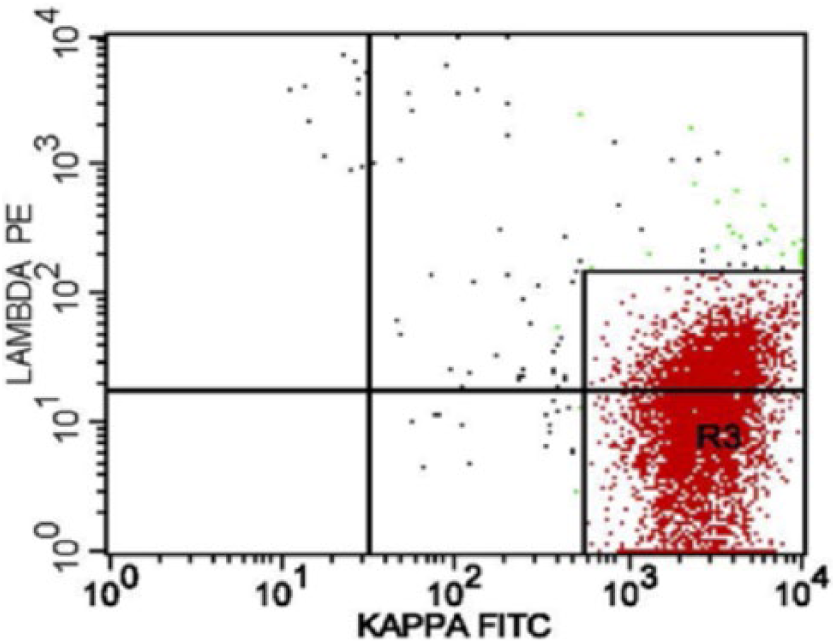
Kappa flow cytometry.
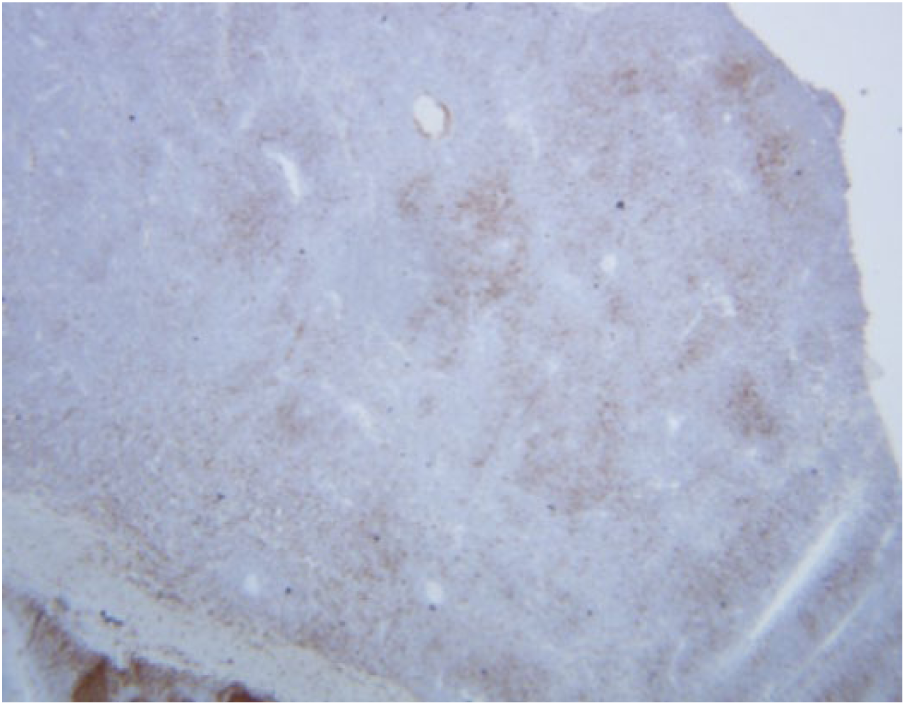
Nuclear positivity for cyclin D1, 20×.
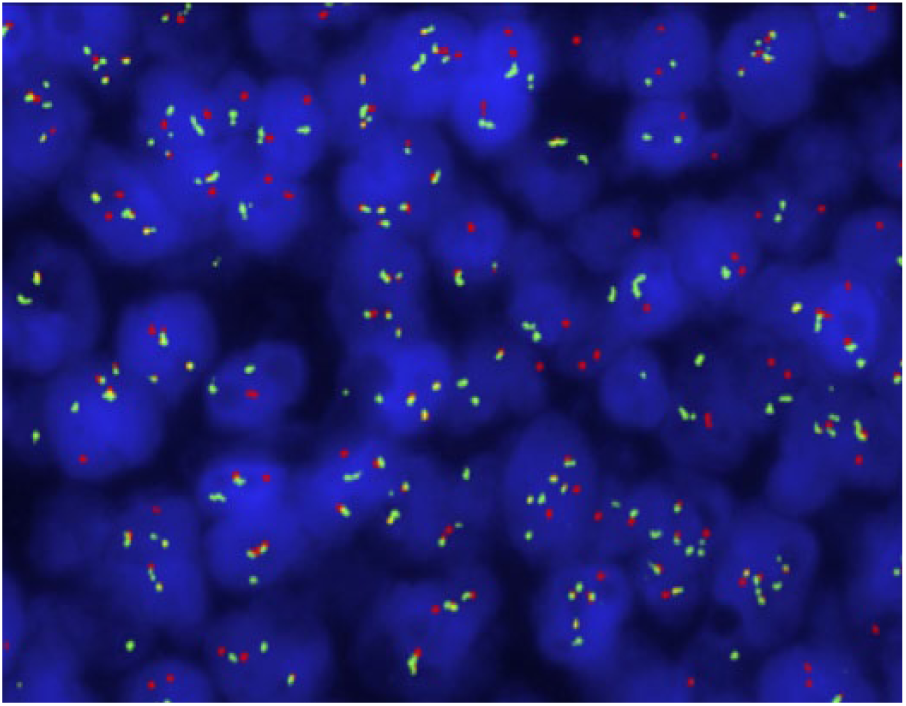
Image of paraffin embedded tissue fluorescent in situ hybridization (FISH) demonstrating CCND1/IGH fusion. The red signal represents the CCND1 locus and the green signal represents the IGH locus. Abnormal nuclei will show 1 red (unrearranged CCND1), 1 green (unrearranged IGH), and 2 or more fusion signals which are either yellow or closely adjacent red and green signals representing the derivative chromosomes involved in the t(11;14).
He completed radiation to the oropharynx and bilateral necks with complete resolution of the mass (Figure 7). The treatment consisted of 3000 cGy to the oropharynx and bilateral level II and then a boost to the oropharynx of 600 cGy for a total dose of 3600 cGy in 18 fractions. He tolerated radiation with minimal mucositis, xerostomia, and decreased taste sensation. He returned to his pre-treatment weight.
Sagittal and axial computed tomography of the neck with contrast 10 months after treatment completion showing resolution of the mass.
Three years after completion of treatment, he noticed a mass in his right temple. Positron emission tomography scan showed a single FDG avid lymph node in the parotid/preauricular region with a SUV of 5.3 (Figure 8). Fine needle aspiration of the mass showed recurrent pleomorphic MCL. Histology and flow cytometry showed the same characteristics as the initial tongue base tumor. Due to the isolated nature of the recurrence, the patient received 36 Gy of radiation to the right parotid/preauricular lesion with complete resolution of the mass. The patient tolerated the radiation well with mild xerostomia. He has had no evidence of disease 17 months after completion of this radiation. He had no evidence of systemic involvement and has not required any treatment with chemotherapy.
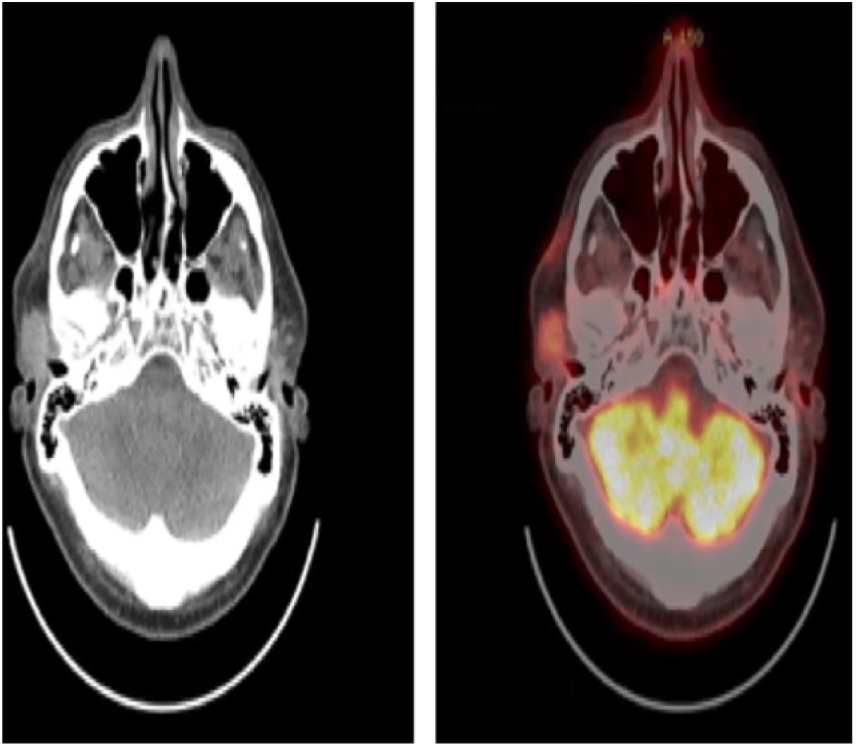
Axial computed tomography and positron emission tomography showing FDG-avid recurrence in the right preauricular region. FDG indicates fluorodeoxyglucose.
Discussion
Pleomorphic MCL is a rare non-Hodgkin B-cell lymphoma. It occurs in the mantle zone of lymphoid follicles, which is an outer ring of small lymphocytes around the germinal center. Mantle cell lymphoma typically arises within lymph nodes, but can exhibit extranodal involvement in 25% of cases, which includes presentation in Waldeyer ring. 3 Extranodal sites show sheets or nodules of small cells and can be difficult to distinguish from other types of small cell lymphoma. Therefore, suspicion and specific immunohistochemistry, flow cytometry, and cytogenetics are required for diagnosis. The pleomorphic variant is an aggressive subtype with a high karyotype complexity and proliferation index. 9
The diagnosis in this case was confirmed with the translocation t(11;14)(q13;q32) as seen in Figure 7. This results in deregulation of the CCND1 gene at 11q13 by the regulatory sequences of IGH at 14q32. This leads to cyclin D1 over-expression as in Figure 5 and impaired lymphocyte maturation. The nuclear heterogeneity, prominent nucleoli, and pale cytoplasm seen in Figure 2 classify the tumor as pleomorphic variant.
The positivity for cyclin D1, CD20, and CD5 markers in this case are characteristic of MCL. 13 The negativity for CD10 and CD23 is also typical for MCL.13,14 Bcl-6 is germinal center marker and MUM-1 is present on post-germinal center B-cells. 13 Both are typically negative in MCL so the positivity in this case is an example of the phenotypic heterogeneity seen in MCL. 13 Gualco et al 13 reported Bcl-6 positivity in 12% and MUM-1 in 35% of 127 cases of MCL. MUM-1 and Bcl-6 positivity are associated in MCL.13,15 However, MUM-1 is a marker of Bcl-6 positive germinal center cell maturation toward plasma cells, and therefore, MUM-1 and Bcl-6 are mutually exclusive in normal, germinal center B-cells. 13 Germinal center tumors and Bcl-6 positivity are associated with a longer overall survival. 15 MUM-1 and non-germinal center tumors are associated with a shorter overall survival. 15 Older age, >60 years, is another negative prognostic factor.16,17 Higher Ki-67 labeling indices are also associated with a poor prognosis, and in this case, it was moderate at 30%. This moderate index may have mitigated the poor prognosis that would have been expected from his older age, immunohistochemical findings, and typically aggressive pleomorphic subtype. 18
It is important to distinguish between the different types of lymphomas because of prognostic and therapeutic significance. When lymphoma is suspected, biopsy specimens should be sent fresh rather than in formalin so that appropriate immunotyping and genotyping can be done. Confirming the proper diagnosis will facilitate appropriate treatment with radiation and/or chemotherapy.
MCL is aggressive and commonly presents at an advanced stage. It therefore has been historically been treated with chemotherapy. 19 However, in more recent years, radiation therapy has been used for local control as well as palliation. A case series by Haque et al 20 supports that MCL is radio-sensitive, even in chemorefractory patients. There was a 69.1% complete response after relatively low doses of radiation therapy (median 30.6, range 18-40 Gy) in 39 patients with 68 sites of disease. 20 Rosenbluth and Yahalom 19 reported a 64% complete response in 21 patients with 38 sites treated with a mean 30 Gy (range 10.5-45 Gy) in mostly chemorefractory patients, including 4 patients with pharyngeal sites. In 2017, Debaja and The International Lymphoma Radiation Oncology Group (ILROG) also found that radiation therapy for recurrence after chemotherapy had lower rates of failure at the original disease site. 17 In this cohort, most treatment failures occurred at a different location than the original site of the disease, as seen in this case. 17
Upfront radiation for low-stage MCL can also provide local control and improved overall survival.16,17,19,21 ILROG reported that patients who received radiation therapy upfront as the only modality or for recurrence after chemotherapy had lower rates of failure at the original disease site. 17 Radiation therapy alone can be considered for patients who cannot tolerate or are not candidates for chemotherapy due to underlying morbidity. 17 In the ILROG cohort, most early-stage MCL also presented in head and neck sites (75%) like this case. 17
When there is disseminated disease, systemic chemotherapy or systemic chemotherapy consolidated with stem cell transplantation is the appropriate treatment. 22 Chemotherapy typically entails a rituximab- and/or doxorubicin-containing regimen. 17 Additional agents can be included in the regimen in relapsed or refractory disease including bortezomib, ibrutinib, and lenalidomide. 22 Even in disseminated disease, radiation can be offered in addition to chemotherapy for local and symptom control. 20
Although radiation helps with local control, it may not help overall treatment outcome. Dabaja et al 17 found no significant difference in either 10-year overall survival or 10-year freedom from progression in the chemotherapy alone, radiation alone, or chemoradiation groups in their 13-institution ILROG study. The median time to relapse was 38 months in all treatment groups. 17 The patients who presented with early-stage MCL had a relatively long overall survival compared with advanced stage MCL, regardless of which form of treatment they received. 17 In contrast, a study using the National Cancer Data Base by Leitch et al 16 showed a statistically significant higher 3-year overall survival for chemoradiation (79.8%) over radiation alone (72.5%) or chemotherapy alone (67.8%).
Regardless of the treatment offered, confirming the proper diagnosis in cases like this will also facilitate the appropriate close clinical follow-up. Because pleomorphic MCL portends a poor prognosis and tends to recur in different locations, continued close surveillance is needed with physical examination and imaging.
Footnotes
Acknowledgements
The authors thank Evan Kulbacki, MD, and Catherine Rehder, PhD, for their pathology input and Louis Diehl, MD, for his medical oncology input. The initial aspects of this case were presented at the International Conference on Head and Neck Cancer, July 2012 in Toronto, ON, Canada.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors contributed to the writing, editing and approval of this case report. MR made revisions based on the reviewer’s comments.
