Abstract
Background:
Paraneoplastic hyper-eosinophilia associated with metastatic lung adenocarcinoma is a rare finding and has been associated with a poor prognosis when present. Early hyper-eosinophilia appearing following non-small cell lung cancer (NSCLC) treatment with immune checkpoint inhibitors (ICI) has been previously reported with contradictory outcomes.
Case summary:
We present the case of an elderly man with newly diagnosed metastatic lung adenocarcinoma and baseline hyper-eosinophilia, treated with pembrolizumab, and showing evidence of significant and rapid disease progression suggestive of hyper-progressive disease, worsening baseline hyper-eosinophilia, and a fatal outcome within 1 month of therapy initiation.
Conclusion:
Pre-treatment hyper-eosinophilia could represent a predictive factor of an unfavorable response to ICI treatment in cases of NSCLC. Additional similar cases are needed to draw a more conclusive relationship.
Background
Eosinophilia is defined as a peripheral blood AEC greater than 500 cells/cu mm, and hyper-eosinophilia is the presence of an AEC ⩾1500 cells/cu mm in peripheral blood on 2 separate occasions at least 1 month apart or pathologic confirmation of tissue hyper-eosinophilia. It can be seen in allergic, immunologic, or infectious processes. 1
Hyper-eosinophilia associated with metastatic lung adenocarcinoma has been described as a rare paraneoplastic syndrome after specific work-up for underlying known causes has been done and ruled out. 2 The prognostic significance of paraneoplastic hyper-eosinophilia has been variable throughout the literature and sometimes contradictory with regard to an array of solid and hematologic malignancies. It has been associated with a better prognosis in certain types of cancers like colorectal and esophageal squamous cell carcinoma, but with a poor outcome in patients with Hodgkin’s lymphoma. 1 Concerning lung cancer,3,4 and specifically non-small cell lung cancer (NSCLC), with the most common sub-type being lung adenocarcinoma, 2 hyper-eosinophilia has been consistently associated with extensive metastasis, poor response to therapy and overall a worse prognosis, particularly when a paraneoplastic process is functioning as the cause. 3
Pembrolizumab, one of the immune checkpoint inhibitors (ICI) used in the treatment of lung cancer, have recently revolutionized treatment in oncology, showing evidence of improved survival compared to conventional platinum-based chemotherapy in lung malignancy. 5 Early hyper-eosinophilia appearing following NSCLC treatment with ICI has been previously reported with contradictory outcomes.1,6
We present the case of an 82-year-old male with metastatic lung adenocarcinoma and worsening baseline hyper-eosinophilia, who developed a dramatic disease progression after being treated with pembrolizumab, a programmed-death receptor-1 (PD-1) inhibitor, with failure of his hyper-eosinophilia to respond to treatment.
Case Report
An 82-year-old man presented on December 1st for work up of recurrent left pleural effusions. A Positron emission tomography-computed tomography (PET-CT) done 3 days later showed an FDG-avid lobulated left lung mass with pleural-based metastatic nodules, mediastinal and left lung hilar metastatic lymph nodes as well as left adrenal involvement (Figure 1). Biopsy of the pleural masses showed a poorly differentiated lung adenocarcinoma, stage IV with positive Programmed Death-Ligand-1 (PD-L1) expression in 90% of tumor cells, and negative Anaplastic Lymphoma Kinase (ALK) and Epidermal Growth Factor Receptor (EGFR) mutations. On December 4th, a peripheral blood count showed white blood cell count (WBC) of 28 700/cu mm, with 17% eosinophils, and an absolute eosinophil count (AEC) of 4879, consistent with hyper-eosinophilia. Work up for infectious and vasculitic causes of eosinophilia was negative. On December 5th, he received the first dose of pembrolizumab 200 mg.
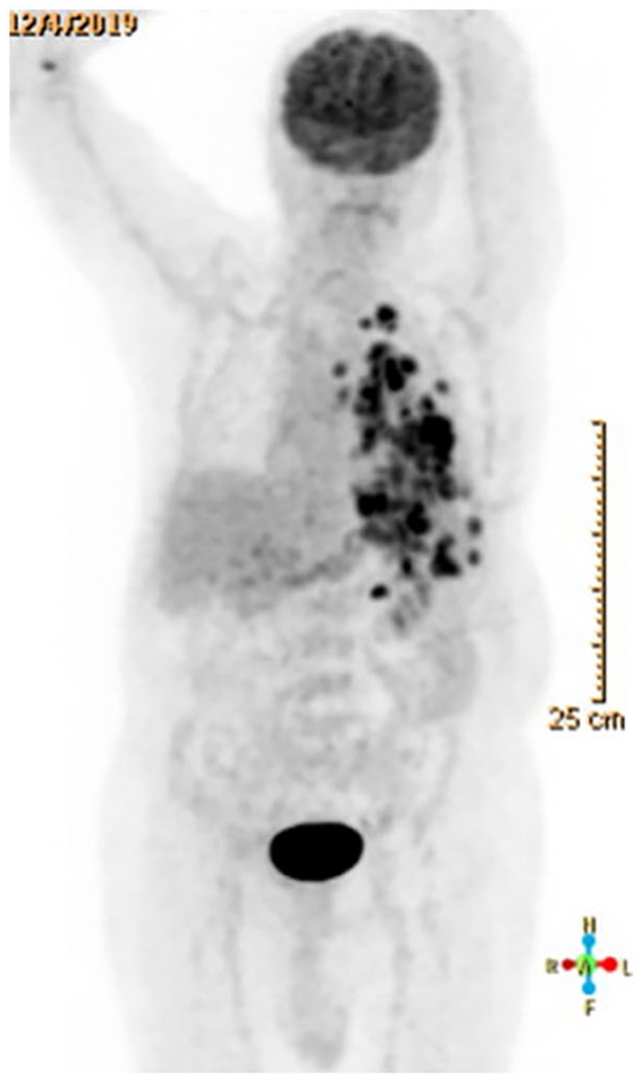
Positron emission tomography (December 4th) showing disease involvement of left lung, left pleura, mediastinal, and left lung hilar lymph nodes and left adrenal gland.
He presented back on December 15th for worsening dyspnea. Repeat chest CT showed an increase in the left lower lobe consolidation with appearance of multiple bilateral sub-centimetric pulmonary nodules (Figure 2). Repeat plain radiograph 2 days later showed complete opacification of the left hemithorax. A bronchoscopy was done in an attempt to look for any mucus plugs resulting in lung collapse with no significant improvement of the consolidation.

Baseline chest computed tomography done on December 1st (left) and follow up imaging on December 15th (right) with evident increase in the size of the left lower lobe consolidation and multiple left pleural-based masses.
A CT chest was done on December 17th (12 days after Pembrolizumab) for worsening dyspnea, showed further increase in the left lung consolidation and enlargement of the pleural-based masses (Figure 3). CT-guided biopsy of one of the new masses was positive for adenocarcinoma with PDL-1 positivity in 70% of the cells. A remarkably worsening in his baseline hyper-eosinophilia reaching 5115/cu mm was also noted. The patient was treated with prednisone 1 mg/kg daily for fear from immunotherapy toxicity. He then received his second dose of pembrolizumab 200 mg on December 26th. Of note, his eosinophil count continued to increase progressively to reach 6237/cu mm, despite his tapered steroid therapy (Figure 4).

December 17th: Imaging pre (left) and post (right) bronchoscopy, 12 days after pembrolizumab dose, showing complete left hemi-thorax opacification and confirmed by pathology to be tumor hyper-progression.

Baseline eosinophilia before and after administration of pembrolizumab.
A chest CT on January 3rd showed progression of the previously seen metastatic pleural nodules with evidence of lymphangitic spread, multiple new metastatic liver lesions and ill-defined multiple thoracic vertebral lytic lesions, not present on imaging 2 weeks prior. In view of his rapidly progressing disease, goals of care were agreed to be supportive only, and he passed away 2 weeks later.
Discussion
Our patient represents the first case to show worsening baseline hyper-eosinophilia before and after ICI therapy initiation, in parallel to a rapidly progressive NSCLC, not responding to systemic steroid therapy, and leading to premature death.
In fact, an atypical response to ICI therapy has been reported in up to 10% of cases, leading to a rapid tumor growth rate that is out of proportion compared to the one before therapy initiation. This response to immunotherapy has been termed as hyper-progressive disease (HPD). 7
Additionally, contradictory theories regarding the exact role of eosinophils have been reported. Some have shown that eosinophils have the ability to invade tumor microenvironments, contributing to a better prognosis in certain types of cancers, 1 others suggest that eosinophilia is associated with tumor dissemination to bone marrow or from local stimulation of the local tissue by the tumor itself through circulating factors secreted by the tumor itself such as Interleukin-5, Granulocyte Colony Stimulating Factor (G-CSF), and Granulocyte/Macrophage Colony Stimulating Factor (GM-CSF). 8
Other routinely available laboratory tests (ie, baseline serum lactate dehydrogenase and relative lymphocyte count) were also studied and successfully implemented in a predictive model of favorable prognosis in melanoma patients treated with pembrolizumab. 9
All these heterogenous and sometimes contradictory results raise the question about the possible presence of a certain threshold or peak of eosinophil counts before, during, and/or after ICI therapy that must be taken into consideration, and above which, a deleterious effect on host cells or response to immunotherapy is exerted. 10 Another hypothesis is the possible co-dependence of eosinophils on other factors, not commonly taken into consideration, like in the example of CD8-T cells, to produce an adequate anti-tumor response, as was demonstrated previously in a mouse model. 11 Predictors and biological markers of this fearful disease acceleration remain unknown and desperately needed to prevent both ICI toxicity and/or premature discontinuation of treatment. 7
Conclusion
Our patient’s rapidly progressing course and the appearance of new metastatic lesions in distant organs within days is suggestive of HPD, but it remains unclear if pre-treatment hyper-eosinophilia represents a predictive factor of an unfavorable response to ICI treatment in cases of NSCLC as in this case. Additional similar cases are needed to draw a more conclusive relationship.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contribution
SAK and HW were responsible for manuscript writing and literature review. MK was responsible for additional literature review. SAK was responsible for manuscript editing and critical feedback before the final form submission.
Informed Consent
A written consent form was secured to publish the findings of this case study.
