Abstract
Bevacizumab is a vascular endothelial growth factor–directed humanized monoclonal antibody used to treat many types of cancer and some eye diseases. Due to inhibition of angiogenesis, many adverse reactions such as bowel necrosis, nasal septal perforation, and renal thrombotic microangiopathy have been described. However, its association with interstitial pneumonitis is scarcely reported in the literature. We report a case of a 79-year-old woman with metastatic colon cancer who presented with cough and dyspnea on exertion the day after initiation of bevacizumab. She was found to have bilateral airspace opacities on imaging. Infectious and cardiogenic etiologies of dyspnea were ruled out. Due to the temporal relationship with the initiation of chemotherapy, she was suspected to have developed bevacizumab-induced interstitial pneumonitis. She improved rapidly with high-dose steroids. Follow-up imaging showed resolution of infiltrates. This is the first reported case in the literature that directly links bevacizumab to interstitial pneumonitis.
Introduction
Targeting the tumor microenvironment has been adopted by scientists since decades in the development of new drugs against cancer. It was found that the growth of tumor cells was strongly dependent on the formation of new blood vessels called neoangiogenesis, supplementing them with oxygen and nutrients and aiding in the removal of waste products as well. Upon the different angiogenic activators involved in cancer proliferation, the family of vascular endothelial growth factor (VEGF) and their receptors have been one of the most interesting targets for scientists and clinicians.1,2 Bevacizumab, a recombinant humanized monoclonal immunoglobulin-1 (IgG1) antibody directed against VEGF, has been widely used in the treatment of many cancers, including colorectal and breast cancers, and its applications has broadened further to treat many eye diseases including age-related macular degeneration.3,4 It functions as a competitive inhibitor of the tumor-secreted VEGF by preventing its association with its receptors on neighboring endothelial cells. 5 As a result, several adverse reactions have been associated with the use of this inhibitor, including, but not limited to, hypertension, thromboembolism, proteinuria, and impaired wound healing. 6 Well-established bevacizumab-associated pulmonary toxicities include hemoptysis and pulmonary hemorrhage. 7 Here, we report a rare case of bevacizumab-induced interstitial pneumonitis in a patient with metastatic colon cancer receiving capecitabine and bevacizumab.
Case Report
A 79-year-old woman with stage IV colon cancer, metastatic to the lungs and the adrenal glands, presented to the emergency department with dry cough and worsening dyspnea on exertion of 1-day duration. She was diagnosed with colon cancer in 2015 and underwent left-sided colectomy and then was treated with adjuvant chemotherapy (5-fluorouracil [5-FU] for a total of 6 cycles). In 2018, she developed left lower extremity edema and was diagnosed with deep venous thrombosis of the left femoral vein and left great saphenous vein and was placed on rivaroxaban. Extensive workup showed metastatic disease to the lungs, adrenal glands, and pelvic lymph nodes. Biopsy of one of the nodes confirmed adenocarcinoma of the colon. She was started on the combination of capecitabine 1000 mg twice a day (day 1 to day 14 every 21 days) and bevacizumab 7.5 mg/kg (day 1 every 21 days) and received the first dose of bevacizumab 1 week after starting capecitabine; she has never been on those drugs before. Less than 24 hours after the bevacizumab dose, she started having a dyspnea on exertion that was worsening and was associated with a dry cough and fatigue.
She was tachycardic and tachypneic on admission (heart rate was 122 bpm, respiratory rate was 24 breaths/min). Her blood pressure was 163/110 mm Hg. Her Sa
Her admission blood work was significant for mild leukocytosis (white blood cell count of 13.42/μL, with 82% neutrophils) (see Table 1). Chest radiograph showed bilateral basilar opacities (see Figure 1). Computed tomography (CT) of the chest with intravenous contrast redemonstrated bilateral airspace opacities with a slight central distribution (see Figure 2).
Admission laboratory values including hematology and chemistry.

Abbreviations: BUN, blood urea nitrogen; eGFR, estimated glomerular filtration rate; RBC, red blood cell; WBC, white blood cell.
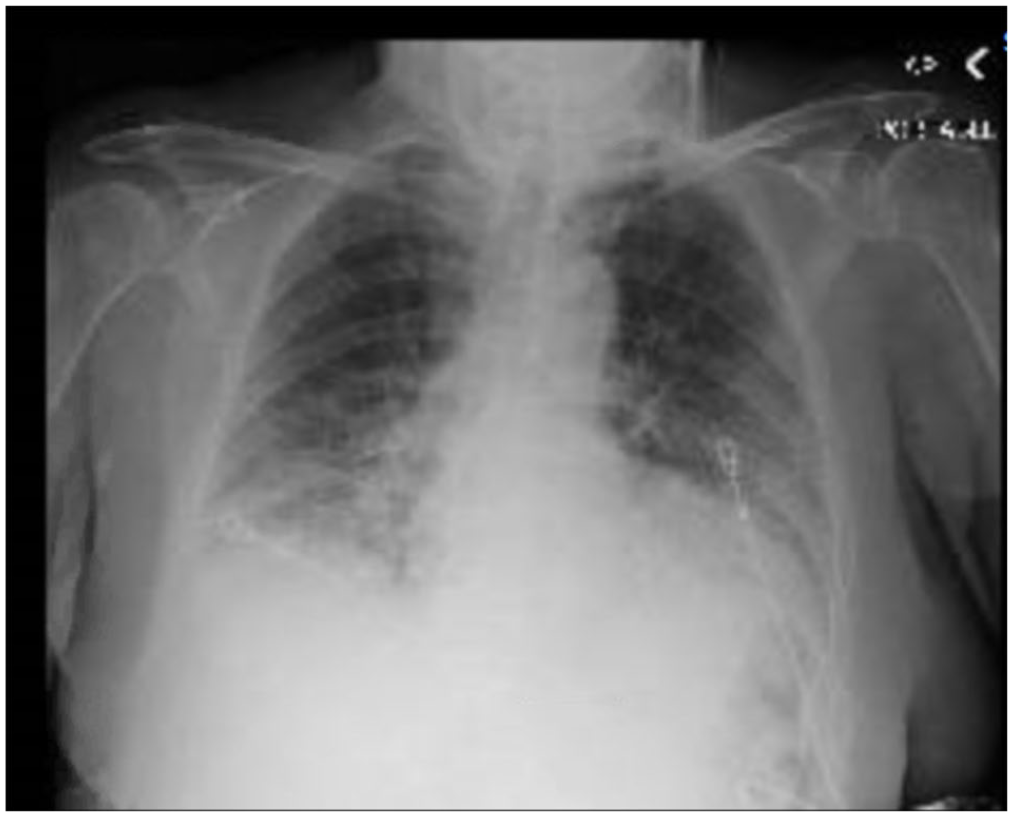
Chest radiograph on admission showing basilar opacities, blunting of the costophrenic angles, and reticular markings.

Computed tomography of the chest on admission showing bilateral ground glass opacities with a slight central distribution.
She was started on broad-spectrum antibiotics due to suspicion of multifocal pneumonia. All cultures and gram stains for infectious etiologies including common bacteria, fungi, legionella, viruses, and Pneumocystis were negative. Intravenous antibiotics were discontinued when cultures came back negative; patient received antibiotics a total of 5 days. Bronchoscopy and bronchoalveolar lavage (BAL) were performed, and cytological examination of BAL fluid sample showed rare mildly atypical cells; it favored reactive mesothelial cells, histiocytes, and bronchial cells in a background of marked acute inflammation. Cultures of BAL were negative for bacteria, mycobacteria, or fungal pathogens. Pneumocystis jiroveci was not detected by polymerase chain reaction. Transthoracic echocardiogram showed a left ventricular ejection fraction of 55%, a grade I diastolic dysfunction, and moderate pulmonary hypertension. On day 6 of hospitalization, the patient was started on intravenous methylprednisolone 60 mg given every 8 hours with the aim to treat presumed interstitial pneumonitis. Subsequently, the patient’s condition improved significantly, and she was extubated within 2 days of corticosteroid therapy initiation. Post-extubation, bilevel positive airway pressure–assisted ventilation was used as needed and she was subsequently transitioned to daily oral prednisone 40 mg. Arterial blood gas analysis on 3 L nasal cannula then showed a pH of 7.54, P

Chest radiograph on the day of discharge showing improvement of the basilar opacities.
Discussion
The close association of this patient’s acute symptoms with the administration of the VEGF inhibitor renders this case unique and interesting. In a case report describing interstitial pneumonitis in a patient treated with the combination of bevacizumab and pegylated liposomal doxorubicin for metastatic breast cancer, cough and dyspnea started after 3 courses of treatment. 8 Another case has reported the diagnosis of acute lung injury in a patient with metastatic breast cancer to the lung right after the initiation of the second cycle of bevacizumab and doxorubicin. 9 Finally, acute interstitial pneumonitis (AIP) was described just after 2 doses of the combination FOLFIRI + bevacizumab in a patient treated for metastatic colon cancer. 10 Bevacizumab is widely used and is considered a relatively safe drug, but nonetheless reports are available regarding rare but serious lung injuries associated with this drug, including chronic interstitial pneumonia, alveolar hemorrhage, and AIP.1,10 The term “acute lung injury” has been used to describe those entities in a simplified way.
Our patient was only exposed to 1 dose of bevacizumab and developed a dry cough with worsening shortness of breath on exertion the next day. In addition, she has been having capecitabine daily for at least 1 week. No cases of acute lung injury have been described with the sole use of fluoropyrimidines such as capecitabine or 5-FU. However, the entity has been described when the latter was used with oxaliplatin in the FOLFOX regimen used for the treatment of colon cancer 11 and gastric cancer. 12 Another chemotherapeutic agent known to be associated with lung injury is the nucleoside analog gemcitabine in the treatment of pancreatic adenocarcinoma 13 and the microtubule inhibitor docetaxel in the treatment of prostate and breast cancer. 14 It is not well understood how VEGF inhibition can lead to lung toxicities; however, studies in vitro showed that the protein VEGF can protect against epithelial apoptosis via a mechanism involving thrombospondin-1, and hence, decrease in its concentration may contribute to the proliferation of fibrosis. 15 , 16 It is likely in this case that the acute lung injury was the result of an idiosyncratic reaction to bevacizumab; however, the likelihood that this was caused by a cumulative toxic dose of capecitabine, which is not even known to cause any lung toxicities, is still possible but highly unlikely. Capecitabine was implicated in pulmonary toxicity when it was administered with oxaliplatin in a patient with colorectal cancer, 17 the latter being known to cause lung injury. Ruling out an infectious etiology was essential because patients on chemotherapy are usually immunosuppressed and an opportunistic pneumonia is high on the differential diagnosis in this setting. Moreover, the resolution of the infiltrates after a course of corticosteroids supports the diagnosis of pneumonitis over other etiologies such as multifocal pneumonia or congestive heart failure exacerbation.
In a retrospective cohort study comparing the incidence of adverse reactions from the most frequently used monoclonal antibodies, bevacizumab was the least to cause adverse pulmonary reactions. Advanced age and low serum albumin were significant risk factors for the development of those reactions. Risk factors associated with pneumonitis during chemotherapy include older age, low-performance status, and a decreased normal lung area on CT scan of the chest. 10
Conclusion
This patient’s acute respiratory failure may have been caused by an inflammation in the lung triggered by 1 dose of bevacizumab giving the pattern of interstitial pneumonitis on CT of the chest. The ineffectiveness of antibiotics along with the clinical improvement seen after corticosteroids initiation was in favor of an inflammatory process causing the symptoms. Although this is a very rare adverse reaction of bevacizumab, early detection is essential and crucial for preventing morbidities and mortalities, as well as unnecessary treatments that might be harmful to patients who already have a compromised performance status.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
HS and PS participated in writing and editing the manuscript. NN and NS participated in reviewing and editing the manuscript. ED revised the article critically and approved the final version of the manuscript.
Ethical Approval
This is a case report and does not meet the Federal Policy for the Protection of Human Subjects definition of Research. Hence, no ethics approval is required.
Informed Consent
A verbal consent over the phone was obtained from the patient’s daughter.
