Abstract
Background:
Corticosteroids are common treatments in certain diseases that cause acute respiratory failure (ARF) and are sometimes administered empirically for patients with critical ARF. Associations between changes in clinical parameters following initiation of steroid pulse therapy and mortality in patients with ARF have not been previously investigated.
Methods:
This was a single-center and retrospective cohort study. Parameters on the day of methylprednisolone pulse therapy initiation (day 1) and the day following the end of methylprednisolone therapy (day 4) in patients who were admitted because of ARF and underwent methylprednisolone pulse therapy between October 2008 and July 2021 were reviewed.
Results:
A total of 98 patients were included in our analysis, and 45 (46%) died at our hospital. Change in lactate dehydrogenase (LDH) from day 1 to day 4 (ΔLDH) was significantly higher in the in-hospital death group than in the survival group (−68 IU/L in the survival group versus 46 IU/L in the in-hospital death group, p < 0.01). Multivariate logistic analyses showed that age >75 years old (odds ratio (OR), 3.88; 95% confidence interval (CI), 1.38–10.9; p < 0.01), previously diagnosed interstitial lung disease (OR, 3.43; 95% CI, 1.10–10.7; p = 0.03), ΔLDH > 0 (OR, 6.47; 95% CI, 2.30–18.2; p < 0.01), and ΔSequential Organ Failure Assessment score > 0 (OR, 3.06; 95% CI, 1.10–8.51; p = 0.03) were significantly associated with in-hospital mortality.
Conclusions:
This study showed that elevation of serum LDH level during methylprednisolone pulse therapy was a predictive factor for high in-hospital mortality in patients with ARF.
Keywords
Background
Acute respiratory failure (ARF) is a major critical condition that is managed in intensive care units. The in-hospital mortality of ARF patients was reported to be 44%. 1 ARF is caused by various diseases, including cardiogenic pulmonary edema, community-acquired pneumonia, acute respiratory distress syndrome (ARDS), and acute exacerbation (AE) of chronic obstructive pulmonary disease or interstitial lung disease (ILD). 2 Appropriate treatments differ depending on the causes of ARF. Especially, the evidence of high-dose steroid therapy or anti-inflammatory therapy remains controversial. A previous study reported better long-term outcomes in AE-interstitial pulmonary fibrosis (IPF) without high-dose corticosteroids therapy. 3 However, corticosteroids are common treatments in certain diseases that cause ARF, such as drug-induced lung injury (DILI), ARDS, and some forms of idiopathic interstitial pneumonia (IIP).2,4–6 Moreover, high-dose corticosteroids, including steroid pulse therapy, have commonly been used in patients with AE-ILD. 7 Early administration of corticosteroids was shown to be effective in moderate-to-severe ARDS, and early treatments intervention, including with immunosuppressive agents, improved outcomes in acute interstitial pneumonia.6,8 However, it is difficult to determine the causes of ARF during the acute phase of the disease. 9 Therefore, in clinical setting, corticosteroids, including steroid pulse therapy, are sometimes administered empirically for severe ARF.
Various prognostic factors have been identified in patients with ARF. Previously reported poor prognostic factors include advanced age, low body mass index, high acute physiology and chronic health evaluation (APACHE) II score, renal failure, hepatic failure, and use of a high fraction of inspired oxygen (FiO2).10,11 Serum lactate dehydrogenase (LDH) is a common enzyme expressed in all living cells, with high levels in heart, lung, liver, muscle, and blood cells. 12 Furthermore, serum LDH can become elevated in ILD patients, reflecting diffuse lung injury. 13 Elevated serum LDH level was reported to be a predictor of poorer outcomes in certain diseases that cause ARF, including AE-IPF 14 and connective tissue disease-associated interstitial lung disease (CTD-ILD), 15 mycoplasma pneumonia, 16 and coronavirus disease 2019. 17 Therefore, elevation of serum LDH level during empiric corticosteroids therapy is expected to reflect the progression of lung injury even following the intervention and may predict poorer prognosis. However, the association between change in serum LDH level following treatment initiation and prognosis of ARF has not been examined.
The aim of the present study was to investigate the association between change in serum LDH level and mortality in patients with ARF undergoing corticosteroids therapy.
Methods
Study design and patients
We conducted a single-center, retrospective cohort study at our hospital in Japan. This study was approved by the ethics committee of our hospital. We retrospectively reviewed the medical records of patients who were admitted because of ARF and underwent methylprednisolone pulse therapy between October 2008 and July 2021. Patients with ARF were defined as those who developed dyspnea within 30 days before the admission, had newly emerging diffuse ground glass opacities (GGOs) or consolidations in radiological tests, and received mechanical ventilation for hypoxia. We defined patients with ARF as those who developed dyspnea within 30 days according to the definition of AE-IPF. 4 Methylprednisolone pulse therapy in the present study was defined as 1000-mg intravenous methylprednisolone administration for three consecutive days. Patients who interrupted methylprednisolone pulse therapy, who developed ARF primarily because of bacterial infection or heart failure, who started methylprednisolone pulse therapy before the introduction of mechanical ventilation, or who lacked clinical data were excluded.
Data collection
The etiologies of ARF were diagnosed by clinicians. Diagnosis of AE-IIP was made for patients who were previously diagnosed with IIP or had radiologically chronic interstitial appearances and developed acute respiratory deterioration with diffuse GGOs or consolidations. CTD-ILD was diagnosed when patients with CTD developed acute respiratory deterioration with diffuse GGOs or consolidations. We diagnosed patients with DILI when they had received suspected drugs for lung injury and had no other causes of pneumonia. ARF with unknown etiology was defined as ARF without a definite diagnosis as the cause of ARF. Sequential Organ Failure Assessment (SOFA) score was calculated according to the previous study. 18 The following variables were collected: demographic data, past medical history, and clinical and biological findings on the day of methylprednisolone pulse therapy initiation (day 1) and the day following the end of methylprednisolone therapy (day 4). The primary outcome in this study was in-hospital mortality.
Statistical analysis
The baseline and clinical features of patients on day 1, and changes in clinical parameters between on day 1 and day 4 were compared between the survival and in-hospital death groups. Next, we performed the Wilcoxon signed-rank tests to compare parameters between on day 1 and day 4 in each group. Categorical variables were presented as counts and percentages, and continuous variables as medians and interquartile ranges (IQRs). We conducted the Mann–Whitney U test to compare continuous variables, and Fisher’s exact test to compare the proportions of categorical variables between the groups. Finally, we performed univariate and multivariate logistic regression analyses for variables, which were predefined based on previous studies.9,10 Previously diagnosed ILD appeared to influence baseline of serum LDH level. 11 We selected changes in LDH from day 1 to day 4 (ΔLDH) and ΔSOFA score, but did not include partial pressure of arterial oxygen (PaO2)/FiO2 ratio (P/F ratio), serum bilirubin, or creatinine because they were elements of the SOFA score. We defined 75 years old as the cutoff age according to a previous study 9 and the Japanese healthcare insurance system. The results are presented as odds ratios (ORs) and 95% confidence intervals (CIs). p-Value < 0.05 was considered statistically significant. We performed all statistical analyses using EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), a user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). 19
Results
Patients’ cohort
During the inclusion period, 116 patients were admitted to our hospital because of ARF and underwent methylprednisolone pulse therapy. Eighteen patients were excluded because of the following: methylprednisolone pulse therapy before initiation of mechanical ventilation initiation (n = 3), cessation of methylprednisolone pulse therapy (n = 1), and lack of data (n = 14). Consequently, 98 patients were included in our analysis (Figure 1).

Flow chart of the study.
Baseline features of the study population
Forty-five patients (46%) in the study population died at our hospital. The numbers of patients who had been weaned from the mechanical ventilation were 51 (96%) and 1 (2.2%) of the survival group and in-hospital death group, respectively. In the survival group, median of ventilation-free days at 28 days was 20 (IQR, 15−22 days). Table 1 shows comparisons of baseline features on admission between patients in the survival group and in-hospital death groups. Median age was 77 years old, and 88% and 57% of patients were >65 and >75 years old, respectively. Age (median 78 versus 75 years old, p = 0.01) and percentage of patients with previously diagnosed ILD (38% versus 17%, p = 0.02) were significantly higher in death group.
Baseline features of patients.

Data are presented as median (IQR) or absolute values (percentage). AE: acute exacerbation; ARDS: acute respiratory distress syndrome; ARF: acute respiratory failure; BMI: body mass index; COP: cryptogenic organizing pneumonia; COPD: chronic obstructive pulmonary disease; CTD-ILD: connective tissue disease-associated interstitial lung disease; DILI: drug-induced lung injury; IIP: idiopathic interstitial pneumonia; IQR: interquartile range; PCP: Pneumocystis jirovecii pneumonia.
Comparisons of clinical parameters
Table 2 shows the clinical features and treatments on day 1. There was no significant difference in serum LDH level between the two groups (median 350 IU/L in the survival group versus 408 IU/L in the in-hospital death group, p = 0.21). No other clinical parameters were significantly different between the two groups on day 1. Table 3 shows the comparison of each clinical parameter of day 1 and day 4. Serum LDH level decreased significantly from 350 U/L (IQR, 286–472 U/L) on day 1 to 310 U/L (IQR, 245–397 U/L) on day 4 in the survival group (p < 0.01), but increased from 408 U/L (IQR, 306–513 U/L) on day 1 to 411 U/L (IQR, 356–633 U/L) on day 4 in the in-hospital death group (p < 0.01). Median SOFA score also improved significantly from 4 on day 1 to 3 on day 4 in the survival group (p < 0.01), while it deteriorated from 5 on day 1 to 6 on day 4 in the in-hospital death group (p = 0.046). Changes in clinical parameters from day 1 to day 4 are shown in Table 4. At first, ΔLDH were significantly higher in the in-hospital death group than in the survival group (−68 IU/L in the survival group versus 46 IU/L in the in-hospital death group, p < 0.01). Additionally, Δbilirubin (−0.3 mg/dL versus −0.2 mg/dL, p < 0.01), Δcreatinine (−0.04 mg/dL versus 0.02 mg/dL, p = 0.04), and ΔSOFA score (−1 versus 0, p < 0.01) were also statistically different between the two groups.
Clinical parameters on the day of methylprednisolone pulse therapy initiation (day 1).
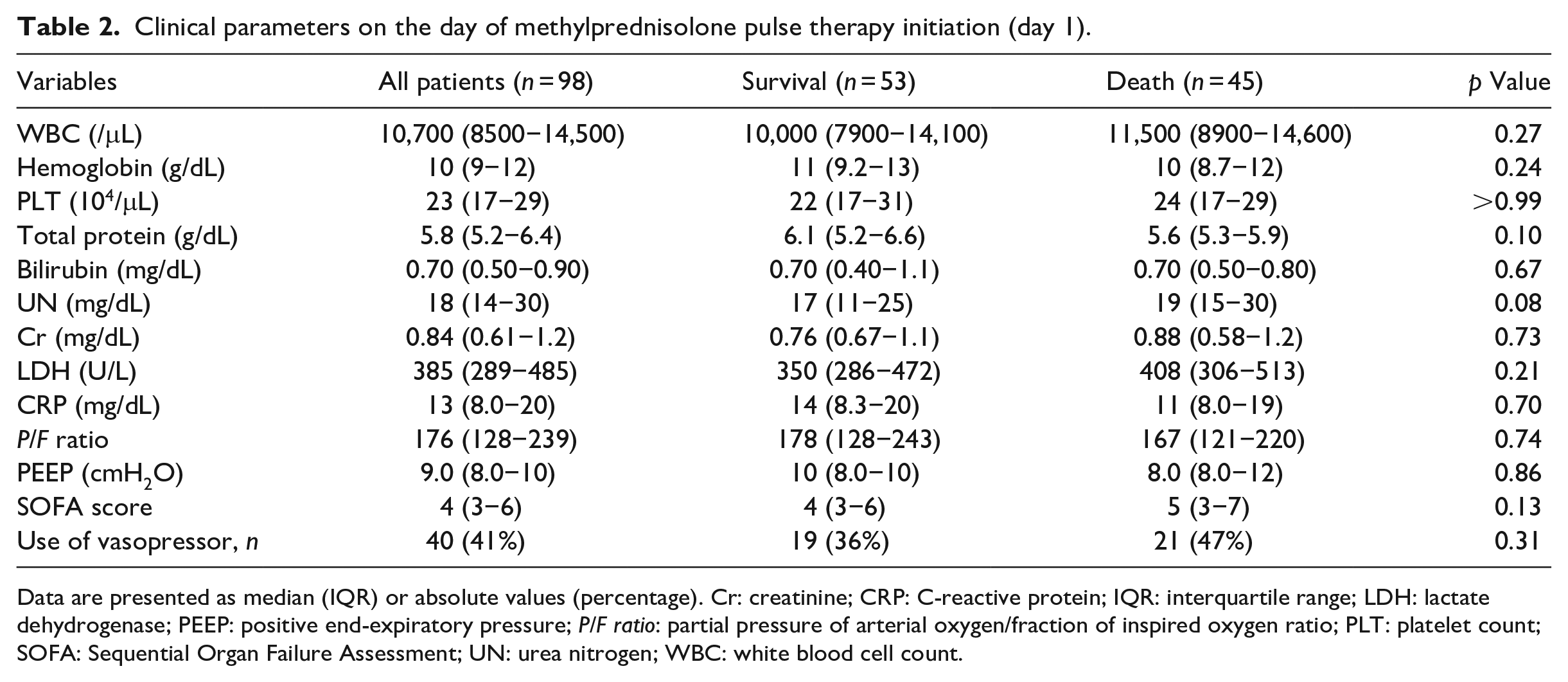
Data are presented as median (IQR) or absolute values (percentage). Cr: creatinine; CRP: C-reactive protein; IQR: interquartile range; LDH: lactate dehydrogenase; PEEP: positive end-expiratory pressure; P/F ratio: partial pressure of arterial oxygen/fraction of inspired oxygen ratio; PLT: platelet count; SOFA: Sequential Organ Failure Assessment; UN: urea nitrogen; WBC: white blood cell count.
Comparisons of clinical parameters between methylprednisolone pulse therapy initiation (day 1) and the day following the end of methylprednisolone pulse therapy (day 4).

Data are presented as median (IQR). Cr: creatinine; CRP: C-reactive protein; IQR: interquartile range; LDH: lactate dehydrogenase; PEEP: positive end-expiratory pressure; P/F ratio: partial pressure of arterial oxygen/fraction of inspired oxygen ratio; PLT: platelet count; SOFA: Sequential Organ Failure Assessment; UN: urea nitrogen; WBC: white blood cell count.
Comparison of clinical parameters between day 1 and day 4 in the survival group.
Comparison of clinical parameters between day 1 and day 4 in the in-hospital death group.
Changes in clinical parameters from methylprednisolone pulse therapy initiation (day 1) to the day following the end of methylprednisolone pulse therapy (day 4).
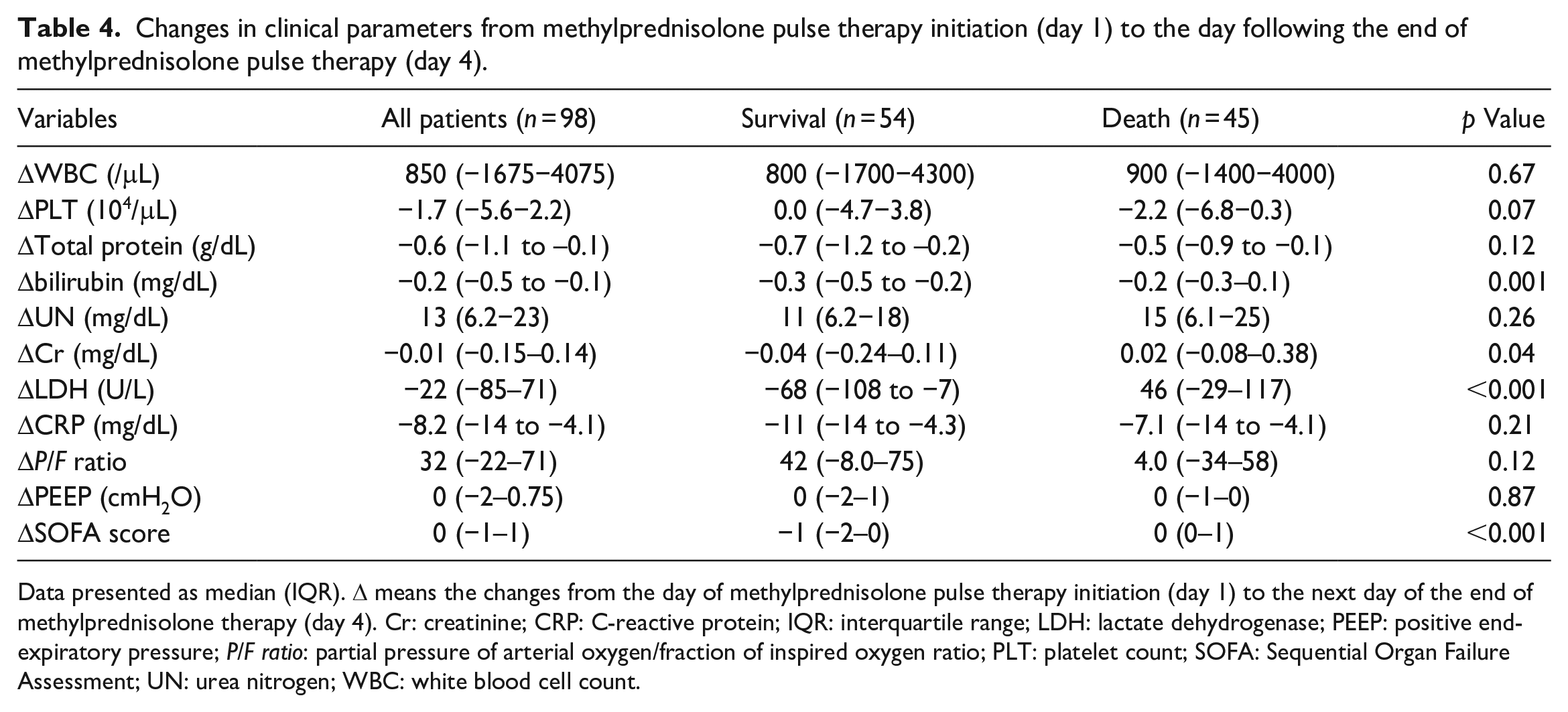
Data presented as median (IQR). Δ means the changes from the day of methylprednisolone pulse therapy initiation (day 1) to the next day of the end of methylprednisolone therapy (day 4). Cr: creatinine; CRP: C-reactive protein; IQR: interquartile range; LDH: lactate dehydrogenase; PEEP: positive end-expiratory pressure; P/F ratio: partial pressure of arterial oxygen/fraction of inspired oxygen ratio; PLT: platelet count; SOFA: Sequential Organ Failure Assessment; UN: urea nitrogen; WBC: white blood cell count.
Variables associated with in-hospital mortality
Table 5 shows univariate and multivariate logistic analyses for risks of in-hospital mortality. The variables included in the analyses were age >75 years old, previously diagnosed ILD, ΔLDH > 0, and ΔSOFA score > 0. Both univariate and multivariate logistic analyses showed that all four variables were significantly associated with in-hospital mortality.
Univariate and multivariate logistic regression for in-hospital mortality.
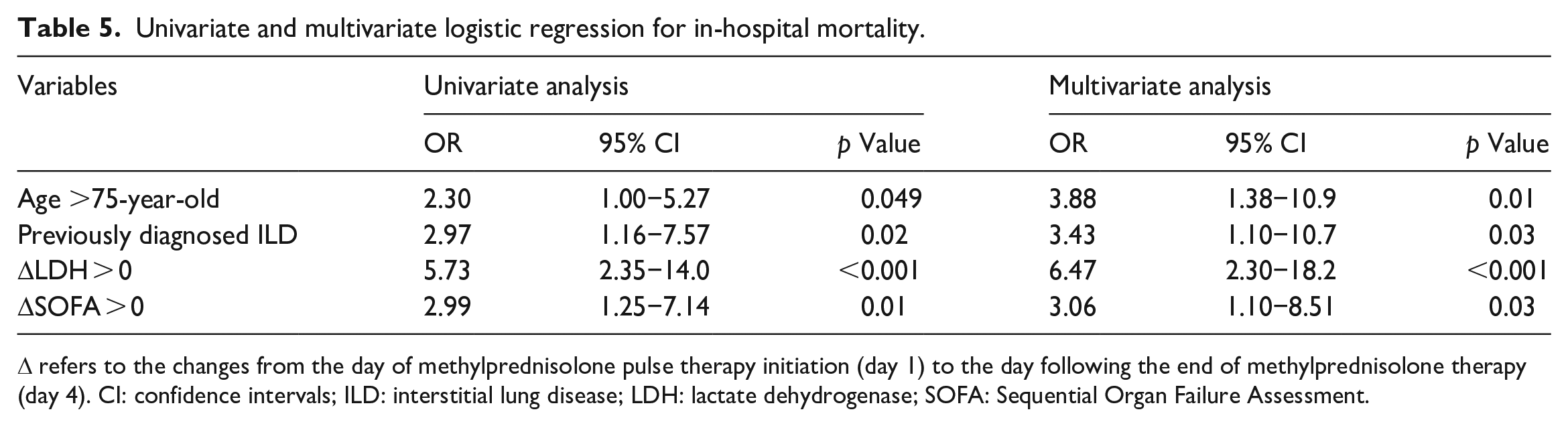
Δ refers to the changes from the day of methylprednisolone pulse therapy initiation (day 1) to the day following the end of methylprednisolone therapy (day 4). CI: confidence intervals; ILD: interstitial lung disease; LDH: lactate dehydrogenase; SOFA: Sequential Organ Failure Assessment.
Discussion
This study showed that elevation of serum LDH level during methylprednisolone pulse therapy was a predictive factor for high in-hospital mortality in patients with ARF. Serum LDH level represented the severity of lung injury and therefore predicted prognosis in some pulmonary diseases in previous studies.14–17 However, the prognosis of patients who receive corticosteroids therapy for ARF depends on both disease severity upon admission and the responsiveness to corticosteroids. In fact, P/F ratio or serum LDH level alone at baseline were not significantly associated with mortality in a previous study in which most of patients subsequently received corticosteroids pulse therapy. 20 Therefore, we evaluated changes in clinical parameters during methylprednisolone pulse therapy to consider not only baseline disease severity, but also early treatment responsiveness. Consequently, in this study, change in serum LDH level during methylprednisolone pulse therapy was shown to predict mortality. To the best of our knowledge, only one single-center, retrospective study has investigated associations between changes in clinical parameters and prognosis in pulmonary diseases. The Japanese study showed that the increase in serum LDH level from admission to 2 weeks postadmission was associated with the 90-day mortality in patients with AE-IPF. 21 However, this prior study had the following limitations: (1) treatments during the 2-weeks period varied, and (2) it was clinically too late to predict prognosis 2 weeks after admission. In contrast, our results showed that mortality can be predicted early during corticosteroids therapy. Elevation of serum LDH level during methylprednisolone pulse therapy reflects the nonresponsiveness to corticosteroids and therefore encourage us to consider whether we should add or change therapeutic intervention at an early stage of the disease. Clinicians should be on the alert for elevations of serum LDH level during methylprednisolone pulse therapy in patients with ARF.
The present study also identified the following three prognostic factors associated with poor prognosis: higher age, previously diagnosed ILD, and elevation of SOFA score during methylprednisolone pulse therapy. First, higher age has previously been shown to be a poor prognostic factor in ARF and various diseases that cause ARF.10,22 However, most patients included in our study were much older; 57% of patients were aged >75 years old, and the median age was 77 years old. In contrast, in the previous international and multicenter ARDS study, the median age of patients was only 63 years old (IQR, 50–73 years old). In another study on ARF from Taiwan, only 30% of patients were aged >75 years old. Therefore, our results suggest that higher age is also a prognostic factor, even in much older patients. Second, previously diagnosed ILD was a reasonable prognostic factor, based on the finding that AE-IPF has poor responsiveness to corticosteroids therapy and poor prognosis. 4 Among our patients who were previously diagnosed with ILD, a few may have had IPF, although we could not determine the definite forms of ILD. The previous studies of prognostic factors of ARF did not examine the prognostic impact of previously diagnosed with ILD.10,11,20 Thus, this study is valuable in alerting clinicians to previously diagnosed ILD when treating ARF. Third, changes in SOFA score can predict prognosis in patients with severe ARF. SOFA score reflects dysfunction in many systems and organs including the circulatory and respiratory systems, liver, and kidney. APACHE II score, another disease severity of disease classification system, has been reported to be a prognostic factor in patients with ARF.10,20 The SOFA score contains fewer elements compared with the APACHE II score, but the two scoring systems were reported to have similar performance for predicting mortality in critically ill patients. 23 Unlike the previous study, this study evaluated not only the baseline score but also the change after treatment. The prognosis of patients who receive corticosteroids therapy for ARF may depend on disease severity and responsiveness to corticosteroids. Thus, the change of score is a noteworthy prognostic marker.
The present study had three limitations. First, this was a single-center, retrospective, small-sized study, and sample size calculation was not performed. There may have been some bias because of the retrospective nature and small sample size. In addition, only four valuables were considered in multivariate analysis. Second, the causes of ARF included various diseases. Although methylprednisolone pulse therapy is not the standard therapy for ARDS or Pneumocystis jirovecii pneumonia, some patients underwent methylprednisolone pulse therapy empirically because of critical ARF and were diagnosed with these diseases later. In addition, we could not identify the cause of ARF in some cases. Although the indications or doses of steroid pulse therapy are differed depending on the diseases that caused ARF, they appeared to reflect actual clinical observations. Third, the patients who had already undergone corticosteroids or immunosuppressive therapy were included. In addition, the data on the additional immunosuppressive therapy, such as intravenous cyclophosphamide, was not collected. It may be difficult to evaluate the pure effect of corticosteroids for ARF from this study.
Conclusions
The present study showed that elevation of serum LDH level during methylprednisolone pulse therapy was a predictor for high in-hospital mortality in patients with ARF.
Footnotes
Author contributions
K.K. designed the study, analyzed data, and drafted the original manuscript. S.M. critically reviewed and revised the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The datasets analyzed in the present study are available from the corresponding author on reasonable request.
Consent for publication
The ethics committee of Osaka Police Hospital waivered the need for informed consent for this study because data were collected retrospectively and anonymized.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
This study was conducted according to the Helsinki Declaration and was approved by the ethics committee of Osaka Police Hospital (Institutional Review Board number 1537). The ethics committee of Osaka Police Hospital waivered the need for informed consent for this study because data were collected retrospectively and anonymized.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
