Abstract
Background:
Lemierre’s syndrome is a rare but life-threatening condition characterized by septic thrombophlebitis, most often due to Fusobacterium necrophorum. Although uncommon, Staphylococcus aureus can also cause atypical and severe presentations.
Case Presentation:
This case describes an unusual presentation of Lemierre’s syndrome in an 18-year-old male, associated with Staphylococcus aureus and complications including left-sided secondary spontaneous pneumothorax, septic pulmonary emboli, and pleural effusion. The patient initially presented with fever, headache, periorbital swelling with double vision, neck pain, and acute onset left-sided chest pain with shortness of breath. Initial evaluations revealed thrombosis in the superior ophthalmic veins, cavernous sinuses, and bilateral internal jugular veins, along with left-sided pneumothorax and diffuse cavitating nodules in both lung fields. Blood cultures confirmed Staphylococcus aureus resistant to macrolides. Treatment includes high-flow oxygen, intravenous vancomycin followed by oxacillin, and anticoagulation therapy. At 3-month follow-up, the patient had fully recovered with resolution of pneumothorax and inflammatory markers.
Conclusion:
This case highlights the importance of recognizing atypical presentations of Lemierre’s syndrome and the need for prompt multidisciplinary intervention to prevent fatal outcomes.
Introduction
Lemierre’s syndrome is a rare and potentially fatal condition characterized by septic thrombophlebitis, typically arising from oropharyngeal infections. 1 Its estimated annual incidence is around 1 in 1 000 000, predominantly affecting adolescents and young adults.1,2 It most commonly involves Fusobacterium necrophorum, but also other pathogens, including Staphylococcus aureus, have been implicated in atypical presentations.2,3 This syndrome often progresses rapidly, with bacteremia leading to septic emboli affecting multiple organ systems, particularly the lungs. While classic cases are associated with sore throat and pharyngitis, atypical presentations without oropharyngeal symptoms pose a diagnostic challenge.1,2 Complications such as pneumothorax and cavitating pulmonary lesions are rare but serious manifestations that necessitate prompt recognition and intervention. 2
Case Presentation
An 18-year-old previously healthy male presented to the emergency department (ER) after being referred from a private hospital for further evaluation and management of left-sided pneumothorax. The patient’s history revealed the development of a boil above the right eye 5 days prior, which was associated with pus formation and swelling that resolved spontaneously. This was followed by the onset of fever with chills, high-grade in nature, accompanied by headache over the same period. Over the last day, the patient experienced worsening symptoms, including left-sided chest pain aggravated with deep breathing, shortness of breath, neck pain, and periorbital swelling associated with diplopia. Initial evaluation at the referring facility suggested cerebral venous sinus thrombosis (CVST) and left-sided pneumothorax, prompting referral to the department of respiratory medicine for specialized care. Notably, there was no history of sore throat, cough, or cold.
On examination, the patient was conscious and coherent (GCS: E4V5M6) but appeared lethargic and febrile with a body temperature of 100.3°F. Vital signs revealed a respiratory rate of 19 breaths/min, oxygen saturation (SpO2) of 96% on room air, heart rate of 110 beats/min, and blood pressure of 130/90 mmHg. Cardiac and neurological examination was normal, while respiratory examination showed bilaterally audible breath sounds with diminished intensity in the left infra-axillary and infra-scapular regions.
Initial investigations demonstrated normal renal and liver function tests, with negative results for HIV and HBsAg. Complete blood picture on admission revealed a leukocytosis with a white blood cell count of 33 700/µL, hemoglobin of 14.3 g/dL, and platelet count of 353 000/µL. These parameters showed improvement during the hospital stay, with the white blood cell count decreasing to 10 101/µL by discharge. Notably, neutrophil predominance was observed, ranging from 85% on admission to 69% at discharge, with a gradual increase in lymphocyte percentage from 11% to 24% over the same period. Inflammatory markers, including an erythrocyte sedimentation rate (ESR) of 140 mm in the first hour and a C-reactive protein (CRP) level of 73 mg/L, were significantly elevated. Fever panel and urine cultures were negative, while blood cultures yielded methicillin-sensitive Staphylococcus aureus (MSSA), resistant only to macrolides (erythromycin and azithromycin). The isolate was sensitive to oxacillin, vancomycin, clindamycin, doxycycline, linezolid, and levofloxacin (Table 1).
Antibiotic Sensitivity Profile of Staphylococcus aureus Isolate.

Chest radiography identified a left-sided pneumothorax with blunting of the costophrenic angle (Figure 1). On High-Resolution Computed Tomography (HRCT), left-sided hydropneumothorax was noted (Figure 2). Diffusely scattered nodules are visible in both lung fields, with cavitation present in some nodules, particularly in the right upper lobe. The cavitating lung lesions demonstrated thin walls in some regions, which raised the possibility of pneumatoceles associated with Staphylococcus aureus infection. Therefore, both pneumatoceles and septic emboli were considered in the radiological differential diagnosis. A computed tomography pulmonary angiography (CTPA) was not obtained at the referring facility, limiting definitive visualization of embolic filling defects within the pulmonary arteries. Therefore, septic emboli could not be confirmed angiographically. Further, the pleural effusion was minimal on imaging, and thoracentesis could not be performed in our resource-limited setting. As a result, pleural fluid biochemistry and culture were not obtained. MRI brain and orbits with contrast revealed thrombosis of the bilateral superior ophthalmic veins, which extended into both cavernous sinuses. Subsequently, thrombosis of the bilateral internal jugular veins was identified, suggesting a sequential progression of thrombus from ophthalmic veins to cavernous sinuses and finally internal jugular veins (Figure 3). Further findings included thrombosis of the bilateral internal jugular veins. A transthoracic echocardiogram (TTE) was performed, which did not reveal any vegetations or valvular abnormalities. Given the absence of clinical suspicion and negative TTE, a transesophageal echocardiogram (TEE) was not performed.
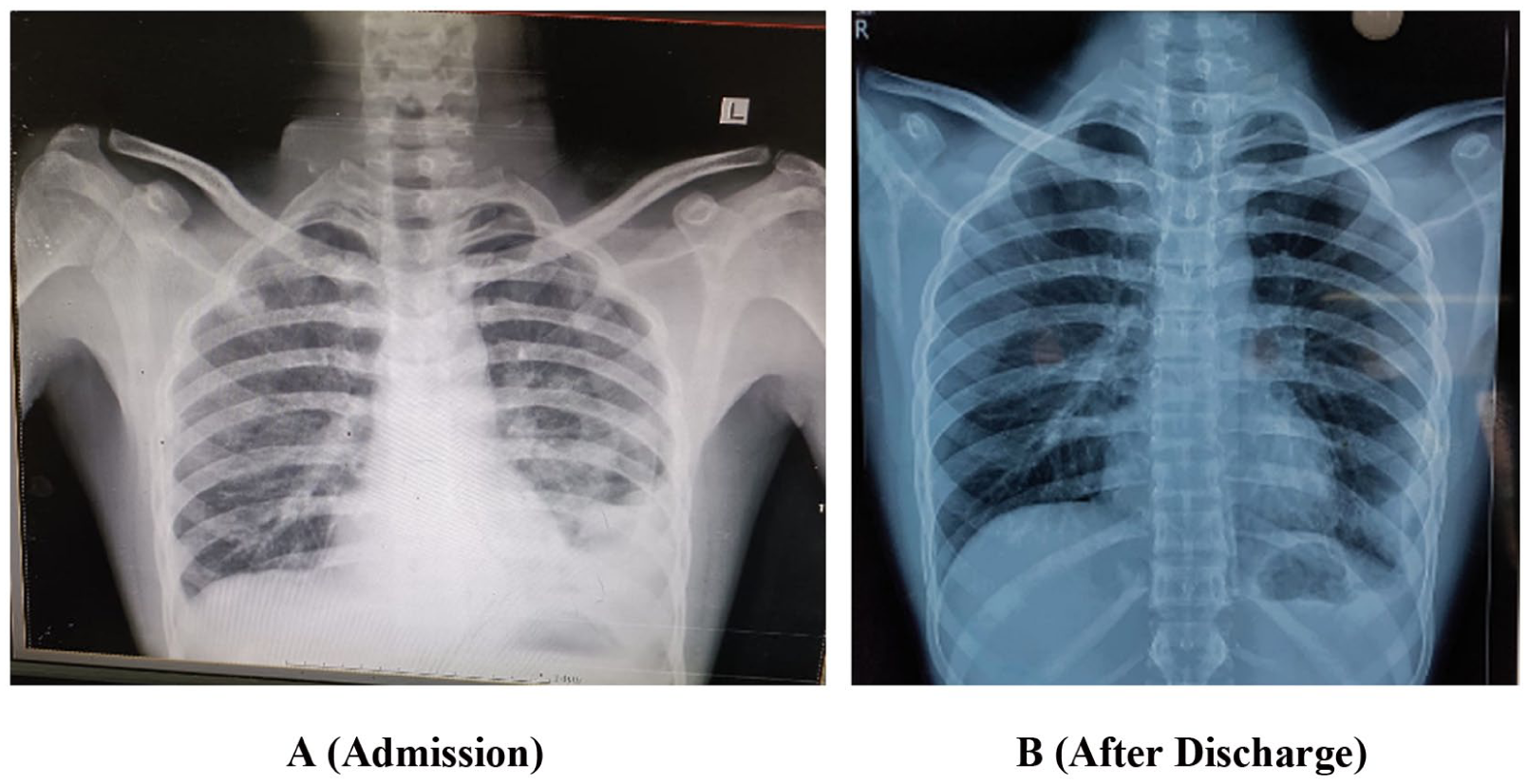
Left sided pneumothorax during admission and resolution of disease can be seen after discharge A (Admission) B (After Discharge).
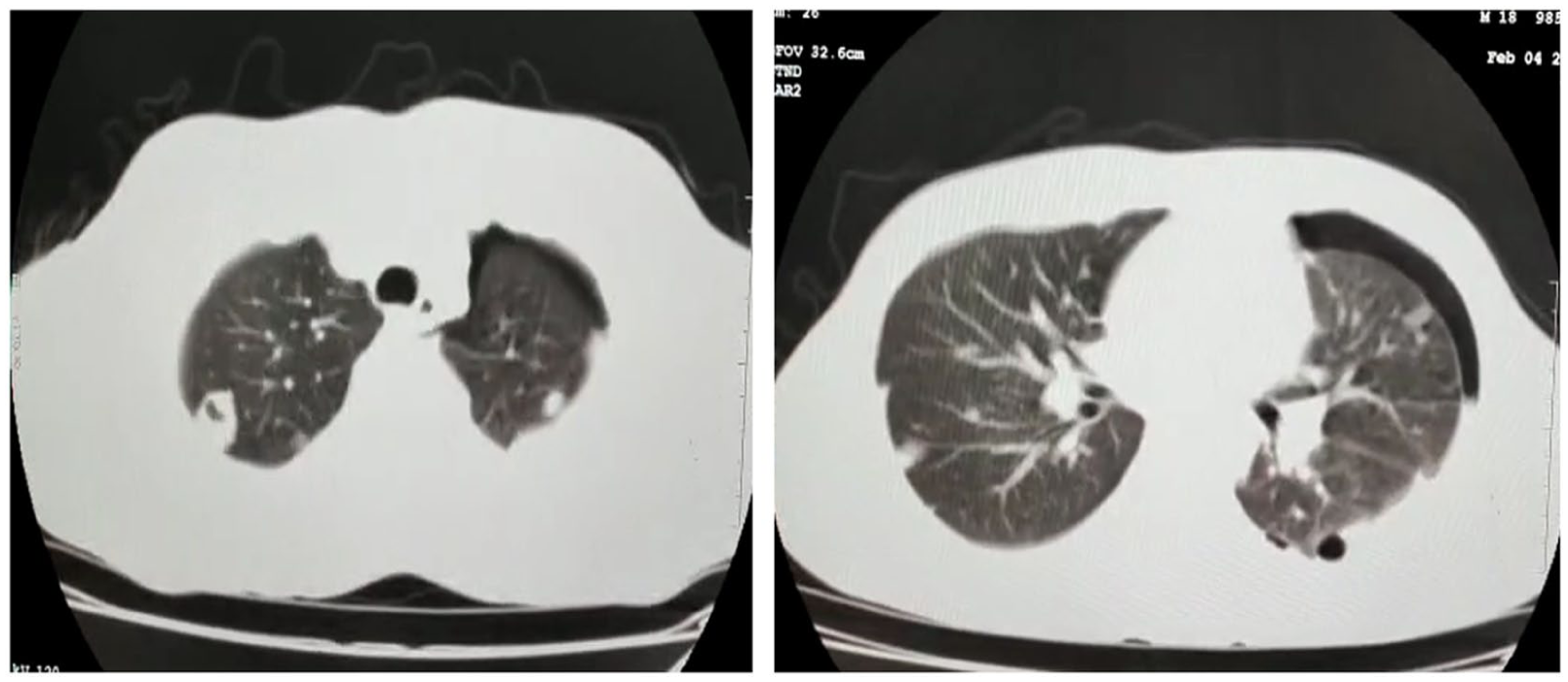
Hydropneumothorax was noted. Diffusely scattered nodules in bilateral lung fields with few of the nodules showing cavitation in right upper lobe.

MRI brain and orbits with contrast showing dilated, tortuous superior ophthalmic veins with thrombosis, involving bilateral cavernous sinuses and internal jugular veins, along with mild perifocal fat stranding.
The patient was diagnosed with secondary spontaneous pneumothorax and septic thrombophlebitis due to Staphylococcus aureus bacteremia. Management included high-flow oxygen therapy for pneumothorax and intravenous antibiotic therapy. The patient was empirically initiated on intravenous vancomycin (15 mg/kg every 12 hours) due to concern for community-acquired MRSA, pending culture and sensitivity. Once blood cultures confirmed MSSA, oxacillin was added for targeted therapy. Vancomycin was discontinued after 5 days, and therapy was de-escalated to oxacillin monotherapy for a total intravenous duration of 3 weeks. In view of the absence of respiratory distress and radiological signs of tension physiology, conservative management with high-flow oxygen and close monitoring was chosen. Anticoagulation was initiated with low-molecular-weight heparin (enoxaparin) at 1 mg/kg subcutaneously every 12 hours, continued for a week during hospitalization. On discharge, the patient was transitioned to an oral anticoagulant (rivaroxaban 20 mg once daily) for a further 4 weeks. This regimen was chosen given the extent of thrombosis involving the ophthalmic veins, cavernous sinuses, and internal jugular veins. The role of anticoagulation in Lemierre’s syndrome remains controversial, but in this case it was employed due to extensive thrombus burden and intracranial venous involvement. Supportive care included fever management with paracetamol 1 g every 8 hours, pain control with NSAIDs as needed, and close monitoring of vital signs, inflammatory markers, and coagulation parameters. After 3 months follow-up, the patient had resumed normal daily activities with no recurrence of symptoms, demonstrating successful recovery.
Discussion
Lemierre’s syndrome is a rare and potentially fatal condition which predominantly affects young, healthy individuals aged 19 to 22 years, with a male-to-female ratio of 2:1. 4 Despite its rarity, the incidence of Lemierre’s syndrome has been increasing, likely due to the restricted use of antibiotics for upper respiratory tract infections. The disease follows a rapid and often irreversible course, where delayed diagnosis can lead to serious complications and increased mortality.5,6
Lemierre’s syndrome is primarily caused by Fusobacterium necrophorum or Fusobacterium nucleatum, both gram-negative anaerobic bacteria.1,2 These organisms invade the pharyngeal mucosa already compromised by a preceding bacterial or viral infection. Other pathogens, such as Streptococcus species, Bacteroides species, Staphylococcus aureus, and Klebsiella pneumoniae, have also been implicated.3,6 The disease typically begins as an oropharyngeal infection that progresses to mucosal damage and subsequent Fusobacterium superinfection. 2 This infection spreads to the lateral pharyngeal space, leading to septic thrombophlebitis of the internal jugular vein. The virulence of F. necrophorum is attributed to its production of hemagglutinin, which promotes venous thrombus formation and subsequent septicemia and embolization. Immunocompromised individuals and environmental factors further increase susceptibility to this syndrome.1,2
The clinical presentation often involves acute pharyngitis, high fever, rigors, neck pain, and tenderness, typically developing 4 to 7 days after the initial illness, with or without respiratory symptoms. 2 However, the current case deviated from this pattern, with no history of sore throat, cough, or cold. Instead, the patient presented with a boil above the right eye, which resolved but was followed by systemic symptoms and complications. The infection likely spread directly into the lateral pharyngeal space, leading to thrombophlebitis of the internal jugular vein – a hallmark of Lemierre’s syndrome.7,8 Therefore, this case represents an atypical variant of Lemierre’s syndrome caused by MSSA. Complications such as septic emboli, sepsis, and thrombus extension usually manifest 1 to 2 weeks after the onset of symptoms. Metastatic infections most commonly involve pleuropulmonary sites (92%-97%) but may also affect joints, muscles, bones, and the liver.7,8
Diagnosis requires a high index of clinical suspicion supported by appropriate imaging and microbiological studies. Contrast-enhanced CT (CECT) is the gold standard investigation, capable of identifying intraluminal thrombi and metastatic septic emboli.2,4 The pulmonary findings on HRCT required careful consideration of pneumatoceles versus septic emboli. Pneumatoceles appear as thin-walled, air-filled cystic spaces typically seen in Staphylococcus aureus pneumonia, whereas septic emboli usually present as peripheral, pleural-based nodules that may show cavitation.2,4 In this case, several HRCT-visualized lesions demonstrated thin walls, suggesting pneumatoceles. however, septic emboli could not be excluded due to extensive venous thrombosis and bacteremia. Blood cultures frequently identify F. necrophorum, although the current case revealed Staphylococcus aureus resistant to macrolides. 9 Doppler ultrasound may assist in identifying internal jugular vein thrombosis when CECT is unavailable, while MRI may be used to delineate soft tissue and intracranial involvement.2,4 Further, imaging revealed left-sided secondary spontaneous pneumothorax, minimal pleural effusion, scattered lung nodules with cavitation (notably in the right upper lobe), and thrombosis in the superior ophthalmic veins, cavernous sinuses, and bilateral internal jugular veins. In our case, the differential diagnoses considered included right-sided infective endocarditis, necrotizing pneumonia, and pulmonary tuberculosis. Right-sided endocarditis was excluded by a negative transthoracic echocardiogram and the absence of valvular lesions. Necrotizing pneumonia was initially suspected due to the presence of cavitary lung lesions, but this was ruled out given the simultaneous involvement of the internal jugular veins and cavernous sinus thrombosis, findings that were more consistent with septic emboli. Pulmonary tuberculosis was also considered but excluded based on negative sputum AFB/GeneXpert testing and the rapid radiological progression, which was inconsistent with tuberculosis.
Management of Lemierre’s syndrome involves a multidisciplinary approach aimed at eradicating the infection, controlling complications, and providing supportive care. Empiric antibiotic therapy is the cornerstone, with broad-spectrum agents such as piperacillin-tazobactam or carbapenems targeting anaerobes like Fusobacterium necrophorum and aerobes like Staphylococcus aureus. Therapy is tailored once culture sensitivities are available, with MRSA-specific agents like vancomycin or linezolid often required. Antibiotic treatment typically continues for 4 to 6 weeks. 10 Anticoagulation therapy is considered in cases of extensive thrombosis or cavernous sinus involvement to prevent thrombus propagation, using agents like LMWH or DOACs. 10 In cases with abscesses, such as peritonsillar or pulmonary abscesses, surgical or percutaneous drainage may be necessary. Management of complications like pneumothorax involves chest tube placement for large pneumothoraces or drainage for pleural effusions.2,4,10 Supportive care, including oxygen therapy, fluid resuscitation, and antipyretics, is essential, with critical care interventions such as mechanical ventilation required in severe cases. 10 Regular monitoring of inflammatory markers and imaging guides treatment efficacy. A multidisciplinary team, including infectious disease, pulmonology, and otolaryngology specialists, is crucial to manage the complex manifestations of Lemierre’s syndrome. Early recognition, aggressive treatment, and education on timely care are vital for optimal outcomes.
Our case has several limitations due to our rural, resource-restricted setting. CT pulmonary angiography was not available, which prevented direct visualization of pulmonary arterial thrombi. Pleural fluid sampling and analysis could not be performed due to the minimal effusion and lack of diagnostic infrastructure. These constraints limit precise characterization of the pulmonary lesions and pleural space pathology. However, interpretations were made using the best available clinical and HRCT data.
Conclusion
This case illustrates an atypical form of Lemierre’s syndrome caused by Staphylococcus aureus, highlighting skin infection as a rare portal of entry. Clinicians should remain alert to such unusual presentations, as early recognition, targeted antimicrobial therapy, and timely multidisciplinary management are key to preventing severe complications and ensuring favorable outcomes.
Footnotes
Acknowledgements
Special thanks to Squad Medicine and Research (SMR) for their guidance and help in publication.
Ethical Considerations
In our university, Ethics approval was not required for case reports and case series.
Consent to Participate
Informed consent was obtained from the patient for the research and the publication.
Author Contributions
Raj S D – Idea, conceptualization, supervision, writing draft, approved final draft. Suvvari TK – Project Administration, resources, formal analysis, writing draft and revision of draft, approved final draft. Vallurupalli V – conceptualization, resources, formal analysis, writing draft and revision of draft, approved final draft. Koneru SK – conceptualization, resources, formal analysis, writing draft and revision of draft, approved final draft. Pottabathini R – resources, data collection, writing draft, revision of draft and approved final draft. Nimmalapudi SSD – resources, data collection, writing draft, revision of draft and approved final draft. Thomas V and Singh T – Idea, Project Administration, formal analysis, resources, writing draft and revision of draft, approved final draft.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
