Abstract
Introduction:
Mitragyna speciosa (commonly known as kratom) has both opioid and stimulant-like effects. Recently, Thailand decriminalized the possession and sale of kratom which led people in many areas to seek income from the sale of kratom at a time of widespread unemployment due to COVID-19. Here, we report a patient with post-COVID syndrome who developed a mixed cholestatic-hepatocellular liver injury secondary to kratom.
Case presentation:
A 23-year-old Thai man was seen for an evaluation of fatigue and nausea which was soon followed by pruritus, dark urine and jaundice. The patient had no known underlying disease but had been treated with mild COVID-19 pneumonia in the past 2 months. He reported taking kratom recreationally for 2 weeks as a treatment for his post-COVID insomnia. Kratom was purchased from a friend and used in a homemade iced cocktail called “4 × 100” consisting of Coca-Cola, tea made from boiled kratom leaves, and diphenhydramine-containing cough syrup which has been popular in Southernmost provinces of Thailand. His lab workup showed his total bilirubin to be 10.6 mg/dL, aspartate aminotransferase was 642 U/L, and alanine aminotransferase was 1635 U/L. Extensive workups including viral etiologies was negative. Abdominal ultrasound revealed normal liver and no cirrhosis. The case was managed conservatively for 5 days in the hospital by giving intravenous fluid and stopping all medications. Urine toxicology screening confirmed the presence of mitragynine and diphenhydramine. He was in a stable condition with normalized liver function tests at 3 months after discharge.
Conclusion:
The COVID-19 pandemic has posed unprecedented challenges to health consequences and this case highlights the importance of kratom as potential cause of acute liver injury. Future studies should accumulate further case series and identify kratom-user subgroups or the polydrug patterns of kratom use that are at heightened risk of severe liver injury.
Introduction
Kratom is a tropical evergreen tree (Mitragyna speciosa) which has been used in herbal medicine in Southeast Asia since the nineteenth century for its stimulant effects. 1 Mitragynine and 7-hydroxymitragynine are the key compounds which could act on μ-opioid receptors. 2 Kratom has been used recreationally for alleviating pain, anxiety, depression, or opioid withdrawal. Originally, it was consumed as tea or chewable leaves. However, a tea-based cocktail known as “4 × 100” became popular among some young people along the border of Malaysia and southern Thailand in the past decade.3 -7 This cocktail composed of carbonated soft drinks, extracted tea made from boiled kratom leaves, diphenhydramine-containing cough, and ice. The addition of carbonated soft drinks helps to reduce the bitter taste of the kratom juice. While the mechanism of this homemade cocktail is unknown, it is speculated that diphenhydramine may facilitate the binding of opioids to the opioid receptor or augment the more opioid available for specific opioid receptors.8,9 Moreover, several cases report of kratom-related fatality had been increasing reported in the past decade.10,11
There have been several reports demonstrating a substantial increase in substance use and abuse during the COVID-19 (Coronavirus disease 2019) pandemic.12,13 In additions, the COVID-19 pandemic has posed unprecedented challenges to the global healthcare crises, persistent, and prolonged effects after acute COVID-19 called long COVID or post-acute COVID-19 syndrome impact on the affected patients’ daily functioning, and ability to work relative to pre-illness levels. 14 Literatures on various adverse effects from kratom including liver toxicity, seizure, abstinence syndrome, and even fatal complications have been emerging in the past 5 years after the possession and sale of kratom have been decriminalized in many countries.15 -18 Herein, we illustrate a patient with long COVID who developed acute mixed cholestatic-hepatocellular liver injury secondary to kratom used as a homemade iced cocktail called “4 × 100.” An informed written consent was obtained from the patient for publication of this case report which has been approved by the ethics committee of Theptarin Hospital, Bangkok, Thailand (IRB No. 6/2021).
Case Presentation
A 23-year-old Thai man was seen for an evaluation of fatigue and nausea which was soon followed by pruritus, dark urine and jaundice. He denied diarrhea or steatorrhea but noticed pale stool. The patient had no known underlying disease but had been treated with mild COVID-19 pneumonia in the past 2 months. At that time, he did not receive oral medications or steroid. Normally, he consumed 2 to 3 bottles of beer every other day for 3 years. He denied taking other medications and no recent travel history. However, the patient reported taking kratom recreationally as a treatment for his post-COVID insomnia and anxiety. Kratom was purchased from his friend and used as a homemade iced cocktail called “4 × 100” that consists of Coca-Cola, tea made from boiled kratom leaves, diphenhydramine-containing cough syrup, and ice cubes as shown in Figure 1. He consumed 600 mL of the cocktail on averages every night with his friends for 14 days before the onset of illnesses. In the past 2 months after recovery from COVID-19, he also increased his usual alcohol intake from every other day to every day with a couple of beers and a half bottle of Thai spirits (distilled alcoholic beverages which contains 38% alcohol) weekly. The estimated amount of alcohol intake was 27 standard drinks (270 g of alcohol) per week. His last drink was 1 week before he came to the hospital.

A homemade iced cocktail called “4 × 100” that consists of Coca-Cola, tea made from boiled kratom leaves, diphenhydramine-containing cough syrup and ice cubes (A) Fresh kratom leaves 25 to 40 leaves and cough syrup as main ingredients for cocktail (B) Preparing boiled water with cough syrup (C) Brewing kratom tea with room-temperature water (D) Cooling down the pot once all ingredients for 4 × 100 have been cooked together.
At presentation (10 days after symptoms), the patient was afebrile and had a regular heart rate of 85 beats/minute, blood pressure of 119/75 mm Hg, and oxygen saturation of 99% on room air breathing. Cardiac and pulmonary examinations were unremarkable. He had epigastric tenderness on right upper quadrant but no rebound or guarding. His skin was dry, and there was a marked icteric sclera. Neurologic examination findings were within normal limits. Laboratory studies showed the following (reference ranges provided parenthetically): hemoglobin 15.4 g/dL (13.5-17.5 g/dL); leukocyte count 7450 cells/µL (3400-9600 cells/µL); platelets 244 000/µL (150 00 to 400 000/µL); sodium 136 mmol/l (135-145 mmol/L); potassium 4.2 mmol/L (3.5-5.1 mmol/L); creatinine 0.81 mg/dL (0.74-1.15 mg/dL); glucose 96 mg/dL (70-100 mg/dL); albumin 4.4 g/dL (3.5-5.2 g/dL); prothrombin time 13.1 seconds (10.4-12.1 seconds); international normalized ratio 1.15 (0.8-1.1). Urine examination revealed dark urine with the presence of urobilinogen and bilirubin. Liver function tests revealed marked elevated transaminase and direct bilirubin (aspartate aminotransferase 642 U/L (normal <40 U/L); alanine aminotransferase 1635 U/L (normal <41 U/L); alkaline phosphatase 285 U/L (40-129 U/L); total bilirubin 10.6 mg/dL (0-0.3 mg/dL); direct bilirubin 9.2 mg/dL (0-0.3 mg/dL). Extensive workups including viral etiologies (hepatitis A, B, C, E) as well as markers for autoimmune hepatitis were negative. Abdominal ultrasound revealed overall decreased echogenicity of the liver without cirrhosis. Liver biopsy was not performed. A Roussel Uclaf Causality Assessment Method (RUCAM) which is a well-known validated tool for assessing the causality of adverse drug reactions 19 suggested probable causality of Kratom-associated liver injury (score of 7) as shown in Table 1. Urine qualitative toxicology tests revealed only mitragynine and diphenhydramine.
Causality Assessment by Roussel-Uclaf Causality Assessment Method (RUCAM) Showed Probable Drug-Induced Liver Injury in This Case.
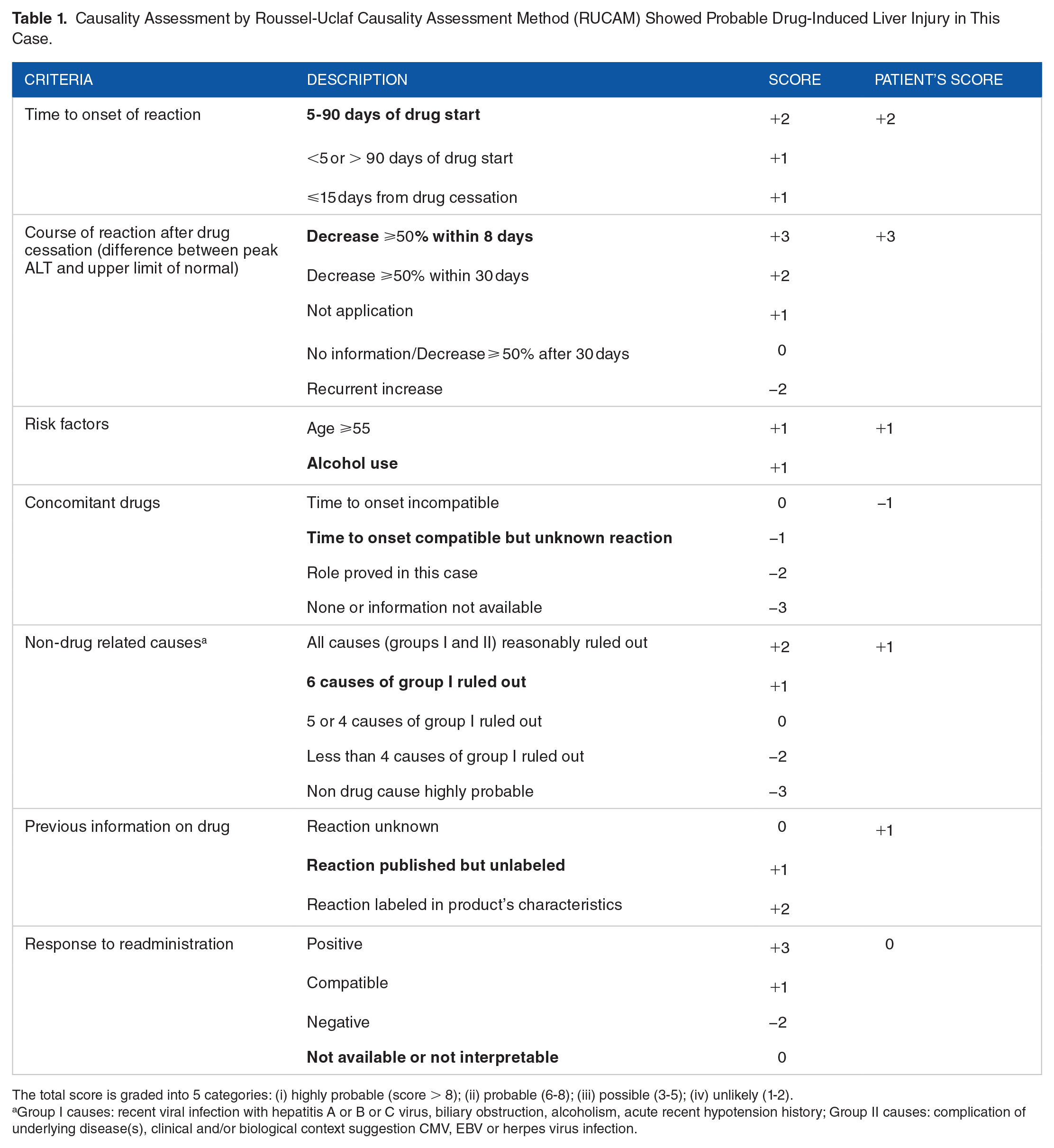
The total score is graded into 5 categories: (i) highly probable (score > 8); (ii) probable (6-8); (iii) possible (3-5); (iv) unlikely (1-2).
Group I causes: recent viral infection with hepatitis A or B or C virus, biliary obstruction, alcoholism, acute recent hypotension history; Group II causes: complication of underlying disease(s), clinical and/or biological context suggestion CMV, EBV or herpes virus infection.
During hospitalization of 1 week with supportive treatments by giving intravenous fluid and stopping all medications, there was a persistent decrease in transaminase levels and cholestasis as shown in Figure 2. Within 72 hours after admission, cholestasis markers reduced with faster speed (reduction for almost 3 times) when compared with hepatocellular necrosis markers (reduction only about half of the initial values). Only oral ursodeoxycholic acid was given as a therapeutic agent for intrahepatic cholestasis. He was in a stable condition with normalized liver function tests at 3 months after discharge.

Clinical course of liver function tests from initial presentation to 2 months after symptoms: (A) Aminotransferase and alkaline phosphatase levels and (B) Total bilirubin and direct bilirubin levels.
Discussion
The increasing use of kratom paralleled the increasing number of reports of related toxicity and deaths in the past decade.12,13 Traditionally, kratom is an indigenous tropical tree in parts of Thailand, Malaysia, Myanmar, Indonesia, Papua New Guinea and the Philippines. In 1979, Thailand moved kratom to category V under the Narcotics Drugs Act, making it illegal to use, buy, sell, grow, or harvest kratom. 3 The new preparation of kratom consumption patterns among youth under the name “4 × 100” cocktail (a mixture of kratom tea, cough syrup containing diphenhydramine or codeine, a caffeine-containing soft drink, and ice cubes) as a way to achieve euphoric state emerged in 2010 seconds and illicit drugs are added in some cases such as anxiolytics, alcohol, anti-depressants, or tramadol.3 -5 Our present case highlights the need to recognize kratom as potential cause of acute hepatotoxicity among pandemic period. However, we acknowledged the limitation in missing quantitative data of mitragynine concentration in our present case. Other concomitant medications or alcohol intake in this present case might also contribute for the development of liver injury. More clinical cases report and research attention of kratom-related liver injury are required to establish the solely effect of Kratom with hepatotoxicity.
Since the early 2010 seconds, several case reports and mechanistic studies identified kratom as a potential cause of herbal-induced liver injury. The underlying mechanism of cholestatic liver injury remained unknown but mitochondrial dysfunction in cholangiocytes, oxidative stress, and altered bile acid homeostasis was proposed based on animal studies.15,20 Latency periods from exposure to the onset of illnesses varied from 2 to 49 days (median of 21 days) and cholestatic jaundice was a common presenting symptom. 15 Interestingly, only minority of kratom users experienced clinically apparent liver injury and some patients developed liver injury after only one-time kratom use. Therefore, the interplay between genetic and environmental factors might lead to kratom-induced liver injury as found in other drug-induced liver injury (DILI). 21 The other components of “4 × 100” cocktail could also contribute to the risk of having liver injury but this postulation has not been well established based on previous literatures. Future studies should accumulate further case series and identify kratom-user subgroups that are at heightened risk. Managements of kratom-associated liver injury are similar to general treatments for DILI (rapid discontinuation of the offending suspected agents/drugs, use of ursodeoxycholic acid in a cholestatic injury, therapeutic trial of N-acetylcysteine in a hepatocellular injury pattern). 22 A favorable clinical course of kratom-induced liver injury have been reported in most cases. Brief admissions followed by outpatient management have been successful in most cases including the present one. Only 2 case reports of fulminant hepatic failure requiring liver transplantation have been documented due to kratom ingestion.23,24
Recently, Thailand decriminalized the possession and sale of kratom, which led many people to explore this as new income avenue in time of high unemployment rate due to Covid-19. This situation is reminiscent of liberal cannabis policies in the United States which increased the prevalence of cannabis-related disorders. 25 Even though there is growing evidence that kratom is safe if used as pure kratom products or brewed herbal decoction in small doses and for a limited period of time, Kratom abuse and addiction as shown in our present case warrant ongoing monitoring of the liberal kratom use during the current pandemic. Substance use has spiked due to pandemic-related mental stress. The number of kratom users outside of Southeast Asia has also increased significantly in recent decades as an alternative treatment option for pain and opioid use disorder.26,27 The socioeconomic impact of the pandemic on society has been uneven, with more vulnerable groups bearing a disproportionately larger share of the negative impacts. The emergence of the second pandemic from the psychotropic substances might start to worsen the situation in the near future. Substance users are not only at an increased-risk for contracting COVID-19, but are also likely to suffer worse health complications and display poorer treatment outcomes than those who do not use substances. 28
Conclusion
In summary, the polydrug patterns of kratom use could lead to severe liver injury. The COVID-19 pandemic has posed unprecedented challenges to health consequences and this case highlights the need to recognize kratom as potential cause of acute liver injury.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr. Tinapa Himathongkam for her professional English editing.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Contributions
TY and NS conceptualized and designed the work. TY analyzed and interpreted the patient data, drafted the manuscript and revised the manuscript. KS and HT revised the manuscript critically for important intellectual content. All authors agree to be accountable for all aspects of the work. All authors read and approved the final manuscript.
