Abstract
Background:
Myocarditis is considered a serious adverse event after COVID-19 infection. The risk and severity of myocarditis after COVID-19 disease decreased significantly in the vaccinated population. We present a case of cardiac magnetic resonance proven fulminant myocarditis following COVID-19 disease in a young female who was previously vaccinated with 2 doses of the BIBP (Sinopharm) vaccine.
Case summary:
A 29-year-old female was referred to the hospital with acute chest pain, dyspnea, and nausea. Her electrocardiogram revealed ST-segment elevation in anterolateral leads with reciprocal changes in inferior leads. She was primarily diagnosed with ST-elevation myocardial infarction following spontaneous coronary artery dissection (SCAD) according to her age and gender. Her coronary angiography was normal. RT-PCR nasopharyngeal swab was positive for SARS-COV-2 infection. According to her history and excluding coronary artery diseases, she was clinically diagnosed with myocarditis and received corticosteroids, IVIG, and colchicine. She was discharged in a favorable condition after 11 days of hospitalization. Cardiac magnetic resonance imaging confirmed the diagnosis of myocarditis according to the updated lake Louise criteria. On her 4-month follow-up, she was asymptomatic, and her echocardiography showed improvement in biventricular function.
Discussion:
The diagnosis of myocarditis caused by COVID-19 infection may be challenging as the symptoms of myocarditis, and COVID-19 disease may overlap. It should be considered when patients have acute chest pain, palpitation, elevated cardiac biomarkers, and new abnormalities in ECG or echocardiography. Cardiac MRI is a non-invasive gold standard modality for diagnosing and follow-up of myocarditis and should be used in clinically suspected myocarditis. The long-term course of myocarditis following COVID-19 disease is still unclear, but some evidence suggests it may have a favorable mid-term outcome.
Introduction
Since COVID-19 emerged, many studies have reported cardiac complications of COVID-19 disease. These complications include Myocardial infarction, thrombotic events, Arrhythmias, Heart failure, and Myocarditis.1 -3 A sizable portion of cardiac complications seems to be Myocarditis.4 -7
Myocarditis is a potential life threatening adverse event of both COVID-19 infection and vaccine.7 -10 The overall incidence of Myocarditis due to COVID-19 infection is unclear, but it is estimated to range from 0.004% to 0.04% among different groups11,12; however, Myocarditis in hospitalized patients was reported more frequent in previous studies (0.1%-1%).13 -15 The most common reported symptoms of COVID-19 associated Myocarditis were chest pain, dyspnea, palpitation, fever, nausea, vomiting, and syncope.1,16,17 The diagnosis of COVID-19-related myocarditis is based on clinical presentation, physical examination, elevated cardiac enzymes, and the use of imaging modalities such as echocardiography and cardiac MRI. 4 The cardiac biopsy is the myocarditis gold standard diagnostic tool, but it is used infrequently.18 -20 According to the potential progression of acute Myocarditis to cardiogenic shock, malignant arrhythmias, chronic heart failure, myocardial fibrosis, and even death, timely diagnosis and treatment is crucial.21,22 Myocarditis can increase mortality in patients with COVID-19 disease, The overall mortality rate of COVID-19 related myocarditis is estimated to be between 13%and 19%,1,23 and it has a worse prognosis than myocarditis caused by COVID-19 vaccination. The incidence of SARS-COV-2 cardiac complications, including Myocarditis, decreased significantly in vaccinated groups. According to a recent study, compared to the non-vaccinated population, the risk of myocarditis is reduced in half if someone experiences COVID-19 disease after receiving at least 1 dose of the COVID-19 vaccine.24,25 We present a case of acute fulminant Myocarditis in a fully-vaccinated female after get infected with COVID-19 disease.
Case Presentation
A 29-year-old female was presented to the emergency department with chest pain and nausea. Her symptoms began suddenly 3 days before the presentation. She complained of retrosternal chest pain that radiated to her left arm and shoulder. The pain was not positional or pleuritic and not related to her physical activity. She also complained of the flu-like syndrome, including mild fever, rhinitis, and myalgia, accompanied by mild dyspnea and diaphoresis 1 day before her chest pain began, but she denied having any cough. She was vaccinated with 2 doses of the BIBP (Sinopharm) vaccine, and her last injection was 3 months before the onset of her symptoms. She had a history of hypothyroidism, using levothyroxine for her condition, but no other history of cardiovascular or metabolic disease. She had no risk factor for coronary artery disease. The intensity of her pain increased gradually from the onset of the symptoms; thus, she decided to consult with her primary physician. Her primary physician ordered an ECG due to her acute-onset chest pain. Her electrocardiogram showed ST-segment elevation in the anterolateral leads with reciprocal ST-depression changes in the inferior leads. (Figure 1) then she was referred to our tertiary heart center for further evaluation. Upon her arrival at the emergency department, her vital sign was stable. Her blood pressure was 120/70 mmHg, pulse rate was 80 beats/minute, respiratory rate was 18 breaths/minute, body Temperature was 36.8°C, and SPo2 was 96% without supplemental oxygen. On physical examination, she was conscious and oriented, no cardiac murmur was heard, lung examination was normal, and other examinations were unremarkable. She was transferred to the Cath lab with the initial diagnosis of ST-segment elevation myocardial infarction caused by suspected spontaneous coronary artery dissection (SCAD) according to her age and gender. She was started on nitrate, beta-blocker, ondansetron, statin, dual anti-platelet agents including aspirin and clopidogrel and morphine sulfate; then, she was undergone coronary angiography (CAG).

Initial electrocardiogram showed ST-segment elevation in the lateral limb leads (I and aVL). Note the reciprocal ST-segment depression change in inferior leads.
There was no evidence of coronary artery obstruction or stenosis in her angiogram (Figure 3). Her chest pain continued, despite the administration of pain relief agents and normal coronary angiogram; therefore she was admitted to CCU for additional evaluation and observation. She continued to receive nitrates for her chest pain and ondansetron for her nausea, but other medications were discontinued. Her initial laboratory tests revealed a normal complete blood count and kidney function but an elevation of cardiac troponin I and creatine kinase-MB (85 U/L, normal <28 U/L) (Table 1).
Key laboratory values.
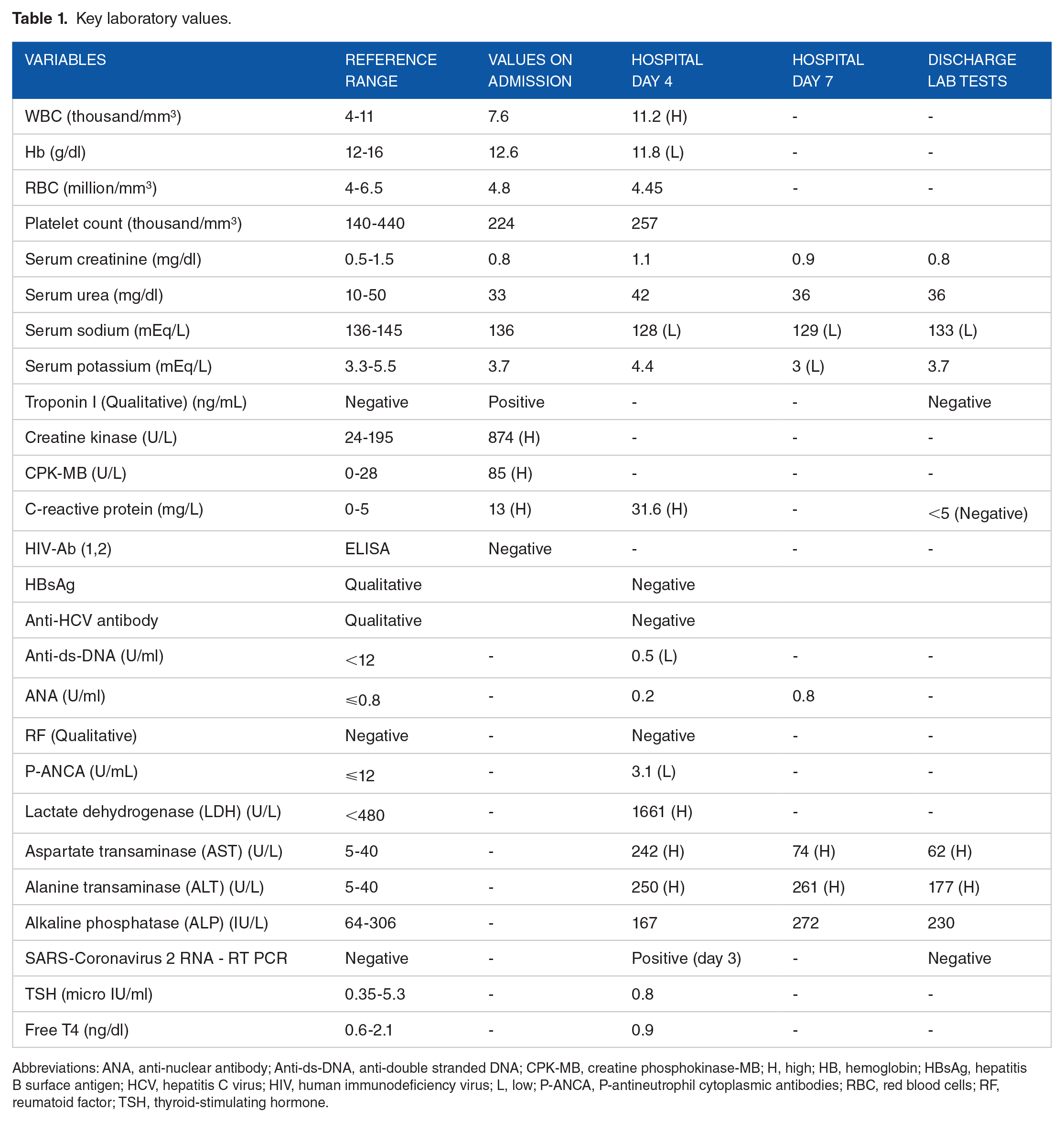
Abbreviations: ANA, anti-nuclear antibody; Anti-ds-DNA, anti-double stranded DNA; CPK-MB, creatine phosphokinase-MB; H, high; HB, hemoglobin; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; HIV, human immunodeficiency virus; L, low; P-ANCA, P-antineutrophil cytoplasmic antibodies; RBC, red blood cells; RF, reumatoid factor; TSH, thyroid-stimulating hormone.
A bedside echocardiogram was performed, which revealed a left ventricular ejection fraction (LVEF) of 45% and Antero-apical hypokinesia without valvular or pericardial abnormalities. During her second day of hospital admission, her blood pressure suddenly dropped to 88/53 mmHg, and oxygen saturation fell to 87%, requiring inotrope and supplemental oxygen to maintain blood pressure and oxygen saturation, respectively. Her dyspnea worsened, and her body temperature rose to 38.6°C. Nitrate was discontinued according to her reduced blood pressure. Her ECG showed sinus tachycardia with prominent ST-segment elevation in anterolateral leads and new right bundle branch block (RBBB). (Figure 2) Second echocardiography was performed; Her LVEF dropped to 15% with global hypokinesia, especially in the anterior circulation, and significant LV enlargement. (Table 3) She was suspected of missed or new myocardial infarction complicated with cardiogenic shock, so she was transferred to the Cath-lab and underwent her second CAG. Similar to her previous coronary angiogram, there were no signs of obstruction or stenosis, and also there was no angiographic evidence of SCAD. (Figure 3) According to the CDC definition of probable acute myocarditis, she was clinically diagnosed with possible acute myocarditis and was scheduled to receive IVIG, colchicine, and corticosteroid.

Second electrocardiogram showed prominent ST-elevation in anterior leads and new right bundle branch block (RBBB).
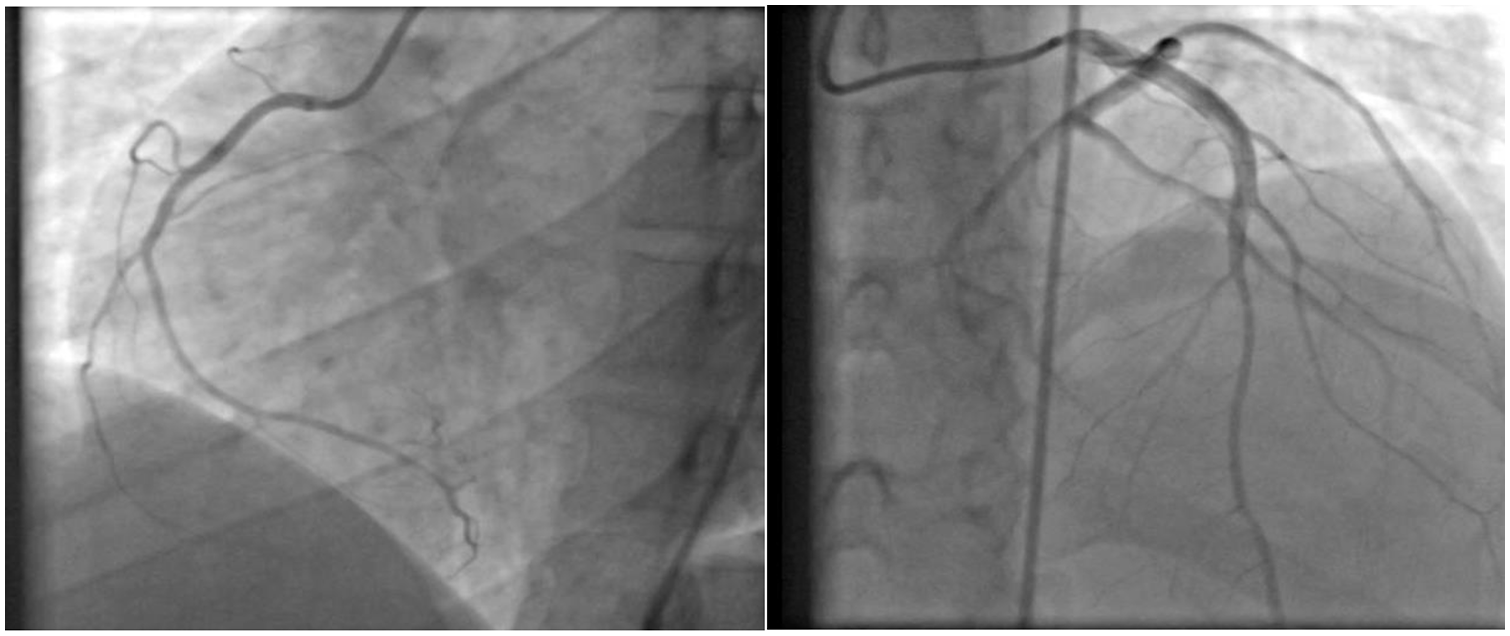
Initial (left) and second (right) coronary angiography revealed no sign of stenosis or obstruction.
Because of her flu-like symptoms at the time of the COVID-19 pandemic, a nasopharyngeal swab was obtained and tested for SARS-COV-2 virus using the RT-PCR method on the third day of her admission; the result was positive. Viral serology and autoantibody screening tests were performed, and they were all negative. The spiral chest CT scan revealed bilateral pleural effusion, pericardial congestion, significant cardiomegaly, and peripheral/hilar ground glass opacity, indicating possible COVID-19 lung involvement (Figure 4); therefore pulmonology consultation was requested, and then she was started on antiviral agent remdesivir and was continued to receive corticosteroid. The following day the patient’s condition continued to deteriorate and her skin became icteric; her liver function test showed abnormal AST, ALT, and bilirubin due to possible congestive hepatitis; therefore, her remdesivir was discontinued. She was transferred to the intensive care unit. During the ICU admission her condition gradually improved, her blood pressure back into normal ranges without inotropes, the elevated hepatic and cardiac blood test gradually reduced, and her LVEF increased to 35% on following days. She was discharged with oral prednisolone taper (50 mg once daily for 2 weeks, then 25 mg once daily for 1 week and 5 mg twice daily for 2 weeks), colchicine 1 mg once daily for 3 months and captopril 12.5 mg 3 times a day after 11 days of hospitalization. She was recommended for exercise restriction for 6 months.

Spiral chest CT scan revealed bilateral pleural effusion, significant cardiomegaly, and interlobular septal thickening, which indicates cardiogenic pulmonary edema. The presence of peripheral/hilar ground glass opacity and reversed halo sign indicates COVID-19 lung involvement.
6 days after her discharge a cardiac magnetic resonance imaging (CMR) was performed. LVEF was 41.6%, and severe hypokinesia was noted in the mid to apical anterior, inferolateral, and anterolateral walls of the left ventricle. Left ventricular hypertrophy (LVH) was not reported. The right ventricular ejection fraction was 55%, and there was no evidence of regional wall motion abnormalities or systolic dysfunction in the right ventricle. There was also no sign of edema on T2 STIR weighted imaging, but T2 mapping showed prolongation of T2 relaxation time in multiple segments of the left ventricle, which may be considered diffuse inflammation/edema (Table 2).
Cardiac MRI findings of patient.

Abbreviations: LVEDVI, left ventricle end diastolic volume index; LVEF, left ventricle ejection fraction; LVESVI, left ventricle end systolic volume index; LVH, left ventricular hypertrophy; ms, millisecond; RVEDVI, right ventricle end diastolic volume index; RVEF, right ventricle ejection fraction; RVESVI, right ventricle end systolic volume index.
Abnormal T1 relaxation time and increased extracellular volume (ECV) were observed in the left ventricle’s interventricular septum and lateral wall, which indicates scar/necrosis in these segments.
Mid-wall delayed enhancement was seen in the inferoseptal wall of the left ventricle, and subepicardial delayed enhancement was seen in the anterior and lateral wall of the left ventricle. (Figures 5 and 6)

Late gadolinium enhancement distribution of patient′s left ventricle based on AHA 17 segment model for myocardial segmentation. 26

Cardiac magnetic resonance imaging showed subepicardial scar on anterior and lateral wall of the left ventricle. (Arrow).
On her 4-month follow-up, her general condition was favorable, she complained of fatigue and intermittent mild non-typical chest pain but no symptoms similar to admission time. Her ECG pattern was regular, and her echocardiography revealed normal LV size and mild LV systolic dysfunction (EF: 45%). Her biventricular function improved significantly; there was no valvular abnormality except for persistent mild to moderate mitral regurgitation. (Table 3) She was scheduled for a follow-up CMR 6 months after discharge to evaluate mid-term cardiac risks.
Echocardiographic parameters changes during hospital admission and on follow up.

Abbreviations: LVEDD, left ventricle end diastolic diameter; LVEF, left ventricle ejection fraction; LVESD, left ventricle end systolic diameter; MR, mitral regurgitation; PE, pericardial effusion.
Discussion
Herein we present a case of acute fulminant myocarditis caused by COVID-19 infection in a Fully-vaccinated female. Cardiovascular complications, including myocarditis, were previously reported after COVID-19 infection,2,27,28 but to the best of our knowledge this is the first case report of COVID-19-related fulminant myocarditis in an individual who was previously vaccinated with 2 doses of the COVID-19 vaccine. Myocarditis is defined as myocardial cell inflammation characterized histopathologically by non-ischemic myocyte necrosis and mono-lymphocytic infiltration.29,30 Myocarditis can affect the myocardium in both focal and diffuse patterns and can be presented in acute, subacute, and chronic types. 31 Various etiologies can develop myocarditis, including viral and bacterial infections, Auto-immune disease, allergic reactions, drugs, and toxic substances.32,33 The most frequent cause of acute myocarditis in developed countries is viral infections. 33 H1N1, adenoviruses, Epstein-Barr virus, parvovirus B-19, HHV-6, coxsackieviruses, and also beta-coronaviruses including MERS and SARS-COV-2 are among the viruses that can induce myocarditis.4,34 Studies on COVID-19 complications revealed that myocarditis is a common complication among patients with severe disease, but the exact mechanism is still not clear. SARS-COV-2 can bind and enter host cells via angiotensin-converting enzyme II receptor (ACE2 receptor). The heart myocardium is among the tissues with high expression of ACE2 receptor; hence Myocardial Direct Viral invasion via ACE2 receptor is one of the possible pathophysiologies to explain this entity. Another potential mechanism is an exaggerated host immune response to the virus, characterized by an excessive cytokine release and cell-mediated cardiotoxicity caused by T-cells.35,36 There is also some evidence of autoimmune response to myocardium caused by molecular mimicry of cardiac auto-antigens such as myosin and the viral capsid proteins. 37 Clinically diagnosis of myocarditis is based on the patient’s presentation, Elevation of cardiac biomarkers, Abnormal ECG findings, and exclusion of other diagnoses, including coronary artery disease. Endomyocardial biopsy (EMB) is the gold-standard technique for diagnosing myocarditis. The use of EMB is limited now due to its low sensitivity caused by sampling error, poor intra-observer variability, and acceptable sensitivity and specificity of non-invasive modalities such as Cardiac MRI.38 -41 Cardiac MRI is a safe technique that can be used for diagnosis, follow-up, and differentiation of myocarditis to other types of cardiomyopathy. Currently, CMR-based updated lake Louise criteria (LLC) are used for diagnosing myocarditis with sensitivity and specificity of 88% and 96%, respectively. It is based on 2 main parameters: (1) Elevated T2 relaxation time or T2 weighted imaging, which indicates myocardial edema (2) Presence of one or more T1-based criteria, including native T1 relaxation time hyper intensity, high extracellular volume (ECV), and late gadolinium enhancement. Both criteria must be met to establish the diagnosis of myocarditis. 42 Our patient’s CMR study showed diffuse myocardial inflammation/edema according to the prolonged T2 relaxation time and the sign of necrosis due to elevated native T1, Therefore she diagnosed myocarditis based on updated LLC. The pattern of myocardial involvement might help to distinguish the etiology of myocarditis. Previous studies suggest that myocarditis caused by COVID-19 infection and COVID-19 vaccine have a similar pattern with predilection involvement of the mid to basal inferior and inferolateral segment of the myocardium.5,24,43 Moreover, septal LGE was reported to be more frequent in COVID-19-related myocarditis, which signifies worse outcomes in these patients. Limited data exist on the long-term prognosis of patients who recovered from COVID-19-associated myocarditis. Some studies reported favorable clinical outcomes on short to mid-term follow-ups but remaining cardiac magnetic resonance imaging abnormalities, especially the presence of late gadolinium enhancement (LGE).22,44,45 The presence of LGE after the acute phase of myocarditis may indicate myocardial necrosis/fibrosis and might be associated with sudden cardiac death and increased overall mortality in patients with myocarditis 46 ; hence repeated cardiac MRI 4 to 8 months after recovery is recommended for evaluating the patient’s condition. 47 Our patient was scheduled for a follow-up CMR study 6 months after discharge to assess the mid-term cardiac risks.
Footnotes
Acknowledgements
Researchers appreciated the Clinical Research Development Units of Kamali and Rajaee Hospitals in Alborz University of Medical Sciences.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contribution
F.S. participated in patient management, study design, and supervision. P.S. and E.J. collected the patient’s data from her medical records and history, P.S. drafted the manuscript, and E.J. and F.S. revised the manuscript.
All authors read and confirmed the final version of the manuscript.
Consent
Written informed consent were obtained.
