Abstract
A 70-year-old man was referred to our department for evaluation of nephrotic syndrome. Renal biopsy revealed membranous nephropathy (MN). Immunohistochemical analysis demonstrated IgG4-positive staining in the glomeruli and interstitial cells. The presence of serum anti-phospholipase A2 receptor (PLA2R) antibody and enhanced staining of PLA2R in the glomeruli was noted. Computed tomography unidentified the extrarenal lesions of IgG4-related disease. He was diagnosed with PLA2R-associated MN possibly complicated with IgG4 related kidney disease (IgG4-RKD). Storiform fibrosis, a typical manifestation of IgG4-RKD, was not apparent. We herein describe a case of serologically and histologically confirmed PLA2R-associated MN with IgG4+ cell infiltration into the interstitium without any signs of IgG4-RD.
Background
Immunoglobulin G4-related disease (IgG4-RD) is a fibroinflammatory condition that could affect multiple organs. 1 The kidney is one of the most frequently affected organs in IgG4-RD. 2 The most common renal manifestation in IgG4-RD is tubulointerstitial nephritis (TIN), 3 but glomerular diseases have also been reported.3-6 Of these, membranous nephropathy (MN) was the most common, and approximately 7% of patients with MN had accompanying IgG4-related kidney disease (IgG4-RKD). 5 Tissue IgG subclass analyses revealed that IgG4 in the glomeruli was often positive, and was sometimes predominant in the patients with MN in the setting of IgG4-RKD.3,7 However, the target antigen/antibody in IgG4-RKD has not been reported yet. M-type phospholipase A2 receptor (PLA2R) is a major target antigen for primary MN. 8 Recently, a case of IgG4-RKD with histologically positive PLA2R staining in the glomeruli has been reported. 9 In addition, a case of IgG4-RD with serologically confirmed PLA2R-associated MN has been reported.10,11 However, these cases had extrarenal lesion of IgG4-RD.9-11 We herein describe a case of serologically and histologically confirmed PLA2R-associated MN with IgG4+ cell infiltration into the interstitium without any signs of IgG4-RD.
Case Report
A 70-year-old man with nephrotic syndrome was admitted to our department for an evaluation. He had routine health check-ups every 6 months, but no remarkable abnormalities in blood examinations were seen up to a year before referral. Six months before referral, his serum albumin level decreased from 4.3 to 3.2 mg/dL, but further examination was not performed. Thereafter, lower extremity edema appeared a month before referral. As the symptoms persisted, he visited his family doctor. The laboratory data revealed massive proteinuria (urine protein-to-creatinine ratio: 12.5 g/g creatinine) and hypoalbuminemia (1.7 mg/dL). He was diagnosed with nephrotic syndrome and was referred to our department.
A physical examination on admission showed a blood pressure of 148/97 mmHg and a regular pulse rate of 82 beats/minute. Laboratory test results are shown in Table 1.
Laboratory data on admission.

Abbreviations: β-2MG, β-2 microglobulin; ANA, antinuclear antibodies; anti-DNA Ab, anti-DNA antibody; anti-SS-A Ab, anti-SS-A antibody; anti-SS-B Ab, anti-SS-B antibody; Alb, albumin; ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BS, blood sugar; BUN, blood urea nitrogen; C3, complement component 3; C4, complement component 4; CH50, complement activities; ; Cr, creatinine; CRP, C-reactive protein; Hb, hemoglobin; HbA1c, glycosylated hemoglobin; HBs-Ag, anti-hepatitis B surface antigen; HCV-Ab, anti-hepatitis C virus antibodyIg, immunoglobulin; iP, inorganic phosphate; NAG, N-acetyl-β-D-glucosamidase; Plt, platelet; RBC, red blood cell; T-Chol, total cholesterol; TP, total protein; UA, uric acid; WBC, white blood cell count.
A renal biopsy was performed, and light microscopy demonstrated a diffuse thickening of glomerular capillary walls with spike formation and interstitial infiltration of lymphocytes and plasma cells (Figure 1a–c). Global sclerosis was found in 19.2% (5 out of 26) of the glomeruli. Immunofluorescence showed granular staining along the glomerular basement membrane (GBM). It was positive for IgG (3+), C3 (trace), C4 (1+), and C1q (1+), but negative for IgA and IgM (Figure 1d–i). The serum IgG4 level was elevated to 225 mg/dL (normal range: 11-121 mg/dL). Immunohistochemistry for IgG4 showed an increase in IgG4-positive infiltrating cells in the interstitium (>10/high-power field [HPF]) and a diffuse granular positivity along the GBM (Figure 1j and k), suggesting MN accompanied by IgG4-RKD. Tubulointerstitial fibrosis was found in 40% of the area; however, storiform fibrosis, a typical form of IgG4-RKD, was not apparent (Figure 1c). Immunohistochemical staining for PLA2R showed enhanced granular staining along the GBM (Figure 1l and m), indicating PLA2R-associated MN. Contrast enhanced computed tomography was performed to examine the extrarenal lesions of IgG4-RD, but no other lesion was identified. Castleman’s disease, vasculitis, Sjogren’s syndrome, and sarcoidosis were ruled out as differential diagnoses of IgG4-RD.

Photographs of the glomeruli and interstitial lesion in the kidney. (a) Periodic acid-Schiff staining of the renal biopsy specimen showed diffuse thickening of capillary walls. Original magnification: ×400. (b) Periodic acid methenamine silver staining showed bubble-like appearance on the capillary wall (arrow). Original magnification: ×1000. (c) Azan staining revealed 40% of tubulointerstitial fibrosis, but storiform fibrosis was not apparent. Original magnification: ×100. Immunofluorescence for IgG (d), IgA (e), IgM (f), C3 (g), C4 (h), and C1q (i). Original magnification: ×400. Immunohistochemistry for IgG4 showed an increase in IgG4+ plasma cells in the interstitium (j) and a diffuse granular positivity along glomerular capillary walls (k). Original magnification: ×400. Immunohistochemistry for PLA2R showed enhanced granular staining along glomerular basement membrane (l). Original magnification: ×1000. (m) Enhanced granular staining along glomerular basement (arrow) at a higher magnification in a fragment of (l).
The clinical course is shown in Figure 2. We initiated 40 mg/day (0.8 mg/kg/day) of prednisolone 13 days after performing renal biopsy. The level of proteinuria decreased to 1-3 g/day over the following month, and then the dose of prednisolone was tapered down. To decrease maintenance dose of prednisolone, 100 mg/day (2 mg/kg) of cyclosporine was added 41 days after initiating prednisolone, but was discontinued because of the elevation of liver enzymes. As the level of proteinuria did not change, 500 mg (320 mg/m2) of rituximab was administered twice 3 months after initiating prednisolone. Seven months after initiating prednisolone, the patient achieved complete remission. Retrospectively, the level of anti-PLA2R antibody in the sera was collected before starting prednisolone, and 1, 3, and 7 months after initiating prednisolone. Samples were examined using enzyme-linked immunosorbent assay (EUROIMMUN, Germany). The ratio to standard control (20 RU/mL) were 7, 2, <0.7, and <0.7, respectively.
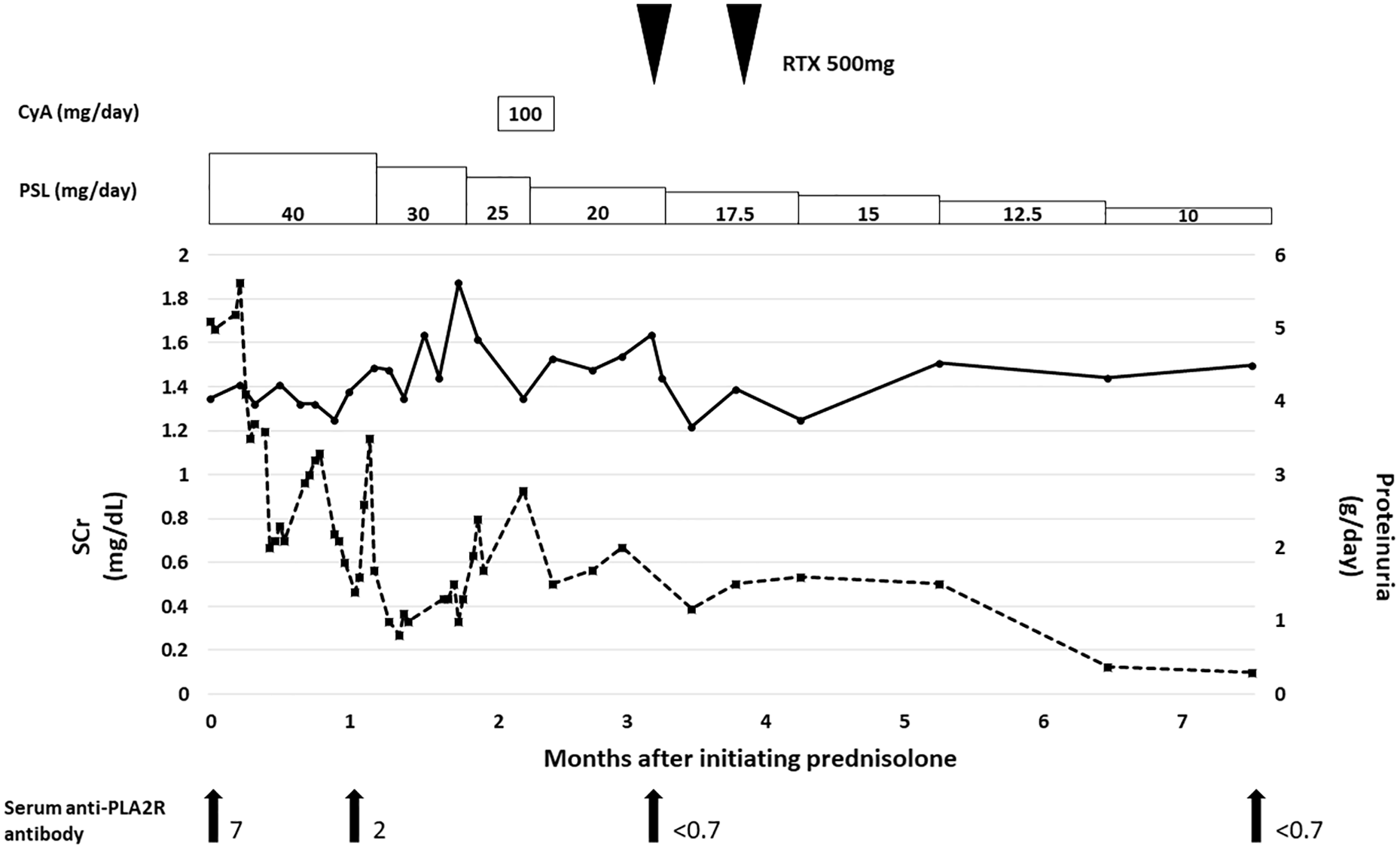
Clinical course after initiation of treatment. The level of serum creatinine and proteinuria are shown in a solid line and dotted line, respectively. The boxes indicate oral administration of prednisolone and cyclosporin A. Arrowheads indicate administration of rituximab. Arrows indicate the time of measuring the level of serum anti-PLA2R antibody. The ratio to standard control (absorbance of patient’s serum/absorbance of standard control) in each point was shown. Standard control includes 20RU/mL of anti-PLA2R antibody.
Discussion
MN is the leading cause of nephrotic syndrome in adults, 12 which is divided into primary and secondary MN based on the etiologies. PLA2R is by far the most frequent podocyte-antigen in primary MN, and IgG4 is a predominant subclass in anti-PLA2R autoantibody. 8 Immunohistochemical analysis using our anti-IgG4 antibody, which could detect the IgG4-predominant (PLA2R/THSD7A-associated) form of MN, 13 showed enhanced granular expression of IgG4 along the GBM, indicating PLA2R/THSD7A-associated MN. The existence of serum anti-PLA2R antibody and enhanced granular expression of PLA2R along the GBM confirmed the diagnosis of PLA2R-associated MN.
In the present case, IgG4-RD was suspected because of the high serum level of IgG4. However, the patient had no symptoms of active IgG4-RD, and computed tomography showed no sign of IgG4-RD. Immunohistochemistry for IgG4 revealed that interstitial IgG4-positive cell infiltration (>10/HPF), the previous diagnostic criteria for IgG4-RD. 14 TIN with storiform fibrosis, a typical renal manifestation of IgG4-RKD, was not found. TIN is often found in patients with MN in the setting of IgG4-RKD, but was not necessarily found in all cases. 7 An interesting case of IgG4-RKD in which MN preceded TIN was reported. 15 Therefore, the current case might be diagnosed with MN prior to developing TIN as IgG4-RKD. Recently, a new classification criterion for IgG4-RD was published from ACCR/EULAR. 16 We could not evaluate the renal specimen with IgG4+/IgG+ ratio because of the lack of remaining renal specimens. Therefore, this case may not meet the new criterion of IgG4-RD, and the diagnosis of PLA2R-associated MN with “renal limited IgG4-RD” is questionable.
To seek whether other similar cases exist in the MN patients, we sequentially examined 17 renal specimens from patients diagnosed with MN in our hospital (Hamamatsu University Hospital; from 2015 to present). IgG4 in the glomeruli was positive in 13 patients. A patient’s sample revealed diffuse IgG4-positive cell infiltration in the interstitium and suspected to have MN possibly with IgG4-RKD. Meanwhile, 2 renal specimens showed focal IgG4-positive cell infiltration in the interstitium, but the number was less than 10/HPF. The result suggested that IgG4-positive cell infiltration was sometimes found in MN, but patients with significant IgG4-positive cell infiltration were rare. The role of IgG4-positive interstitial cells in MN has yet to be elucidated. IgG is produced by B cells and plasma cells. 17 Recently, the potential of proteasome inhibitor bortezomib in treating patients with rituximab-resistant PLA2R-associated MN has been reported. 18 It suggested that plasma cells also produce autoantibodies against podocyte antigens. However, the kidneys may not be main location of IgG4 production because IgG4-positive interstitial cells are rare even in MN with IgG4-positive staining in the glomeruli. Future studies to reveal which cell-types produce autoantibodies against podocyte antigens and where they produce these antibodies are warranted. It should be mentioned that our case had high levels of IgG4 in the serum; thus, the increased number of IgG4-positive cells in the interstitium might just reflect its elevation in the serum.
It was difficult to determine the extent of disease activity of MN with IgG4-RKD in the current case. In general, the response to steroid treatment of TIN with IgG4-RKD was fast and good, 19 but that of MN was diverse and the information was limited (Table 2). Conversely, steroid monotherapy for MN is not a recommended option in guidelines from Kidney Disease: Improving Global Outcomes. 20 However, it has been reported that steroid monotherapy in Japanese patients with MN showed relatively good response compared to that in patients in other countries. 21 In addition, our previous cohort study in Japanese patients demonstrated that the response of immunosuppressive therapy in the patients with PLA2R-associated MN was good (the rate of remission of proteinuria 6 months and 1 year after the initiation of treatment was approximately 80% and 90%, respectively [unpublished data]). Therefore, steroid monotherapy was chosen as the initial treatment in the current case. The differences of treatment response between PLA2R-associated MN and MN with IgG4-RD should be further examined.
Summary of reported cases of MN with IgG4-RKD.

A rare case of IgG4-RD with PLA2R-associated MN has been reported. 10 The patient did not have IgG4-RKD, but had serum anti-PLA2R antibody with immunostaining of IgG subclasses showing IgG1 predominance in the glomeruli. In contrast, IgG4 in the glomeruli was often positive, and was sometimes predominant in patients with MN accompanied by IgG4-RKD.3,7 The IgG4 positivity/predominance in the glomeruli in these patients could be explained by 2 possibilities. First, there might be IgG4-predominant autoantibodies other than anti-PLA2R antibody in the patients with MN as IgG4-RKD. So far, IgG3 subclass of anti-superoxide dismutase 2 antibody has been detected in a patient of MN accompanied by IgG4-RKD, 22 but IgG4 subclass of autoantibody associated with IgG4-RKD has not been discovered yet. Although THSD7A might be the target antigen in MN accompanied by IgG4-RKD, there have been no such reports. Second, there might be misinterpretation of IgG4 positivity in MN. It could be possible to differentiate between primary and secondary MN if the staining condition of the anti-IgG4 antibody is well-established. However, a small amount of IgG4 subclass of autoantibody might be detected by highly sensitive anti-IgG4 antibodies or highly sensitive detection methods even in secondary MN. In fact, it has been reported that immunofluorescence for IgG4 in the glomeruli was positive in renal specimens of many patients with lupus MN, a typical form of secondary MN. 23 In addition, in our previous cohort study, IgG4 in glomeruli was positive in the patients with lupus MN by immunofluorescence of cryosection, but was negative by immunohistochemistry of formalin-fixed sections from the same patients, even though the anti-IgG4 antibody was same (unpublished data). Recently, new target antigens of MN such as Neural epidermal growth factor-like 1 protein, Semaphorin 3B, Protocadherin 7, Neural cell adhesion molecule 1, and High temperature recombinant protein A1 were discovered by the combination of laser microdissection and tandem mass spectrometry.24-28 In addition, Exostosin 1 and Exostosin 2 were found in autoimmune type of secondary MN patients. 29 It is possible that these markers are associated with MN in the setting of IgG4-RKD.
This report has 2 limitations. First, we could not assess the IgG4+/IgG+ plasma cell ratio, which is a criterion of the new classification criteria for IgG4-RD published from ACCR/EULAR, 16 due to the lack of remaining renal specimens. Second, we could not identify which cell types were IgG4-positive infiltrating cells in the interstitium.
Conclusions
We described a case of serologically and histologically confirmed PLA2R-associated MN with IgG4+ cell infiltration into the interstitium without any signs of IgG4-RD. As serum anti-PLA2R autoantibody might present in MN patients with IgG4-positive cell infiltration in the interstitium as well as IgG4-RKD, the measurement of serum antibody level should be considered in MN patients with IgG4-positive cells infiltration in the glomeruli and interstitium.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
K.I. prepared the abstract, case presentation, Table 1, and figures. T. I. prepared the remaining parts of manuscript, performed immunohistochemistry, and revised the abstract/case presentation/tables and figures. C.E. collected and examined the data of immunohistochemistry for IgG4 staining. T.M. performed immunofluorescence. H.F., D.N., Y.U., M.K., H.Y., and R.F. reviewed and corrected the manuscript.
Ethical Statement
Written informed consent for patient information to be published was provided by the patient.
