Abstract
Background:
Quantification of the M-type phospholipase A2 receptor antibodies (anti-PLA2R) is now an essential tool for diagnosis and management of primary membranous nephropathy (MN). Since October 2018, Hôpital Maisonneuve-Rosemont (HMR) has been designated as Quebec’s reference center for serum anti-PLA2R antibody testing by the Institut National d’Excellence en Santé et Services Sociaux (INESSS), the regulatory body on drugs and tests usage in Quebec.
Objectives:
To describe the 2-step method of serum qualitative and quantitative anti-PLA2R antibody testing during its first year of use in Quebec and analyze its diagnostic value in the province’s population.
Design:
Retrospective cohort study.
Setting:
Single-center academic teaching hospital in Quebec, Canada.
Patients:
All patients who had a serum anti-PLA2R antibody test analyzed at HMR from October 1, 2018, to October 1, 2019, were included in the study.
Measurements:
Serum anti-PLA2R antibodies were screened by indirect immunofluorescence tests. If results were positive or undetermined, it was followed by a quantitative enzyme-linked immunosorbent assay (ELISA) test. Both tests were based on a commercial kit developed by the same company.
Methods:
We calculated sensitivity, specificity, predictive value, and likelihood ratio for both tests, using kidney biopsy findings performed at HMR as the gold standard.
Results:
In Quebec, a total of 1690 tests were performed among 1025 patients during the study year. A small proportion of these patients (8%) were followed at HMR. Patients tested at HMR and in the rest of Quebec had similar characteristics. Test validity was only characterized for patients tested at HMR. Sensitivity and specificity were, respectively, 58% and 100% for the qualitative test, and 71% and 100% for the quantitative test. The combined net sensitivity was 42% and the net specificity 100%. The net positive and negative predictive value were 100% and 84% respectively, whereas the net negative likelihood ratio was 0.58.
Limitations:
As the detailed analysis was only possible in the small proportion of patients clinically followed at HMR, there is a possible selection bias. Another potential selection bias was the focus on patients who were selected to have a kidney biopsy, probably because of more severe disease, higher probability of glomerulonephritis, or lesser number of comorbidities. Given the retrospective nature of this study, there was no systematic kidney biopsy or serum PLA2R antibody testing performed. Finally, we were unable to provide detailed information on the timing between immunosuppressive therapy and anti-PLA2R results.
Conclusions:
Serum anti-PLA2R antibody testing was widely used in Quebec during its first year of availability. A 2-step approach, using a qualitative test first, followed by a quantitative test if the results are positive or undetermined, appears efficient to avoid useless quantitative testing in negative patients and to better characterize undetermined results on immunofluorescence.
Trial registration:
Due to the retrospective nature of this study, no trial registration was performed.
Introduction
Primary membranous nephropathy (MN) is a leading cause of nephrotic syndrome in the adult Caucasian population worldwide, representing around 20% to 30% of cases. 1 It is characterized by accumulation of immune deposits on the outer aspect of the glomerular basement membrane. The clinical presentation and natural history of MN are highly variable, with up to one third of patients entering remission spontaneously without treatment, 2 but many others experiencing progressive decline in kidney function, even with appropriate treatment. 3 The understanding of the pathophysiology and treatment of MN has advanced considerably in the last decade, in particular since the discovery by Beck and colleagues that M-type phospholipase A2 receptor antibodies (anti-PLA2R) are implicated in about 70% to 80% of cases of primary MN. 4 The PLA2R antigen is expressed on podocytes where it serves as a target for autoantibodies.
Since its original description, serum anti-PLA2R antibody testing has been validated in several cohorts of MN patients. The pooled sensitivity and specificity were 65% and 97%, respectively, in a recent meta-analysis. 5 It is now well recognized as a diagnostic tool for MN, and a serology-based approach to MN diagnosis has been proposed in the literature. 6 Acknowledging this body of literature, the Institut National d’Excellence en Santé et Services Sociaux (INESSS), Quebec’s regulatory body on drugs and tests usage, approved serum anti-PLA2R antibody testing at Hôpital Maisonneuve-Rosemont (HMR) in October 2017.
The objective of the present study was to provide, for the population of Quebec, a 1-year assessment of serum anti-PLA2R antibody testing. This study also aimed to compare results from HMR patients with those from the rest of the province and to analyze the diagnostic value of this test using patients who underwent a kidney biopsy at HMR.
Methods
Study Design
This study is a retrospective cohort study of all the serum anti-PLA2R antibody tests performed at HMR for the first year after the inception of the test (October 1, 2018, to October 1, 2019). The HMR is the only reference center for serum anti-PLA2R antibody testing in the province of Quebec and performed all tests in the province since October 1, 2018. The study was approved by our institution’s Research Ethics Committee, in agreement with the Declaration of Helsinki.
Data Collection
Data were collected for 2 distinct cohorts. First, data for all patients followed in other hospitals who had a serum anti-PLA2R antibody test analyzed at HMR were collected in the central laboratory database. Data collection was minimal: age, sex, site of test prescription, number of tests performed, and each test’s results. Second, for patients with both serum anti-PLA2R antibody testing and nephrology follow-up at HMR, more detailed characteristics were obtained from their medical records (demographics, comorbidities, medications, laboratory results, and kidney biopsy results, including PLA2R staining). This cohort was used to determine the test validity.
PLA2R Testing
Serum anti-PLA2R antibody from all submitted samples was initially screened by indirect immunofluorescence tests (IIFT) (EUROIMMUN, Lübeck, Germany) according to the manufacturer’s instructions. If this qualitative test was positive or undetermined, a quantitative test was performed (Figure 1). Quantitative serum anti-PLA2R antibody was measured by enzyme-linked immunosorbent assay (ELISA) test using previously validated commercial kit developed by the same company as the IIFT. Quantitative tests were considered negative with a value of <14 relative units (RU)/mL, weakly positive between 14 and 19.9 RU/mL, and positive if >20 RU/mL.
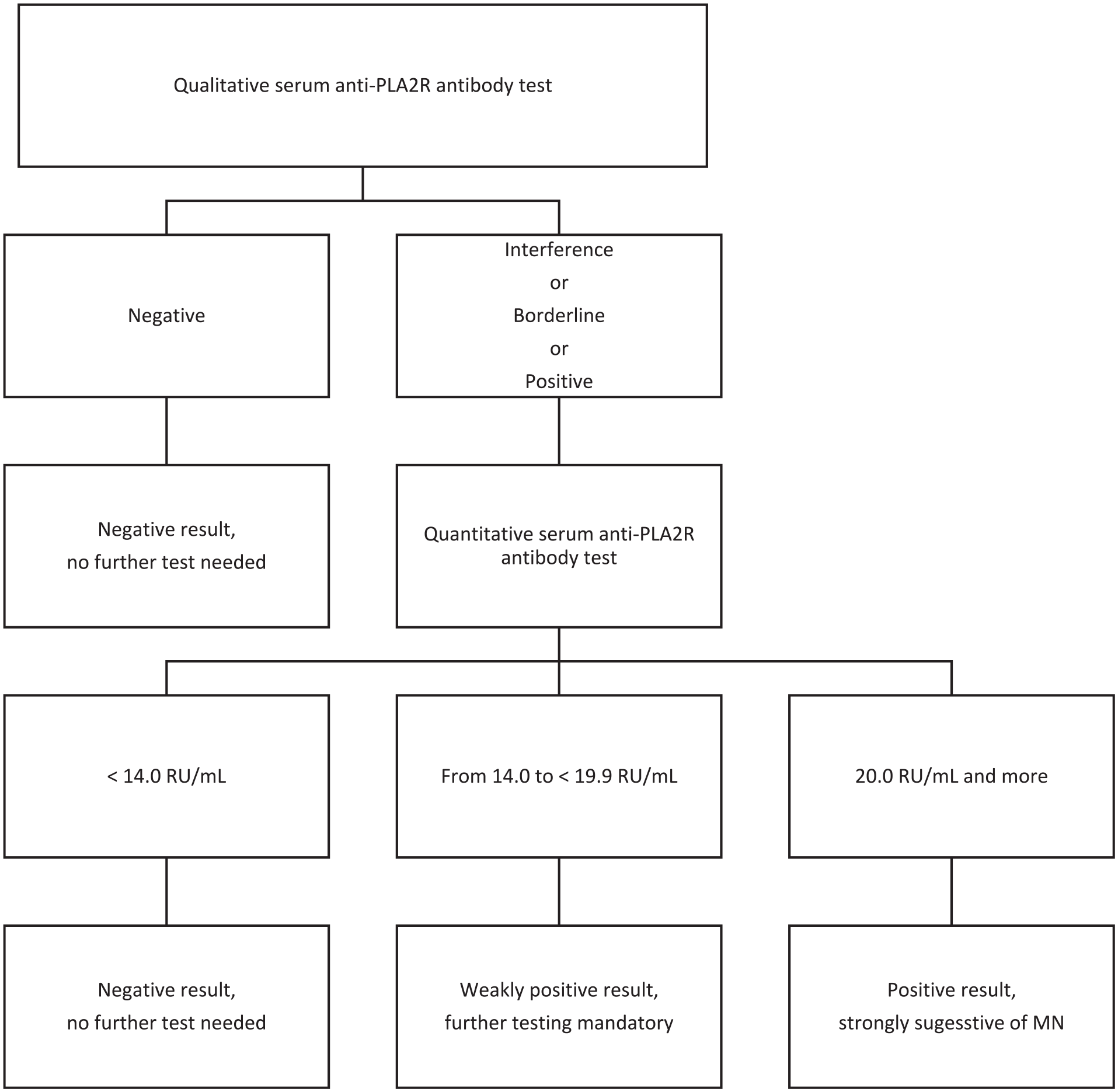
Serum Anti-PLA2R antibody testing algorithm.
The PLA2R immunohistochemistry on kidney biopsy was performed on 2-μm-thick, formalin-fixed, paraffin-embedded tissue sections mounted on charged slides. Tissue slides were dried and melted in an oven at 56°C for 60 minutes. Slides were stained with an anti-PLA2R rabbit antibody (catalog no. HPA012657; 1/500 titer; Sigma-Aldrich, Darmstadt, Germany) on a Ventana BenchMark ULTRA system (Roche, Rotkreuz, Switzerland). The staining protocol included online deparaffinization, heat-induced epitope retrieval with Ventana Cell Conditioning 1 solution (CC1) for 24 minutes, and incubation with the primary antibody for 8 minutes at 35°C. Antigen-antibody reactions were visualized using Ventana OptiView Universal DAB Detection Kit (Roche, Rotkreuz, Switzerland). Staining was interpreted as positive when a granular pattern, coarse and/or fine, diffuse or focal, and global or segmental was observed.
Covariate Definitions
Every serum anti-PLA2R antibody test was classified according to their referring hospital. Teaching hospitals were defined as the main tertiary or quaternary university-affiliated hospitals in Quebec. Community hospitals were defined as primary or secondary hospitals operating mostly without medical students and residents. For tests performed at HMR, we were able to differentiate outpatient serum anti-PLA2R antibody tests, prescribed in the nephrology clinic, and inpatient serum anti-PLA2R antibody tests, prescribed on a hospital ward. Chronic kidney disease (CKD) was evaluated with the estimated glomerular filtration rate (eGFR) level, calculated with Chronic Kidney Disease–Epidemiology Collaboration (CKD-EPI) equation. 7 The severity of CKD was classified according to the Kidney Disease: Improving Global Outcome (KDIGO) CKD guidelines. 8
Statistical Analysis
Age, sex, and test results were first compared between HMR patients and the rest of the province. The characteristics of HMR patients were stratified based on their qualitative serum anti-PLA2R antibody test result. For HMR patients for whom kidney biopsy results were available, sensitivity, specificity, predictive value, and likelihood ratio were calculated for both qualitative and quantitative serum anti-PLA2R antibody tests. We also calculated the net sensitivity and net specificity of using both tests in sequence. Of note, patients who showed interference on the first qualitative test were considered positive and tested further with the quantitative test. PLA2R staining on kidney biopsy was used as the gold standard to determine the validity of the serum testing. Baseline categorical variables were analyzed with Fisher and χ2 tests, and a Kruskal-Wallis test was used with continuous variables. A 2-sided P value of <.05 was considered statistically significant. All statistical analyses were performed using SAS 9.4 (SAS Institute, Cary, North Carolina).
Results
Prescription Settings
During the first year of serum anti-PLA2R antibody testing, 1025 patients were tested, 79 (8%) of which were followed at HMR (Table 1). A total of 1690 serum anti-PLA2R antibody tests were performed on those patients. A majority of those tests (58%) were prescribed at teaching hospitals (vs 41% in community hospital). The top 10 prescribing hospitals were in highly populated areas and/or were considered reference centers with specialized glomerulonephritis or transplant clinics (Figure 2). Among the tests prescribed at HMR, we were able to determine that 81% were prescribed from the outpatient clinic, the rest being prescribed from the emergency room or the hospital wards.
Comparison of Patient Characteristics and Serum Anti-PLA2R Antibody Results Between HMR and Other Hospitals in Quebec.
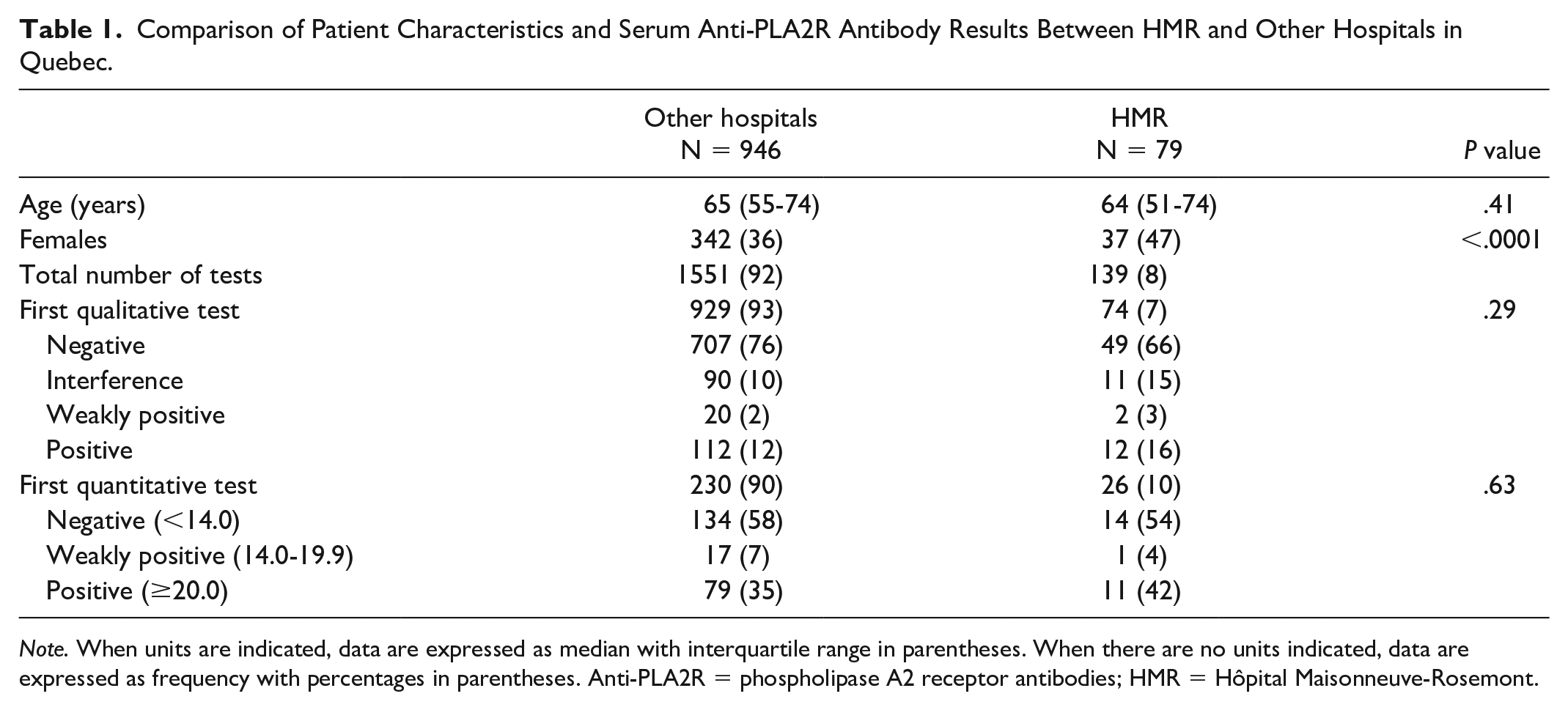
Note. When units are indicated, data are expressed as median with interquartile range in parentheses. When there are no units indicated, data are expressed as frequency with percentages in parentheses. Anti-PLA2R = phospholipase A2 receptor antibodies; HMR = Hôpital Maisonneuve-Rosemont.

Serum anti-PLA2R antibody testing by prescribing center across Quebec.
Comparison Between HMR and the Rest of the Province
Patients being tested for serum antibodies at HMR had a similar median age than those tested in the rest of Quebec (65 [interquartile range (IQR) = 55-74] vs 64 [IQR = 51-74] years old for HMR; P = .41). There were fewer females tested in the rest of Quebec compared with HMR (36% vs 47%; P < .01). Regarding both qualitative and quantitative serum anti-PLA2R antibody tests, the rate of negative or positive results was similar between the province and HMR.
HMR Patient Characteristics by Serum Anti-PLA2R Status
Among the patients who were tested and followed at HMR, we were able to analyze the differences between patients with negative, undetermined, and positive results (Table 2) on qualitative testing. There were significantly fewer patients with diabetes in the group with positive results (only 1 patient [8%] was known for diabetes). There was no difference in CKD stage, use of renin-angiotensin-aldosterone system blockade, and previous immunosuppressant use (received in a 3-month window before and after testing) among all groups. Laboratory parameters were similar in all groups, except from serum albumin which was significantly lower in the group with positive serum anti-PLA2R antibody testing. More patients in the positive group had a kidney biopsy compared with the other groups (Table 3). A glomerular PLA2R staining was performed in 82% of those biopsies and was positive 78% of the time. Results from the serum and glomerular anti-PLA2R tests were concordant (both positive and both negative) on average in 75% of cases in all groups. Most patients (68%) had their kidney biopsy performed the same year than the anti-PLA2R antibody testing, and 29% the year before.
Hôpital Maisonneuve-Rosemont Patient Characteristics by Serum Anti-PLA2R Antibody Status.

Note. When units are indicated, data are expressed as median with interquartile range in parenthesis. When there are no units indicated, data are expressed as frequency with percentages in parentheses. eGFR, estimated glomerular filtration rate using CKD-EPI equation; CKD, chronic kidney disease, staged according to KDIGO guidelines; presence of hematuria is defined as more than 2 red blood cells per high-power field; RBC, red blood cells; HPF, high-power field. Data are complete except for the following entries: CKD stage (11 missing), albumin (2 missing), albumin-to-creatinine ratio (19 missing), protein-to-creatinine ratio (14 missing), 24-hour urine collection (59 missing), and hematuria (5 missing). Anti-PLA2R = phospholipase A2 receptor antibodies; KDIGO = Kidney Disease: Improving Global Outcome.
PLA2R Glomerular Staining in Hôpital Maisonneuve-Rosemont Patients by Serum Anti-PLA2R Antibody Status.

Note. When units are indicated, data are expressed as median with interquartile range in parentheses. When there are no units indicated, data are expressed as frequency with percentages in parentheses. Anti-PLA2R is for anti M-type receptor of the phospholipase A2 receptor autoantibodies.
Validity Among the HMR Population
Kidney biopsies were performed for 38 of the 79 patients followed at HMR. This allowed us to define the validity and predictive value of serum anti-PLA2R antibody testing in HMR patients using PLA2R staining on kidney biopsy as the gold standard (Table 4). Sensitivity and specificity were, respectively, 58% and 100% for qualitative serum anti-PLA2R antibody testing, and 71% and 100%, respectively, for the quantitative one. The net sensitivity of the sequential testing was 42% and the net specificity 100% (see contingency tables in the Supplemental Material). The net positive and negative predictive value were 100% and 84%, respectively, whereas the net negative likelihood ratio was 0.58.
Parameters of the Serum Anti-PLA2R Antibody Testing at Hôpital Maisonneuve-Rosemont Using PLA2R Staining on Kidney Biopsy as a Gold Standard.

Note. Prevalence of membranous nephropathy was estimated at 25% for patient undergoing qualitative testing, as it is the prevalence of MN amongst patients presenting with nephrotic syndrome reported in the literature.1,9 Anti-PLA2R = phospholipase A2 receptor antibodies; MN = membranous nephropathy; CI = confidence interval.
Discussion
This study allowed us to depict the use of the serum anti-PLA2R antibody testing in its first year of availability in Quebec and evaluate the usefulness of the test in this province’s population. Many studies have shown the utility of serum anti-PLA2R antibody testing in the diagnosis and monitoring of MN. 10 The HMR is the only center in Quebec that performs serum anti-PLA2R antibody testing. It uses a 2-step approach with qualitative IIFT testing first, followed by quantitative ELISA testing if the qualitative test is either positive or undetermined. This approach has the advantage of avoiding quantification for all patients and of precisely confirming positivity with a titer. Moreover, the titer might be useful for patient follow-up after diagnosis and treatment. This stepwise approach also allows a characterization of undetermined results with interference on IIFT. Of note, before its implantation in Quebec, only qualitative testing was available in the province (but performed in Alberta).
Serum anti-PLA2R antibody testing was used across all the province of Quebec, in many different centers, from teaching to community hospitals and regional clinics. This demonstrates a widespread use of serum anti-PLA2R antibody testing across nephrologists in Quebec in the management of MN. Furthermore, the testing rate and incidence of MN measured with the 2018 Quebec population (8,388 million) were consistent with the published incidence previously reported in the literature and demonstrated appropriate test utilization 11 : There were 201 tests per million and 11 new cases per million.
Patients with positive serum anti-PLA2R antibody results followed at HMR were less likely to have diabetes (compared with negative anti-PLA2R results), suggesting diabetic patients had higher risk of another disease than MN, such as diabetic nephropathy. The serum albumin was lower in patients with positive results, likely related to a more severe nephrotic syndrome associated with MN diagnosis. Moreover, patients with negative test results had a higher probability of diabetic nephropathy and nephroangiosclerosis, 2 diseases less associated with very low serum albumin.
There is equipoise as to whether patients with positive anti-PLA2R test should undergo a kidney biopsy. 6 Patients with positive serum anti-PLA2R antibody testing at HMR, however, underwent a kidney biopsy more often than patients with negative results. Several reasons could explain such findings. First, negative PLA2R test, along with a negative glomerulonephritis workup, might have convinced clinicians that a biopsy was futile. Second, nephrologists might have ordered serum anti-PLA2R antibody testing when there was a high probability of an alternate diagnosis (eg, diabetic nephropathy), and were reassured by a negative test result. Finally, many nephrologists still offer a kidney biopsy to patients with a positive serum anti-PLA2R antibody, as they consider the biopsy gives prognostic information essential to initiate immunosuppressive treatment. Other reasons to perform kidney biopsy despite positive testing include the need to assess the degree of chronicity in patients with impaired kidney function or suspicion of a secondary process.
Among patients who had a biopsy with PLA2R glomerular staining, results were concordant in about 75% of cases with serological testing. Thus, in a nonnegligible proportion of cases, a negative serological testing was followed by a positive staining on renal biopsy, a finding very useful to clinicians. This scenario denotes a quiescent MN, as there are no antibodies in circulation, but the antigen expressed in the kidney characterizes previous disease activity.
Using the kidney biopsy results as gold standard, we were able to assess serum anti-PLA2R antibody testing parameters in the HMR population. All patients who tested positive or undetermined for the qualitative test and had a kidney biopsy were diagnosed with MN. The net sensitivity of the qualitative and quantitative serum anti-PLA2R antibody testing in our cohort (42%) was lower than in the literature, 11 but the net specificity (100%) was excellent. This might be explained by the fact that patients with known MN having serum anti-PLA2R antibody testing for their follow-up were included in the study. Such patients could have a recent diagnosis of MN on a kidney biopsy and a negative follow-up serum anti-PLA2R antibody serology as they entered in remission after receiving immunosuppressive therapy. Their first positive titer might have been quantified before the start of serum anti-PLA2R antibody testing in Quebec, and thus outside of the study period, and unavailable to us. Given this lower net sensitivity, kidney biopsy is necessary in patients with high suspicion of MN. The sensitivity of our quantitative test, using a cutoff of 14.0 RU/mL and greater, was 71%, which is concordant to what was previously described in the literature. 12 Interestingly, using a cutoff of 2.0 RU/mL as advocated by some authors, 12 the sensitivity would be 100%, with a specificity of 100%. This cutoff change would have improved the net sensitivity to 58% while maintaining the net specificity at 100%. In general, lowering the cutoff of a quantitative test hinders its specificity; however, applying the HMR algorithm with a lower cutoff would have improved net sensitivity while having no effect on the net specificity.
Our study has some important strengths. First, this is one of the largest cohorts of serum anti-PLA2R antibody testing reported in the literature. It is also of value for nephrologists across Quebec and the rest of Canada, as it represents a real-life sample of our population as all patients having serum anti-PLA2R antibody testing during the year of study were included, without exclusion. Also, HMR is one of the rare centers internationally that performs a sequential approach with a qualitative IIFT test first before a quantitative ELISA test. This diagnostic algorithm allows a better characterization of undetermined results on IIFT and the possibility to observe the antibody titer evolving over time. As the test kits used at HMR are the main commercially available assays for anti-PLA2R measurement, and the only one approved in Canada, our data can be compared with other quantitative studies.
Unfortunately, our study also has several limitations. Information about most patients who had their serum anti-PLA2R antibody test while being followed somewhere else than HMR were minimal. Hence, only HMR patients were included in the most detailed analysis, introducing a potential selection bias. However, HMR is a center that not only attends to a very populous and diversified region of Montreal but also acts as a reference center for many surrounding hospitals and regions, which enhances the generalizability of our findings. In addition, the known characteristics of HMR patients were very similar to those of other centers, implying that those 2 groups are somewhat similar. The group of patients at HMR was much smaller, reducing the sample size. Another potential selection bias was the focus on patients who were selected to have a kidney biopsy, probably because of more severe disease, higher probability of glomerulonephritis, or lesser number of comorbidities. Given the retrospective nature of our study, we were not able to ask for a mandatory biopsy or serum PLA2R antibody testing for more patients, as it was at the physician’s discretion. Finally, we were unable to provide detailed information on the timing between immunosuppressive therapy and anti-PLA2R results.
Conclusions
In conclusion, serum anti-PLA2R antibody testing was diffusely used at an appropriate rate in Quebec during its first year of availability at HMR. A 2-step approach, using a qualitative test first and a quantitative test afterward if results are positive or undetermined on IIFT, combined with a cutoff of at least 2.0 to 14.0 RU/mL, seems like an efficient approach to avoid using quantitative testing in negative patients and to optimize testing specificity.
Supplemental Material
sj-pdf-1-cjk-10.1177_20543581211052729 – Supplemental material for Qualitative and Quantitative Dosage of the Anti M-Type Phospholipase A2 Receptor Autoantibody: One-Year Experience in Quebec’s Reference Center
Supplemental material, sj-pdf-1-cjk-10.1177_20543581211052729 for Qualitative and Quantitative Dosage of the Anti M-Type Phospholipase A2 Receptor Autoantibody: One-Year Experience in Quebec’s Reference Center by Simon Leclerc, Karim Benkirane, Annie-Claire Nadeau-Fredette, Naoual Elftouh, Jean-Philippe Lafrance, Vincent Pichette, Virginie Royal, Caroline Lamarche and Louis-Philippe Laurin in Canadian Journal of Kidney Health and Disease
Footnotes
Acknowledgements
We would like to thank Nathalie Samson for her help with data collection. All expenses related to this study were paid by the Division of Nephrology, Hôpital Maisonneuve-Rosemont. L.-P.L. is a Fonds de recherche du Québec-Santé Junior 2 Scholar. A.-C.N.-F. is a Fonds de recherche du Québec-Santé Junior 1 Scholar.
Ethics Approval and Consent to Participate
Data collection for this study was approved by CIUSSS-EMTL Hôpital Maisonneuve-Rosemont Research Ethics Committe.
Consent for Publication
All authors have provided consent for publication.
Availability of Data and Materials
The datasets used and/or analyzed for this study are not available upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
