Abstract
One of the solutions to decrease the frequency of sudden cardiac arrest (SCA) in athletes is cardiovascular screening in the form of pre-participation tests (PPT). It is especially frustrating when SCA affects an athlete who has recently undergone PPT without any alarming findings. In our case, a 44-year-old ambitious amateur duathlete who underwent PPT within a year experienced SCA at home after an intensive weekend of training. It was related to a coincidence of new disease and other risk factors. PPT cannot be viewed as an unconditional green light for training and competition.
Keywords
Introduction
Prevention of sudden cardiac arrest (SCA) is the foundation of sports cardiology. Despite being relatively infrequent events, SCAs in athletes are always a dramatic circumstance for otherwise healthy and fit people pursuing good lifestyle and to their families. They also draw a huge media attraction creating a false picture of detrimental effects of physical activity. 1 One of the solutions to decrease the frequency of SCA in athletes is cardiovascular screening in the form of pre-participation testing (PPT). 1 While these are mandatory in competitive or professional (mainly youth) athletes in some countries (eg. Poland, Italy), they are only sporadically required in master, amateur athletes. There is also none recommended set of tests to perform. 1 The simplest and cheapest variant of PPT include medical history and physical examination, more elaborated ones include resting electrocardiogram (ECG), transthoracic echocardiography (TTE), and exercise testing. 1 Unfortunately, there is no single diagnostic test in cardiology capable of identifying all potential cardiac causes of SCA. Each component of the PPT targets specific aspects of cardiovascular pathology. Among these, resting ECG remains the most diagnostically valuable tool when considering the balance between diagnostic accuracy, accessibility, and cost-effectiveness. 1
Recent studies show that the frequency of SCA in athletes remains stable across years, but there is an improved survival of those suffering from SCA due to more frequent use of automated external defibrillators (AEDs) leading to overall lower sudden cardiac death rate (SCD).2 -4 The decision to undergo PPT is left to master athletes themselves, which may be costly and logistically demanding. For these reasons it is especially frustrating when SCA affects an athlete who has recently undergone extensive PPT without any alarming findings. It is instructive for future decisions making to understand what might have been the cause of SCA despite obtaining green light for competitive sport participation by the athlete. Was it related to a human error, false negative PPT results, new onset of a disease which was not present during PPT or a rare coincidence of multiple factors, which individually would rarely lead to SCA? We present a case of an asymptomatic middle-age competitive duathlete who suffered SCA within a year from negative PPT.
Case
A 44-year-old man suffered a sudden cardiac arrest unaccompanied by earlier symptoms during working from home while sitting in the chair on Monday late afternoon. His wife called 112 and started cardiopulmonary resuscitation (CPR). When paramedics arrived, he was still unresponsive with ECG trace showing torsade de pointes. Sinus rhythm was restored with 200 J defibrillation (Figure 1), however he required 4 more shocks in the hospital emergency department. Gas analysis showed hypokalaemia of 2.84 mmol/l and the patient was administered i.v. potassium, magnesium, and 300 mg of amiodarone, which led to permanent sinus rhythm restoration after the fifth 200 J shock. ECG in the hospital showed mild upslope ST-segment depression up to 0.5 mm in leads V4 to V6 with QTc of 406 milliseconds. Coronary angiography performed immediately did not disclose the presence of significant atherosclerotic changes. Contrary, TTE demonstrated generalized, but asymmetric left ventricular hypertrophy (LVH) up to 15 mm in the basal antero-septal segment of the left ventricle with mild left ventricular outflow tract (LVOT) obstruction up to 26 mmHg. The left ventricular parameters were – left ventricular diastolic dimension (LVDd) – 52 mm, left ventricular ejection fraction (LVEF) – 65%, and left atrial volume index (LAVI) of 37 ml/m2. There were no signs typical for infiltrative diseases on CMR and the patient denied the use of anabolic steroids. Transoesophageal echocardiography (TEE) excluded the presence of a subaortic membrane and confirmed the existence of mild resting LVOT obstruction.
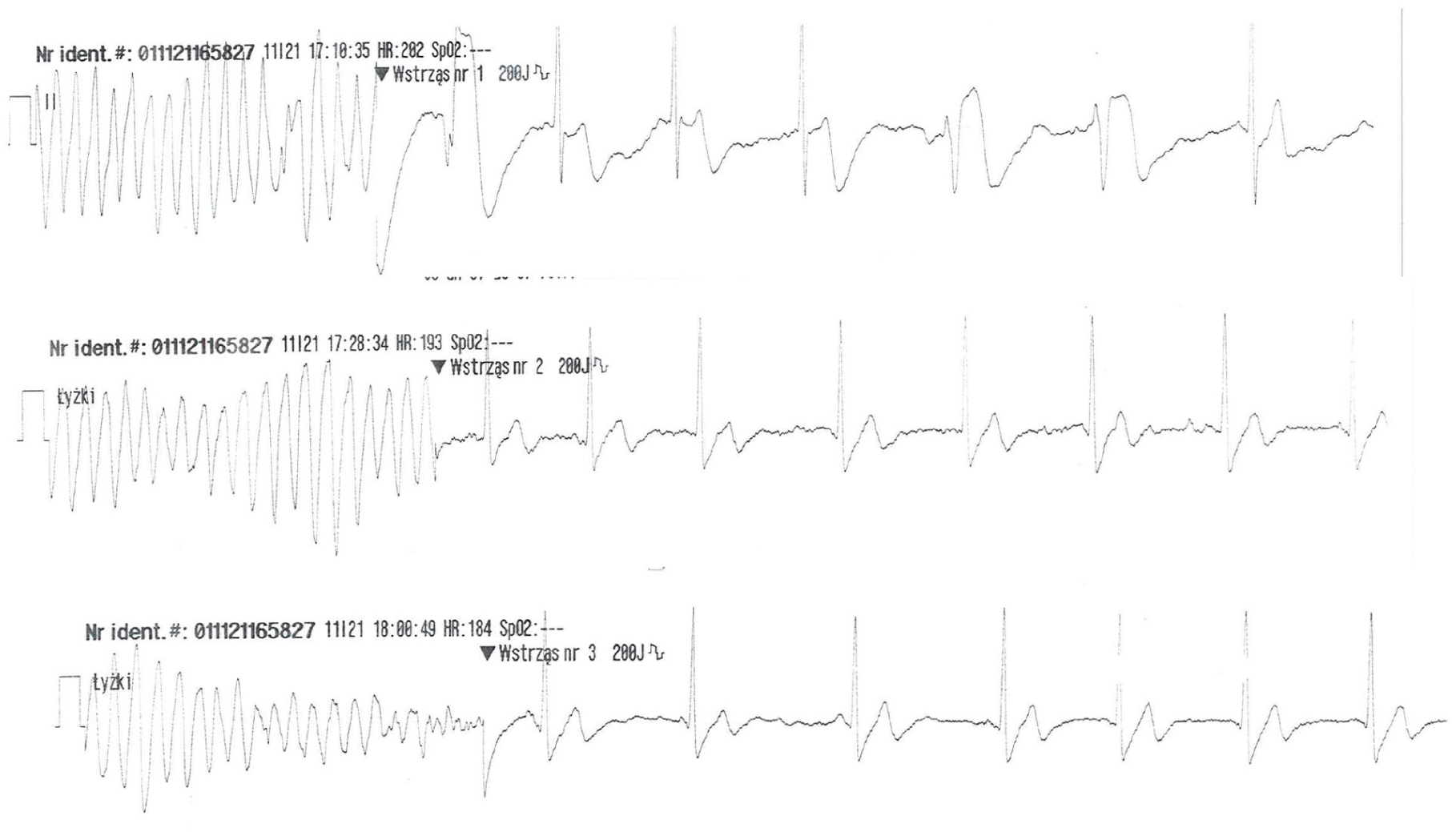
First 3 runs of torsade de pointes in the patient in 50 minutes with sinus rhythm return after 200 J defibrillation shocks.
During hospital stay the patient remained in a stable clinical condition with stable heart rate and blood pressure and absence of neurological deficits. Holter monitoring revealed only mild ventricular arrhythmia (up to around 100 single ventricular extrasystoles per day). Except initial hypokalemia, which was not present in previous or following laboratory tests, other laboratory tests, including D-dimers and troponin were unremarkable. Cardiac magnetic resonance (Figure 2) confirmed the presence of LVH between 13 and 16 mm in the basal interventricular septum with LVOT obstruction and the presence of non-ischemic late gadolinium enhancement (LGE) in the head of the posterior papillary muscle (less than 3% of the left ventricular mass), which led to the diagnosis of a hypertrophic obstructive cardiomyopathy (HOCM). A test with adenosine did not demonstrate the presence of an accessory pathway. Finally, the patient underwent successful subcutaneous implantable cardioverter defibrillator (S-ICD) insertion in the secondary prevention of SCD.
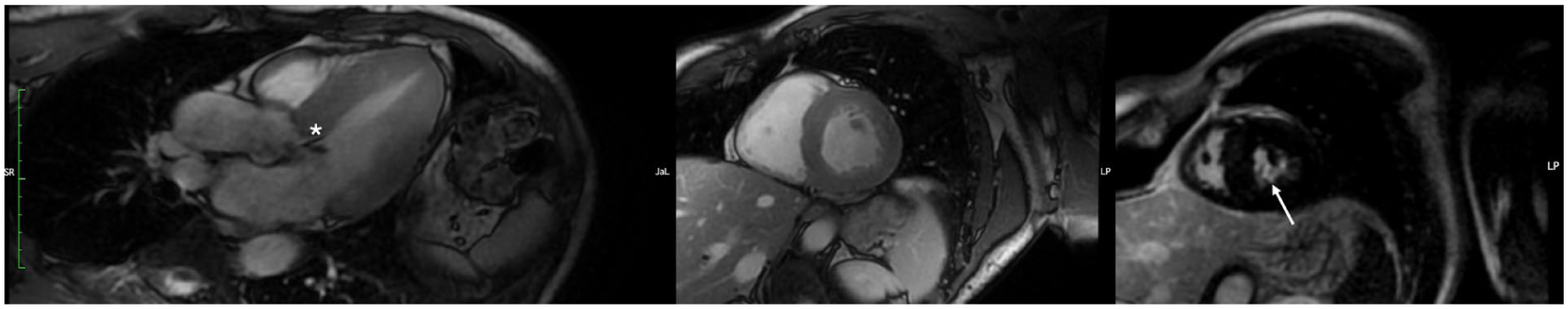
Cardiac magnetic resonance images. Left: cine end-systole image in 3-chamber view with visible left ventricular outflow tract obstruction (asterisk). Middle: cine end-diastole short-axis view showing mild left ventricular hypertrophy. Right: late gadolinium enhancement (LGE) image in short-axis showing small non-ischemic LGE in the head of the posterior papillary muscle (arrow).
This amateur athlete has started to be engaged in intensive sport trainings about 8 years prior to the index event with training volume of about 10 hours per week. He underwent PPT less than a year earlier before the start of the season, which disclosed absence of family history of sudden cardiac death (SCD), absence of symptoms, and the presence of a mild hypertension on physical examination. At that time ECG showed sinus rhythm 56 beats per minute (bpm) without any pathological findings. There were no signs of left ventricular hypertrophy or HCM on ECG. Similar ECG pattern was found at discharge from the hospital (Figure 3). Echocardiogram performed at the time of initial screening showed mild LVH up to 12 to 13 mm without LVOT obstruction, which could be easily explained by intensive athletic training and an untreated hypertension. The left ventricular dimensions were – LVDd – 51 mm and LVEF – 65% with LAVI – 25 ml/m2. He received 40 mg telmisartan with good effect and normalization of blood pressure (BP), subsequently changed to ramipril 2.5 mg in the hospital. Holter ECG at that time demonstrated the presence of only 11 ventricular and 23 supraventricular monomorphic premature contractions without severe forms of arrhythmia. He was advised to supplement electrolytes. Patient’s pre-season cardiopulmonary exercise test (CPET) at that time showed V02 max of 56 ml/minute/kg (a slight drop from 59-60 ml/minute/kg in previous 2 years).

Initial electrocardiogram performed within a year before the index event (left) and at discharge from the hospital showing no pathological findings (right).
In a year between the PPT and SCA the athlete completed several starts, ran a total of 1400 km and cycled 13 000 km without symptoms. Two months before SCA he experienced Covid-19 (in late 2020), only to finish marathon 2 months later slightly below 3 hours and 30 minutes. On weekend preceding the event he completed a 3-hour cycling training on Saturday and a 2.5-hour running session on Sunday once more without symptoms. He admitted omitting electrolyte supplementation after trainings on those 2 days. He also stated that Monday morning was very stressful, but again asymptomatic. He denied the use of doping, alcohol, and was a non-smoker with good body weight.
Further tests after the hospital discharge revealed dynamic raise of LVOT obstruction on stress TTE to 105 mmHg during maximal exercise with normal BP response to exercise (137/90 to 186/86 mmHg) and lack of symptoms. There was no significant arrhythmia during exercise. The patient was offered a beta-blocker which he did not tolerate. Due to his good physical fitness and only mild LVH other forms of LVOT obstruction reduction were not considered. Genetic diagnosis of HCM using commercially available Illumina TruSight Cardiomyopathy Sequencing Panel including exonic regions of 174 cardiomyopathy genes did not reveal the presence of any pathological mutations. Examination of first-degree relatives did not demonstrate any signs of HCM.
His S-ICD was set to intervene at 240 bpm and he was gradually allowed to return to moderate training. He remained asymptomatic with no S-ICD interventions during almost 4 years of follow-up. He trains for several hours a week with heart rate up to 150 bpm for 2 hours cycling at 200 W or up to 130 to 135 bpm during longer rides with lower power. Occasionally he runs fast with heart rate up to 170 bpm. His follow-up includes annual S-ICD control, resting ECG, TTE, and periodic exercise test, which remain stable. The only problem brought by the patient is a post-traumatic stress with any unpleasant feelings in the body triggering anxiety necessitating continuous psychiatric and psychological help. A timeline of events is presented in Figure 4.

Timeline of events.
Discussion
The “rule of 7” in the context of plane crashes refers to a pattern where accidents often involve a series of 7 consecutive errors or failures. This idea is not a formal rule but a concept discussed by Malcolm Gladwell in his book “Outliers.” The “rule of 7” suggests that plane crashes are rarely caused by a single event, but rather by a chain reaction of smaller, often overlooked, mistakes, or lapses. It may be similar with SCA in athletes. Most of them perform hundreds of exercise sessions and participate in numerous competitive events without symptoms before the SCA, which may be a standalone, once in a lifetime incident. In our athlete the event might have been caused by a coincidence of at least several factors listed in a random order: (1) hypertrophic cardiomyopathy, (2) left ventricular outflow tract obstruction, (3) hypokalemia/electrolyte imbalance, (4) life stress, (5) overtraining, (6) male sex, (7) master sport category (age above 40), (8) recent COVID-19 infection, (9) hypertension. Our case can also be explained by a Swiss Cheese model of accident causation, which illustrates that, although many layers of defense lie between hazards and accidents (slices of cheese), there are flaws in each layer (holes) that, if aligned, can allow the accident to occur.
While it cannot be fully proven the hypothetical sequel of events which led to the SCA in this patient included an intensive training season causing decreased immunity and leading to COVID-19 infection and exacerbation or unmasking of previously subclinical HCM mimicking mild left ventricular hypertrophy related to hypertension. 5 The risk was potentiated by LVOT obstruction putting an extra strain on the left ventricular muscle during exercise along with a weekend of intensive training with electrolyte imbalance before the SCA and high psychological stress. Indeed, alterations in potassium concentrations, leading to hypo- or hyperkalemia, can be observed following sudden cardiac arrest (SCA). Hypokalemia may result from cellular shifts following the correction of acidosis, while hyperkalemia is often a consequence of cellular breakdown, ischemia, and renal injury following prolonged resuscitation efforts. These electrolyte imbalances are important considerations in the post-resuscitation management of SCA patients. Thankfully, patient’s life was saved due to prompt bystander CPR performed by his wife and prompt paramedics arrival with immediate defibrillation of the pulseless rhythm.
This case also demonstrates that under scrutinous monitoring, with adequately programmed S-ICD in place and with exercise intensity limits it is possible to return to sport without pharmacological treatment and intervention even in the case of HCM with dynamic LVOT obstruction. In fact, recent studies demonstrated that patients with HCM with or without LVOT obstruction can safely engage in moderate or even high-intensity exercise under adequate treatment and monitoring performed in the reference centers.6 -8 While the clinical trial is still ongoing, one registry reported that exercise was safe for athletes with an implanted S-ICD.9,10 It is also important to emphasize that management after SCA should involve psychological or psychiatric consultations for the survivors and their family members as these events often lead a significant psychological distress.
Conclusions
We presented a case of an ambitious master athlete in whom a coincidence of several factors contributed to the SCA. The case provides context for historical instances of negative PPT followed by subsequent SCA in athletes, which often can be viewed as once-in-a-lifetime events triggered by emergence of new risk factors not identified during PPT. Therefore, PPT cannot be regarded as an unequivocal indicator for unrestricted participation in training or competition. Ongoing surveillance and monitoring are equally essential. The case also demonstrates that after removal of precipitating and concomitant factors contributing to the SCA and with an ICD in place a return to sport is feasible without a significantly increased risk of recurrent life-threatening events.
Learning Objectives
Pre-participation testing, even in amateur master athletes, should be performed regularly, especially after events which can potentially contribute to change of the cardiovascular status. Despite that measures some events cannot be predicted. It always calls for reasonable training and competition and an easy access to automated external defibrillators (AED).
Electrolyte imbalance is an important and frequently undervalued factor contributing to SCA in patients with various cardiac conditions. This case demonstrates that often it is more than one risk factor leading to SCA and that SCA may be a rare coincidence of several accumulating risk factors.
Return to sport is possible after SCA in patients with HCM and LVOT obstruction with S-ICD in place and an adequate precautionary measure with close follow-up.
Footnotes
Consent for Publication
The patient and the patient’s family were informed that patient information and images would be submitted for publication and gave their consent.
Author Contributions
This manuscript has not been published and is not under consideration for publication elsewhere. All authors have (a) contributed substantively to the conception, design, or analysis and interpretation of the data, (b) contributed substantively to the drafting of the manuscript or critical revision for important intellectual content, (c) given final approval of the version to be published in Clinical Medicine Insights: Cardiology, and (d) agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
