Abstract
Background:
The effect of Sodium Glucose Co-Transporter 2 inhibitors on cardiovascular, renal, dyslipidemia, and inflammatory markers has not been analyzed simultaneously. The goal is to determine if SGLT2 inhibitors significantly reduce cardiovascular and renal mortality, and improve these health outcomes.
Methods:
PubMed, Cochrane Library and MEDLINE databases were used to conduct a comprehensive literature search from inception to September 2023. Randomized control trials with follow-up for at least 8 weeks, with a group taking SGLT2 inhibitors being compared with a group taking either placebo or other medication, in which cardiovascular outcomes, renal outcomes, lipid biomarkers, and inflammatory markers were reported as the primary outcomes were included. The statistical analyses were conducted using Review Manager with a random-effects model.
Results:
Thirteen studies comprising 92 920 patients were analyzed for several outcomes. The analysis revealed a significant reduction in overall mortality, death due to heart failure, cardiovascular causes, and renal causes in the SGLT2 inhibitor group as compared to placebo. Pooled results also revealed a significant reduction in the frequency of renal replacement therapy and renal composite endpoint in patients on SGLT2 inhibitors. There was also a significant reduction in IL-6, TNF-a, systolic blood pressure in the same group. On the other hand, Flow Mediated Dilation (FMD), lipid profile, and the incidence of amputations and fractures showed no significant associated SGLT2 inhibitor therapy.
Conclusion:
Our analysis indicates that diabetics and non-diabetics suffering from cardiovascular and renal diseases experience a significant reduction in morbidity and mortality from SGLT2 inhibitor therapy, leading to a better prognosis of such conditions in the long-term.
Keywords
Introduction
Non-communicable diseases (NCDs) encompass a vast category of health conditions, including diabetes, chronic kidney disease, and various cardiovascular disorders, mainly heart failure. The mortality rate associated with NCDs is significantly elevated in low-income and middle-income countries (LICs and MICs), posing a substantial obstacle to addressing health disparities in both developing and developed nations. 1 Cardiovascular diseases account for most NCD deaths, about 17.9 million people annually, while diabetes and its renal complications account for 2.0 million deaths. 2
The introduction of sodium-glucose cotransporter-2 inhibitors (SGLT-2 inhibitors) in recent years has shown a significant shift in the management of diabetes, with their implications extending beyond glycemic control. Although sodium glucose co-transporter type 2 (SGLT-2) inhibitors were initially introduced as glucose-lowering medications, it was later discovered that their cardio-protective effect combined with their safety associated with renal health is the most important treatment effect of these agents. 3 Various clinical trials demonstrate that the advantages of SGLT2 inhibitors extend across diverse patient subgroups. This includes individuals both with and without type 2 diabetes, with heart failure (both preserved or reduced ejection fraction) and those at different stages of chronic kidney disease (CKD). 4 Additionally, post-hoc analyses indicate that in CKD patients it lowers the risk of anemia and hyperkalemia in CKD patients. 4
The EMPA-OUTCOME trial, which utilized empagliflozin (SGLT-2 receptor inhibitor), demonstrated notable decreases in major adverse cardiac events (MACEs), cardiovascular mortality, and hospitalization for heart failure (HF) when added to standard-of-care therapy for individuals with type 2 diabetes (T2DM) at a high cardiovascular risk. The significant shift in primary composite outcome that is, major adverse cardiovascular events, was due to a markedly lower rate of cardiovascular death that is, 38% relative risk reduction. 5
While some trials have compared their efficacy in treating diabetes as well as comorbid heart conditions, none of these studies have systematically assessed and compiled the effects of SGLT2 inhibitors on cardiovascular outcomes, renal outcomes, dyslipidemia, and inflammatory markers associated with chronic conditions. Assessing these parameters in synchrony would give rise to a better interpretation of outcomes, and ultimately, would elicit a well-rounded analysis of the safety and efficacy of these drugs. Thus, our study aims to assess the outcomes and effects of treatment with SGLT2 inhibitors of patients having heart failure, diabetes mellitus, chronic kidney disease, and dyslipidemia. Our study’s goal is to find out if SGLT2 inhibitors play a significant role in lowering mortality due to cardiovascular or renal outcomes and decreasing lipid abnormalities and serum inflammatory markers.
By encompassing a broad spectrum of individuals, our findings aim to be applicable across various demographics, facilitating a more generalized assessment of the impact of SGLT-2 inhibitors. We have also incorporated the latest trials that haven’t been included in previous studies ensuring that the findings are current and relevant.
Methods
Data Sources and Search Strategy
This meta analysis was performed according to the PRISMA guidelines to enhance the clarity and reliability of systematic reviews and meta-analyses in the medical and healthcare literature. 6 PubMed, Cochrane Library, and MEDLINE databases were used to conduct a comprehensive literature search from inception to September 2023. The detailed search strategy is given in Supplemental File Table 1. We further reviewed the relevant articles from the reference section of the originally retrieved articles.
Study Selection and Eligibility Criteria
After completing the literature search, all the redeemed articles were exported to the Endnote reference library software to remove any duplicates. The 2 independent investigators (A.I. and S.H.H.) first reviewed the articles at the title and abstract level and then screened the full text of the relevant articles. Discrepancies during the screening process were resolved through discussions. The studies were included if they met our predefined inclusion criteria including (1) randomized control trials with follow-up for at least 8 weeks, (2) patients were on SGLT2i, (3) studies in which a group taking SGLT2i was compared with a group taking either placebo or other medication, (4) studies in which cardiovascular outcomes, renal outcomes, lipid biomarkers, and inflammatory markers were reported as the primary outcomes. All the case series, case reports, and non-English articles were excluded. We also excluded the articles that didn’t report any of the listed outcomes as their primary outcomes.
Outcomes of Interest, Data Extraction, and Quality Assessment
We extracted the baseline demographic characteristics from the included studies. The primary outcomes assessed in this meta-analysis included: mortality due to cardiovascular causes, death from heart failure, hospitalization due to heart failure, composite renal endpoints, renal replacement therapy, and death from any cause. Secondary outcomes included total cholesterol levels, low-density lipoprotein (LDL), triglyceride levels, flow-mediated dilation (FMD), IL-6, TNF-a, amputation, and fracture. The quality assessment of the included studies was conducted using the Cochrane Risk of Bias tool by 2 independent reviewers (M.O.L and A.R) and any disparities were resolved through discussion. 7
Statistical Analysis
The statistical analyses were conducted using Review Manager (RevMan version 5.4; Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014). In order to address the expected heterogeneity in study design and outcome definitions, we employed a random-effects model. Evaluation of continuous outcomes involved the use of standard Mean differences, while the risk ratios were used to assess dichotomous outcomes with 95% confidence intervals. Study weights were determined using the inverse variance method, which assigns greater weight to studies with larger sample sizes and smaller standard errors, thereby improving the precision of pooled estimates. P-values were derived from Z-tests based on the pooled effect estimates and their standard errors. Results attaining statistical significance were those with a P-value ⩽ .05. We also assess the statistical heterogeneity in the outcomes using the Higgins I2 statistic, with significance attributed to values exceeding the 50% threshold. Sensitivity analysis entailed a comprehensive leave-one-out analysis to pinpoint trials responsible for heterogeneity surpassing 50%. For outcomes reported by more than 10 studies, a visual examination of funnel plots was conducted to discern potential publication bias.
Results
Literature Search and Patient Characteristics
Our extensive systematic search initially yielded 14 734 studies, dating from inception to December 2023. These articles were subject to deduplication, after which there were 7098 records remaining. Subsequently, these articles were rigorously evaluated at the title-and-abstract level by independent investigators, followed by manual full-text review of 527 articles. Ultimately, our systematic search concluded with 31 studies selected for inclusion within this analysis,8-38 after the exclusion of 496 studies due to the lack of relevant outcomes reported (n = 341), or the incompatibility of the patient inclusion criteria (n = 155). The complete PRISMA flowchart is illustrated in Supplemental Figure 1, outlining the details of the screening process of the systematic review.
A total of 92 920 patients were featured in this comprehensive systematic review and meta-analysis, including 47 544 patients receiving SGLT2i drugs, and 45 376 patients receiving a control/placebo regimen. The mean age of the SGLT2i cohort was 64.36 years, whereas the mean age for the controlled cohort was 63.91 years. There were a total of 8 different types of SGLT2i drugs included: dapagliflozin (n = 14), empagliflozin (n = 7), canagliflozin (n = 5), sotagliflozin (n = 2), ertugliflozin (n = 1), luesogliflozin (n = 1), tofogliflozin (n = 1), and ipragliflozin (n = 1). Complete information regarding the baseline demographic and clinical characteristics are available in Table 1.
Baseline Characters.

Heart Failure Medication: (ACEI, ARBs, ARNI, Beta Blockers, Calcium Channel Blockers, Ivabradine.
Risk of Bias and Assessment of Quality
As all included studies were RCTs, the Cochrane Risk of Bias Tool for Randomized Controlled Trials (RoB-2) was employed for appraisal purposes. Generally, the included studies exhibited an overall moderate risk of bias. Notably, performance bias (due to lack of blinding of participants and/or personnel) and selection bias (due to inadequate allocation concealment) were the most commonly observed concerns [Supplemental Figures 2 and 3]. These biases may have introduced some degree of overestimation or underestimation of treatment effects, particularly in subjective or clinician-assessed outcomes. However, as over 75% of the assessments were rated as low or negligible risk across all domains, the overall impact on the pooled estimates is expected to be minimal.
Comparison of Clinical Outcomes
In this extensive meta-analysis, the following clinical outcomes were compared between cohorts receiving SGLT2i versus no SGLT2i, including: (i) systolic BP, (ii) renal composite endpoint, (iii) renal replacement therapy, (iv) hospitalization for HF, (v) death from HF, (vi) death from renal causes, (vii) death from cardiovascular causes, (viii) death from any cause, and (ix) FMD.
The differences in post-intervention systolic BP were reported by 9 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (MD: −1.41; 95% CI [−2.53 to −0.30]; P = .01; Figure 1). The differences in renal composite endpoint were reported by 6 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.65; 95% CI [0.54-0.78]; P < .00001; I2 = 59%; Figure 2A). The differences in the utilization of renal replacement therapy were reported by 3 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.72; 95% CI [0.59-0.88]; P = .002; Figure 3). The rate of hospitalization for HF was reported by 10 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.71; 95% CI [0.67-0.75]; P < .00001; Figure 4). The mortality from HF was reported by 5 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.80; 95% CI [0.76-0.86]; P < .00001; Figure 5). The mortality from renal causes was reported by 4 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.27; 95% CI [0.11-0.68]; P = .005; Figure 6). The mortality from cardiovascular causes was reported by 11 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.88; 95% CI [0.81-0.95]; P = .0007; Figure 7). The all-cause mortality was reported by 11 studies, which indicated the statistically significant superiority in the SGLT2i cohort in comparison to the control group (RR: 0.89; 95% CI [0.84-0.95]; P = .0006; Figure 8). The differences in FMD were reported by 3 studies, which indicated the lack of statistically significant differences between the SGLT2i cohort in comparison to the control group (MD: 1.25; 95% CI [−0.57 to 3.08]; P = .18; I2 = 73%; Figure 9A). Overall, it was found that SGLT2i drugs provide noteworthy improvements with respect to clinical outcomes in patients with a wide array of comorbidities.

Forest plot of systolic BP: (MD: −1.41; 95% CI [−2.53 to −0.30]; P = .01).

(A) Forest plot of renal composite: (RR: 0.65; 95% CI [0.54-0.78]; P < .00001; I2 = 59%) and (B) post sensitivity forest plot of renal composite: (RR: 0.58; 95% CI [0.52-0.66]; P < .00001; I2 = 0%).

Forest plot of renal replacement therapy: (RR: 0.72; 95% CI [0.59-0.88]; P = .002).

Forest plot of hospitalization for HF: (RR: 0.71; 95% CI [0.67-0.75]; P < .00001).

Forest plot of death from heart failure: (RR: 0.80; 95% CI [0.76-0.86]; P < .00001).

Forest plot of death from renal causes: (RR: 0.27; 95% CI [0.11-0.68]; P = .005).
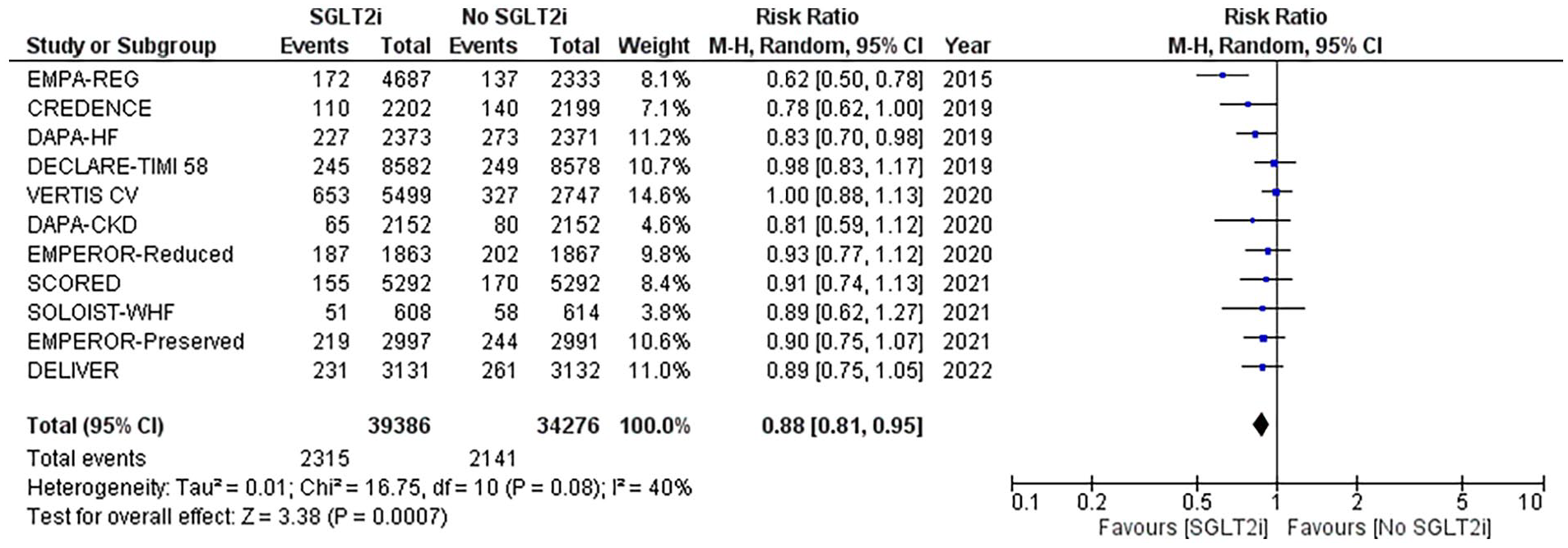
Forest plot of death from CVS causes: (RR: 0.88; 95% CI [0.81-0.95]; P = .0007).

Forest plot of death from any cause: (RR: 0.89; 95% CI [0.84-0.95]; P = .0006).

(A) Forest plot of FMD: (MD: 1.25; 95% CI [−0.57 to 3.08]; P = .18; I2 = 73%) and (B) post sensitivity forest plot of FMD: (MD: 0.40; 95% CI [−0.41 to 1.21]; P = .33; I2 = 0%).
Comparison of Lipid Markers
In this extensive meta-analysis, the following laboratory parameters were compared between cohorts receiving SGLT2i versus no SGLT2i, including: (i) serum total cholesterol, (ii) serum triglycerides, (iii) serum LDL.
The differences in total cholesterol values were reported by 6 studies, which indicated the lack of statistically significant differences between the SGLT2i cohort in comparison to the control group (MD: −0.80; 95% CI [−11.07 to 9.47]; P = .88; I2 = 78%; Figure 10A). The differences in triglyceride values were reported by 4 studies, which indicated the lack of statistically significant differences between the SGLT2i cohort in comparison to the control group (MD: −5.43; 95% CI [−17.06 to 6.19]; P = .36; Figure 11). Similarly, the differences in LDL values were reported by 5 studies, which indicated no statistically significant differences between the 2 groups (MD: 2.05; 95% CI [−2.33 to 6.43]; P = .36; Figure 12) Overall, it was found that SGLT2i drugs do not significantly affect the lipid markers.
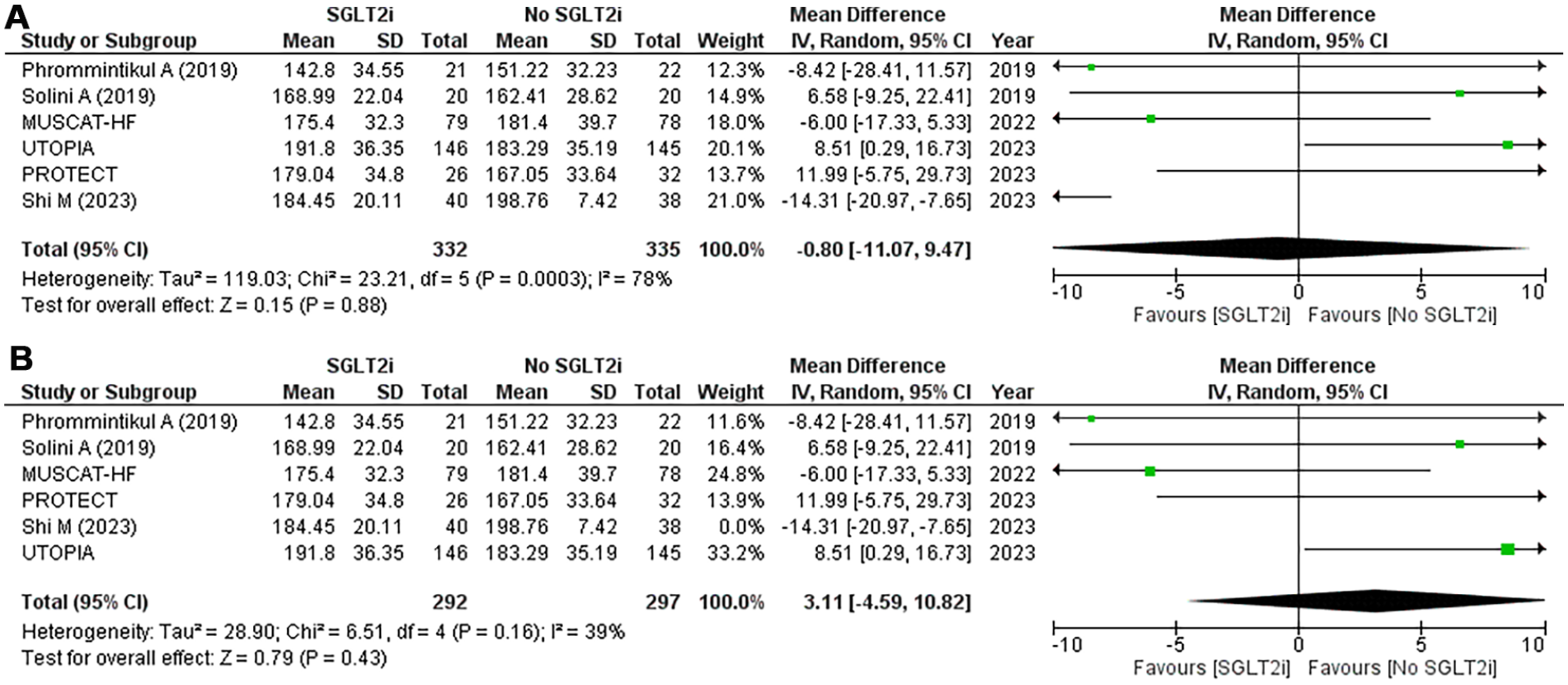
(A) Forest plot of total cholesterol levels: (MD: −0.80; 95% CI [−11.07 to 9.47]; P = .88; I2 = 78%) and (B) post sensitivity forest plot of total cholesterol levels: (MD: 3.11; 95% CI [−4.59 to 10.82]; P = .43; I2 = 39%).

Forest plot of triglycerides: (MD: −5.43; 95% CI [−17.06 to 6.19]; P = .36).

Forest plot of LDL: (MD: 2.05; 95% CI [−2.33 to 6.43]; P = .36).
Comparison of Inflammatory Biomarkers
The following laboratory parameters were analyzed between cohorts receiving SGLT2i versus no SGLT2i, including (i) serum TNF-a and (ii) serum IL-6.
The differences in serum TNF-a values were reported by 3 studies, which indicated a statistically significant distinction between the SGLT2i cohort in comparison to the control group (MD: −0.20; 95% CI [−0.28 to −0.12]; P ⩽ .00001; Figure 13). The differences in serum IL-6 values were reported by 4 studies, which indicated a statistically significant difference between the SGLT2i cohort in comparison to the control group (MD: −0.2; 95% CI [−0.27 to −0.12]; P ⩽ .00001; I2 = 45%; Figure 14).

Forest plot of TNF-a: (MD: −0.20; 95% CI [−0.28 to −0.12]; P ⩽ .00001).

Forest plot of IL-6: (MD: −0.2; 95% CI [−0.27 to −0.12]; P ⩽ .00001).
Overall, it was found that SGLT2i drugs provide noteworthy improvements with respect to inflammatory biomarkers in patients with a wide array of comorbidities.
Comparison of Adverse Events
In this extensive meta-analysis, the following post-interventional adverse events were compared between cohorts receiving SGLT2i versus no SGLT2i, including: (i) incidence of fracture and (ii) incidence of amputation. The incidence of Fracture was reported by 6 studies, which indicated the lack of statistically significant differences between the SGLT2i cohort in comparison to the control group (RR: 1.04; 95% CI [0.95 to 1.15]; P = .38; Figure 15). The incidence of amputation was reported by 8 studies, which indicated the lack of statistically significant differences between the SGLT2i cohort in comparison to the control group (RR: 1.06; 95% CI [0.91 to 1.22]; P = .46; Figure 16). Overall, it was found that SGLT2i drugs exhibit a desirable safety profile.
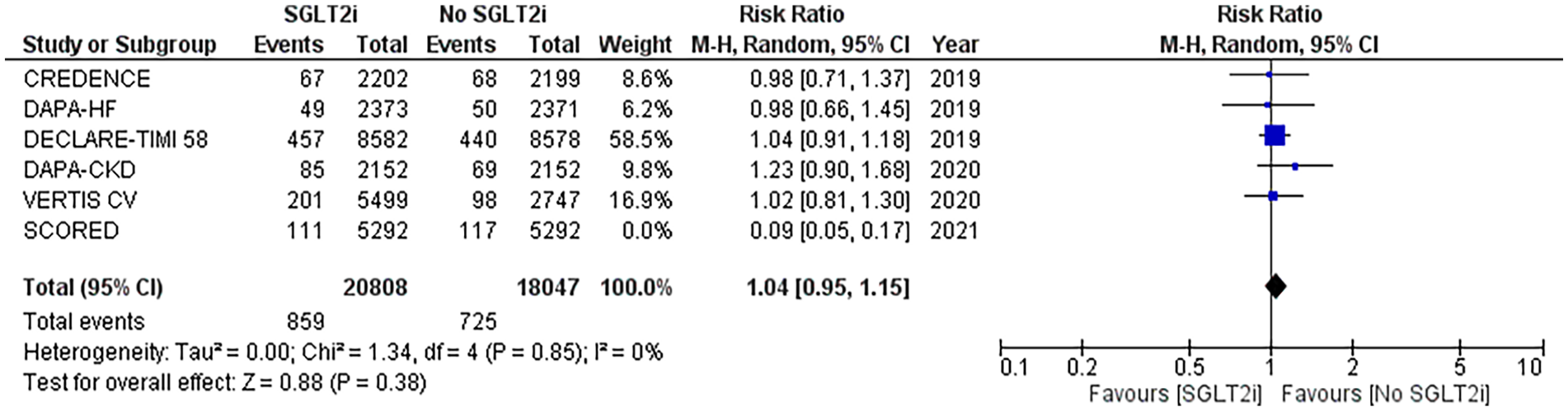
Forest plot of fractures: (RR: 1.04; 95% CI [0.95 to 1.15]; P = .38).

Forest plot of amputations: (RR: 1.06; 95% CI [0.91 to 1.22]; P = .46).
Assessment of Heterogeneity
The following outcomes of interest were flagged to have suboptimal levels of heterogeneity (I2 ⩾ 50%), including: (i) renal composite endpoint, (ii) FMD, and (iii) total cholesterol levels. In the first 2 cases, sensitivity analysis technique using the leave-one-out method was successful in eradicating the heterogeneity completely (I2 = 0%), whereas in the latter case, heterogeneity was reduced to an acceptable standard (I2 = 39%).
Broadly, no particular trends were identified with regards to 1 specific study causing the perceived heterogeneity. However, 2 of the excluded studies, including Shi et al 28 and Sposito et al 20 had significantly smaller sample sizes, 40 and 48 respectively, in comparison to some other larger studies. The wide variation in the sample sizes of included studies may account for inappropriate weight distribution or study strength within the meta-analysis, thus possibly resulting in heterogeneity. Alternatively, other causes of heterogeneity may revolve around the complex set of included patients with a diversified set of comorbidities, including HF, diabetes mellitus, CKD, and more. Ultimately, this may cause subtle differences within the efficacy profile of SGLT2i drugs, hence potentially leading to the heightened levels of heterogeneity, as observed within these outcomes (Figures 9B and 10B).
A distinct peculiarity was observed within the study conducted by Mc Causland et al, 33 responsible for causing a significant level of heterogeneity within the renal composite endpoint outcome (Figure 2B). The overall mean age of the included study population was 64.36 years for the SGLT2i cohort and 63.91 years for the controlled cohort, whereas McCausland et al. had an above-average age range of 74.5 years. This introduces considerable variation within the analysis, as older HF patients are highly susceptible to a poorer prognosis and worsened clinical outcomes, mostly due to the increased rates of multimorbidity, and polypharmacy. Therefore, it is plausible to assume that age-level variation is an important source of heterogeneity that must be interpreted and accounted for.
Assessment of Publication Bias
The evaluation of publication bias was conducted on all outcomes of interest with ⩾10 studies via the visual assessment of funnel plots, generated on the random-effects model, for the following outcomes, including: (i) hospitalization for HF and (ii) death from cardiovascular causes. All outcomes demonstrated asymmetrical funnel plots, and therefore are suspected to be affected by publication bias (Supplemental Figures 4 and 5).
Discussion
Heart failure is a life-threatening condition that is associated with high morbidity and mortality, poor life quality, and high costs. It affects approximately 64 million people worldwide. 39 Recent data indicates that 32% to 43% of patients with chronic HF have coexisting diabetes mellitus. 40 The relationship between heart failure and chronic kidney disease, diabetes mellitus, and atherosclerosis has been well-established in the past.40,41 SGLT2 inhibitors are a class of drugs widely used to treat diabetes, heart failure, and the complications associated with them. 42 While some trials have compared their efficacy in treating diabetes as well as comorbid heart conditions, 43 others have compared their efficacy in preventing the progression of CKD. 44 Some have also assessed their effects on dyslipidemia. 45 However, none of these studies have systematically assessed and compiled the effects of SGLT2 inhibitors on cardiovascular outcomes, renal outcomes, dyslipidemia, and inflammatory markers associated with chronic conditions. Thus, our study aimed to assess the outcomes and effects of treatment with SGLT2 inhibitors of patients having heart failure, diabetes mellitus, chronic kidney disease, and dyslipidemia. Our study’s goal was to find out if SGLT2 inhibitors played a significant role in lowering mortality due to cardiovascular or renal outcomes and decreasing lipid abnormalities and serum inflammatory markers in a vast array of patients. We comprehensively assessed and analyzed all the outcomes and found SGLT2 inhibitors to be positively correlated in reducing mortality from any cause as well as any adverse cardiovascular or renal outcomes, and inflammatory markers. On the other hand, our analysis revealed no relationship between SGLT2 inhibitors and dyslipidemia or fractures and amputations. Our findings are of paramount importance because they assessed the correlation of SGLT2 inhibitors with various systemic outcomes in diabetic patients on a larger scale than in the previous literature. More importantly, our research confirms and validates that SGLT2 inhibitors can lead to reduced mortality in patients with complicated and uncomplicated type 2 diabetes mellitus as well as non-diabetic heart patients, thus warranting this class of drug as an even more effective treatment regimen.
SGLT2 transporters are widely expressed in the proximal tubules of the kidney and are mainly responsible for glucose and sodium co-reabsorption from the kidney. SGLT2 inhibitors function by blocking these transporters, resulting in glucosuria and osmotic natriuresis and diuresis, leading to a decrease in the fluid overload that commonly occurs in heart failure patients. The efficacy of SGLT2 inhibitors in treating heart failure, diabetes mellitus, and other comorbid conditions can be well-explained with this mechanism of action. 46 Our analysis revealed that patients on SGLT2 inhibitors have a lower rate of hospitalization and mortality from heart failure and a lower rate of mortality from any other cardiovascular condition as well. This outcome can be explained by the inhibition of SGLT2 transporters, leading to natriuresis and osmotic diuresis and thus lowering the fluid load of the body. 47 Our findings are in line with the study by Packer et al 11 who found a reduction of 5.3% in hospitalization and mortality due to heart failure in the group receiving Empagliflozin as compared to the group receiving placebo. Another study by Solomon et al 15 also revealed similar findings and found a reduction of 3.1% in the mortality and hospitalization of the heart failure patients being treated with Dapagliflozin as compared to a placebo. Our analysis also evaluated systolic blood pressure to note whether SGLT2 inhibitors have any effect on hypertension. Validating the findings of Katakami et al 35 that found systolic BP to decrease by 5.6 mmHg from the start to the end of the treatment, our analysis also revealed a highly significant trend when it came to lowering hypertension with this drug. The proposed mechanism is the decreased absorption of sodium due to SGLT2 inhibitors, leading to increased natriuresis, and consequently, a lower BP. 48
Besides evaluating the cardiovascular outcomes, we also analyzed the renal outcomes reported by the trials that used SGLT2 inhibitors as treatment. Our analysis revealed that the use of SGLT2 inhibitors decreases mortality from any renal causes, as well as the need for renal replacement therapy. The reno-protective effects of SGLT2 inhibitors can be explained by the reduction of inflammation, oxidative stress, and a reduced intraglomerular pressure that prevents tubular injury.49,50 Another renal outcome that our analysis assessed was a reduction in the renal composite that was defined as a ⩾40% decrease in eGFR to <60 ml/min 1.73 m2, ESRD, or death from any renal cause. This renal outcome is an important prognostic factor in evaluating the development of chronic kidney disease in diabetics. The proposed mechanism for decreased eGFR in patients on SGLT2 inhibitors is explained by the action of SGLT2 inhibitors in preventing the reabsorption of sodium and glucose in the proximal convoluted tubule, this results in a concomitant increased delivery of sodium to the juxtaglomerular apparatus that releases ATP into the interstitium which gets broken down into adenosine. Adenosine is the primary regulator of GFR and acts on the vasoconstricting A1 receptors of the afferent arteriole, resulting in a lower eGFR and improving the prognosis of CKD in heart failure and T2DM patients. 51 Our study confirms the findings of Perkovic et al 16 and Bhatt et al 13 that revealed a 30% less incidence of renal outcomes development in patients in the SGLT2 Canagliflozin treatment group compared to the placebo and a hazard ratio of 0.71 (4.6-1.08) in patients on SGLT2 Sotagliflozin treatment group compared to the placebo, respectively.
One prognostic factor that other meta-analyses done on the subject lacked was inflammatory markers. Assessment of these markers in patients with diabetes mellitus is important because they directly contribute to the development of fatal complications such as atherosclerosis, myocardial infarction, heart failure, as well as ischemic stroke. 52 We assessed IL-6 and TNF alpha which are acute phase reactant proteins and indicate systemic inflammation. 53 Surprisingly, our analysis found these inflammatory markers to decrease with the usage of SGLT2 inhibitors. The proposed mechanism is that SGLT2 inhibitors decrease inflammatory markers indirectly by reducing insulin and uric acid which in turn lower IL-6 as well as other inflammatory markers. 47 These findings align with the findings of Benedikt et al 27 that revealed that patients on Empagliflozin have a percent change in mean from the baseline equal to −33.72 as compared to the placebo-treated patients having a percent change in mean from the baseline equal to −36.60 in IL-6 levels.
Although certain outcomes demonstrated small effect sizes, their statistical significance and consistency across diverse patient populations underscore their clinical relevance. In the context of chronic cardiovascular and renal conditions, even modest improvements in surrogate markers—when sustained—may contribute to meaningful long-term risk reduction. These findings support the role of SGLT-2 inhibitors as a viable therapeutic option, particularly in high-risk individuals with multiple comorbidities. The reproducibility of these effects across studies further strengthens their validity and suggests potential value in informing clinical decision-making and optimizing patient outcomes.
In addition to evaluating the cardiovascular and renal outcomes, our study also evaluated the effect of SGLT2 on fractures, amputations, dyslipidemia, and flow-mediated dilation (FMD) which is an endothelial dysfunction marker. While some studies52,54 revealed a decreased risk of amputation and fractures in patients who are on SGLT2 inhibitors, others revealed an increased risk of fractures and amputations in patients on treatment.8,9 This trend of varying risks of amputations and fractures on treatment with SGLT2 inhibitors strengthens the stance that this class of drug has no appreciable effects on amputations and fractures. We evaluated and analyzed dyslipidemia and found it to follow a similar trend as well. Dyslipidemia is an important risk factor for the development of complications like atherosclerosis and myocardial infarction, and thus it was important to evaluate any effects that SGLT inhibitors may have on dyslipidemia. 54 We evaluated dyslipidemia by the prominent lipid markers LDL, HDL, triglycerides, and total cholesterol. Studies like Katakami et al 35 found a change of 0.01 ± 0.79 in mean cholesterol levels of the patients being treated by Topigloflozin from the start of the treatment till the end and Solini et al 22 found a change an increase of 0.12 in the mean triglyceride levels in Dapagliflozin-treated group. On the other hand, studies by Ejiri et al 30 and Phrommintikul et al 31 indicated a significant decrease in lipid markers like triglycerides changed from baseline and decreased by a mean of 2.3 in the treatment group and LDL cholesterol decreased by a mean of 5.31 on treatment as compared to the placebo group, respectively. Another significant factor that our analysis evaluated is the endothelial dysfunction marker, flow-mediated dilation (FMD). Studies like Sposito et al 20 indicated a significant post-treatment change in FMD (P = .0001) while other studies like Kishimoto et al 37 reported no significant change from baseline (5.2% ± 2.6% vs 5.2% ± 2.6%, P = 0.98) on treatment with Ipragliflozin. The fact that our analysis could not find an appreciable risk of fractures, amputations, dyslipidemia, and change in FMD levels associated with this drug, further validates the efficacy and safety of this drug in treating T2DM and its complications.
While our meta-analysis supports the cardiovascular and renal benefits of SGLT-2 inhibitors, not all studies have reported consistent results. Kristensen et al 55 observed that reductions in major adverse cardiovascular events (MACE) were largely driven by empagliflozin and canagliflozin, with dapagliflozin showing a neutral effect in DECLARE-TIMI. 58 The VERTIS-CV trial 9 found no significant reduction in MACE with ertugliflozin compared to placebo. Similarly, Lo et al 56 reported heterogeneity in renal outcomes, particularly among patients without established chronic kidney disease. Fei 57 noted that while SGLT-2 inhibitors were beneficial overall, the cardiovascular mortality benefit was attenuated in non-diabetic populations. In the EMPA-KIDNEY trial, Herrington et al 58 reported that the renal benefit was most pronounced in patients with eGFR < 60 ml/min/1.73 m², indicating a limitation in broader applicability.
Some important factors make our study unique and important in the discourse of evaluating SGLT2 inhibitors as a potential treatment for complicated and uncomplicated diabetes mellitus as well as for other cardiovascular conditions. One of the most significant strengths of our study is that it had a diverse population sample size of 92 920 which increased the statistical power and allowed us to comprehensively assess these outcomes in varying diverse populations. Another important strength of our study, which the current literature also lacks is that it systematically combined and analyzed the effect of SGLT inhibitors on all the renal outcomes, cardiovascular outcomes, inflammatory markers, as well as markers of dyslipidemia. Evaluation of all these outcomes helped us analyze the effect of SGLT2 inhibitors more stringently and allowed us to reach valuable conclusions about its efficacy and safety as a drug regimen in the treatment of T2DM and its complications. Moreover, meta-analyses done on the subject lacked recent studies. Our study combined the previous and recent data and analyzed it together to reach more conclusive results. Collectively, the strengths of this study speak volumes about its credibility as an important piece of scientific literature that should be considered invaluable in evaluating drug regimens when it comes to treating complicated conditions such as diabetes mellitus.
Despite being a unique and well-defined piece of literature, our study had some limitations, that had little to no effects on the outcomes. One limitation is that due to having a larger sample size from diverse populations, the patients had varying baseline parameters and outcomes. Even though this was done initially to evaluate the patients on a larger scale and make the data statistically significant, due to this variation in parameters, our outcomes presented significant heterogeneity. Another limitation of this study is that most of the trials we included used different SGLT2 inhibitors, dosages, follow-up durations, and placebos. Our study also was limited in terms of accounting for any confounding factors like smoking, obesity, physical inactivity, or any other factor that may have been contributing to the worsening of the outcomes in the placebo-treated patients. Collectively, these limitations may have a slight effect on the outcomes but are not significant enough to account for any drastic changes in the evaluated outcomes.
Our study has evaluated all the major outcomes of SGLT2 inhibitors in heart failure and diabetic patients, indicating it as an effective treatment for such conditions. However, future studies are welcome to be more elaborate on the subject and minimize the limiting factors like confounding factors and heterogeneity that are present in our study. While SGLT2 inhibitors demonstrate significant benefits in various populations, their use in patients with contraindications, such as severe chronic kidney disease (CKD) or type 1 diabetes, requires careful ethical consideration. In these populations, the risk of adverse outcomes, including diabetic ketoacidosis and renal complications, may outweigh the potential benefits. Thus, clinicians must carefully evaluate individual patient factors, and further research is needed to clarify the safety and efficacy of these drugs in such high-risk groups.
More importantly, to truly determine the efficacy of SGLT2 inhibitors, there are bigger, more important questions like whether it reverses the pathological cardiac remodeling in heart failure patients or whether it has any prominent effects in preventing or treating cardiac arrhythmias, that are yet to be answered. Hence, a more streamlined approach that evaluates functional capacity in heart failure and electrophysiology associated with these chronic conditions can help answer these bigger, more important questions that can truly lead us to know whether SGLT2 inhibitors can be accepted as a widely used treatment for such complications. Future researchers are encouraged to research these bigger questions that can help us understand this miracle class of drugs better.
Conclusion
Our systematic review and meta-analysis of 92 920 patients that were on SGLT2 inhibitor therapy primarily assessed these drugs as an effective treatment in patients with heart failure and diabetes mellitus. Our study analyzed the effects of SGLT2 inhibitors on cardiovascular outcomes, renal outcomes, levels of inflammatory markers, and dyslipidemia. Our results showed us that there is significant prognostic improvement in heart failure, renal replacement therapy, hypertension, and levels of IL-6 and TNF-alpha. Our analysis did not reveal SGLT2 inhibitors to be significantly related with increase or decrease in cholesterol, triglycerides, HDL, LDL, fractures, amputations, and FMD, thus indicating the safety of this drug in diabetic and heart failure patients. Our research indicates that heart failure patients with other comorbidities significantly benefit from SGLT2 inhibitor therapy, leading to a better prognosis of such conditions in the long-term.
Supplemental Material
sj-docx-1-cic-10.1177_11795468251347777 – Supplemental material for Evaluating the Safety and Efficacy of SGLT-2 Inhibitors on Reducing Cardiovascular and Renal Mortality, Morbidity and Inflammatory Outcomes in Various Patient Populations: A Systematic Review and Meta-Analysis of 92 920 Patients
Supplemental material, sj-docx-1-cic-10.1177_11795468251347777 for Evaluating the Safety and Efficacy of SGLT-2 Inhibitors on Reducing Cardiovascular and Renal Mortality, Morbidity and Inflammatory Outcomes in Various Patient Populations: A Systematic Review and Meta-Analysis of 92 920 Patients by Syed Hamza Haider, Areeka Irfan, Samir Mustafa Sheikh, Muhammad Omar Larik, Alishba Raza and Yumna Khabir in Clinical Medicine Insights: Cardiology
Footnotes
Author Note
I take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation.
Author Contributions
Syed Hamza Haider: Conceptualization, Data Curation, Methodology, Investigation, Formal Analysis, Writing- Reviewing, and Editing, Supervision. Areeka Irfan: Data Curation, Visualization, Writing- Reviewing and Editing, Formal Analysis, Project Administration. Samir Mustafa Sheikh: Writing- Original draft preparation, Data Curation Omar Larik: Writing - Original draft preparation, Data Curation.: Alishba Raza: Data Curation, Methodology. Yumnah Khabir: Writing- Original draft preparation, Data Curation.
Information Pertaining to Writing Assistance
No funded writing assistance was used.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
