Abstract
Purpose
This study was to investigate the association between the use of Sodium-glucose Cotransporter-2 inhibitors (SGLT2i) or angiotensin receptor-neprilysin inhibitor (ARNI; ie, Sacubitril + valsartan, Product name ENTRESTO) and the risk of atherosclerotic cardiovascular disease (ASCVD) in patients with coexisting diabetes and heart failure. Specifically, the study compared outcomes between patients using SGLT2i or valsartan + sacubitril and those not using these medications.
Methods
This study utilized data from the National Health Insurance Research Database (NHIRD) from 2017 to 2018. The case group consisted of 8691 patients with coexisting diabetes and heart failure who did not use SGLT2i or Entresto, while the control group consisted of 8691 patients with coexisting diabetes and heart failure who used SGLT2i or Entresto. The primary outcome was ASCVD, including a composite of cardiovascular death and hospitalization for worsening heart failure. Secondary outcomes included all-cause death, cause of cardiovascular death, and recurrence of heart failure, non-fatal myocardial infarction, non-fatal stroke (including ischemic stroke and hemorrhagic stroke) and new renal replacement therapy.
Results
The study found that the use of SGLT2 inhibitors or ARNI was associated with a lower risk of ASCVD in patients with coexisting diabetes and heart failure.
Conclusion
The study suggests that the use of SGLT2 inhibitors, alone or in combination with Entresto, may be effective in reducing the risk of ASCVD and its associated adverse outcomes in patients with diabetes and heart failure. This finding has important implications for the management of these conditions.
Introduction
Atherosclerotic cardiovascular disease (ASCVD) is a leading cause of mortality and morbidity for individuals with diabetes and coronary heart disease. It is also very costly to manage, costing over $37 billion annually1,2
Heart failure is a common and serious condition that affects millions of people worldwide, and it is often associated with diabetes and ASCVD. 3 Patients with heart failure and diabetes are at increased risk of adverse outcomes, including hospitalization, cardiovascular events, and death.4–6 Therefore, it is important to identify effective treatments that can reduce the risk of ASCVD and improve outcomes in this patient population.
Different pathophysiologic factors engage in ASCVD, thereby creating a perfect environment for atherosclerosis.7 Compared to those without, type 2 diabetic patients have a smaller diameter of the coronary artery, higher atheroma volume, and greater burden of atherosclerotic plaques, due to various factors such as hyperglycemia and hyperinsulinemia. Hyperglycemia increases the risk of ASCVD in diabetic patients and it is a predictor of heart disease, stroke, cardiovascular disease (CVD), and mortality. Insulin resistance also increases cardiovascular risk, with insulin-resistant individuals having high rates of dyslipidemia, hypertension, and impaired glucose tolerance, all of these contribute to disease progression and complexity. 8 People with type 2 diabetes may have impaired insulin signaling and lipid abnormalities. Indeed, hyperglycemia increases the formation of atheroma in diabetic dyslipidemia. The epidemics of ASCVD, diabetes and obesity point to common molecular mechanisms. Moreover, there is an increase in inflammatory mediators and markers in obesity and high rates of cardiovascular events in type 2 diabetes. Other events that might have a role to play include reactive oxygen species, endothelial dysfunction, hypercoagulability, and vascular calcification.
Sodium-glucose cotransporter 2 (SGLT2) inhibitors and angiotensin receptor-neprilysin inhibitors (ARNIs; Brand Name ENTRESTO, Generic Name Sacubitril/Valsartan, First approved July 7, 2015; ARNI was launched in Taiwan in, 2017) (Appendix) are two classes of medications that have been shown to improve outcomes in patients with heart failure and diabetes. 9 However, the association between these medications and the risk of ASCVD in this patient population is not well understood. 10 Therefore, the objective of this study was to investigate the association between the use of SGLT2 inhibitors or ARNI and the risk of ASCVD in patients with coexisting diabetes and heart failure.11,12 This study improve outcomes in patients with coexisting diabetes and heart failure13,14 has significant implications for the management of ASCVD, By investigating the association between the use of sodium-glucose cotransporter-2 inhibitors (SGCT2i) or angiotensin receptor-neprilysin inhibitor and the risk of ASCVD in this patient population, the study may provide valuable insights into the effectiveness of these medications in reducing the risk of ASCVD. This could lead to improved treatment options and better outcomes for patients with these conditions.
Methodology
Data Source
The study data was sourced from the National Health Insurance Research Database (NHIRD), which contains the medical records of approximately 99% of the Taiwanese population. The information includes hospital admissions, comprehensive inpatient and outpatient health care information, diagnostic codes and prescription medications status since 1995. Diseases were classified according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and Tenth Revision, Clinical Modification (ICD-10-CM). To protect the patients’ privacy, all personal identification numbers were encrypted by the National Health Insurance before the data were released. The Taiwan National Health Research Institutes encrypts patient personal information to protect privacy and provides researchers with anonymous scrambled identification numbers associated with relevant claims information, including sex, date of birth, medical services received, and prescriptions. Therefore, patient informed consent is not required for authorized researchers to access this research database. The researchers of this study are not possible to contact any studied patient for obtaining informed consent. No informed consent form is used in this study. Furthermore, the Institutional Review Board (IRB) of China Medical University (MOHW110-TDU-B-212-124004) also specifically waived the requirement of informed consent. This study was conducted in accordance with the Declaration of Helsinki.
Study Population
From the Claims data of the whole insured population, this study investigated the association between the non-use of SGLT2i or Entresto and the use of SGLT2i or Entresto with the risk of ASCVD in patients with coexisting diabetes and heart failure patients from 2017 to 2018. The index date for the case group was the first date of the initial use of SGLT2i or Entresto (n = 46 030), while the index date of the control group was a random date within the study period. The exclusion criteria were patients who received hemodialysis before the index date, were less than 20 years old, missing data or had withdrawn from the insurance program before the index date (n = 21 412) (Figure 1).

Flow chart diagram showing numbers of the enrollment.
Construction of the Propensity Score
The control group was propensity score-matched according to gender, age (at every 5 years), baseline comorbidities and the index year at a ratio of 1:1 to the case group. The study design involved using data from the National Health Insurance Research Database (NHIRD) to identify patients with coexisting diabetes and heart failure who used either SGLT2 inhibitors or ARNI. Patients who used both SGLT2 inhibitors and ARNI were excluded from the study. The case group consisted of patients who did not use SGLT2i or ARNI, while the control group consisted of patients who used either SGLT2i or ARNI. The primary outcome was ASCVD, including a composite of cardiovascular death and hospitalization for worsening heart failure.
By excluding patients who used both SGLT2i and ARNI, the study aimed to minimize the potential confounding effects of combining different classes of medications. However, it is important to note that this approach may limit the generalizability of the study results to patients who use both SGLT2i and ARNI.
Main Outcome and Comorbidities
The primary outcome of this study was ASCVD, including a composite of cardiovascular death and hospitalization for worsening heart failure. The secondary outcomes were all-cause death, cause of cardiovascular death, recurrence of heart failure, non-fatal myocardial infarction, non-fatal stroke (including ischemic and hemorrhagic stroke), and new renal replacement therapy. We searched for comorbidities that were potentially linked to ASCVD, including hypertension, hyperlipidemia, atrial fibrillation, and chronic kidney disease.
Statistical Analysis
We used SAS version 9.4 (SAS Institute Inc., Cary, NC) software data and survival curves were plotted by R software to perform he data analysis for this study, with a P-value of <.05 considered statistically significant. The t test, Chi-squared test and Mann–Whitney U test were used to examine differences of categorical variables between non-exposure and SGLT2i/Entresto group including sex, age, comorbidities and mean outcome follow-up. The crude hazard ratio (cHR) adjusted hazard ratio (aHR) and 95% confidence intervals (CIs) were calculated for ASCVD and outcome disease among the sampled patients by Cox proportional hazards regression model. The regression coefficient is that when X increases by 1 unit, the relative risk of ASCVD decreases. The aHRs of ASCVD were calculated in the different multivariate cox proportional models included sex, age and comorbidities. The Kaplan–Meier method was used to estimate the time-to-event.
Results
As shown in Table 1, a total of 17 382 patients with diabetes and heart failure were included in this study after propensity score matching. The case group contained 8691 patients with diabetes and heart failure without the use of SGLT2i or Entresto and the control group contained 8691 patients with diabetes and heart failure who used SGLT2i or Entresto. Approximately half of the patients were female and almost all patients were aged over 60 years old. There were no significant differences between case and control groups regarding sex, age and comorbidities. The performance of mean follow-up within the ASCVD, all-cause death, non-fatal myocardial infarction, non-fatal stroke and new renal replacement therapy were both significantly different between the case and control groups.
Comparison of Baseline Characteristics in the Non-Exposure and SGLT2i or Entresto Cohorts.

at-Test.
bMann–Whitney U test.
Table 2 presents the risk of ASCVD among the sampled patients. After adjusting for age, sex and comorbidities, the use of Entresto patients with coexisting diabetes and heart failure had a significantly higher risk of ASCVD. The non-use of Entresto or SGLT2i patients with coexisting diabetes and heart failure cohort were selected as reference (aHR, 5.89; 95% CI, 3.94-8.80; estimate, 1.77; standard error, 0.2). Compared to the non-use of Entresto or SGLT2i patients with coexisting diabetes and heart failure, patients with diabetes and heart failure who used Entresto or SGLT2i had a significantly increased risk of ASCVD in the multivariable analysis (aHR, 1.84; 95% CI, 1.42-2.39; estimate, 0.61; standard error, 0.13). Patients with diabetes and heart failure who used Entresto and SGLT2i had a significantly greater risk of ASCVD than the non-use of Entresto or SGLT2i patients with coexisting diabetes and heart failure (aHR, 5.13; 95% CI, 2.93-8.99; estimate, 1.64; standard error, 0.29).
Hazard Ratios (HRs), and 95% Confidence Intervals (CIs) for ASCVD Among the Sampled Patients.

PY: person-years; IR: incidence rate, per 10 000 person-years; cHR, crude hazard ratio; aHR: adjusted hazard ratio.
aHR: multivariable analysis including sex, age, comorbidities.
* P-value < .05; ** P < .01, *** P < .001.
Table 3 shows the risk factors resulting in the development of ASCVD with a different multivariable Cox model. Controlling for age, sex and comorbidities, patients with diabetes and heart failure who use Entresto had a higher risk of ASCVD than patients with diabetes and heart failure who used SGLT2i (aHR, 4.89; 95% CI, 3.16-7.58). The multivariable Cox model showed that patients with diabetes and heart failure who used SGLT2i or Entresto had a significantly greater risk of ASCVD than patients with diabetes and heart failure who did not use SGLT2i or Entresto (aHR, 1.84; 95% CI, 1.42-2.39). Patients with diabetes and heart failure who used Entresto and SGLT2i at the same time had a significantly higher risk of ASCVD than the patients with diabetes and heart failure who did not use Entresto or SGLT2i (aHR, 4.93; 95% CI, 2.8-8.67) after adjusting for sex, age and comorbidities.
Hazard Ratios of Atherosclerotic Cardiovascular Disease in Different Multivariable Cox Models.
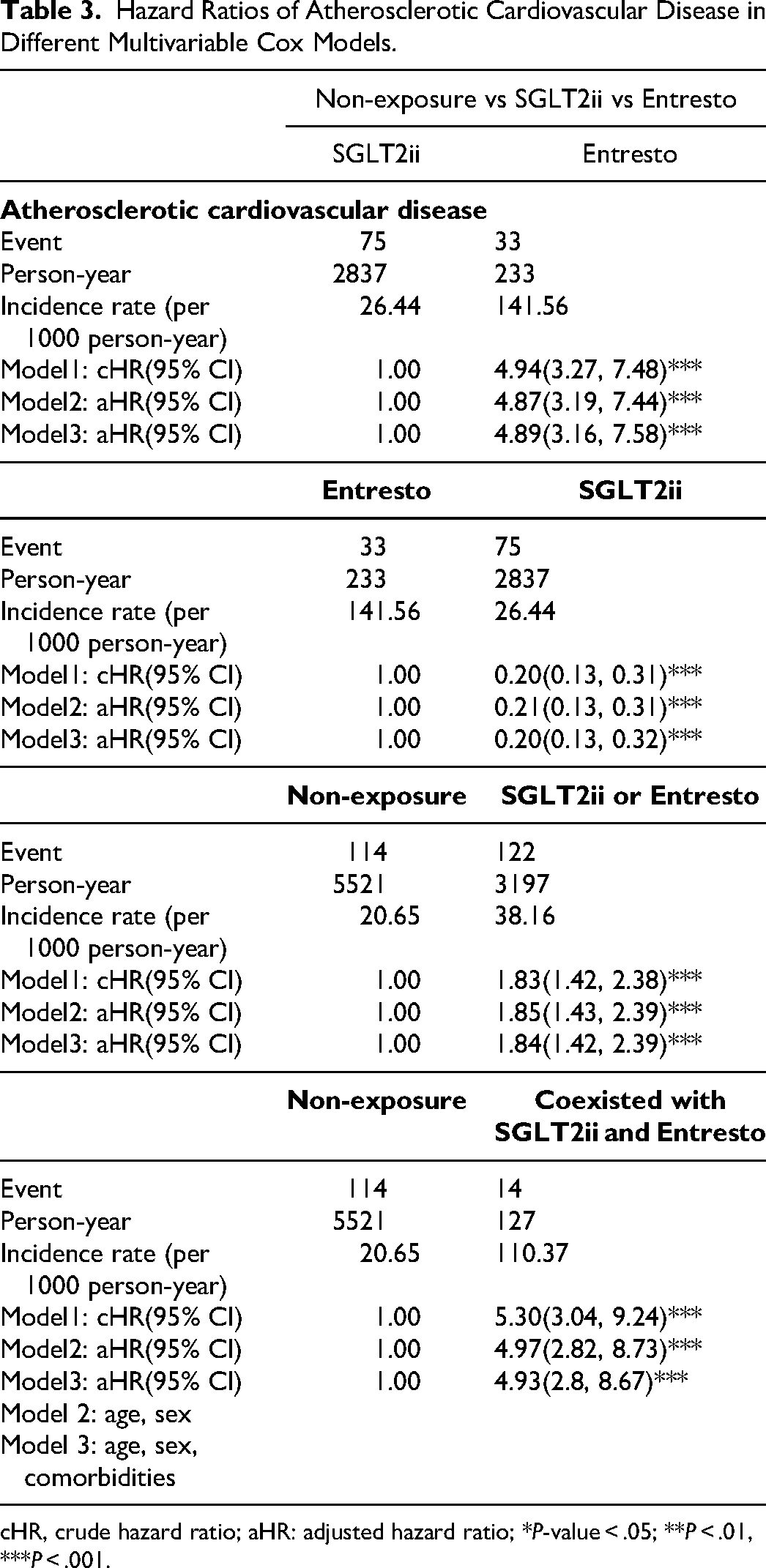
cHR, crude hazard ratio; aHR: adjusted hazard ratio; *P-value < .05; **P < .01, ***P < .001.
According to the data in Table 4, that the SGLT2i group and ARNI group both had better results than the control group in terms of heart failure hospitalization risk, CVD death risk, and total death risk. In addition, in the multivariate Cox proportional hazards regression model, the aHR of the SGLT2i group and ARNI group was also lower than that of the control group. Therefore, we can conclude that combined SGLT2i and ARNI therapy can significantly reduce ASCVD risk in patients with heart failure.
Hazard Ratios (HRs), and 95% Confidence Intervals (CIs) for Outcome Disease Among the Sampled Patients.

PY: person-years; IR: incidence rate per 1000 person-years; cHR: crude hazard ratio; aHR: adjusted hazard ratio.
The different risks among the sampled patients were also examined as shown in Table 4. Patients with diabetes and heart failure who used SGLT2i or Entresto had a higher risk of all-cause death (aHR,2.33; 95% CI, 1.96-2.78), non-fatal myocardial infarction (aHR, 1.6; 95% CI, 1.09-2.33), non-fatal stroke (aHR, 1.47; 95% CI, 1.08-2.02), cause of death in cardiovascular (aHR, 1.82; 95% CI, 1.05-83.14) and recurrence of heart failure (aHR, 1.81; 95% CI, 1.35-2.41) by multivariable analysis selecting the non-use of Entresto or SGLT2i patients with coexisting diabetes and heart failure cohort was selected as a reference. Patients with diabetes and heart failure who used SGLT2i had a significantly reduced risk of all-cause death (aHR,0.34; 95% CI, 0.26-0.45), non-fatal stroke (aHR, 0.37; 95% CI, 0.2-0.68), new renal replacement therapy (aHR, 0.35; 95% CI, 0.19-0.62), cause of death in cardiovascular (aHR, 0.15; 95% CI, 0.07-0.34) and recurrence of heart failure (aHR, 0.22; 95% CI, 0.13-0.36) by multivariable analysis selecting the patients with diabetes and heart failure cohort who used Entresto as reference.
According to Figure 2, diabetes and heart failure patients who received Entresto treatment had a significantly higher cumulative incidence of ASCVD than coexisting diabetes and heart failure patients who received SGLT2i treatment (log-rank test, P < .001). As shown in Figure 3, the Kaplan–Meier curve for the cumulative risk of ASCVD in diabetes and heart failure patients who received Entresto and SGLT2i was significantly higher than the cohort who did not receive ARNI and SGLT2i (log-rank test, P < .001).

The cumulative incidence of ASCVD in patients with both DM and HF.

The cumulative incidence of ASCVD in patients with both DM and HF.
Discussion
Benefits of Combining SGLT2i and ARNI
Our study aimed to investigate the potential benefits of combining SGLT2 inhibitors and ARNI therapies in reducing ASCVD risk among patients with both diabetes and heart failure. 15 By utilizing the National Health Insurance Research Database, which offers comprehensive healthcare information for a significant portion of the Taiwanese population, we aimed to provide insights into managing these conditions effectively.
In our analysis of 17 382 propensity score-matched patients, we observed distinct differences in ASCVD risk among those treated with SGLT2 inhibitors, Entresto, or a combination of both, 16 compared to those not receiving these treatments. The findings indicated a substantial reduction in cardiovascular death and heart failure hospitalization risk with combined SGLT2i and ARNI therapy. This contributes to the growing evidence supporting the synergistic effects of these treatments.17,18 SGLT2 inhibitors have shown multifaceted impacts on cardiovascular health, including diuretic and antihypertensive effects.19,20 Our study aligns with prior research indicating they might induce favorable metabolic shifts like improved lipid mobilization and glycemic control, 21 potentially explaining the reduced risk of all-cause death, stroke, and heart failure recurrence observed. 22
The benefits of ARNI therapy stem from counteracting maladaptive processes in heart failure, such as RAAS (renin–angiotensin–aldosterone system) and natriuretic peptide system activation.23 ARNIs inhibit neprilysin, preventing natriuretic peptide breakdown, leading to vasodilation, diuresis, and reduced sympathetic tone. Our study underscores ARNI therapy's value in mitigating cardiovascular risks in diabetes and heart failure patients. 19 While offering valuable insights, our study has limitations to consider. 24 The retrospective nature and reliance on claims data warrant cautious result interpretation. Also, the observed ASCVD risk reduction must be contextualized with potential adverse events and individual patient profiles.25,26
ASCVD Significance for Diabetic Individuals with Coronary Heart Disease
ASCVD's significance for diabetic individuals with coronary heart disease, systematic risk factor assessment is crucial for prevention and management.27,28 This study focused on SGLT2i and ARNI use, investigating their association with ASCVD risk in coexisting diabetes and heart failure patients.19,29 The analysis revealed reduced risk with SGLT2i use, and the combination of therapies improved management. SGLT2i and ARNI might have diverse mechanisms, but their combination enhances cardiovascular prognosis and myocardial function.11,30 The finding in Figures 2 and 3 suggests patients on Valsartan/Sacubitril had higher ASCVD incidence than SGLT2i users, and combining them increased ASCVD risk. SGLT2 inhibitors reduce heart failure and cardiovascular risks by glucose control, blood pressure reduction, and weight loss. ARNIs improve heart function and decrease risks by blocking the RAAS.31–34 The researchers conducted a comparison between patients using SGLT2 inhibitors or ARNI and those not using these medications. However, the study design excluded patients who used both SGLT2 inhibitors and ARNI in order to minimize potential confounding effects of combining different classes of medications. The study period was limited to two years (2017-2018) (Appendix, Taiwan Health insurance payment regulations).
The benefits of certain medications, especially those with mechanisms that involve structural changes in the heart, might take longer to manifest fully. The combination of SGLT2 inhibitors and ARNIs shows promise in reducing cardiovascular death and heart failure hospitalization, indicating a potential avenue for optimizing outcomes.35,36 More research, including randomized trials and long-term studies, is vital to fully understand the clinical implications and safety of this approach. As we refine cardiovascular risk management, studies like ours contribute to improved patient care and outcomes and It is important to note that these findings are based on observational data and do not prove causation. Further research is needed to fully understand the potential benefits and risks of combined SGLT2 and ARNI therapy for reducing ASCVD risk in patients with diabetes and heart failure.
Limitations of the Study
Firstly, the study was retrospective in nature, meaning that the researchers analyzed data that had already been collected rather than conducting a new study. This type of study design can be subject to biases and limitations in the data available.
Secondly, that the observed risk reduction in ASCVD needs to be considered within the context of potential adverse events and individual patient profiles. While the combination of SGLT2 inhibitors and ARNIs appears to hold promise in reducing cardiovascular death and heart failure hospitalization, it is important to consider the potential risks and benefits for each individual patient. The Entresto group with sample sizes of n = 6 for non-fatal myocardial infarction, n = 15 for non-fatal stroke, n = 16 for new renal replacement therapy, and n = 10 for cause of death in CAD, it is crucial to highlight the potential limitations associated with these small sample sizes for conducting robust multivariate modeling.
Finally, the study does not prove causation between the use of SGLT2 inhibitors or ARNIs and reduced ASCVD risk. The study was observational in nature, meaning that the researchers did not randomly assign patients to treatment groups. Instead, our analyzed data from patients who were already receiving treatment with SGLT2 inhibitors or ARNI and compared them to patients who were not receiving these treatments. This type of study design can be subject to confounding factors that may influence the results. Therefore, further research is needed to fully understand the potential benefits and risks of combined SGLT2 and ARNI therapy for reducing ASCVD risk in patients with diabetes and heart failure. The fact that ARNI was launched in Taiwan in 2017 (Appendix) means that the study is limited in terms of the duration of exposure to this medication, which means that patients who started taking ARNI during this time period would have had less exposure to the medication than patients who had been taking it for longer. This may limit the ability of the study to fully capture the long-term effects of ARNI on ASCVD risk reduction. However, it is worth noting that the study also included patients who were taking SGLT2 inhibitors, which have been available for longer than ARNI. Therefore, the study may still provide valuable insights into the relationship between these medications and ASCVD risk reduction, even if the duration of exposure to ARNI is limited.
Conclusion
Our study reinforces the value of ARNI therapy in mitigating cardiovascular risks in patients with diabetes and heart failure. While our study provides valuable insights into the potential benefits of combining SGLT2i and ARNI therapies, it is important to acknowledge certain limitations. The retrospective nature of our analysis and the reliance on claims data necessitate cautious interpretation of the results. Additionally, the observed risk reduction in ASCVD needs to be considered within the context of potential adverse events and individual patient profiles.
Our study contributes to the evolving understanding of effective therapeutic strategies for managing ASCVD risk in patients with diabetes and heart failure. 37 The combination of SGLT2 inhibitors and ARNIs appears to hold promise in reducing cardiovascular death and heart failure hospitalization, suggesting a potential avenue for optimizing patient outcomes. Further research, including randomized controlled trials and long-term follow-up studies, will be crucial to fully elucidate the clinical implications and safety of this combined approach. And the combination of SGLT2 inhibitors and ARNIs may have a synergistic and additive impact on cardiovascular outcomes, including reducing the risk of cardiovascular death and heart failure hospitalization. These findings are supported by several studies, but further research is needed to confirm their efficacy and determine the optimal treatment strategies for patients with diabetes and heart failure.
Footnotes
Abbreviations
Acknowledgements
This study is supported in part by Taiwan Ministry of Health and Welfare Clinical Trial Center (MOHW110-TDU-B-212-124004), China Medical University Hospital (DMR-111-105). The authors are grateful to Health Data Science Center, China Medical University Hospital for providing administrative, technical and funding support. The funders had no role in the study design, data collection and analysis, the decision to publish, or preparation of the manuscript. No additional external funding was received for this study.
Availability of Data and Material
We accessed the database from the Taiwan National Health Research Institutes. We are not eligible to duplicate and disseminate the database. For further access to the database, please contact the Ministry of Health and Welfare (Email: stcarolwu@mohw.gov.tw) for assistance. Taiwan Ministry of Health and Welfare Address: No.488, Sec. 6, Zhongxiao E. Rd., Nangang Dist., Taipei City 115, Taiwan (R.O.C.). Phone: + 886-2-8590-6848.)
Author Contributions
CH Lin and MH Lin had full access to the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Dr CH Lin and MH Lin contributed equally to this study and are considered Correspondence authors.
Concept and design: YW Lin, CH Lin, Chun-Hsiang Lin, MH Lin.
Acquisition, analysis, or interpretation of data: CH Lin, Chun-Hsiang Lin
Drafting of the manuscript: YW Lin, Chun-Hsiang Lin, CH Lin, MH Lin.,
Critical revision of the manuscript for important intellectual content: YW Lin, CH Lin, Chun-Hsiang Lin, MH Lin.
Statistical analysis: CL Lin
Administrative, technical, or material support: YW Lin, CH Lin, MH Lin,.
Obtained funding: MH Lin.
Supervision: CH Lin and MH Lin.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
To protect the patients’ privacy, all personal identification numbers were encrypted by the National Health Insurance before the data were released. The Taiwan National Health Research Institutes encrypts patient personal information to protect privacy and provides researchers with anonymous scrambled identification numbers associated with relevant claims information, including sex, date of birth, medical services received, and prescriptions. Therefore, patient informed consent is not required for authorized researchers to access this research database. The researchers of this study are not possible to contact any studied patient for obtaining informed consent,All identification numbers were replaced with surrogate numbers for insured people to protect the privacy. This study was approved by the Research Ethics Committee of China Medical University and Hospital (MOHW110-TDU-B-212-124004).No consents are required from the study population.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix: Health Insurance Payment Regulations in Taiwan

|
National Health Insurance Sacubitril + Valsartan (Entresto) Price/Pill/2.63 USD(76 NT dollars) |
| 1. For chronic heart failure that meet each criteria below since 2017 March 11th |
| (1) New York Heart Association (NYHA) class II, III, or IV symptoms, and with reduced ejection fraction (LVEF<=35%) |
| (2) Required to take a stable dose of a beta-blocker and an ACE inhibitor (or ARB) at least 4 weeks with symptoms |
| 2. Two pills per day |
| 3. Should not combine with ACEI; When switching to ARNI from an ACEI (but not an ARB), a 36-hour washout period is needed; Patients with history of angioedema are also not allowed to use. |
| 4. The Entresto Launching in Taiwan National Health Insurance reimbursement Mar.2017 |
Data source; https://www.nhi.gov.tw/QueryN_New/QueryN/Query1,Taiwan National Health Insurance Administration, 2017.
