Abstract
Background:
Transcatheter aortic valve replacement (TAVR) has been widely used to treat patients with aortic stenosis (AS). The anchoring of most transcatheter heart valves (THV) depends on friction with the native aortic valve (AV).
Methods:
A total of 9 patients with severe AS accepted TAVR using Xcor system with transapical access in our center. Clinical outcomes were collected at baseline, before discharge, and at the 30-day follow-up.
Results:
All patients achieved procedural success, postprocedural transesophageal echocardiography showed that all of the patients had no/trace paravalvular leakage. The mean AV pressure gradient decreased from 50 mmHg (range 18-76 mmHg) to 10 mmHg (range 8-14 mmHg) (P < .001). At the 30-day follow-up, all patients had an improvement of ⩾1 New York Heart Association functional class (P < .001). The average 6-minute walk distance (377.2 [range 330.0-430.0] m vs 276.1 [range 245.0-320.0] m, P < .001) and Kansas City cardiomyopathy questionnaire score (53.4 [range 45.0-62.0] vs 38.9 [range 35.0-43.0], P < .001) were both improved.
Conclusions:
Our early experience shows that the Xcor system is safe and feasible in the treatment of patients with severe AS.
Introduction
Transcatheter aortic valve replacement (TAVR) is a minimally invasive procedure in which the transcatheter heart valve (THV) is inserted into the aortic root and released to replace the native pathological aortic valve (AV). Since the first TAVR was successfully completed in 2002, 1 its safety and efficacy have been confirmed by several large randomized controlled trials.2,3 However, most of the current TAVR systems rely on the unique properties of the THV material by using friction to anchor the implanted THV in the native AV.4,5 Because the native AV is not removed and often presents an asymmetrical morphology, the THV is often pulled or squeezed by the original structures after implantation, resulting in an increased risk of poor coaxiality, THV displacement, and even THV malposition.4,5 Finally, complications such as device embolism, a new permanent pacemaker implant, and decreased THV durability may affect the long-term prognosis of the patient.4,5
The Xcor System (Saint Medical Technology Co., LTD., Nanjing, China) is a new-generation THV with a uniquely designed “supporting arm” that is loaded with 6 arms in the middle area of the THV. This design provides a full fit between the support arm and the aortic root for accurate positioning and strong anchoring. This single-center study was designed to evaluate the early outcomes of the Xcor system in 9 patients with severe aortic stenosis (AS).
Methods
Study population
From September 2022 to June 2023, a total of 9 patients with severe AS were accepted for TAVR using Xcor system with transapical access at Xijing Hospital. Inclusion criteria were (1) age ⩾ 65 years; (2) severe AS assessed by transthoracic echocardiography or patients with low flow velocity and a low-pressure gradient assessed for severe AS using the dobutamine stress test; (3) New York Heart Association (NYHA) functional class ⩾ II; and (4) surgical contraindications or high risk. Exclusion criteria included (1) acute myocardial infarction occurring within 1 month before the procedure; (2) preprocedural imaging assessment of aortic root anatomy indicating that conditions were not suitable for transapical TAVR; (3) ascending aortic aneurysm diameter >50 mm; (4) coagulation dysfunction; (5) left ventricular ejection fraction <20%; (6) transthoracic echocardiography showed the presence of an intracardial mass, thrombus, or neoplasm; (7) expected survival time after the procedure was less than 12 months. This study complied with the Declaration of Helsinki and was approved by the Xijing Ethics Commission. All patients provided written informed consent and gave permission for subsequent data collection.
Device
The Xcor system is carried out procedures using transapical access, which includes the innovative supporting arm design. The supporting arm controls exactly how the THV is released into the annulus. As shown in Figure 1, the THV is fixed between the narrowest position of the aortic root and the narrowest position of the annulus through the transapical approach to achieve a full fit between the supporting arms and the aortic root. The Xcor system comprises the prosthesis with a delivery system that includes an implant and a compression cone. The prosthesis is available in 23-, 26-, and 29-mm sizes.

Xcor transcatheter heart valve. (A) The prosthesis. (B) The transcatheter heart valve is fixed between the narrowest position of the aortic root and the narrowest position of the annulus to achieve a full fit between the supporting arms and the aortic root.
Key aspect of the Xcor stents is the above-mentioned expandable arms. Designed with no undercut, allowing therefore for full re-sheating and reposition, their role is to assure the correct positioning, self-alignment and anchoring of the prosthesis. Using a form-fitting principal the arms allow to reduce the maximum radial force of the conical inflow of the stent. Meanwhile, assuring a supra-annular valve function of the 3 bovine pericardial leaflets the expendable arms in conjunction with large cells preserve and facilitate coronary access. Central co-axial self-alignment and the Dacron outer-skirt with a total height of 10 to 11 mm confer an excellent seal preventing paravalvular leakage. All valve sizes can be used with the same 23 French delivery system. Implantation is executed in a top-down and pullback fashion with good tactile feedback once the arms are deployed in the optimal position.
Preprocedural imaging assessment
Transthoracic echocardiography (TTE) was used for preprocedural assessment of the AV, calcification, and the left cardiac system function, and the annulus diameter was measured on the short axis (Figure 2A). Preprocedural computed tomography angiography (CTA) was performed in all patients to assess the anatomy of the AV and adjacent tissues, including measurements of the annulus, left ventricular outflow tract, sinus junction diameter, and coronary artery heights (Figure 2B-E). The angle between the major axis of the aortic root and the left ventricular outflow tract was measured to determine the optimal approach for the Xcor system during TAVR (Figure 2F). Coronary angiography was used to rule out severe coronary artery disease.

Preprocedural imaging assessments. (A) Preprocedural transesophageal echocardiography assessment. (B, C) The aortic annulus and left ventricular outflow tract were assessed using computed tomography angiography scans. (D, E) The left and right coronary artery heights were measured before the procedure. (F) The projection angle was determined.
Procedures
TAVR was performed with the patient under general anesthesia. A contrast catheter was inserted into the aortic root after the right femoral artery was punctured. The apical position was located by digital subtraction angiography (DSA). An incision of about 3 cm was made into the chest through the fourth left lateral intercostal space. The pericardium was incised and suspended. The apex cordis was successfully punctured into the left ventricle; the pressure gradient between the left ventricle and the aorta was measured; and the multifunctional catheter guided the wire into the descending aorta to replace the 2.6-m straight head guide wire into the right femoral artery, and then the Lunderquist guide wire was inserted into the multifunctional catheter. Subsequently, the Xcor system was fed into the AV along the Lunderquist guide wire, and the anchor point was placed at the base of the aortic sinus to release the THV (Figure 3A-D). The pacing reached 180 beats/min, and the prosthesis was released successfully. Immediately after the procedure, DSA and transesophageal echocardiography (TEE) were studied to verify the position and morphology of the THV (Figure 3E and F).

Procedural details using the Xcor system. (A) The supporting arms are released. (B) The prothesis is gradually released. (C) The prosthesis is fully unfolded. (D) Postdilation. (E, F) Digital subtraction angiography and transesophageal echocardiography show the position and morphology of the prosthesis.
Follow-up and data collection
Each case has a dedicated form detailing baseline characteristics, procedural information, and planned follow-up outcomes. Follow-up was carried out through a clinical visit with the patient after a predetermined period of time. TTE was performed at the 30-day follow-up to determine the extent of the improvement of the cardiac functions. In addition, all patients underwent CTA at the 30-day follow-up. Clinical Outcomes were analyzed according to the Valvular Academic Research Consortium-3 criteria. 6 Quality of Life were evaluated using 6-minute walk test and Kansas City cardiomyopathy questionnaire. In addition, the criteria of the Acute Kidney Injury Network was used to evaluate the severity of renal failure. 7
Statistical analysis
Continuous variables are expressed in quartile ranges, and categorical variables are expressed in frequency and percentage. The Student t-test and the Mann-Whitney U test were used to compare the normally distributed continuous variables and the continuously non-normally distributed variables, respectively. Bilateral P-values < .05 were considered statistically significant. All statistical analyses were performed using SPSS software version 26.0 (SPSS, Inc., Armonk, NY, USA).
Results
Baseline characteristics
Detailed patient characteristics are shown in Table 1. Before the procedures, all patients were evaluated by the interdisciplinary cardiac team and were considered unable to undergo surgical aortic valve replacement due to comorbidities or high risk. The median age of the 9 patients with severe AS was 69.0 years (range 65.0-75.0 years); 66.7% were female; and the Society of Thoracic surgery predicted risk of mortality was 6.3% (range 5.1%-7.7%). Except for patient #4, who was New York Heart Association (NYHA) functional class III, the other 8 patients had symptoms of severe heart failure (NYHA functional class IV). Patient #2 and patient #9 each had a history of stroke. The median quality-of-life (QoL) score was 675 (range 450-825).
Baseline characteristics.

Abbreviations: CABG, coronary artery bypass graft; NYHA, New York Heart Association; PCI, percutaneous coronary intervention; PPI, permanent pacemaker implantation; QoL, quality of life; STS, Society of Thoracic Surgeons.
The results of preprocedural imaging measurements are listed in Table 2. A total of 33.3% (n = 3) of the patients had tricuspid AV; 55.6% (n = 5) had bicuspid AV; and only 1 patient (patient #1) had the quadricuspid AV. The mean annular diameter was 24.3 mm (range 20.3-27.2 mm). Furthermore, the median peak flow velocity was 4.5 m/s (range 3.2-6.0 m/s), and the median value of the mean pressure gradient was 50 mmHg (range 18-76 mmHg). Notably, 66.7% (n = 6) of the patients had ⩾ moderate aortic regurgitation, and 55.6% (n = 5) had ⩾ moderate mitral regurgitation.
Preprocedural imaging assessments.

Abbreviation: AV, aortic valve.
Procedural outcomes and THV functions
All (100%) patients were successfully implanted with the Xcor prosthesis. The THV functions immediately after implantation are shown in Table 3. Two 23-mm, one 26-mm, and six 29-mm prostheses were implanted based on the annulus diameter measured by CTA and TEE. All procedures were performed without cardiopulmonary bypass. No patient was converted to open surgery, and no major adverse cerebrovascular events occurred. No coronary artery obstruction or THV displacement occurred. The total operating time was 95 minutes (range 65-115 minutes); the fluoroscopy time was 8.9 minutes (range 7.4-13.0 minutes); and the contrast volume was 96 mL (range 76-132 mL). Postprocedural TEE showed that all (100%) patients had no/trace PVL. In addition, the mean AV pressure gradient decreased from 50 mmHg (range 18-76 mmHg) to 10 mmHg (range 8-14 mmHg) (P < .001).
Procedural details and transthoracic echocardiography evaluation before discharge.
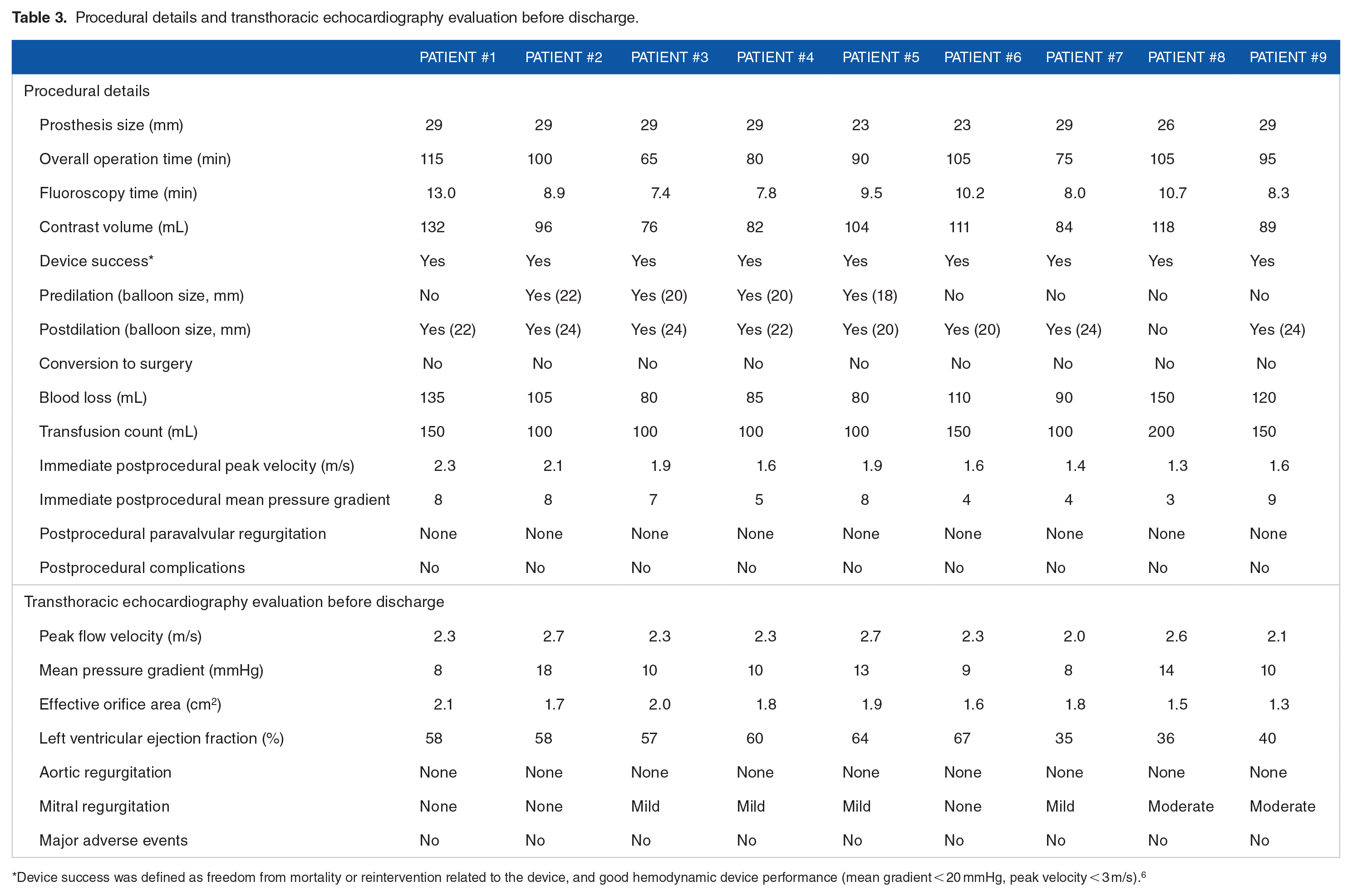
Device success was defined as freedom from mortality or reintervention related to the device, and good hemodynamic device performance (mean gradient < 20 mmHg, peak velocity < 3 m/s). 6
Clinical outcomes
The 30-day follow-up data are shown in Table 4. There were no deaths, neurological complications (including stroke and transient ischemic attacks), or myocardial infarctions during hospitalization. No patients required permanent pacemaker implants during the periprocedural period. Patient #6 suffered stage 2 acute kidney injury after the procedure and recovered quickly after discharge without dialysis. At the 30-day follow-up, all (100%) patients were NYHA functional class II (Figure 4A), and no cases occurred with ⩾ mild PVL (Figure 4B). Notably, the average 6-minute walk distance (377.2 [range 330.0-430.0] m vs 276.1 [range 245.0-320.0] m, P < .001) and Kansas City cardiomyopathy questionnaire score (53.4 [range 45.0-62.0] vs 38.9 [range 35.0-43.0], P < .001) were both improved. (Figure 4C-D).
30-day follow-up clinical outcomes.

Abbreviations: NYHA, New York Heart Association; QoL: quality of life.

Improvements in prognosis at the 30-day follow-up compared to pre-transcatheter aortic valve replacement results. (A) The probability of the New York Heart Association functional class. (B) The probability of paravalvular leakage. (C) The improvement of 6-minute walk distance. (D) The improvement of Kansas City cardiomyopathy questionnaire score.
Discussion
Our study confirms the feasibility of the transapical implantation of the Xcor system in high-risk patients with predominantly severe AS. According to the latest guidelines, TAVR is a reasonable alternative to surgical aortic valve replacement for AS patients due to its advantages of no thoracotomy, no cardiac arrest, and no cardiopulmonary bypass.8,9 However, the existing THVs are fixed in the implanted position by the friction generated between the THV and the aortic root.4,5 However, due to the complexity of the AV anatomy, if the THV is positioned and fixed only by friction, it may be pulled or squeezed by the native AV anatomy to varying degrees after it is implanted.4,5 Importantly, underanchoring may lead to an increased risk of THV displacement, whereas oversizing might lead to annular rupture.4,5 The preceding factors decrease the success rate of TAVR and increase the intraprocedural complications to a certain extent.
Through the implementation of this design, the Xcor system also achieves a self-centered effect, that is, the supporting arms contact the native annulus during release, forming a longitudinal centering effect, ensuring good coaxiality while fully conforming to the direction of the hemodynamics. Importantly, the Xcor system precisely locates the leaflet junction by introducing the supporting arms. When the THV is loaded in a specific orientation, the controlled THV release can be achieved during the procedure to achieve the goal of commissural alignment.
Meanwhile, the Xcor system has a lower THV height, which may avoid the risk of being easily breakable that may exist when a high THV is stretched. Procedural factors like implant depth and procedural manipulations, such as re-sheathing have been shown to interfere with the conduction system and can result in the subsequent need for permanent pacemaker implantation (PPI). In the present study, no case occurred with PPI. It is much lower than observed after implantation of any other TAV device without the consideration of the limited sample size (n = 9). Reported PPI rates for the Evolut PRO (11.8%), ACURATE NEO2 (15.0%), and SAPIEN 3 (13.3%) in prospective studies with a similar high- or extreme-risk population are considerable higher.10-12
Additionally, the incidence of PVL after TAVR is 12% to 30%, which is significantly higher than that of SAVR. 13 More than moderate PVL due to poorer THV fitting is a major risk factor for poor prognosis. 13 Central co-axial self-alignment of Xcor system, along with the inclusion of a Dacron outer-skirt measuring a total height of 10 to 11 mm, bestows an excellent sealing capability, effectively averting PVL. From the outcomes of our study, no patient had no/trace PVL after the prosthesis was implanted. Importantly, the mean pressure gradient decreased significantly after procedures.
In our study with limited sample size, event rates were comparable with other published outcomes of the SAPIEN 3, Evolut PRO, and ACURATE neo2 valves.10 -12 For a high or extreme surgical risk population and suggests that the novel Xcor device performs similarly to other contemporary THVs. Furthermore, all (100%) patients had improved ⩾1 NYHA class at the 30-day follow-up. It is noteworthy that the average 6-minute walk distance (377.2 [range 330.0-430.0] m vs 276.1 [range 245.0-320.0] m, P < .001) and Kansas City cardiomyopathy questionnaire score (53.4 [range 45.0-62.0] vs 38.9 [range 35.0-43.0], P < .001) were both improved. Considering that patients with severe AS often have a heavier burden of calcification, thus we decided to adopt the transapical approach when designing this device to improve the procedural success rate in the clinical trial. However, the surgical incision through the approach is larger than that through the transfemoral artery approach, and the damage to the heart is also greater. Therefore, in the next step, the researchers will focus on developing a transfemoral approach version of the Xcor system. Additionally, transapical approach is currently only available with the Sapien device (Irvine, California, USA) which is balloon-expandable system, and the Xcor system offers a self-expandable option. As we know, the Sapien device allows approach with an 18Fr sheath, whereas the Xcor system requires a 23Fr sheath, so a smaller diameter is preferred for less invasive procedures, and the incidence of related complications are needed to analyze in the further study.
In summary, our early experience shows that the Xcor system is efficacious and safe for the treatment of patients with severe AS, can significantly improve the symptoms and hemodynamics, and exhibits excellent performance in reducing potential PVL, coronary artery obstruction, and conduction block. Meanwhile, the Xcor system is less difficult to operate, and interventional cardiologists may become proficient after a short learning curve. In the future, we plan to conduct further studies by expanding the sample size and extending the follow-up time to confirm its efficacy and reliability.
Study limitations
The study has the following limitations: First, the small sample size caused the Xcor system to temporarily not be validated in a larger patient population. Second, the follow-up time of this study was relatively short, and only the early functional performance after the THV was implanted was observed. Next, extended follow-up is needed to observe the function and durability of the THV. In addition, we conducted a single-center study, so more surgeons from different centers are needed to evaluate the operability and safety of the Xcor system in the future.
Conclusions
We introduced the new-generation Xcor prosthesis and reported our experience and early outcomes with this device in patients with severe AS. Because of the specific design of this device, accurate deployment and reinforced anchoring of the prosthesis produced excellent early outcomes.
Footnotes
Authors’ Contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China (No. 2020YFC2008100); the Development and Transformation of New Technology and Construction of Precision Diagnosis and Treatment System for Transcatheter Interventional Diagnosis and Treatment of Structural Heart Diseases (2022YFC2503400); and the Research on Key Techniques of Minimally Invasive Treatment for Valvular Heart Diseases (2023-YBSF-105); National Natural Science Foundation of China (82370375).
Abbreviations
AS aortic stenosis
AV aortic valve
TTE transthoracic echocardiography
CTA computed tomography angiography
DSA digital subtraction angiography
NYHA New York Heart Association
QoL quality-of-life
TAVR transcatheter aortic valve replacement
TEE transesophageal echocardiography
THV transcatheter heart valve
Availability of data and material
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Ethics Approval
The studies involving human participants were reviewed and approved by Clinicaltrials Organization: Xijing Hospital, Air Force Medical University.
Consent to Participate
The patients/participants provided their written informed consent to participate in this study.
Consent for Publication
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Clinical Trial Registration
ClinicalTrials.gov Protocol Registration System (NCT02917980).
