Abstract
Objective:
To investigate the clinical value of echocardiography in the preoperative assessment, intraoperative monitoring, and postoperative follow-up of patients with aortic valve disease (AVD) undergoing transcatheter aortic valve replacement (TAVR).
Materials and Methods:
This retrospective study included clinical data of 158 patients with AVD who underwent TAVR. Patients were divided into three groups based on the type of AVD. The perivalvular structure of the patient was evaluated preoperatively using echocardiography and compared with multi-slice computed tomography (MSCT) measurements. Echocardiography was used to dynamically monitor the entire surgical process. Each patient was followed up for 1 month.
Results:
The success rate of TAVR was 94.9%. Preoperative measurements of the paravalvular (PVL) structure indicated good agreement between transesophageal echocardiography (TEE) and MSCT. Real-time TEE was used to adjust the location of the artificial valve to reduce leakage before completing TAVR, and the final incidence of moderate and above PVL was 1.3%. Data obtained 1 month after TAVR revealed that both the structure and function of the patient’s heart underwent benign reverse remodeling compared with those before surgery.
Conclusion:
Echocardiography played an important role in the entire procedure and may provide a basis for evaluating the efficacy and prognosis of TAVR.
Keywords
Aortic valve disease (AVD) is a common heart valve disease that includes aortic stenosis (AS) and aortic regurgitation (AR). In China, older patients with AS are more prone to AR. Statistically, AR is the most common type of AVD in China. 1 Earlier, AVD could only be treated through surgical intervention. Ever since Cribier et al. 2 performed the first human transcatheter aortic valve replacement (TAVR) in 2002, it has become an effective alternative treatment for patients with AVD who are at high risk or contraindicated for traditional surgery. Recently, with the rapid advancements in the TAVR technology and equipment in China, the number of TAVR operations has increased annually, benefiting patients with AVD. As a noninvasive examination method, echocardiography is of great value for the early detection of AVD. This study aimed to investigate the value of echocardiography in the preoperative evaluation, intraoperative monitoring, and postoperative follow-up of patients with different types of AVD who underwent TAVR, with the expectation of providing a basis for determining the efficacy and prognosis of TAVR.
Materials and Methods
Inclusion and Exclusion Criteria
This retrospective study included 158 patients with AVD who underwent TAVR at the Cardiology Centre of Yijishan Hospital, Wannan Medical College. The inclusion criteria were as follows: (1) maximum aortic valve (AV) blood flow velocity (AV Vmax) ≥4 m/s, mean AV pressure gradient (AVPG mean) ≥ 40 mmHg, or AV area ≤ 1.0 cm2; (2) severe AR with vena contracta width (VCW) > 6 mm, pressure halving time (PHT) < 200 ms, effective regurgitant orifice area (EROA) > 0.30 cm3, regurgitant volume (RVol) > 60 mL/beat; (3) relevant clinical symptoms (chest tightness and history of syncope) and New York Heart Association (NYHA) cardiac function classification ≥ grade II; (4) contraindications or high risk for surgery or other risk factors; (5) life expectancy > than 1 year; and (6) patients with bioprosthetic structural valve deterioration who are at high risk or contraindicated for re-surgery. The exclusion criteria were as follows: (1) fresh thrombus in the left ventricle, (2) acute myocardial infarction, (3) severe left ventricular (LV) outflow tract obstruction, and (4) postoperative life expectancy < than 1 year. All medical records were selected simultaneously based on the same inclusion and exclusion criteria. The selection process is illustrated in Figure 1.
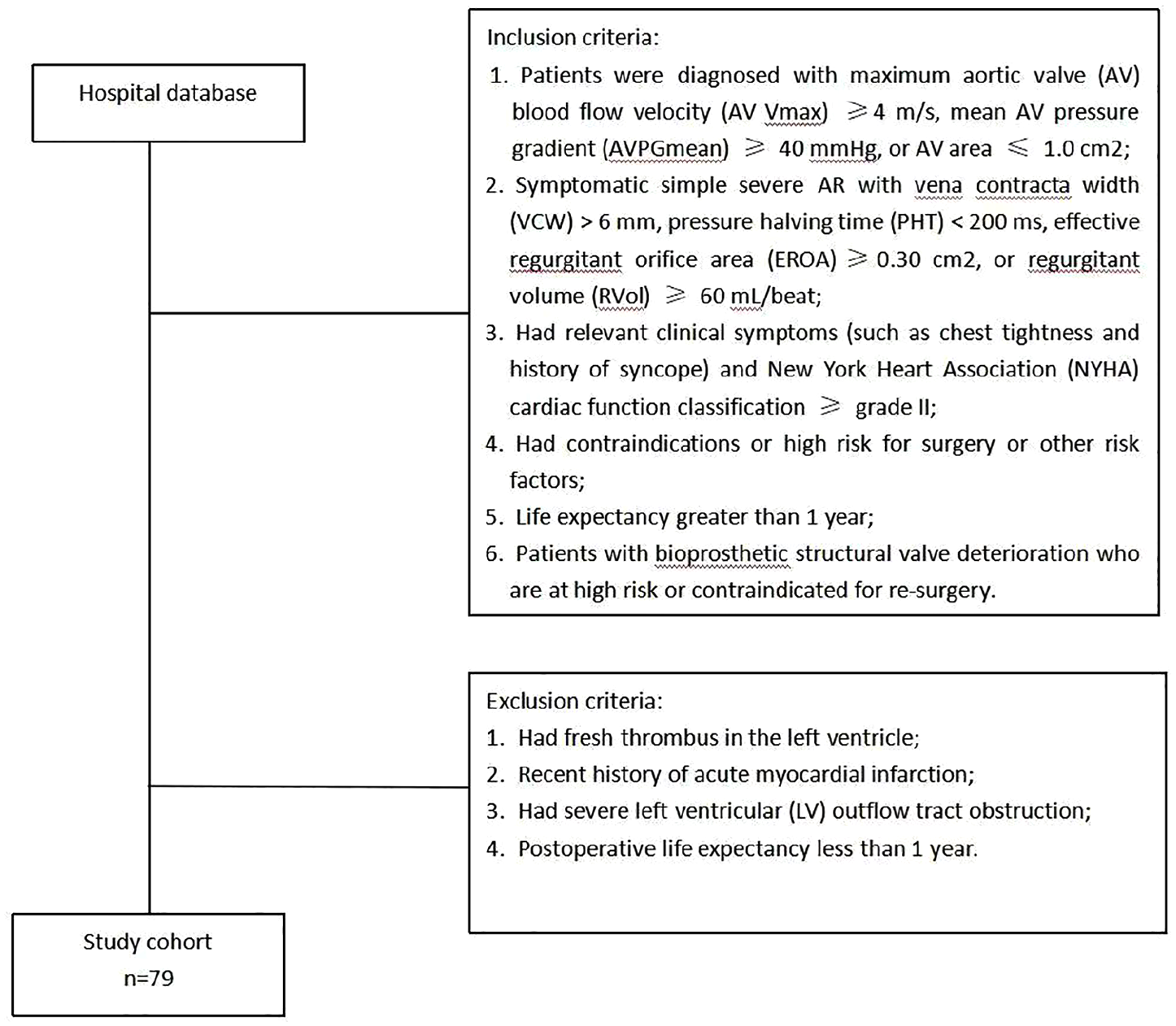
Flow chart demonstrating the inclusion and exclusion criteria in patients with aortic valve disease (AVD).
All patients were implanted with a Vita-Flow valve, a second-generation self-expanding aortic valve independently developed from MicroPort in China, made of an anti-calcification bovine pericardium sewn on a nitinol stent. Four valve sizes were used: 21, 24, 27, and 30 mm.
Ethical Approval
This study was approved by the Medical Ethics Committee of the Yijishan Hospital (IRB no. 2024-0426). The requirement for informed consent was waived by the Medical Ethics Committee owing to the retrospective design of the study and data analysis. All patient information was de-identified for the study. This study was conducted in accordance with the principles of the Declaration of Helsinki. This study conformed to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines. 3
Data Collection
Basic patient data such as age, sex, major preoperative comorbidities, and postoperative complications were obtained and recorded. All patients underwent multi-slice computed tomography (MSCT) 3 to 5 days before the procedure, and experts on the TAVR team analyzed the maximum and minimum diameters, area and circumference of the aortic annulus, and the left and right coronary ostial heights to select the appropriate valve size for each patient.
An EPIQ 7C (Philips Medical, Amsterdam, the Netherlands) ultrasound equipment system was used, along with an S5-1 transthoracic transducer and an X7-2t transesophageal echocardiography (TEE) transducer with a frequency of 1 to 5 MHz.
For the preoperative transthoracic echocardiography (TTE) evaluation, conventional echocardiographic parameter measurements, including LV end-diastolic diameter (LVEDD), interventricular septum thickness (IVST), and posterior wall thickness (PWT) measured in the long-axis view of the left ventricle next to the sternum; LV ejection fraction (LVEF) measured using the biplane Simpson’s method in apical four-chamber and two-chamber views; and pulmonary artery systolic pressure (PASP) measured using the tricuspid regurgitation pressure gradient were obtained. For AV-related parameters, continuous-wave (CW) Doppler was used to measure AV Vmax and AVPG mean in the apical five-chamber view (to record blood flow direction parallel to the ultrasound beam), and for Group II, EROA, RVol, and AR width were used to assess the degree of AR.
Transesophageal echocardiography was performed before the intervention. Under general anesthesia, the sonographer inserted an esophageal ultrasound transducer to obtain long- and short-axis views of the aortic root in the middle esophagus, after which three-dimensional (3D) zoom and full volume imaging were performed. The 3D TEE MultiVue multiplanar positioning and cutting were utilized to obtain the shape of the aortic annulus to measure the area, circumference, and maximum and minimum diameters. The image was adjusted to measure the distance between the aortic opening and valve annulus. Immediate postoperative assessments of the prosthetic valve position, morphology and function, prosthetic AR, and LV function were performed.
Transthoracic echocardiography was performed 1 month after the procedure to assess overall cardiac function and prosthetic valve morphology and function.
Statistical Analyses
All data were analyzed using SPSS 25.0. The measurement data are expressed as (x±s), and count data are expressed as ratio. The paired
Results
Basic Information of Patients in the Three Groups
All the patients underwent TAVR. Fifty-two patients with severe AS (including those with pure severe AS and severe AS combined with mild AR) were included in group I, 62 patients with severe AR (including those with pure severe AR and severe AR combined with mild stenosis) were included in group II, and the remaining 44 patients with AS combined with AR (AS+AR) were included in group III. There were 88 (55.7%) males and 70 (44.3%) females, with an average age of 73.48 ± 8.21 years. Before surgery, there were two cases of severe AR recurrence after the previous surgical AV bioprosthesis, 12 cases of chronic renal insufficiency, 56 cases (35.4%) of moderate-or-more mitral regurgitation, 30 cases of moderate-or-more tricuspid regurgitation (18.9%), and 22 cases (13.9%) of moderate-or-more pulmonary hypertension (Table 1).
The Descriptive Data of the Three Groups of Patients, in the Study Cohort.
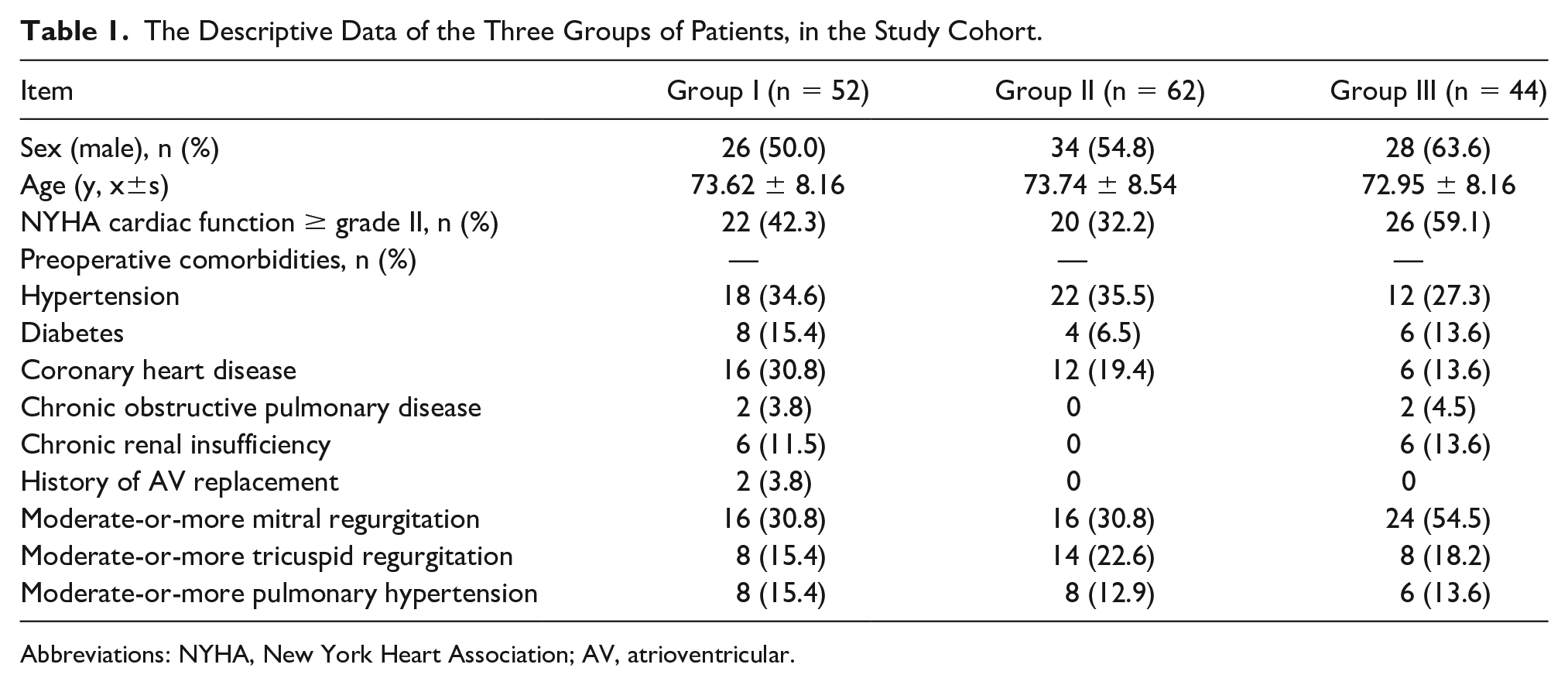
Abbreviations: NYHA, New York Heart Association; AV, atrioventricular.
Surgical Situation
Four patients underwent surgical AV replacement (SAVR) due to hemodynamic instability: two patients underwent SAVR due to prolapse of the prosthetic AV into the left ventricle due to deep implantation, and the operation was terminated in two patients because the prosthetic AV could not be anchored. In the remaining 150 patients, TAVR was successfully performed through the femoral artery. All the patients were monitored by using TEE; four patients underwent TEE-guided transseptal puncture for retrograde crossing of the valve, and 12 underwent valve-in-valve (VinV) implantation. Immediate postoperative evaluation revealed two cases of moderate paravalvular leakage (PVL) in group I, eight cases of mild-to-moderate PVL (group I: group II: group III = 3:2:3), 28 cases of mild PVL (group I: group II: group III = 12:9:7), 16 cases of trace PVL (group I: group II: group III = 6:7:3), and 96 cases of no PVL (group I: group II: group III = 27:38:31).
Preoperative Assessment of the Aortic Root Anatomy by Using RT-3DTEE and MSCT
There were no significant differences between RT-3DTEE and MSCT in preoperative maximum diameter, minimum diameter, area, circumference, and left and right coronary ostial heights of the aortic annulus (all
The Comparison of the Preoperative Assessment of the Aortic Root Anatomy Using RT-3DTEE and MSCT.
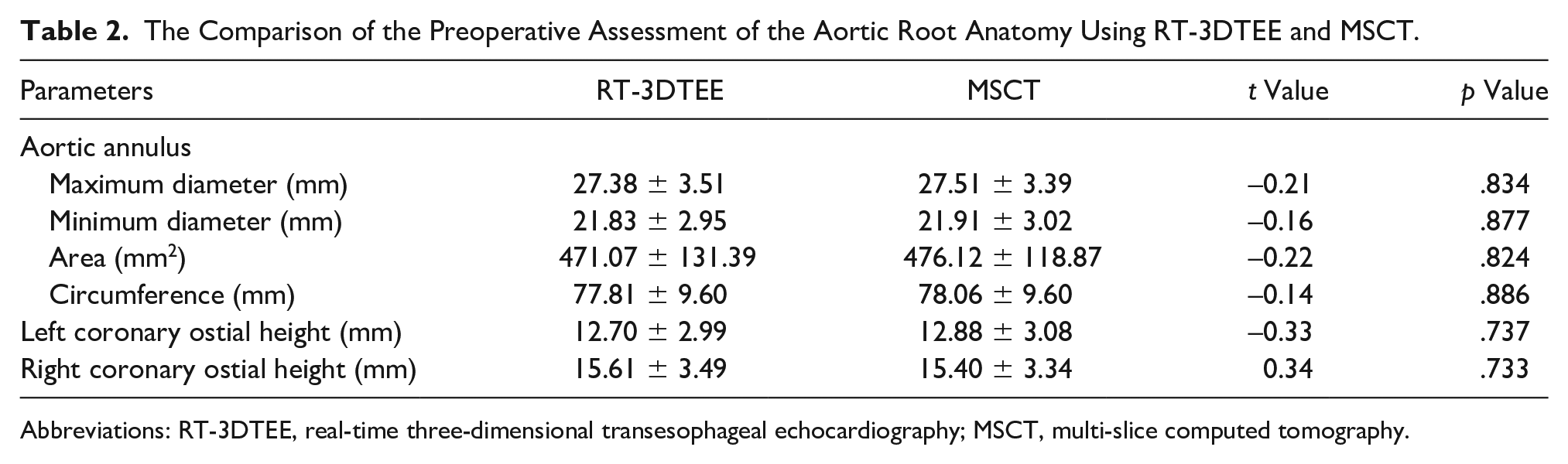
Abbreviations: RT-3DTEE, real-time three-dimensional transesophageal echocardiography; MSCT, multi-slice computed tomography.
Basic Clinical Status of Patients After TAVR
Five patients died within 1 week after surgery (two patients died due to aortic dissection and three patients died due to preoperative renal insufficiency and postoperative multiple organ failure), and 26 patients had a permanent pacemaker implanted (20 of these patients were in group II).
Comparison of Preoperative and 1-Month Postoperative TTE Indices Among the Three Groups
Compared with the preoperative data, the 1-month postoperative TTE indicated that in group I, the AV Vmax and AVPG mean decreased significantly (
The Diagnostic Results, of Patients in Group I, Based on Echocardiography.
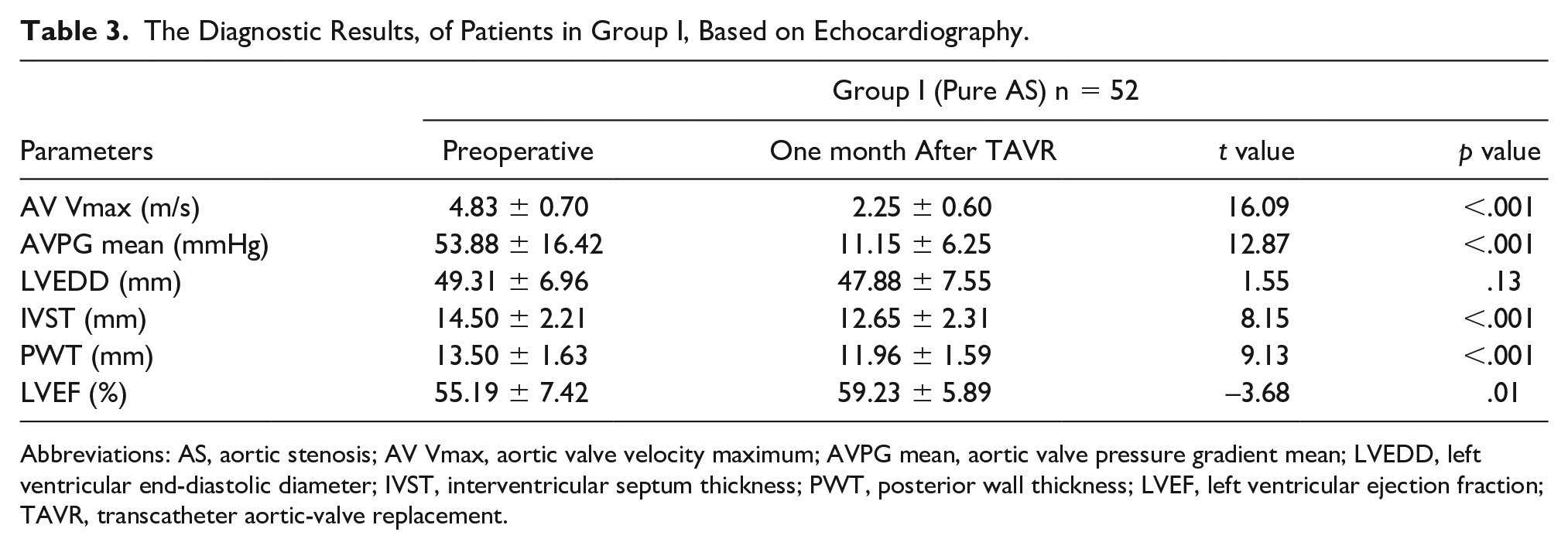
Abbreviations: AS, aortic stenosis; AV Vmax, aortic valve velocity maximum; AVPG mean, aortic valve pressure gradient mean; LVEDD, left ventricular end-diastolic diameter; IVST, interventricular septum thickness; PWT, posterior wall thickness; LVEF, left ventricular ejection fraction; TAVR, transcatheter aortic-valve replacement.
The Diagnostic Results of Patients, in Group II, Based on Echocardiography.
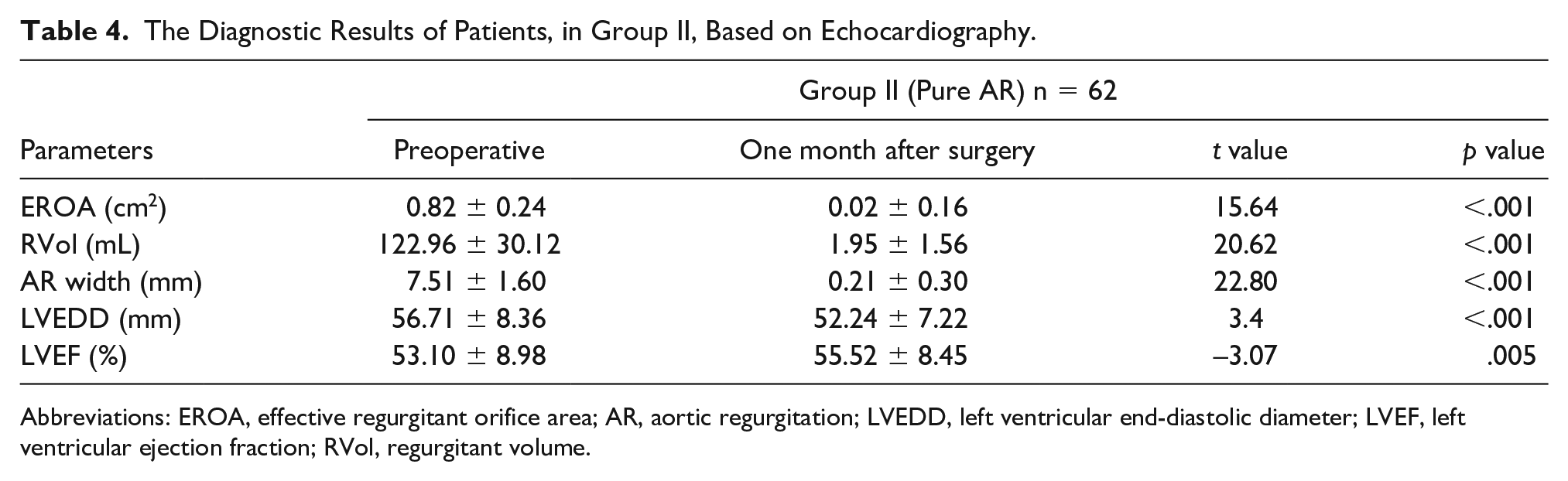
Abbreviations: EROA, effective regurgitant orifice area; AR, aortic regurgitation; LVEDD, left ventricular end-diastolic diameter; LVEF, left ventricular ejection fraction; RVol, regurgitant volume.
The Diagnostic Results, of Patients in Group III, Based on Echocardiography.
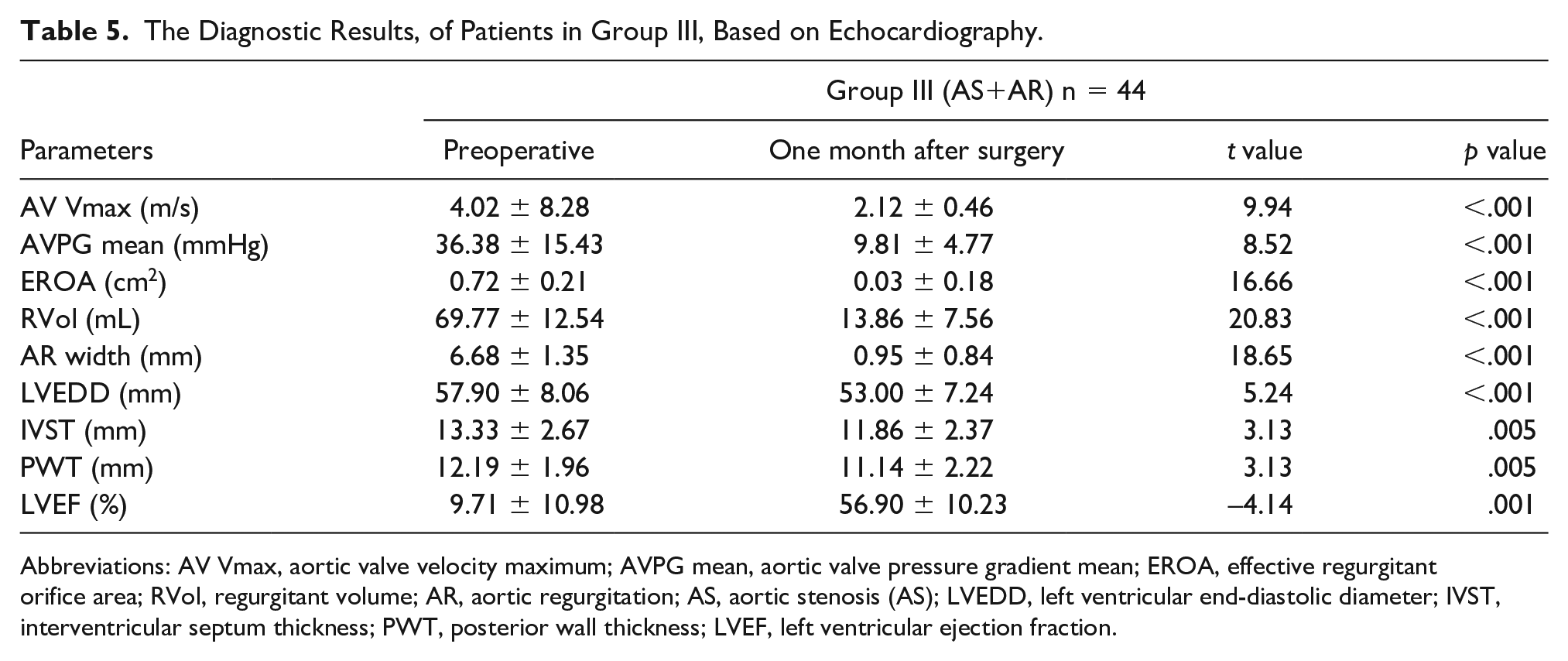
Abbreviations: AV Vmax, aortic valve velocity maximum; AVPG mean, aortic valve pressure gradient mean; EROA, effective regurgitant orifice area; RVol, regurgitant volume; AR, aortic regurgitation; AS, aortic stenosis (AS); LVEDD, left ventricular end-diastolic diameter; IVST, interventricular septum thickness; PWT, posterior wall thickness; LVEF, left ventricular ejection fraction.
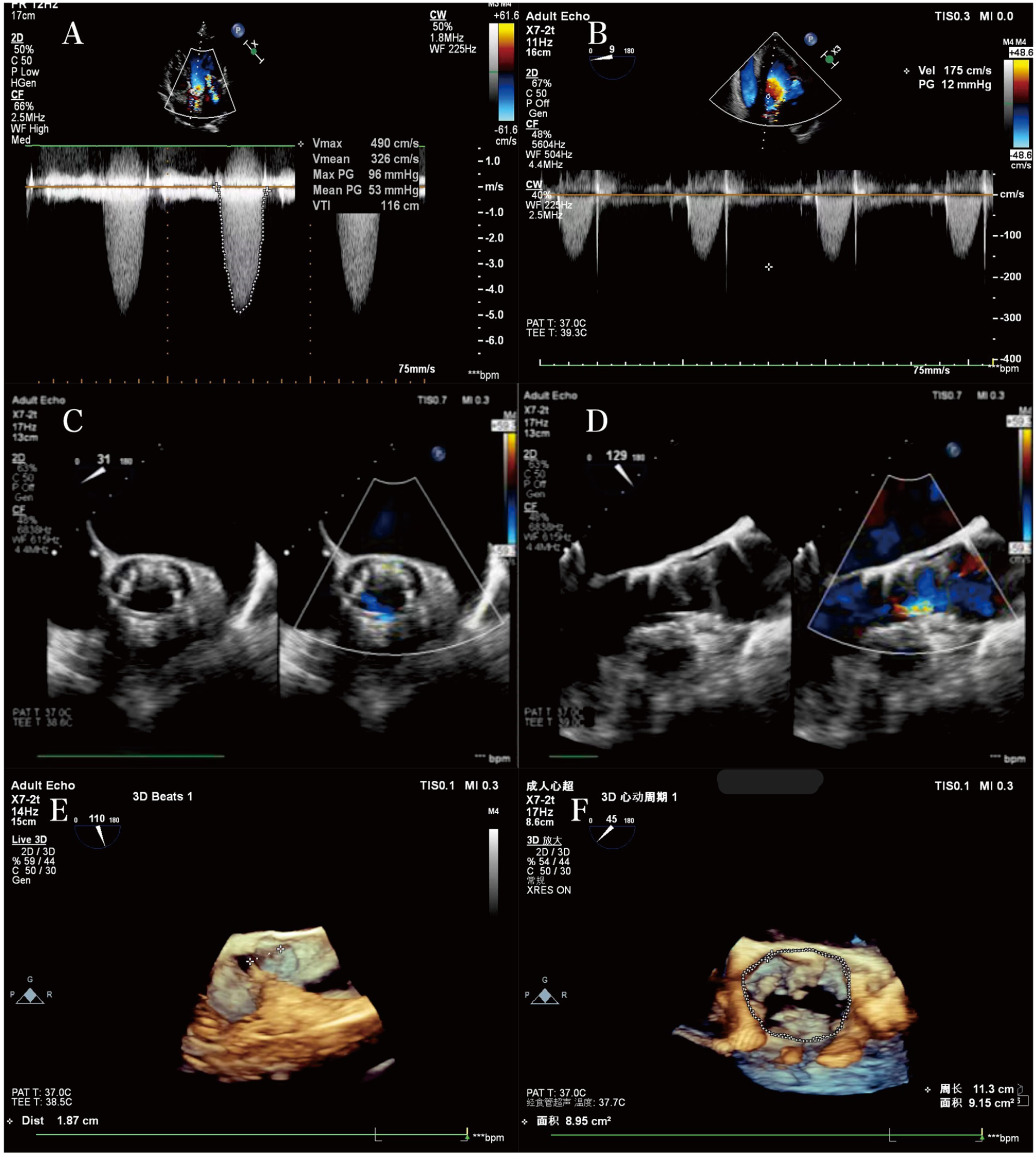
(A) In a patient with severe aortic stenosis (AS), maximum aortic valve (AV) blood flow velocity (Vmax) and mean AV pressure gradient (AVPG mean) are measured by using transthoracic echocardiography (TTE) in apical five-chamber view. (B) AV Vmax and AVPG mean are measured in the five-chamber view of the patient in (A) immediately after surgery. (C, D) A small amount of paravalvular leakage after valve release is detected through intraoperative monitoring. (E, F) Real-time three-dimensional transesophageal echocardiography (RT-3DTEE) measurement of coronary ostial height and aortic annulus circumference and area.
Discussion
Transcatheter aortic valve replacement has been widely recognized as an alternative to SAVR and is mainly indicated in patients with severe AS who are inoperable or have a relatively high surgical risk. 4 In low-risk groups, TAVR and SAVR are not significantly different with regard to the postoperative reduction in LV hypertrophy and LVEF changes in patients with AS 5 ; also, the efficacy of TAVR is similar to that of SAVR,6,7 and TAVR is less invasive. The latest 2021 Expert Consensus on the Clinical Pathway for Transcatheter Aortic Valve Replacement, in China, updated and expanded the indications for TAVR. 8 For simple severe AR, an exploratory trial can be performed by experienced surgeons in cardiology centers. 9 The incidence of moderate-or-more AR in patients > 75 years is 2.85% and the incidence of pure AR is higher than that of pure AS; therefore, AR might be more common in older patients with AVD in China. 10 Li et al. 11 confirmed that transarterial TAVR is safe and effective for the treatment of pure AR. Echocardiography is an essential imaging examination performed prior to TAVR for both AR and AS.
Accurate assessment of the aortic root anatomy before TAVR is crucial for successful surgery. For patients who cannot tolerate CT, echocardiography can be used to assess the anatomy of the aortic root before surgery. 12 This study showed that there was no significant difference in the preoperative aortic annulus, and left and right coronary ostial height measurements between RT-3DTEE and CT measurements in 158 patients. In studies with large sample sizes, more than 50% of patients scheduled for TAVR had chronic kidney disease and approximately 10% of these patients had severe renal insufficiency; therefore, the cumulative dose of contrast agents used for MSCT and TAVR can further aggravate poor renal function, and patients who received renal replacement therapy after TAVR have had poor clinical outcomes. 13 Six patients in this study who had preoperative combined renal insufficiency died due to postoperative multi-organ failure. Therefore, in patients with chronic kidney disease, RT-3DTEE can be used to evaluate the structure of the aortic annulus, thereby minimizing the use of contrast agents. Transesophageal echocardiography can be an effective alternative for TAVR in emergency patients who have contraindications for CT or who do not have time for CT. 14 The good agreement between TTE and MSCT found in this study confirms that sonography can be used instead of CT in special cases, reflecting the importance of echocardiography. In addition, in four patients in this study in whom antegrade crossing of the valve was difficult, retrograde crossing of the valve was successfully performed through transseptal puncture under the guidance of TEE, playing an irreplaceable role in TAVR surgery and reducing exposure time during the operation.
Paravalvular is a common postoperative complication of TAVR, but its incidence has decreased with the use of new-generation valves with PVL prevention technology. Most patients had slight to mild PVL, and the PVL of the self-expanding prosthesis may resolve over time. 12 Among the 158 patients in this study, two patients were changed to SAVR due to severe mitral and tricuspid regurgitation and hemodynamic instability; two patients underwent a long coronary angiography (CAG) + percutaneous coronary intervention (PCI) before TAVR, and therefore, TAVR could not be performed immediately due to the patient’s condition and was performed later; two patients had a large aortic annulus and no valve calcification; therefore, there were four artificial valves that could not be anchored, and SAVR was administered later. Two patients underwent SAVR because of prolapse of the prosthetic AV into the left ventricle due to deep implantation. The remaining 150 patients received self-expanding prostheses through the femoral artery. Among the 150 patients, 12 underwent VinV implantation due to moderate-or-more PVL after the first valve was implanted, and there was almost no PVL after the second valve was implanted. Large amount of PVL seriously affects the prognosis of patients. 15 Therefore, if there is moderate or large perivalvular leakage during intraoperative valve implantation, adjustments can be made to reduce leakage before the completion of TAVR. In addition, if intraoperative TEE monitoring indicates moderate-or-more PVL after the first valve is implanted, timely consideration/performance of VinV implantation or post-balloon dilation is critical for patient prognosis. These situations demonstrate the importance of real-time TEE monitoring during the procedure.
After TAVR, follow-up occurs mainly through TTE, and the focus of follow-up differs for patients with different types of AVD. 16 In this study, 58 of 150 patients had moderate-or-more mitral regurgitation before surgery, decreasing to 26 patients at 1 month after surgery; 30 patients had moderate-or-more tricuspid regurgitation before surgery, decreasing to 16 patients at 1 month after surgery; and the number of patients with high PASP after surgery was lower than that before surgery. In groups I and III, AV Vmax and AVPG mean at 1 month after surgery were lower than those before surgery, and LVEF at 1 month after surgery was higher than that before surgery. After a prosthetic valve is used to replace the original valve, LV afterload significantly decreases, inverse remodeling of the LV myocardium occurs, and LV function improves. Left ventricular ejection fraction in patients with AS improves after TAVR.17,18 Furthermore, IVST and LVPT 1 month after surgery were lower than those before surgery, which is consistent with reverse remodeling of the LV. In groups II and III, the LVEDD, EROA, RVol, and AR widths at 1 month after surgery decreased compared with those before surgery. The lack of significant improvement in LVEF in some patients in group II may be due to long-term aortic insufficiency, high LVEDP, and LV decompensation, which may require longer follow-up to observe improvements in cardiac function. Studies have shown that the lack of LVEF improvement after TAVR may be due to a lack of contractile reserves or the presence of significant pulmonary vascular resistance (PVR). Although patients with preserved preoperative LVEF (>50%) generally do not experience a significant increase in LVEF after TAVR, early improvement can be observed on two-dimensional speckle tracking echocardiography for LV global longitudinal strain (≥1.5% absolute increase).19–22
The incidence of new-onset or pre-existing atrial fibrillation after SAVR is high and more patients have permanent pacemakers implanted after TAVR.23,24 With continuous advances in the delivery system, accumulation of experience, and improvements in surgical skills, the incidence of TAVR complications is expected to gradually decrease.
Limitations
This study has several limitations owing to its research design and sample of patients by convenience. First, this was a single-center retrospective study, and the number of included cases was limited. Currently, few research data exists on the use of TAVR for the treatment of simple AR through the femoral artery approach. Second, the postoperative follow-up period was short. Third, the patients were not re-examined postoperatively with MSCT in combination with echocardiography to evaluate changes in prosthetic valve morphology and cardiac function.
Conclusion
Echocardiography plays an important diagnostic role in the preoperative assessment, intraoperative monitoring, and postoperative follow-up of patients with different types of AVD receiving TAVR. This limited evidence may provide a basis for evaluating the efficacy and prognosis of TAVR. However, further studies with large sample size and longer follow-up period is required to validate these findings.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by funding from the “Peak” Training Program for Scientific Research of Yijishan Hospital, Wannan Medical College. KGF 2019G14 and Start-up Fund Project of Special Scientific Research for Introducing Talents in Yijishan Hospitals in 2024 YR202418.
Ethics Approval
Ethical approval for this study was obtained from the Institutional Review Board (IRB) of the Yijishan Hospital (IRB no. 2024-0426).
Informed Consent
Informed consent was not sought for the present study because all case data was de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
