Abstract
For several decades, atherosclerosis has attracted the attention of researchers around the world. Even being a major cause of serious cardiovascular disease and events, atherosclerosis is still not fully understood. Despite the fact that the main players in the pathogenesis of atherosclerosis are well known, many mechanisms of their implementation and interactions remain unknown. The same can be said about the risk factors for atherosclerosis. Many of them are known, but exactly how they work remains to be seen. The main objective of this review is to summarize the latest data on sex as a biological variable in atherosclerosis in humans and animals; to determine what we do not still know about how sex affects the process of growth and complications of atherosclerosis. In this review, we summarized data on sex differences at 3 atherosclerotic aspects: inflammation, vascular remodeling, and plaque morphology. With all overviewed data, we came to the conclusion on the atheroprotective role of female sex.
Introduction
To date, heart disease is the major reason for death regardless of sex, ethnicity, and race. So far, this situation remains unchanged, despite the fact that there is an awareness of the mechanisms leading to ischemic heart disease formed, as well as many years of innovations in both medical and interventional cardiovascular (CV) care. Because of ischemic cardiovascular disease (myocardial infarction (MI) and stroke included), about 650 000 people die every year. Statistics show that every year, over 600 000 people in America have their first myocardial infarction, and 200 000 people have recurring events. 1 The fact that the structure of the prevalence of cardiovascular disease (CVD) varies depending on sex has been known for a very long time. 2
In 1772, the English physician William Heberden described angina pectoris as follows: “I have seen nearly a hundred people under this disorder [of the breast], of which there have been three women. . .all the rest were men.” 3 In 1896, Sir William Osler gave a lecture at Johns Hopkins University and, quoting Heberden, also added: “In my own series of 40 cases of true angina, there was only one woman.” 4
Epidemiological information still proves that, unlike men, young women are less likely to develop cardiovascular disease and have decreased rates of myocardial infarction. However, women catch up closer to 70 years of life and surpass them closer to the ninth decade (see Figure 1). On the contrary, the nature of the onset of the disease is not repeated with a stroke. It has been shown that women have the highest incidence of stroke before the age of 70. At the age of 70 men are leading, with age, women begin to outperform men again. 5 Male stroke mortality rates (adjusted for age) are the same in whites and higher in blacks, Hispanics, and Asian-Pacific islanders. It is noteworthy that in comparison with men, White and Black women have a chance to experience a second stroke during the following 5 years after the first event. 6

Diagram of CVD incidence in males and females with aging (based on data from Woodward 5 ).
Ischemic cardiovascular disease is most often triggered by atherosclerosis, a process in which lipid-saturated plaques are shaped within the vascular network. Long-term studies dedicated to atherogenesis mechanisms and plaque development in numerous preclinical animal models have demonstrated that atherosclerosis is a diffuse chronic inflammatory disease of the vascular network. Reacting to affection caused by various CV risk factors (eg, hypertension or diabetes), endothelial cells (EC) covering the vascular network get nonfunctional and drop their usual anti-inflammatory properties, as well as antithrombotic properties. 7
Subsequently, inflammatory cells enter the vascular network and absorb oxidized lipids, turning into foam cells that make up the plaque core and are covered with a fibrous cap. This fibrous cap stabilizes the plaque. Inflammatory plaque cells release factors that attract extra immune cells and suppress their excretion, thus leading to a chronic inflammatory condition and plaque development.8,9 Bigger plaques (stenosis >70%) that block the bloodstream eventually cause symptoms such as angina or lameness, which become chronic. Nevertheless, inflammatory plaque cells also secrete matrix metalloproteinases capable of destroying the fibrous membrane and ultimately stimulate plaque rupture, acute ischemia, and thrombosis, which are well-known as the key reason for most MIs and strokes. The key role of inflammation in atherosclerosis, discovered by preclinical studies, has resulted in the testing of anti-inflammatory therapy as a way of prevention of cardiovascular events. 10
Atherosclerosis
Inflammation of blood vessels, accumulation of lipids, thickening, and fibrosis of intima, remodeling and rupture or erosion of plaques causing MI and ischemia, are the main processes in the mechanism of the occurrence and development of atherosclerosis. All data on the role of inflammation in the formation of atherosclerosis are mainly obtained from studies on animal models.11-13 Several recent studies demonstrate in detail how inflammatory mediators affect the development of atherosclerosis. Speaking about the structure of atherosclerotic plaque, we can say that it consists of macrophages, mast cells (Mcf), T cells, B cells, and dendritic cells (DCs); its location is the mucous membrane of the vascular walls. The plaques form a necrotic nucleus consisting of lipids, and a fibrous cap of smooth muscle cells (SMCs) and collagen. 14 When the disease reaches the later stages of its development, immune cells are able to build up on the lumen side of the plaque, disrupting blood circulation. When circulating cholesterol and apolipoprotein B (Apob) levels rise, they undergo various changes to produce modified low-density lipoprotein (LDL), which is metabolized by macrophage scavenger receptors to produce macrophage foam cells inside plaques. 15 Most of the foam cells are formed from mononuclear phagocytic cells, but vascular ECs and SMCs may also contain lipids. Internalization of oxidized LDL (oxLDL) leads to the appearance of cholesterol crystals, which trigger the inflammasome. This, in turn, stimulates the release of mediators (among them interleukin (IL)-1, IL-6, IL-18, chemokines, eicosanoids, proteinases, and oxidases), which contribute to plaque inflammation, formation of scars on the tissues, and remodeling.16,17
Inflammation in Atherosclerosis: Sex Differences
Sex hormone effects on inflammation with aging and menopause
Changes in immune18-22 function associated with age include impaired ability to respond to new antigens, unstable memory responses, low-severity chronic inflammation, and a growing tendency to autoimmune reactions because of the growth of antibody and autoantibody levels. To date, there are very few studies aimed at investigating the effect of sex hormones on the immune reaction in aging with atherosclerosis (except for studies of brain inflammation, aging, and stroke).23,24
Oral hormone replacement therapy (HRP) (raising the level of estrogen to a relatively high dose) leads to a decrease in the level of antibodies in menopausal women. Based on this information, it can be assumed that these changes are associated with age, and not only with the level of estrogen. Menopause is considered the final menstrual period without repeated menstruation for 1 year. 25 The average age of menopause in Western culture ranges from 49 to 52 years. A drop in the level of estrogen (ie, estradiol) occurs only in the last 6 months before and after menopause. Testosterone, in turn, slowly decreases with age in both women and men. After menopause, the main endogenous source of estrogens is estrone, which is synthesized in adipocytes using aromatase. 26
In addition, menopause is linked with dropping in CD4+ and CD8+ T cells and natural killer cell activity. Moreover, both aging and menopause were linked with elevated levels of cytokines IL-1, IL-6 (IL-1ß elevated levels of IL-6), IL-18, TNF, and C-reactive protein (CRP). They are biomarkers of an inflammatory response and are linked with an elevated risk for the treatment of heart failure in individuals with atherosclerosis. 27 Consequently, during menopause, the protective role of estrogen in reducing the activation of innate immune cells disappears. And this, in turn, leads to an elevation in the level of pro-inflammatory cytokines, while the levels of autoantibodies and immune complexes continue to grow due to the continued low level of estrogen. On the contrary, in the vast majority of men, the levels of androgens that contribute to the activation of innate immune cells and pro-inflammatory cytokines decrease after 50 years without an elevation in autoantibodies, which women have due to estrogen. This may partially answer the question of why atherosclerosis and hypertension increase in women after menopause. 28
Inflammation role in the development of atherosclerosis
The most severe following events of atherosclerosis are mediated by plaque rupture and thrombosis, which leads to myocardial infarction, stroke, and related mortality. It has been found that in humans, more vulnerable dental plaques have a greater burden of inflammation.29,30 In particular, both in the lipid-rich nucleus and vulnerable plaques, in contrast to stable plaques, there are much more T cells and macrophages. Due to the fact that plaques avoided tearing in the majority of preclinical atherosclerotic models, many studies have focused specifically on the process of inflammation of plaque as a surrogate endpoint of plaque destabilization. 31
When circulating monocytes enter into the subintimal part through the damaged endothelium of the vessel, where they are able to differentiate into macrophages, uptake modified low-density lipoprotein, and turn into lipid-saturated foam cells, plaque inflammation becomes stronger. Being located in the plaque, foam cells release matrix metalloproteinases, which, in turn, lead to the destruction of the fibrous membrane which covers the plaque.32,33 Efferocytosis is also inhibited, which results in the buildup of a necrotic nucleus. In addition, activated macrophages release chemokines that subsequently recruit other immune cells that, in turn, release cytokines on their own, thus favoring the pro-inflammatory phenotype of macrophages and expanding chronic vascular inflammation. Ultimately, this results in a fragile morphology of plaques, which are more valuable, and also provoke ischemic events.34,35
There are known differences in immune function that are related to sex and age. Thus, sexual dimorphism in the immune reaction to hyperlipidemia and also CV risk factors may contribute to sex differences in the phenotype of plaques and the frequency of ischemic events.
The role of sex differences in systemic inflammation due to atherosclerosis
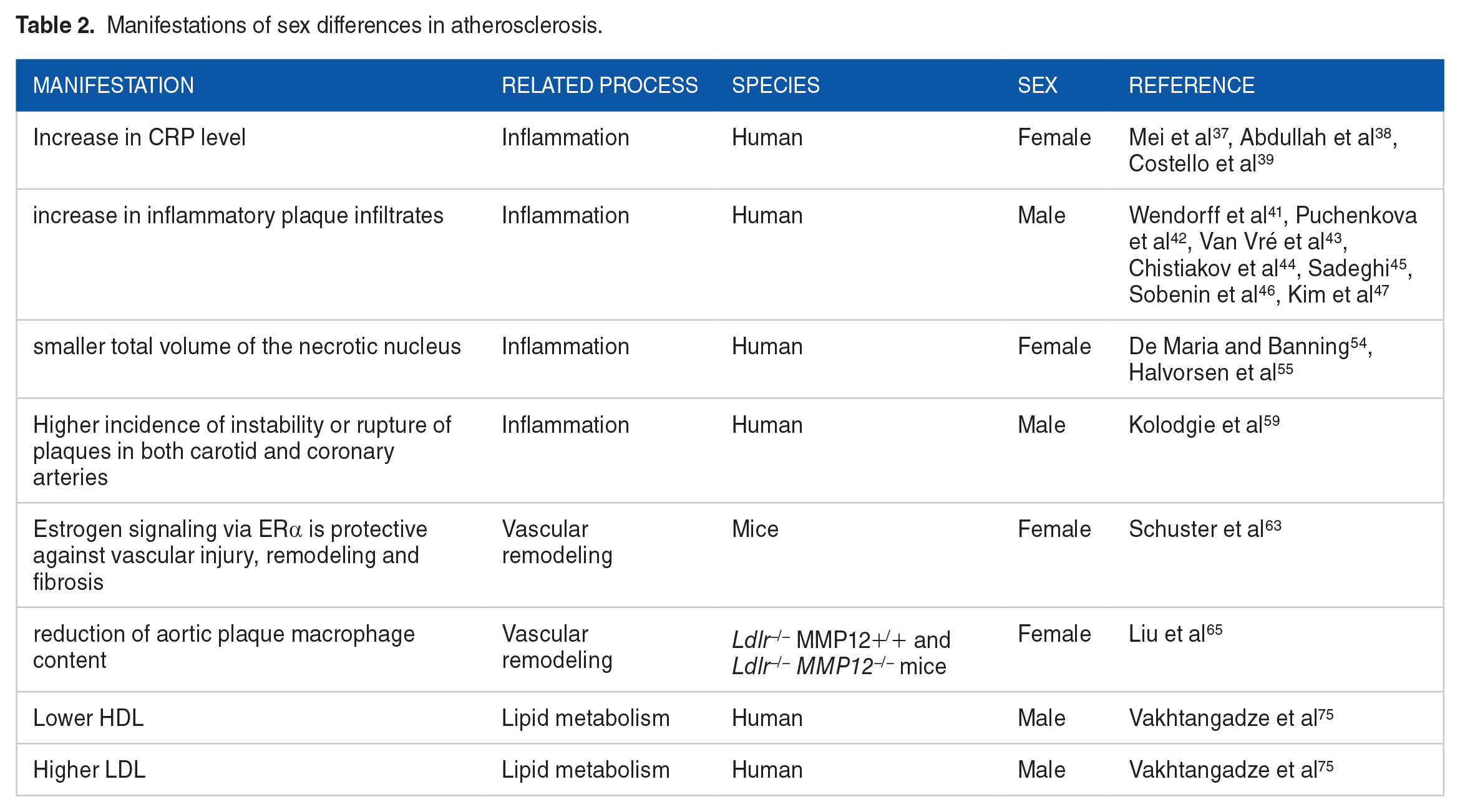
Systemic inflammation in human beings could be assessed by measuring blood-based biomarkers; the inflammation of plaques can be evaluated using arterial wall imaging. CRP is a key sign of systemic inflammation and a harbinger of cardiovascular disease. 36 We represent the data in Table 1. When assessing the NHANES survey, the average level of CRP (2.1 mg/L) was similar for both sexes, but the average value for women (0.55 ± 0.91) was higher than for men (0.41 ± 0.64). In this study, despite higher levels of CRP, women had a lower 10-year likelihood of developing coronary heart disease (CHD) in all categories of CRP. 37 The Dallas Heart Study confirmed these outcomes through a cross-sectional analysis of more than 2700 patients aged 30 to 65 years, the results demonstrated that women had higher CRP levels (median 3.31 mg/L) than men (median 1.8 mg/L). 38 In the GSMS study, children aged 9 to 13 were followed up to the age of 21. Changes in CRP in boys and girls began to diverge around puberty (approximately 15 years old), when the growth rate of the CRP became faster in girls CRP, in contrast to a smaller linear growth in boys. 39 It is noteworthy that the use of oral HRT was also linked with an increased level of CRP. In postmenopausal women, the level of CRP is the same as in men, but any type of HRT (progesterone with or without estrogen) increases the level of CRP. However, one possible explanation is that estrogen elevates CRP by affecting protein synthesis in the liver, other systemic inflammation markers (including IL-6, IL-1β, and TNFa) are also elevated by estrogen. Female sex hormones can provoke systemic inflammation; there are other factors that may also weaken the vascular effects of increased systemic inflammation among women. 40
CRP level and associated parameters comparison.

Sex differences in plaque inflammation
A significant study was conducted that examined 763 samples of carotid endarterectomy. In this study, the male sex was considerably linked to an increase in inflammatory plaque infiltrates using logistic regression.
Numerous researched aimed to study endarterectomy samples have demonstrated that in men plaques have greater CD68 staining (a marker of inflammation linked with macrophages) in contrast to plaques in women of the corresponding age.41,42 It is noteworthy that this was still positive for both asymptomatic individuals and individuals with carotid artery stenosis by more than 90% (the groups were evaluated separately from each other).
In men, plaque macrophages express increased levels of CD86, a co-stimulating molecule promoting interaction with T cells and the development of inflammation. Apparently, there is no sex discrepancy in the total number of T cells located in plaques based on CD3 staining of samples for carotid endarterectomy.43,44
Uptake of 18F-fluorodeoxyglucose (FDG) is a metabolic activity biomarker associated with plaque inflammation, which is measured by PET; and it is known that it is also associated with morphological signs of high risk, for example, CD45 and CD68 staining (total number of leukocytes and macrophages), and the severity of symptoms.45,46 A study conducted among 61 individuals who experienced a transient ischemic attack or mild stroke showed, the absorption of 18F-fluorodeoxyglucose in carotid artery lesions was noticeably higher among men. However, this inequality between the sexes was not observed in any studies of FDG in individuals with symptomatic atherosclerosis of the carotid arteries. 47
In 521 patients without any symptoms who were screened for carotid artery stenosis, men were more likely to have carotid and femoral artery FDG uptake compared with women. Among the tested patients with carotid and femoral artery activity, carotid artery stenosis was detected twice as often in men. It is noteworthy that some differences can be associated with the frequency of metabolic syndrome among men. 48 There are 2 different studies, that have associated FDG absorption with metabolic syndrome; on the other hand, Lee et al have demonstrated that 18F-fluorodeoxyglucose absorption in the carotid artery is higher in men with/without metabolic syndrome. 49
Implications for Sex Differences in Inflammation During Atherosclerosis
The similarity between the immune mechanisms participating in the pathogenesis of myocarditis and atherosclerosis suggests that sex differences in the inflammatory response and remodeling of the heart may also have intersections. On the other hand, mechanical studies aimed at studying sex differences in inflammation in atherosclerosis have not yet been conducted in general.50,51
However, there are a number of questions that need to be answered. Do serum biomarkers such as TNF and IL-6 predict an adverse result in patients with atherosclerosis (both sexes)? Are they the best predictors due to the suppression by estrogen? Is it possible that TLR4/inflammasome/IL-1ß levels predict MI in men more precisely, as testosterone enhances this signaling pathway? Can the correlation between autoantibodies to oxLDL and coronary artery disease be considered to be more dominant in women, where estrogen enhances the reaction of B cells and antibodies/autoantibodies? If the answer is yes, can this then give a partial understanding of why women are at high risk of developing coronary artery disease when they have at least one (or more) autoimmune diseases? What is the reason for arterial hypertension to be higher in women with CHD after menopause than in men, and is there a direct link between hypertension and cardiac inflammation? Does the activation of AngII contribute to hypertension and arrhythmias to a greater extent in men, as estrogen lowers its expression in women? The answers to these questions are extremely important for determining sex differences in the occurrence and development of atherosclerosis and risk factors for the occurrence of MI.
Let us imagine that the sex differences in inflammation found in myocarditis can be applied to atherosclerosis. In this case, it can be hypothesized that MCs and M2 TLR4+ macrophages contribute to increased inflammation and accumulation of foam cells in atherosclerotic plaques in men, where they contribute to remodeling leading to thrombotic MI. On the contrary, in women, the main cause of MIs may be the outcome of deposition of autoantibodies against oxLDL and other autoantibodies which have the form of ICS. 52 The presence of ICS in combination with a smaller vessel size can lead to an elevation in BP (blood pressure) in women and stimulate local inflammation, as well as a shift in the vessel wall, which leads to MI. In women, this process will be speeded up after menopause, as the level of cardioprotective estrogens lowers, but in whom the level of autoantibodies is increased due to the presence of one or more autoimmune diseases. 53
Sex Differences in Plaque Morphology
In virtue of achievements in imaging, it has become possible to assess the individual characteristics of atherosclerotic plaque linked with undesirable events. Individuals who have experienced percutaneous coronary intervention (PCI) and intravascular ultrasound (IVUS) are great candidates, who can help to assess both stenosis and necrotic nucleus degree in the plaque based on its echo permeability.
An examination of individuals with the acute coronary syndrome (ACS) by IVUS showed that women had the same number of lesions. On the other hand, a smaller number of lesions that were not the causative agent of infection; a smaller number of affected coronary arteries, a lower incidence of plaque rupture, and an aggregate volume of the necrotic nucleus (even regardless of the older average age and considerable concomitant pathology). 54 This sex difference in the volume of the necrotic nucleus was also noted in the Tromsø study. 55
The emergence of optical coherence tomography (OCT) in the frequency domain made it possible to obtain images of morphological signs of plague with a higher resolution. In a study conducted among individuals who underwent PCI, among women with stable CAD, plaques had a smaller lipid arc, less often contained crystals of cholesterol, and had less lesion calcification, which are signs of plaque stability. 56 Anyway, plaques were more likely to have erosion, it is a sign that might develop before atherothrombotic events. It was demonstrated that the thickness of the fibrous cap and the frequency of thin-cap fibroatheroma does not depend on sex. Nevertheless, another study showed that stratification by age among men (up to 70 years) had an increased frequency of fibroateroma of the thin membrane. On the contrary, the frequency was higher among women (over 70 years old). 57
Also, sex differences have been found in plaques assessed using MRI. Among the individuals who experienced MRI of the carotid artery with stenosis >50%, a plaque with intraocular hemorrhage, fibrous cap whether it’s thin or torn, or contralateral artery clot with <50% of stenosis was more common among men. 58 Histological analysis of endarterectomy or postmortem tissue performed among men also shows an increased frequency of morphological signs linked with instability or rupture of plaques in both carotid and coronary arteries, in contrast to women. Notably, one study found that plaque hemorrhages were linked with a higher frequency of relapsing events among both men and women. 59 These differences are summarized in Figure 2.

Sex differences of atherosclerotic plaques.
Studies conducted among people have shown that women often have less stress on atherosclerotic plaques and fewer signs of high-risk plaques in contrast to men. As a rule, this remains genuine even in the context of clinical events, both in active lesions and non-culprit lesions. 60 Since most studies were conducted on young individuals and middle-aged individuals, there is still a question about whether the presence of plaques or their morphology promotes the mechanism of catching up with the increase in the number of cases in older women.
Vascular Remodeling Differences
Female sex hormones, such as estrogen, progesterone, and androgen can bind to receptors, both intracellular and extracellular, through ligand-independent, ligand-dependent, genomic or non-genomic mechanisms. Estrogen response elements (EREs) or androgen response elements (AREs) are necessary parts of the intracellular pathway because binding to them results in the inhibition or stimulation of gene transcription factors. 61 Extracellular and intracellular estrogen receptors (ERs), such as ERα, ERβ, and G protein-coupled receptor 30 (GPR30), can be found in the immune cells, both innate and adaptive immune systems, as well as on cardiovascular cells like vascular ECs, VSMCs, cardiac fibroblasts and cardiomyocytes. In female coronary artery VSMCs, ERs expression is higher, than in those cells in males, but their expression lowers with age after menopause. 62 Thus, in mice, estrogen signaling via ERα is more protective against vascular injury, remodeling, and fibrosis after MI in a cardiomyocyte-specific ERα overexpression model in females than in males. Moreover, estrogen-replacement therapies reduce the size of atherosclerotic plaque and prevent vascular remodeling.63,64
Lowering of expression of adhesion molecules and subsequent inflammatory cell recruitment by premenopausal estrogen potentially contribute to protection against atherosclerotic vascular remodeling. Estrogen lowers the negative impact of MMP12, which relates to arterial stiffening. This is supported by the recent study 65 by Liu et al, which showed the stimulating effect of the uptake of oxLDL on the release of MMP12 by macrophages. This study also revealed that treatment with E2 decreases MMP12 gene expression and secretion in human macrophages. Also, a noticeable reduction of aortic plaque macrophage content was admitted in both Ldlr−/− MMP12+/+ and Ldlr−/− MMP12−/− female mice compared to male mice, all fed a high-fat diet for 16 weeks. This is consistent with the conclusion that decreased estrogen levels after menopause are related to altered vascular function, increased inflammation, and upregulation of other hormonal systems such as the renin–angiotensin–aldosterone system and reduced nitric oxide-dependent vasodilation. 65
Testosterone, being the crucial male hormone, is a hormone of a steroid nature, which belongs to the androgen hormone family and binds to intracellular androgen receptors (ARs). With age, the testosterone level lowers, which is linked to the boost of CVD and CVD-associated mortality. 66
Knockout of AR (ARKO) mice and mice of C57BL/6J background were used to demonstrate the implication of androgen receptors in angiotensin II (Ang II)-induced vascular remodeling. For this purpose, animals were treated with Ang II at 2.0 mg/kg per day for 14 days by a subcutaneously implanted osmotic minipump. In animals with ARKO, the rate of medial thickness and perivascular fibrosis of the coronary artery and aorta was significantly higher than in untreated mice. 67 Also, levels of collagen I and collagen III gene expression as well as superoxide production appeared to be enhanced only in mice with ARKO that were treated with Ang II among all groups. Moreover, the vascular transforming growth factor β (TGFβ) expression in ARKO mice was higher than in male wild-type mice. 68
According to other studies, AR knockout has different effects on cells of various types. Thus, monocyte/macrophage-deficient ARKO Ldlr−/− mice showed a decrease in atherosclerosis and had a significant decrease in macrophage content and collagen deposition as well as increased VSMC content in the aortic root, while mice with an AR knockout in ECs or VSMCs did not manifest any differences in lesion size or changes in vascular wall composition. 69 All mice were fed the high-cholesterol diet for 16 weeks. It was shown in vitro, that AR expression on monocytes promotes their migration, adhesion to ECs, and differentiation into foam cells. Thus, we can suggest the opposite effects of systemic AR deficiency and monocyte/macrophage-specific AR deficiency on atherosclerosis. 70
Lipid Metabolism Differences
One of the cornerstones of atherosclerosis initiation and development is the lipid metabolism shift. However, there are serious differences in the metabolism of cholesterol between men and women, that seem to result in other differences in the disease course. 71 Studies among the US population, it was revealed that total cholesterol is higher among white women, in both sexes among blacks and Asians, and Hispanic women. 72 Moreover, the composition of lipid profiles is also different in men and women. Men are more than twice as likely to have low high-density lipoprotein (HDL) than women in all age groups. The levels of LDL in men are higher in comparison to women. Response to treatment has been shown to vary by sex as well. 73 In the SATURN trial, the female sex was independently associated with greater atheroma regression with statin therapy when LDL levels were ⩽70 mg/dL. 74
During aging, cholesterol profiles are also different in men and women. LDL levels are lower in women than in men until they reach the age of 50 when LDL levels grow in women. HDL is approximately 10 mg/dL higher in women compared to men. Increased incidence of coronary artery disease in older women can be related to these changes in lipid profiles. High cholesterol was confirmed to be a risk factor for CAD in women. A lower HDL value predicts coronary risk in women better than in men.75,76 (Table 2).
Manifestations of sex differences in atherosclerosis.
We summarized the mechanisms of sex-associated differences in Figure 3. While the atheroprotective effect of testosterone, as well as atherogenic effect, remains questionable, the strong evidence of estrogen implication on the vascular remodeling inhibition, and thus, in atheroprotection, was provided. However, the female sex is also associated with higher incidence of systemic inflammation, which contributes to atherogenesis. Another association of female-specific factor with atherosclerosis, is menopause, which is also typically linked to aging. As for male sex, there are more systemic mechanisms associated with atherosclerosis, such as altered lipid profile, and androgen receptor signaling.
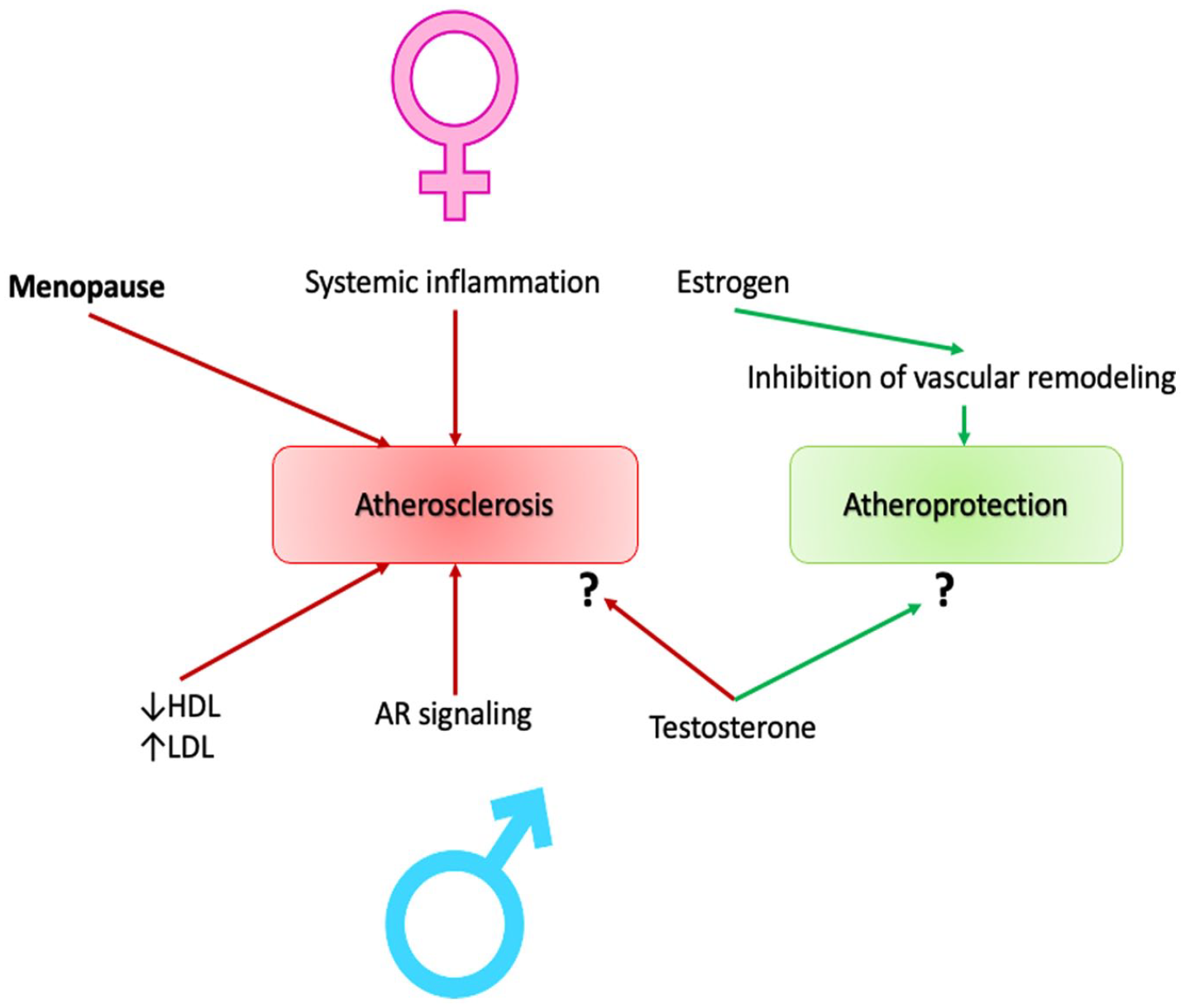
Sex-associated features contributing to atherosclerosis (or atheroprotection) development.
Therapeutic Potential
Estrogen supplementation therapies are being investigated for 3 decades, but their beneficial effects are still disputable. On the other side, hormone therapy may decrease CVD risks, when it started during or shortly after menopause. On the other, severe side effects, like increased risk of breast cancer should be taken into account. 77 Currently prescribed estrogen supplementation schemes have lower doses of estrogens, systemic and vaginal. However, oral supplementation of estrogen enhances the risk of venous thromboembolism. Thus, the decision to the estrogen supplementation use should be made only after an accurate analysis of risks and benefits. 78
Even though testosterone replacement therapy (TRT) is widely investigated, its ability to decrease the risk of atherosclerotic CVD is still controversial. According to several studies, this therapy has beneficial effects, while others state that the impact of TRT is detrimental. Some clinical studies revealed that a lower level of androgens correlates with an increase in cIMT in men from 40 to 70 years of age. 79 On the other hand, a randomized clinical trial 80 dedicated to the evaluation of the long-term effect of testosterone administration on subclinical atherosclerosis in men older than 60 years old, revealed no significant differences in the rate of changes in cIMT after 3 years of daily testosterone treatment in comparison with placebo.
Conclusion
In our review, we went through the various mechanisms implemented in atherogenesis, which are also affected by the sex differences. We united data on inflammation, plaque morphology differences, and vascular remodeling, and all these findings contribute to the atheroprotective role of female. Adding the data on menopause, and the result of clinical studies, we can clearly conclude that female hormones are somehow atheroprotective. The demographic statistics report the base biological differences between men and women and outline the necessity to take sex into account when conducting animal model studies in clinical trials. Long-term observations have not yet given a clear understanding of the cause of sexual dimorphism in CV ischemic events, relapses, and results.
So, we do not have sufficient information (including clinical and preclinical) for a more thorough study of the mechanisms that determine sex as a biological variable in atherosclerosis. Among people, both noninvasive imaging and pathological evaluation show that atherosclerotic plaques appear earlier in men. Moreover, these plaques have a high degree of inflammation and additional unstable signs. The total area that is affected by plaques is larger in men, and the stenosis of individual plaques may be larger in women. It must be the load on the plaques that predicts the development of MI and stroke, and not individual stenosis. Clinical data also highlight the relationship between age and sex: younger men have the more atherosclerotic burden and a high frequency of ischemic events up to the 7th decade, while women keep pace and eventually exceed men in the frequency of myocardial infarction in old age.
Preclinical literature shows that most studies do not consider both sexes, but when this comparison occurs, reliable statistical comparisons between men and women continue to be rather infrequent. Despite the additional costs of such studies, it was found that the size of the plaque is often bigger in young females in the majority of animal models of atherosclerosis (non-human primates excluded). This probably played a role in the formation of the hypothesis that these particular animal models might be not suitable for the identification of sex differences. The analysis of existing information reports that the bigger size of plaque in female animal models gets less obvious with age; on the other hand, some studies show a greater burden of plaque in older male animals. Additional studies on other animals are required to understand whether animals in cages can be a more advantageous model for studying sex as a biological variable in atherosclerosis.
When preclinical studies compare the sexes, the most common measurement is the size of the plaque. Nevertheless, a huge amount of data indicates that in humans, the size of the plaque does not match the rupture of the plaque (
On the contrary, morphology, and plaque inflammation, are the best surrogates for ischemic events, vulnerability of plaque, and mortality. Nowadays, few preclinical studies compare plaque inflammation between men and female. Nevertheless, the existing data demonstrate that this endpoint may reflect the human condition to a greater extent since partners in preclinical models had more inflamed (albeit smaller) plaques. The non-inclusion of both men and female sexes in atherosclerosis mechanistic studies is also a lost chance to discover the basic mechanisms that reveal the differences in this disease due to sex and can have translational significance. One of the potential factors for the clinical trials’ setback in which both sexes participate may be the development of treatment methods focused on the mechanism of the disease, which is confirmed only in one sex in preclinical animal models. Determination of the mechanisms that define sex as a biological variable in atherosclerotic diseases plays a key role in future precision medicine strategies aimed at mitigating the consequences of what is still the key cause of death worldwide.
Footnotes
Acknowledgements
Not applicable.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: research was funded by Russian Science Foundation, grant number 23-45-00031.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Writing—original draft preparation, AVP; writing—review and editing, SG, AYP, VNS, ANO.
