Abstract
Introduction:
Cerebral venous sinus thrombosis associated with vaccine-induced immune thrombotic thrombocytopenia (CVST-VITT) is a severe disease with high mortality. There are few data on sex differences in CVST-VITT. The aim of our study was to investigate the differences in presentation, treatment, clinical course, complications, and outcome of CVST-VITT between women and men.
Patients and methods:
We used data from an ongoing international registry on CVST-VITT. VITT was diagnosed according to the Pavord criteria. We compared the characteristics of CVST-VITT in women and men.
Results:
Of 133 patients with possible, probable, or definite CVST-VITT, 102 (77%) were women. Women were slightly younger [median age 42 (IQR 28–54) vs 45 (28–56)], presented more often with coma (26% vs 10%) and had a lower platelet count at presentation [median (IQR) 50x109/L (28–79) vs 68 (30–125)] than men. The nadir platelet count was lower in women [median (IQR) 34 (19–62) vs 53 (20–92)]. More women received endovascular treatment than men (15% vs 6%). Rates of treatment with intravenous immunoglobulins were similar (63% vs 66%), as were new venous thromboembolic events (14% vs 14%) and major bleeding complications (30% vs 20%). Rates of good functional outcome (modified Rankin Scale 0-2, 42% vs 45%) and in-hospital death (39% vs 41%) did not differ.
Discussion and conclusions:
Three quarters of CVST-VITT patients in this study were women. Women were more severely affected at presentation, but clinical course and outcome did not differ between women and men. VITT-specific treatments were overall similar, but more women received endovascular treatment.
Keywords
Introduction
Vaccine-induced immune thrombotic thrombocytopenia (VITT) is a rare but severe adverse reaction after adenoviral vaccination for SARS-CoV-2. 1 It may cause thromboses at multiple sites and in multiple vascular beds, cerebral venous sinus thrombosis (CVST) being the most frequent and strongly associated with mortality. 2 Previous reports on VITT have shown conflicting results, with some reporting a higher frequency in women,3–5 while others found no difference in frequency of VITT in women and men.2,6 However, reports on patients with CVST associated with VITT (CVST-VITT) consistently found a higher proportion of affected women than men.7,8
There is a lack of data about the clinical characteristics of CVST-VITT in women compared to men. The aim of this study was to compare the presentation, treatment, clinical course, complications, and outcome of CVST-VITT in women and men.
Methods
We used data from an ongoing international registry on CVST-VITT, details of which have been reported previously. 9 In short, this is a registry-based study. Investigators were asked to report consecutive patients who developed CVST within 28 days of any SARS-CoV-2 vaccination.
Data were collected using a standardized electronic case report form (Castor EDC, Ciwit B.V., Amsterdam, The Netherlands). The ethical review committee of the Academic Medical Center in Amsterdam approved this observational cohort study. Each center was responsible for obtaining permission from local authorities if required by national and local law. The study was endorsed by the European Academy of Neurology and European Stroke Organisation.
We included patients reported until January 10, 2023 with possible, probable, or definite CVST-VITT according to the criteria proposed by an expert hematology panel by the British Society for Haematology as described by Pavord et al. 2 For the assessment of anti-platelet factor 4 (anti-PF4) antibodies, we accepted all types of tests, as reported by the investigators. In all cases, CVST was confirmed radiologically or at autopsy, and symptom onset was within 28 days of SARS-CoV-2 vaccination. Coma was defined as Glasgow Coma Scale score <9 points. Non-haemorrhagic lesions were defined as edema or venous infarction. Major bleeding was defined according to International Society of Thrombosis and Haemostasis criteria. 10 For outcome analysis, we dichotomized the modified Rankin Scale at discharge in 0–2 (favorable outcome) and 3–5 (poor outcome). The VITT-specific treatments included immunomodulatory treatment such as intravenous immunoglobulins or plasma exchange, avoidance of heparins, and avoidance of platelet transfusions, unless required for surgery. 11 Female specific risk factors for CVST were defined as oral contraceptive use, pregnancy, or recent childbirth. Conventional risk factors for CVST were defined as infection, previous venous thromboembolism, genetic or acquired thrombophilia, or cancer within the past 10 years. For thrombus load, the number of sinuses or veins that were thrombosed were added. 12
Data analysis
We used descriptive statistics for baseline characteristics, treatment, complications during hospitalization, and outcome. We used Mann–Whitney U test, Chi-square, Fisher’s exact, or Fisher-Freeman-Holton test, as appropriate, to determine significance and considered a two-sided probability value below 0.05 as significant. Confidence intervals were calculated using Wilson’s method. The number of missing values for each variable is reported. Analyses were performed with IBM SPSS Statistics for Windows, version 28.0 (IBM Corp., Armonk, N.Y., USA).
This study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines.
Results
Of the 255 patients entered in the registry, 22 were excluded due to onset of symptoms more than 28 days after, or prior to vaccination, 5 were duplicates, 93 had unlikely VITT, and 2 had VITT after mRNA vaccination, leaving 133 cases for analysis (Figure 1).
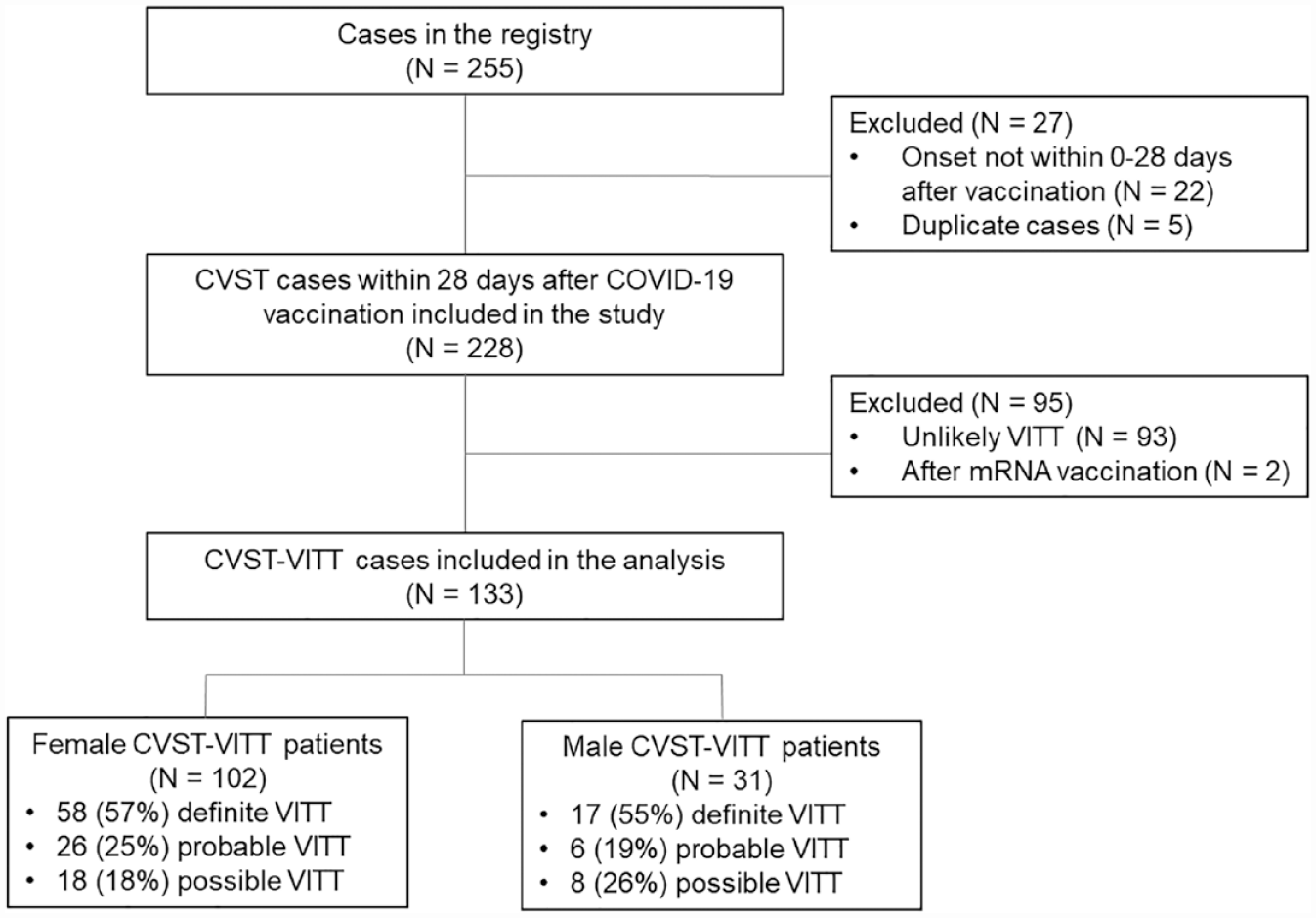
Flowchart of patient selection.
Of the 133 included cases, 102 (77%) were women and 31 (23%) were men. Women were slightly younger than men (median age 42 [IQR 28–54] vs 45 [28–56], respectively). The time from vaccination to symptom onset (9 days [IQR 7–11] vs 10 days [7–11]) and from symptom onset to diagnosis (3 days [IQR 2–6] vs 3 days [1–6]) was similar in women and men. At the time of CVST-VITT diagnosis, 14/102 (14%) women had an intake of oral contraceptives and one was pregnant. Conventional risk factors for CVST did not differ between women and men (Table 1).
Patient characteristics of female and male CVST-VITT patients.
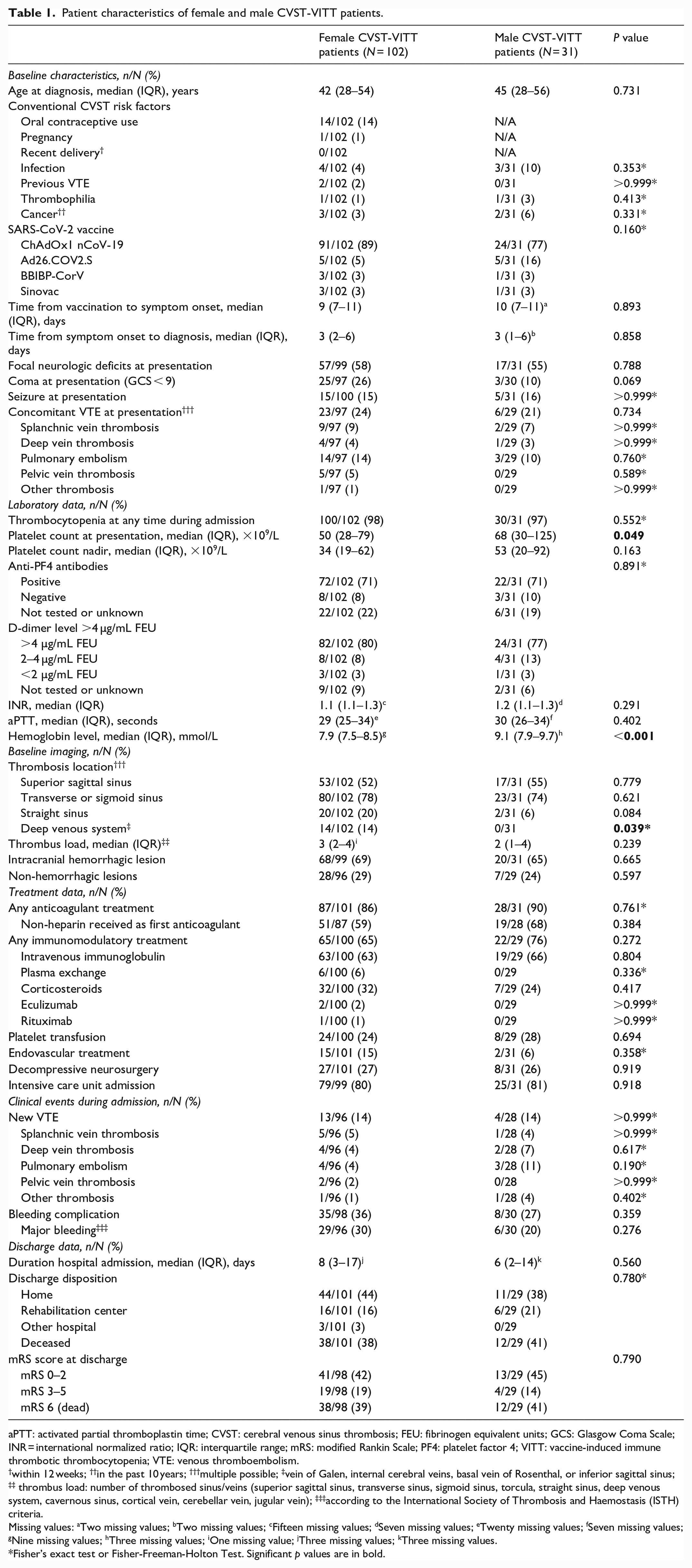
aPTT: activated partial thromboplastin time; CVST: cerebral venous sinus thrombosis; FEU: fibrinogen equivalent units; GCS: Glasgow Coma Scale; INR = international normalized ratio; IQR: interquartile range; mRS: modified Rankin Scale; PF4: platelet factor 4; VITT: vaccine-induced immune thrombotic thrombocytopenia; VTE: venous thromboembolism.
within 12 weeks; ††in the past 10 years; †††multiple possible; ‡vein of Galen, internal cerebral veins, basal vein of Rosenthal, or inferior sagittal sinus; ‡‡ thrombus load: number of thrombosed sinus/veins (superior sagittal sinus, transverse sinus, sigmoid sinus, torcula, straight sinus, deep venous system, cavernous sinus, cortical vein, cerebellar vein, jugular vein); ‡‡‡according to the International Society of Thrombosis and Haemostasis (ISTH) criteria.
Missing values: aTwo missing values; bTwo missing values; cFifteen missing values; dSeven missing values; eTwenty missing values; fSeven missing values; gNine missing values; hThree missing values; iOne missing value; jThree missing values; kThree missing values.
Fisher’s exact test or Fisher-Freeman-Holton Test. Significant
More women presented with coma (26% [95%CI 18–35] vs 10% [3–26]) and in women the platelet count at presentation was lower (50 × 109/L [IQR 28–79] vs 68 [30–125],
The nadir of platelet count was lower in women than in men (median [IQR]: 34 [19–62] vs 53 [20–92]). During hospitalization, occurrence of new venous thromboembolism (14% [95%CI 8–22] vs 14% [6–31]) and bleeding complications (36% [95%CI 27–46] vs 27% [14–44]) had similar proportions in both women and men.
More women received endovascular treatment than men (15% [95%CI 9–23] vs 6% [2–21]). The proportions of VITT-specific treatments such as any immunomodulatory treatment (65% [95%CI 55–74] vs 76% [58–88]), any non-heparins as first anticoagulants (59% [95%CI 48–68] vs 68% [49–82]) or platelet transfusions (24% [95%CI 17–33] vs 28% [15–46]) were similar.
In women and men, functional outcome at discharge for modified Rankin Scale (mRS) 0–2 (41/98 [42%, 95%CI 33–52] vs 13/29 [45% 28–62]), mRS 3–5 (19/98 [19%, 95%CI 13–28] vs 4/29 [14% 5–31]) and in-hospital mortality (38/98 [39%, 95%CI 30–49] vs 12/29 [41% 26–59]) did not differ (Figure 2). Also there were no differences regarding the discharge disposition.
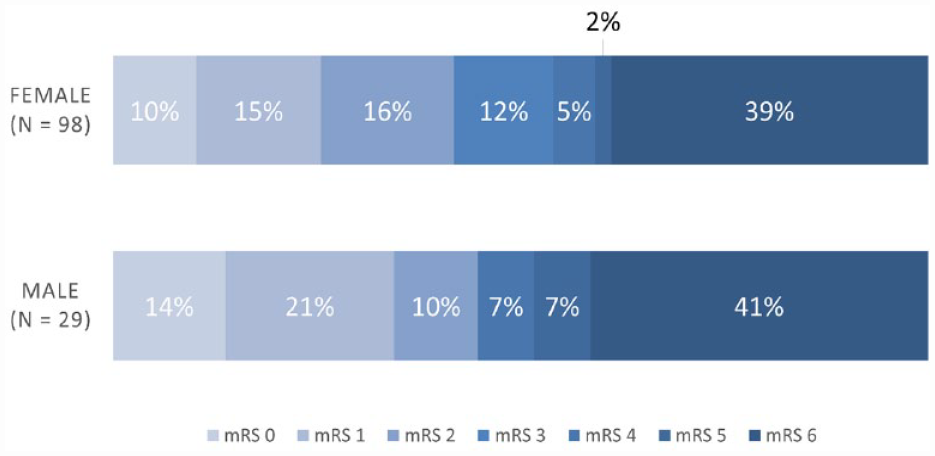
Modified Rankin Scale (mRS) score of women and men with cerebral venous sinus thrombosis due to vaccine-induced immune thrombotic thrombocytopenia (CVST-VITT) at discharge. There are four missing values in the female group and two missing values in the male group.
In an exploratory analysis of cases who were comatose and had a severe thrombocytopenia (platelet count <50 × 109/L) at presentation, 20/133 (15%) patients were selected. Of these, 19 were women and one was a man. The male patient had a recent lumbar puncture, but no other risk factors for CVST. Of the female patients, two used prothrombotic medication (one of which oral contraceptives), one patient was pregnant, one patient had a history of auto-immune disease, and one patient was obese without oral contraceptive use. The other patients had no known risk factors for CVST.
Discussion
The main findings of our analysis of sex differences in our multicenter cohort study are: (1) three quarters of CVST-VITT patients were women, (2) women were slightly younger and women appeared to be more severely affected at presentation with higher frequency of coma and lower admission platelet counts, and (3) VITT-specific treatments, complications during hospitalization, clinical outcome, and in-hospital mortality did not differ between sexes.
The higher proportion of women with CVST-VITT is in-line with previous reports.7,8 A direct pathophysiological link between female sex and risk of CVST-VITT cannot be inferred from our observational study. Furthermore, we cannot rule out a selection bias, for example because healthcare workers, which comprise predominantly women, were more likely to be vaccinated against COVID-19 in an early stage before restrictions on adenoviral COVID-19 vaccinations were widely implemented.13–15 In addition, there might have been a higher awareness of CVST-VITT in female patients since the majority of patients in reports on CVST-VITT were women and might therefore be more likely to undergo investigations in case of suspicion of CVST. In general, women are more likely to suffer from autoimmune disease given the more pronounced immune response to antigens and vaccines with higher antibody production and stronger T-cell activation, which could at least partially explain these findings.16,17 Compared to historical CVST cohorts, a lower proportion of women with CVST-VITT had women-specific risk factors. 9
In our study, the higher proportion of women with coma and lower platelet count at presentation is not explained by a delayed recognition and diagnosis. The number of days between vaccination to symptom onset and symptom onset to diagnosis was similar in both sexes. The higher proportion of women with thrombosis in the deep venous system might explain the higher rate of coma at presentation.18,19 Additionally, the lower platelet count at admission and the lower nadir during hospitalization in women might reflect a more severe disease. However, the cerebral venous sinus thrombus load and haemorrhagic lesions at presentation, the new thromboses and bleeding rates during hospitalization as well as outcome were similar in both women and men. This could partially be explained by the similar VITT-specific treatments in both groups, especially the treatment with intravenous immunoglobulins (IVIG). 11 IVIG was previously shown to be effective in CVST-VITT.1,11 The higher proportion of women treated with endovascular treatment might be explained by the more severe clinical presentation.
Our study has several limitations. First, the overall number of patients was low, precluding detection of significant differences, robust statistical comparisons and outcome analyses. Second, there was no central adjudication of reported data, as they have been collected from clinical routine records. Third, local funding and ethical constraints may have influenced the decision to participate in the study, and hence affected the actual consecutiveness of participating centers and reported cases.
In conclusion, in this international cohort, more women than men were reported with CVST-VITT. More women presented with more severe thrombocytopenia and coma compared to men and the nadir platelet count was lower in women. VITT-specific treatments were overall similar. Despite the more severe clinical presentation in women, clinical course and outcome did not differ between women and men.
Supplemental Material
sj-docx-1-eso-10.1177_23969873231185213 – Supplemental material for Sex differences in cerebral venous sinus thrombosis after adenoviral vaccination against COVID- 19
Supplemental material, sj-docx-1-eso-10.1177_23969873231185213 for Sex differences in cerebral venous sinus thrombosis after adenoviral vaccination against COVID-19 by Adrian Scutelnic, Anita van de Munckhof, Katarzyna Krzywicka, Mayte Sánchez van Kammen, Erik Lindgren, Charlotte Cordonnier, Timothy J Kleinig, Thalia S Field, Sven Poli, Robin Lemmens, Saskia Middeldorp, Sanjith Aaron, Afshin Borhani-Haghighi, Antonio Arauz, Johanna A Kremer Hovinga, Albrecht Günther, Jukka Putaala, Mohammad Wasay, Adriana Bastos Conforto, Diana Aguiar de Sousa, Katarina Jood, Turgut Tatlisumak, José M Ferro, Jonathan M Coutinho, Marcel Arnold, Mirjam R Heldner
Footnotes
Acknowledgements
None
**The Thrombosis with Thrombocytopenia Syndrome Study Group
Kateryna Antonenko1, Joshua Mbroh2, Justine Brodard3, Etrat Hooshmandi4, Vanessa Dizonno5, Annemie Devroye6, Alfonso Ciccone7, Matthias Wittstock8, Julian Zimmermann8, Felix J. Bode9, Mona Skjelland10, Jiangang Duan11, Sini Hiltunen12, Susanna M. Zuurbier13, Marco Petruzzellis14, Aarti R. Sharma15, Abdoreza Ghoreishi16, Ahmed Elkady17, Alberto Negro18, Alexander Gutschalk19, Silvia Schoenenberger19, Simon Nagel19, Alina Buture20, Alvaro Cervera21, Ana Paiva Nunes22, Ana Romina Montané Baños23, Andreas Tiede24, Anemon Puthuppallil25, Anil M. Tuladhar26, Annerose Mengel2, Antonio Medina27, Åslög Hellström Vogel28, Audrey Tawa29, Avinash Aujayeb30, Balakrishnan Ramasamy31, Barbara Casolla32, Beng Lim Alvin Chew33, Bentalhoda Ziaadini34, Boby Varkey Maramattom35, Brian Buck36, Carla Zanferrari37, Carlos Garcia-Esperon38, Caroline Vayne39, Catherine Legault40, Christian Jacobi41, Christian Pfrepper42, Johann Pelz42, Christoph Wahl43, Rolf Kern43, Clement Tracol44, Cristina Soriano45, Daniel Guisado-Alonso46, David Bougon47; Deepti Bal48, Domenico Sergio Zimatore49, Dominik Michalski42, Dylan Blacquiere48, Elias Johansson50,51, Elisa Cuadrado-Godia52, Elyar Sadeghi-Hokmabadi53, Emmanuel Carrera54, Emmanuel De Maistre55, Espen Saxhaug Kristoffersen56, Fabrice Bonneville57, Thomas Geeraerts57, Fabrice Vuillier58, Fabrizio Giammello59, Florindo D’Onofrio60, Francesco Grillo61, François Caparros62, Sophie Susen62, Frank Maier63, Georgios Tsivgoulis64, Giosue Gulli65, Giovanni Frisullo66, Guillaume Franchineau67, Hakan Cangür68, Hans Katzberg69, Hossein Mozhdehipanah70, Igor Sibon71, M. Irem Baharoglu72, Jaime Masjuan73, Jaskiran Brar74, Jean-Francois Payen75, Jim Burrow76, João Fernandes77, Jorge Octavio López Esparza78, Joyce Oen79, Judith Schouten80, Karl Ng81, Sophie Chatterton81, Miriam Wronski81, Katharina Althaus82, Katia Garambois83, Laurent Derex84, Laurent Puy62, Leila Poorsaadat85, Lenise Valler86, Letícia Januzi de Almeida Rocha87, Lisa Humbertjean88, Lucia Lebrato Hernandez89, Luis Murillo-Bonilla90, Lukas Kellermair91, Mar Morin Martin92, Maria Sofia Cotelli93, Maria Hernandez Perez94, Marialuisa Zedde95, Mariana Carvalho Dias96, Marie-Cecile Dubois97, Marta Carvalho98, Masoud Ghiasian99, Meenakshisundaram Umaiorubahan100, Ravi Kumar Karunakaran100, Mehrdad Roozbeh101, Michele Romoli102, Miguel Miranda103, Mohammad Saadatnia104, Monica Bandettini di Poggio105, Moritz J. Scholz106, Robert Kahnis106, Mostafa Almasi-Dooghaee107, Nahid Hoseininejad Mir108, Nasli R. Ichaporia109, Naveen Kumar Paramasivan110, Sapna Erat Sreedharan110, PN Sylaja110, Nicolas Raposo111, Nima Fadakar4, Nyika Kruyt112, Olivier Detante113, Pauline Cuisenier113, Olivier Huet114, Pankaj Sharma115, Paolo Candelaresi116, Pasquale Scoppettuolo117, Peggy Reiner118, Reza Nemati119, Ricardo Vieira120, Rudy Goh121, Seán Murphy20, Serge Timsit122, Shelagh Coutts123, Shyam S. Sharma124, Simerpreet Bal124, Subhash Kaul125, Theodoros Karapanayiotides126, Thomas Cox127, Thomas Gattringer128, Thomas Mathew129, Thorsten Bartsch130, Vahid Shaygannejad131, Veronica Garcia-Talavera132, Vincenzo Palma133, Yıldız Arslan134, Zahra Mirzaasgari135, Zeinab Yavari136, Zohreh Zamani137, Tamam Bakchoul138, Marcel Levi139, Eric C.M. van Gorp140
1Department of Neurology, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
2 Department of Neurology & Stroke, University Hospital Tuebingen, Eberhard-Karls University, Tuebingen, Germany
3Department of Hematology, Inselspital, Bern University Hospital, University of Bern, Bern, Switzerland
4 Clinical Neurology Research Center, Shiraz University of Medical Sciences, Shiraz, Iran
5University of British Columbia, Vancouver, Canada
6 Department of Neurology, University Hospitals Leuven, Leuven, Belgium
7Department of Neurology, Carlo Poma Hospital, Azienda Socio Sanitaria Territoriale di Mantova, Mantua, Italy
8University Medicine Rostock, Rostock, Germany
9Universitätsklinikum Bonn, Bonn, Germany
10Oslo University Hospital, Oslo, Norway
11Department of Neurology and Emergency, Xuanwu Hospital, Capital Medical University, Beijing, China
12Department of Neurology, Helsinki University Hospital and University of Helsinki, Helsinki, Finland
13Department of Neurology, Amsterdam University Medical Centers, location University of Amsterdam, Amsterdam, The Netherlands
14AOU Consorziale Policlinico di Bari, Bari, Italy
15Imperial College School of Medicine, London, United Kingdom
16Stroke Research Group, Head of Stroke Care Unit, Department of Neurology, Vali-e-Asr Hospital, School of Medicine, Zanjan University of Medical Sciences, Iran
17Saudi German Hospital, Jeddah, Saudi Arabia
18Ospedale del Mare, Naples, Italy
19Department of Neurology, Heidelberg University Hospital, Heidelberg, Germany;
20Mater Misericordiae University Hospital, Dublin, Ireland
21Royal Darwin Hospital, Tiwi, Australia
22Lisbon Central University Hospital and Faculdade de Medicina da Universidade de Lisboa
23Queretaro General Hospital, Santiago de Querétaro, Mexico
24Hannover Medical School, Hannover, Germany
25Hamilton General Hospital, Hamilton, Canada
26Radboud University Medical Center, Department of Neurology, Donders Center for Medical Neurosciences, Nijmegen, The Netherlands
27Hospital Universitario Nuestra Señora de Candelaria, Santa Cruz de Tenerife, Spain
28Skåne University Hospital, Lund, Sweden;
29University Hospital of Rennes, Rennes, France;
30 Northumbria Healthcare NHS Foundation Trust, Cramlington, United Kingdom;
31PSG Institute of Medical Sciences and Research, Coimbatore, Tamil Nadu, India;
32Stroke Unit, Hôpital Pasteur 2, URRIS-UR2CA, Unité de Recherche Clinique Cote d’Azur, Cote d’Azur University, Nice, France;
33Department of Neurology, John Hunter Hospital, Newcastle, Australia;
34Neurology Research Center, Kerman University of Medical Sciences, Kerman, Iran;
35Aster Medcity, Kochi, Kerala, India;
36University of Alberta Hospital, Edmonton, Canada;
37Neurology and Stroke Unit, ASST Melegnano e della Martesana - Milan – Italy
38John Hunter Hospital, New Lambton Heights, Australia;
39Tours University Hospital, Tours, France;
40McGill University Health Centre, Montreal, Canada;
41Department of Neurology, Krankenhaus Nordwest, Frankfurt am Main, Germany;
42Leipzig University Hospital, Leipzig, Germany;
43Kempten Hospital, Kempten, Germany;
44CHU Rennes, Rennes, France;
45Hospital General de Castellón, Castelló, Spain;
46Hospital de La Santa Creu I Sant Pau, Barcelona, Spain;
47Annecy Genevois Hospital, Annecy, France;
48The Ottawa Hospital, Ottawa, Canada;
49Policlinico di Bari, Bari, Italy;
50Department of Neurology, Sahlgrenska University Hospital, Gothenburg, Sweden and Department of Clinical Neuroscience, Institute of Neuroscience and Physiology, Sahlgrenska Academy at University of Gothenburg, Sweden
51Department of Clinical Science, Umeå University, Umeå, Sweden;
52Hospital del Mar Medical Research Institute (IMIM), Barcelona, Spain;
53NeuroSciences Research Center (NSRC), Imam-Reza hospital, Tabriz University of Medical Sciences, Tabriz, Iran;
54Hôpitaux Universitaires de Genève, Geneva, Switzerland;
55CHU Dijon, Dijon, France;
56Department of Neurology, Akershus University Hospital, Lorenskog, Norway;
57Toulouse University Hospital, Toulouse, France;
58University Hospital of Besancon, Besancon, France;
59Translational Molecular Medicine and Surgery 36th Cycle, Department of BIOMORF, Stroke Unit, Department of Clinical and Experimental Medicine, University Hospital G. Martino, Messina, Italy;
60San Giuseppe Moscati Hospital, Avellino, Italy;
61University Hospital G. Martino, Messina, Italy;
62Université Lille, INSERM, Centre Hospitalier Universitaire (CHU) Lille, U1172–Lille Neuroscience and Cognition, Lille, France63 Caritas Hospital Saarbrücken, Saarbrücken, Germany;
64Second Department of Neurology, National & Kapodistrian University of Athens, School of Medicine, Athens, Greece;
65Ashford and St Peters Hospital NHS Foundation Trust, Surrey, United Kingdom;
66Department of Neurology, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Rome, Italy;
67Centre Hospitalier Intercommunal de Poissy Saint Germain en Laye, Poissy, France;
68Hospital of the city of Wolfsburg, Wolfsburg, Germany;
69Toronto General Hospital, Toronto, Canada;
70Department of Neurology, Bouali University Hospital, Qazvin, Iran;
71Bordeaux University Hospital, Bordeaux, France;
72Haaglanden Medisch Centrum, The Hague, Netherlands;
73Ramón y Cajal Hospital, Madrid, Spain;
74Surrey Memorial Hospital, Surrey, Canada;
75CHU Grenoble, Grenoble, France;
76Royal Darwin Hospital, Tiwi, Australia;
77Norra Älvsborgs Länssjukhus, Trollhättan, Sweden;
78Centenario Hospital Miguel Hidalgo, Aguascalientes, México;
79Department of Neurology, Antonius Ziekenhuis, Sneek, The Netherlands;
80Rijnstate Hospital Arnhem, Arnhem, The Netherlands;
81Department of Neurology, Royal North Shore Hospital, Sydney, Australia;
82Ulm University Hospital, Ulm, Germany;
83University Hospital of Grenoble, Grenoble, France;
84Hospices Civils de Lyon, Lyon, France;
85Department of Neurology, Arak University of Medical Sciences, Arak, Iran;
86UNICAMP Universidade Estadual de Campinas, Campinas, Brazil;
87Hospital Universitário Professor Alberto Antunes, Universidade Federal de Alagoas (HUPAA/UFAL/EBSERH), Maceió, Brazil
88University Hospital of Nancy, Nancy, France;
89Virgen del Rocio University Hospital, Seville, Spain;
90Instituto Panvascular de Occidente, Guadalajara, Jalisco, México;
91Johannes Kepler University Linz, Linz, Austria;
92Hospital complex of Toledo, Toledo, Spain;
93Neurology Unit ASST Valcamonica, Esine, Brescia-Italy;
94Stroke Unit, Department of Neurosciences, Germans Trias i Pujol University Hospital, Badalona, Spain;
95Azienda Unità Sanitaria Locale-IRCCS di Reggio Emilia, Reggio Emilia, Italy;
96Department of Neurosciences and Mental Health, Hospital de Santa Maria, Centro Hospitalar Universitario Lisboa Norte, University of Lisbon, Lisbon, Portugal;
97University Hospital of Poitiers, Poitiers, France;
98Department of Neurology, Centro Hospitalar Universitario São João and Department of Clinical Neurosciences and Mental Health, Faculty of Medicine, University of Porto, Portugal;
99Sina Hospital, Hamadan University of Medical Sciences, Hamadan, Iran;
100Institute of Neurosciences, SIMS hospital, Chennai, Tamil Nadu, India;
101Brain Mapping Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran;
102Neurology and Stroke Unit, Department of Neuroscience, Bufalini Hospital, Cesena, Italy;
103Hospital de Cascais Dr. José de Almeida, Cascais, Portugal;
104Isfahan Neurosciences Research Center, Isfahan University of Medical Sciences, Isfahan, Iran;
105IRCSS Ospedale Policlinico San Martino, Genoa, Italy;
106Vivantes Auguste-Viktoria-Klinikum, Berlin, Germany;
107Firoozgar hospital, School of Medicine, Iran University of Medical sciences, Tehran, Iran;
108Department of Neurology, Lorestan University of Medical Sciences, Khorramabad, Iran;
109Sahyadri Superpeciality Hospital, Pune, Maharashtra, India;
110Sree Chitra Tirunal Institute for Medical Sciences and Technology, Trivandrum, Kerala, India;
111Department of Neurology, Centre Hospitalier Universitaire de Toulouse, Toulouse, France;
112Leiden University Medical Centre, Leiden, The Netherlands;
113Department of Neurology, CHU Grenoble Alpes, Grenoble, France;
114Hospital de la Cavale Blanche, CHRU de Brest, Brest, France;
115Institute of Cardiovascular Disease, Royal Holloway University of London, London, United Kingdom;
116Neurology and Stroke Unit, Cardarelli Hospital, Naples, Italy;
117Department of Neurology, Cliniques Universitaires Saint-Luc, Université Catholique de Louvain, Hippocrate 10, 1200, Brussels, Belgium;
118Lariboisière Hospital, Neurology Department, Assistance Publique Hopitaux de Paris, France;
119Department of Neurology, Bushehr University of medical science, Bushehr, Iran;
120Universidade Federal do Cariri, Juazeiro do Norte, Brazil;
121Department of Neurology, Royal Adelaide Hospital, Adelaide, Australia
122Department of Neurology & stroke unit, Hôpital de la Cavale Blanche, CHRU de Brest (University Hospital), Université de Bretagne Occidentale, Inserm1078, Brest, France;
123Foothills Medical Centre, Calgary, Canada;
124Edinburgh Medical School, University of Edinburgh, Edinburgh, Scotland;125KIMS Hospital, Hyderabad, Telangana, India;
1262nd Department of Neurology, Aristotle University of Thessaloniki, School of Medicine, AHEPA University Hospital, Greece;
127University Hospital Southampton NHS Foundation Trust, Southampton, United Kingdom;
128Department of Neurology and Division of Neuroradiology, Vascular and Interventional Radiology, Department of Radiology, Medical University of Graz, Austria;
129St John’s Medical College Hospital, Bengaluru, Karnataka, India;
130Dept. of Neurology, University Medical Center Schleswig-Holstein, Campus Kiel, Germany;
131Isfahan University of Medical Sciences (IUMS) and Isfahan Neurosciences Research Center (INRC), Isfahan, Iran;
132Unidad medica de alta especialidad No. 25. Monterrey Nuevo León, Mexico;
133Department of Neurology, Stroke Unit, Ospedale del Mare, ASL Napoli 1 Centro, Napoli, Italy;
134Medicana İzmir International Hospital, Izmir, Turkey;
135Department of Neurology, Firoozgar Hospital, School of Medicine, Iran University of Medical Sciences, Tehran, Iran;
136Neurology, public Imam Hossein Hospital, Kermanshah, Iran;
137Department of Neurology, Firoozabadi Hospital, Iran University of Medical Sciences, Tehran, Iran;
138Institute for Clinical and Experimental Transfusion Medicine, Medical Faculty of Tuebingen, University Hospital of Tuebingen, Tuebingen, Germany;
139Department of Vascular Medicine, Amsterdam UMC, University of Amsterdam, Amsterdam, The Netherlands;
140Department of Viroscience, Erasmus Medical Center Rotterdam, Rotterdam, The Netherlands.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: A.S. has received a grant from Swiss Heart Foundation; C.C. has received speaker honoraria from Boehringer Ingelheim, personal fees for advisory board participation from AstraZeneca and Biogen, and personal fees for steering committee participation from Biogen and Bristol Myers Squibb; T.J.K. has received educational meeting cost assistance from Boehringer Ingelheim; T.S.F. receives in-kind study medication from Bayer Canada and advisory board honoraria from HLS Therapeutics; S.P. received research support from BMS/Pfizer, Boehringer-Ingelheim, Daiichi Sankyo, European Union, German Federal Joint Committee Innovation Fund, and German Federal Ministry of Education and Research, Helena Laboratories and Werfen as well as speakers’ honoraria/consulting fees from Alexion, AstraZeneca, Bayer, Boehringer-Ingelheim, BMS/Pfizer, Daiichi Sankyo, Portola, and Werfen (all outside the submitted work); R.L. reports fees paid to his institution for consultancy by Boehringer Ingelheim, Genentech, Ischemaview, Medtronic and Medpass; E.L. has received academic grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG 942851), Swedish Neurologic Society, Elsa and Gustav Lindh’s Foundation, Wennerströms’ Foundation, P-O Ahl’s Foundation and Rune and Ulla Amlöv’s Foundation for research on CVT; A.C. received speaker grants from Alexion Pharma, Italfarmaco, and Daiichi-Sankyo; M.W. has received consulting fees from Portola/Alexion; C.J. has received speaker honoraria from Alexion, CSL Behring, TEVA and Sanofi Genzyme, personal fees for advisory board participation from Alexion, Roche, Novartis and Merck Serono; H.K. has received personal fees for consulting and data safety monitoring board activities for Octapharma, Grifols, CSL Behring, UCB, Argenx, Takaeda, Alexion and his institution has received clinical trial support from Takaeda; S.N. has received consulting fees from Brainomix and lecture fees from Boehringer Ingelheim and BMS Pfizer; T.G. has received travel grants and speaker honoraria from Boehringer Ingelheim, Bayer, Novartis, BMS / Pfizer and Alexion; A.G. has received personal fees from Bayer Vital, Bristol Myers Squibb, and Daiichi Sankyo; K.J. has received academic grants from the Swedish state under the agreement between the Swedish government and the county councils, the ALF agreement (ALFGBG 965417) for research on CVT; T.T. has received personal fees from Bayer, Boehringer Ingelheim, Bristol Myers Squibb, Inventiva, and Portola Pharma; D.A.S. reports travel support from Boehringer Ingelheim, speaker fees from Bayer, and Advisory Board participation for AstraZeneca; J.M.F. has received personal fees from Boehringer Ingelheim, Bayer, and Daiichi Sankyo as well as grants from Bayer; J.M.C. has received grants paid to his institution from Boehringer Ingelheim and Bayer, and payments paid to his institution for data safety monitoring board participation by Bayer; M.A. reports personal fees from AstraZeneca, Bayer, Bristol Myers Squibb, Covidien, Daiichi Sankyo, Medtronic, Novartis, Pfizer, and Amgen; M.R.H. reports grants from the Swiss Heart Foundation and from the Bangerter Foundation, and Advisory Board participation for Amgen. All other authors have nothing to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by The Netherlands Organisation for Health Research and Development (ZonMw, grant number 10430072110005), the Dr. C.J. Vaillant Foundation, and Hospital District of Helsinki and Uusimaa (grant TYH2022223).
Informed consent
Not applicable
Ethical approval
The ethical review committee of the Academic Medical Center in Amsterdam approved this study. Each center was responsible for obtaining permission from local authorities if required by national and local law.
Guarantor
M.A.
Contributorship
Conceptualization: M.A., M.R.H., J.M.C., J.M.F., A.S, A.M.. Methodology: M.A., M.R.H., A.M., A.S., J.M.F., J.M.C. Validation: A.M., A.S.. Formal analysis: A.S., A.M.. Investigation: All authors. Resources: J.M.C. Data Curation: A.M., K.K., M.S.K. Writing-Original Draft: A.S., A.M., J.M.F., J.M.C., M.R.H., M.A. Writing-Review & Editing: All authors. Visualization: A.S., A.M., J.M.F., J.M.C., M.A., M.R.H.. Supervision: M.A., M.R.H., D.A.S., K.J., S.P., J.P., T.T., J.M.F., J.M.C.. Project administration: A.M., A.S., K.K., M.S.K., S.P., A.S., E.L., A.G., K.J., T.T., M.R.H., M.A., D.A.S., J.M.F., J.M.C. Funding acquisition: J.P., J.M.C., A.S., A.M., M.R.H. and M.A. directly accessed and verified the underlying data reported in the manuscript.
Data sharing
The de-identified, individual participant data that underlie the results reported in this article can be made available to investigators whose proposed use of the data has been approved by the International Cerebral Venous Thrombosis Consortium Leadership. Proposals should be directed to the study’s Principal Investigator (Dr. Jonathan Coutinho, email:
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
