Abstract
The purpose of this study was to determine whether the longitudinal progression of decline in left ventricular ejection fraction (LVEF) in Duchenne muscular dystrophy (DMD) patients is moderated by ADRB1 genotype and whether the efficacy of ß-blocker therapy is influenced by genotype status. About 147 DMD patients (6-34 years.) were analyzed with a focus on β1 adrenergic receptor (ADRB1) genotype variants. Patients were grouped by ADRB1 genotype resulting in Gly389 patients and Arg389 patients. A generalized additive mixed effects model was used to examine differences in the nonlinear trend of LVEF across patient ages between genotype groups and for ß-blocker use. Both genotype groups displayed a progressive decline in LVEF starting around the mean age of ambulation loss (~12 years). However, there was no difference between genotype groups in the progression of decline in LVEF. There was a significant effect of ß-blocker use on longitudinal LVEF, wherein patients on ß-blockers had systematically lower LVEF when compared to patients not on ß-blockers. However, the effect of ß-blocker therapy on LVEF was not affected by ADRB1 genotype. The current study did not demonstrate an influence of patient ADRB1 genotype on longitudinal LVEF in our cohort. Despite previous literature suggesting a positive influence of ß-blocker use on cardiac function in DMD patients and of an ADRB1 genotypic difference in responsiveness to ß-blocker use, we did not observe this in our cohort. Interestingly, our cohort did not demonstrate a positive influence of ß-blocker use on LVEF measures.
Keywords
Introduction
The respiratory component of Duchenne muscular dystrophy (DMD) is primarily characterized by respiratory muscle weakness and severe respiratory derangement.1,2 However, as respiratory care for DMD patients improves with widespread respiratory function monitoring and early respiratory intervention, DMD related cardiovascular disease (CVD) has emerged as a significant source of morbidity and mortality. 3 Indeed, CVD is the primary cause of mortality in over 20% of DMD patients. 4 Unfortunately, due to the physical disabilities engendered by DMD, it is difficult to surveil these patients for early signs and symptoms of CVD, such as a reduced exercise capacity. Without appropriate cardiac monitoring, this lack of symptoms can lead to a late diagnosis of cardiac dysfunction in DMD patients, which delays evaluation, referrals to cardiology units, and the initiation of treatment. 3 As such, the proper diagnosis and treatment of CVD are essential aspects of effective care in DMD patients. 5 While recommendations for cardiac care have been outlined in recent clinical guidelines, 6 the importance of cardiac surveillance in DMD patients remains underappreciated, and clinical management strategies for CVD are quite variable and under-utilized in this population.3,7
Left ventricular ejection fraction (LVEF) is a frequently used clinical measure of left ventricular dysfunction and CVD progression in patients with DMD. 8 A compromised LVEF is a common cardiac finding and a strong predictor of mortality in DMD patients.9,10 As such, the American Academy of Pediatrics suggest clinicians manage concomitant CVD in DMD patients using a similar pharmacotherapy approach to that used for patients with primary CVD; that is, ACE inhibition, angiotensin receptor blockade, ß-blocker therapy and mineralocorticoid receptor antagonists.8,11 ß-blocker therapy in patients with DMD is known to improve both systolic and diastolic function. 12 Moreover, Matsumura et al 13 reported that the incidence rate of CVD primary endpoints (ie, death, declines in cardiac function, and severe arrhythmias) and all-cause mortality risk were lower in DMD patients on ß-blocker therapy. It is important to note that individual responsiveness to ß-blocker therapy may be modulated by a common genetic variation in the β1-adrenoceptor (ADRB1).14,15 A single nucleotide polymorphism (SNP) of the ADRB1 that modulates receptor functionality includes an arginine (Arg) for glycine (Gly) substitution at amino acid 389.16-19 Specifically, the Arg389 variants of the ADRB1 demonstrate enhanced receptor function within the organs and tissues of the cardiovascular system.14,15,20 In fact, primary CVD patients with the Arg389 polymorphism have significantly higher systolic, diastolic, mean arterial blood pressure, and heart rate—contributing to poorer cardiac function and increased mortality risk.19,21-23 Given such influence of ADRB1 functionality on cardiovascular health, the ADRB1-coupled pathway has long been considered a pharmacological target for preserving cardiac function in cardiomyopathies.18,19,21,23,24 Indeed, research has demonstrated individuals (eg, CVD and healthy subjects) with the Arg389 variant are more likely to be “responders” to ß-blocker therapy when compared with carriers of the Gly389 polymorphism25-28
While the relationship between ADRB1 genotype status and responsiveness to ß-blocker therapy has been well-documented in both primary CVD and controls, little is known about this relationship in patients with DMD. If ß-blockers are to be recommended as a therapy in cardiac management in DMD patients, 8 we must determine the influence of ADRB1 genotype on ß-blocker therapy efficacy in this population. Research has demonstrated ß-blockers have an approximately 50% effectiveness rate, suggesting there is a significant genetic component to therapy efficacy. 29 Given the relatively high rate of ineffective ß-blocker therapies, it is not uncommon to “layer” drugs to achieve proper cardiac management. 8 However, previous work suggests that for each additional drug prescribed there is up to an 80% reduction in medication adherence. 30 Further, this clinical approach of “layering” drug therapies may have many potential short- and long-term consequences, consisting of, but not limited to, additional costs to the patient, increased side-effect profiles and healthcare service utilization, and reductions in quality-of-life. 31
Given the well-described relationship between ADRB1 genotype and responsiveness to ß-blocker therapy, it is conceivable that DMD patients expressing the Gly389 variant would have improved LVEF while DMD patients possessing the Arg389 polymorphism would more likely be “responders” to ß-blocker therapy. Therefore, the aims of this study were to examine the influence of ADRB1 genotype on longitudinal measures of LVEF in patients with DMD, and to determine whether this influence is moderated by ß-blocker use. We hypothesized that DMD patients expressing the “less” functional ADRB1 polymorphism (ie, Gly389) will demonstrate a less marked progression of decline in LVEF compared with patients expressing the more functional polymorphism (ie, Arg389). Additionally, we hypothesize that ß-blocker use will further slow the decline in LVEF in both ADRB1 variants, with the Arg389 polymorphism demonstrating enhanced responsiveness to ß-blocker therapy when compared with Gly389 carriers.
Methods
Patients
The data analyzed in this current study were a part of a larger dataset from the Cooperative International Neuromuscular Research Group Duchenne Natural History Study (CINRG-DNHS). All patients included in this study and/or their legal guardians specifically consented to genotyping for research purposes, and the study was approved by local institutional or ethics review boards at each participating institution. One hundred forty-seven patients with a clinical DMD diagnosis (ages 4-30 years at study entry) were included in the data analyzed in the current study and were followed for up to 9 years. This study focused on the functional ADRB1 protein-altering variant at codon 389. Specific genotyping was performed utilizing an Exome Chip (ie, genotyping chip focusing on variants within the gene-coding portions of the genome). Genotyping via an Exome Chip and the data cleaning methods used in the CINRG-DNHS cohorts have been previously described by others. 32
Data analysis
Patient demographics were obtained from the CINRG-DNHS database (ie, age, height, weight, corticosteroid-use, ambulatory status, and clinic site). All reported patient heights were calculated from ulnar length, as have been previously described. 33 Corticosteroid-use was derived from the clinically reported start and stop dates and reported as the total number of years a patient was on corticosteroid treatment. Ambulatory status was defined as ambulatory or non-ambulatory and derived from the clinically reported date of full-time wheelchair reliance. Clinic site was reported as the city in which the patients’ clinic visit took place. Left ventricular ejection fraction was reported as the clinically observed measures at the patients’ clinic visits.
Statistical analysis
Group demographics were compared using a one-way analysis of variance (ANOVA). A Tukey’s honest significant difference (HSD) post-hoc comparison was used to investigate differences between the specific genotype groups. A generalized additive mixed effects model (GAMM) was used to examine the influence of ß-blocker use and ADRB1 genotype variant on the nonlinear trend in longitudinal measures of LVEF after adjusting for patient demographics (age, height, weight, ambulatory status, corticosteroid-use, and clinic site). A GAMM was used because it allows for the fitting of nonlinear splines when adjusting for various covariates. We chose to group ADRB1 genotypes according to a dominant model for the Arg allele, that is, homozygous for Gly at amino acid spot 389 (henceforth named “Gly389 group,” n = 80) versus homozygous or heterozygous for Arg at amino acid 16 (“Arg389 group,” n = 67) because preliminary analyses of the data demonstrated phenotypic similarity of heterozygotes with Arg homozygotes. Additionally, this dominant model is consistent with previous literature.34-36
The GAMM model used in this study was selected through the interrogation of multiple competing models. Included in these models were main effects; the 3-way interaction with ADRB1 genotype, ß-blocker use, and patients’ age; and all relevant lower order 2-way interaction terms. Random effects for patient ID, age, and clinic site were also included in the model. One hundred seventy-five DMD patients were included in the initial model (n = 95 and 80, for Gly389 and Arg389 respectively). However, 28 patients were selected out due to not having a LVEF measurement on file, resulting in 147 patients in the final model (n = 80 and 67, for Gly389 and Arg389 respectively). The selection of group-level main effects and interaction terms was determined using a backward selection method based on the Akaike’s Information Criteria (AIC) score. 37 The final GAMM model was fit using the restricted maximal likelihood (REML) method, cubic regression penalties for nonlinear smooths, and the hyperparameter γ was set to 1.4 to reduce overfitting.38,39 An extra penalty was added to each individual term so it could be penalized to zero, thereby allowing terms to be automatically “selected out” from the GAMM when appropriate.
Results
Subject characteristics
The frequency of genotypes at rs1801253 was as follows: 54.3% and 45.7% for Gly389 and Arg389 respectively and passed the Hardy-Weinberg equilibrium. There were no differences between genotype groups for patient age, height, weight, corticosteroid use, or LVEF at study entry or at the last observed clinic visit (Table 1). Additionally, there were no differences between genotype groups in the percent of non-ambulatory patients at study entry, nor for the mean age of loss of ambulation (50% and 45%, and 9.99 years and 9.97 years, for Gly389 and Arg389 respectively). Furthermore, our cohort included 22 patients who did not receive corticosteroid treatment at any point during the study follow-up (n = 17 and 5, for Gly389 and Arg389 respectively). There were differences between genotype variants when grouped by ß-blocker use for age, height, corticosteroid use, and LVEF at both study entry and at the last observed clinic visit (Table 2). Specifically, DMD patients who expressed either the Gly389 or Arg389 polymorphism but were on ß-blockers were older, taller, and had a longer corticosteroid use history compared with DMD patients expressing either polymorphism but were not on ß-blockers. Additionally, patients with DMD who expressed either the Gly389 or Arg389 polymorphism but were on ß-blockers were heavier at study entry compared with DMD patients expressing either polymorphism but were not on ß-blockers. However, there was no difference between groups for weight at the last observed clinic visit (Table 2).
Subject characteristics at entry into the study and at last observed clinic visit with a left ventricular ejection fraction (LVEF) measurement.

SE: standard error; Arg389: patients who were homozygous or heterozygous for the β1-adrenergic receptor (ADRB1) resulting in at least one arginine substitution at amino acid 389 (n = 67); Gly389: patients who were homozygous for ADRB1 resulting in a glycine substitution at amino acid 389 (n = 80); corticosteroid-use years refers to the total number of years a patient was on corticosteroid treatment. There were no difference between genotype groups at neither entry into the study nor the last observed clinic visit.
Subject characteristics at entry into the study and at last observed clinic visit with a left ventricular ejection fraction (LVEF) measurement grouped by β1-adrenergic receptor genotype (Arg389 and Gly389) and beta-blocker use.

SE: standard error; Gly389 no BB refers to patients with the Gly389 variant not taking beta-blockers (n = 65); Gly389 on BB refers to patients with the Gly389 variant taking beta-blockers (n = 15); Arg389 no BB refers to patients with the Arg389 variant not taking beta-blockers (n = 53); Arg389 on BB refers to patients with the Arg389 variant taking beta-blockers (n = 15). Bolded P-values denote statistical significance (P < .05) and refer to patients on BB and patients not on BB comparisons regardless of genotype. No other comparisons were significant.
Generalized additive mixed model
Generalized additive mixed model
There was no main effect of patient ADRB1 genotype on the longitudinal trend in LVEF (Table 3). There was, however, a significant main effect of patient ß-blocker use on longitudinal LVEF in our cohort (Table 3). Patients with DMD who were on ß-blockers at entry into the study demonstrated systematically lower LVEF when compared with those patients who were not on a ß-blocker at study entry (Figure 1). Patient age displayed a significant main effect on the longitudinal trend in LVEF (P < .001); wherein both genotype groups demonstrated a longitudinal decline in LVEF (Table 3). There was also a significant main effect of patient corticosteroid use history on the longitudinal trend in LVEF (P < .01). Specifically, patients with a longer corticosteroid use history had systematically higher LVEF compared to those with a shorter corticosteroid use history. There were no significant main effects of patient height, weight, or ambulatory status on the longitudinal trend in LVEF (Table 3).
Generalized additive mixed model (GAMM) results for left ventricular ejection fraction (LVEF) measures in patients with Duchenne muscular dystrophy (DMD).
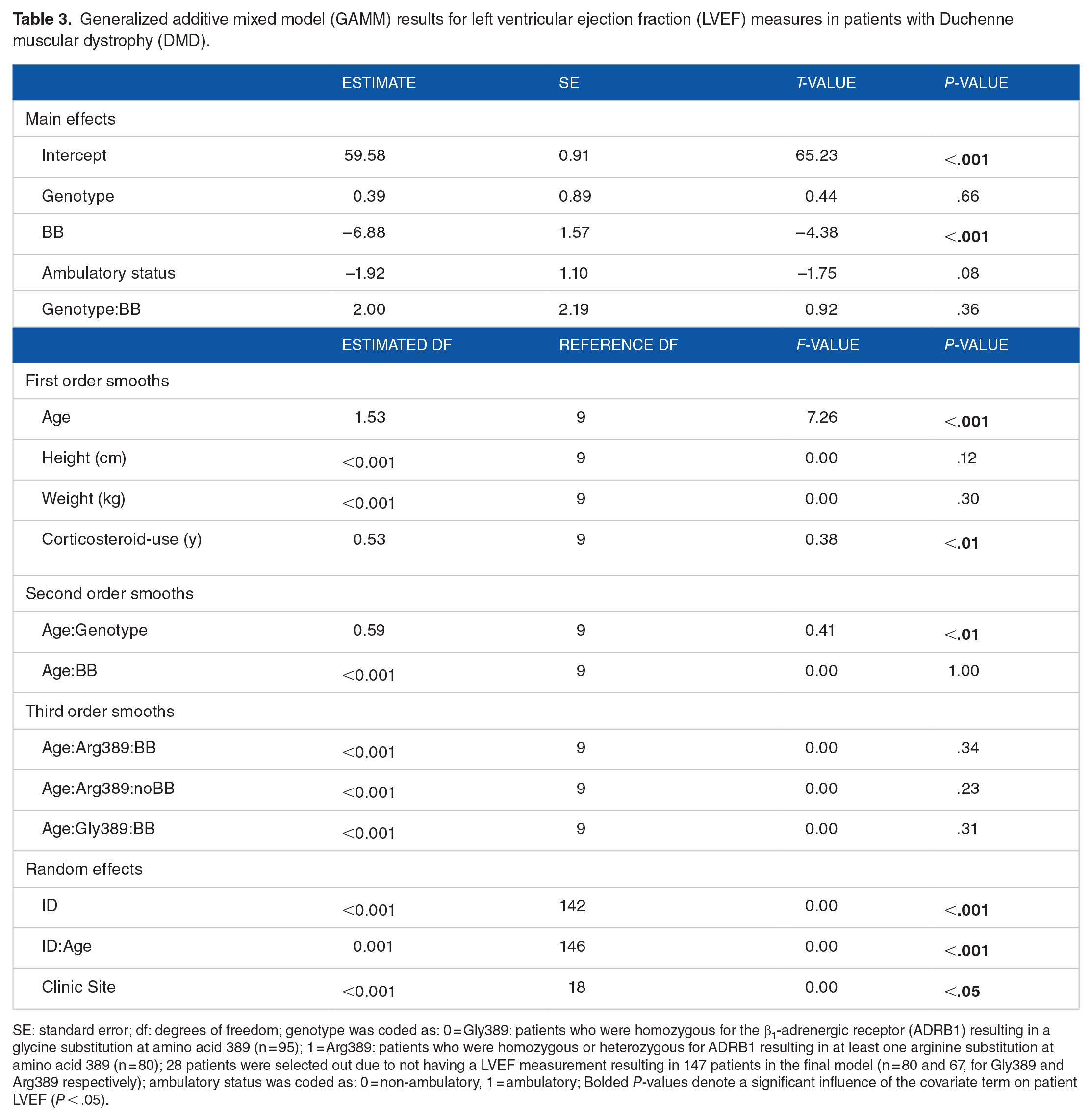
SE: standard error; df: degrees of freedom; genotype was coded as: 0 = Gly389: patients who were homozygous for the β1-adrenergic receptor (ADRB1) resulting in a glycine substitution at amino acid 389 (n = 95); 1 = Arg389: patients who were homozygous or heterozygous for ADRB1 resulting in at least one arginine substitution at amino acid 389 (n = 80); 28 patients were selected out due to not having a LVEF measurement resulting in 147 patients in the final model (n = 80 and 67, for Gly389 and Arg389 respectively); ambulatory status was coded as: 0 = non-ambulatory, 1 = ambulatory; Bolded P-values denote a significant influence of the covariate term on patient LVEF (P < .05).

The nonlinear trend in left ventricular ejection fraction (LVEF) in Duchenne muscular dystrophy (DMD) patients grouped by ß-blocker use. The predicted smoothed LVEF (Panel (A)) and predicted difference in LVEF (Panel (B)) were obtained from GAMM modeling of longitudinal LVEF measures where ß-blocker use, genotype group, age, ambulatory status, corticosteroid-use, weight, height, and clinic site were entered into the model as covariates. The predicted smoothed curves in Panel (A) was produced for ß-blocker use with set numeric predictors for all other covariates (Height = 151.4 cm; Weight = 47.4 kg; Corticosteroid-use = 4.8 years; Ambulatory status = 0.63). The thick lines indicate the ages at which LVEF measures significantly differed between patients on ß-blockers and those not on ß-blockers (4.5 and 29.6 years). Panel (B) shows the predicted difference curve between patients on ß-blockers and those not on ß-blockers. The shaded region indicates the ages at which patients not on ß-blocker therapy had significantly higher LVEF measures compared with patients on ß-blocker therapy (4.5-29.6 years).
There was no interaction between ADRB1 genotype and ß-blocker status on the longitudinal trend in LVEF. Additionally, there was significant individual variation (random effect) in the nonlinear longitudinal trend in LVEF within our cohort of DMD patients (P < .001) (Table 3). Furthermore, clinic site was a significant source of random variation in LVEF in patients with DMD (P < .001) (Table 3).
Discussion
Our original hypothesis was that DMD patients expressing the “less” functional Gly389 polymorphism would demonstrate a less marked progression of decline in LVEF compared with patients expressing the Arg389 polymorphism. Additionally, we hypothesized that ß-blocker therapy would further slow the decline in LVEF in both ADRB1 variants, with the Arg389 polymorphism demonstrating enhanced responsiveness to ß-blocker therapy when compared with Gly389 carriers. The findings of the current study refute this hypothesis insofar as patients with the Gly389 variant did not demonstrate a difference in the progression of decline in LVEF when compared with patients expressing the Arg389 genotype variant. Furthermore, ß-blocker use does not appear to present a positive influence on LVEF in either genotype variant. In fact, DMD patients on ß-blockers had systematically lower LVEF compared with those patients not on ß-blockers. Furthermore, our cohort failed to demonstrate a “responder” effect to ß-blocker therapy based on ADRB1 genotype. These findings suggest that there is neither an influence of ADRB1 genotype variant on LVEF in our cohort of DMD patients, nor does genotype affect responsiveness to ß-blocker therapy.
ADRB1 genotype and longitudinal LVEF measures in DMD
The ADRB1 coupled pathway has long been a pharmacological target for preserving cardiac function in cardiomyopathies.18,19,21,23,24 ADRB1 activity has been shown to influence cardiac: (1) inotropy, lusitropy, and chronotropy40,41; (2) nuclear and perinuclear Ca2+ handling 42 ; (3) reuptake of cytosolic Ca2+43; and (4) cardiomyocyte relaxation.16,44 As such, the less “functional” genotype variant (ie, Gly389) is associated with lower blood pressure, improved cardiac function, and decreased mortality risk in heart failure.19,21-23 Previous literature suggests ADRB1 functionality strongly influences cardiovascular health, and that dampening ADRB1 function may improve outcomes in CVD.19,21-23 Indeed, these beneficial effects of ß-blocker use have also been demonstrated in DMD patients.12,13
In light of the above, clinical care guidelines suggest the use of ß-blocker therapy for the treatment of CVD in DMD patients. 45 However, previous research has identified the efficacy of ß-blocker therapy may largely depend on ADRB1 genotype, with Arg389 carriers more likely being “responders” than Gly389 carriers.25-27 However, our data failed to demonstrate an influence of ADRB1 genotype on patient responsiveness to ß-blocker therapy in our cohort. Specifically, the effectiveness of ß-blocker therapy on slowing the progression of decline in LVEF in DMD patients was not influenced by their genotype.
While the data may suggest an overall detrimental effect of BB on LVEF in DMD, the limitations of the study cannot support such a conclusion. We propose 3 possible factors for our contradictory results wherein ß-blocker use appeared detrimental to cardiovascular function (ie, LVEF) in our cohort of DMD patients. First, the DMD patients in our cohort who were on ß-blockers may in fact be sicker than those patients not on ß-blocker therapy. As such, patients on ß-blockers may have systematically lower LVEF by virtue of a worse clinical prognosis. In fact, patients on ß-blockers were also more likely to be on additional therapies (Supplemental Table 1). Secondly, the variability in cardiac management across clinic sites may account for these findings. As demonstrated in the current study, there was a significant influence of clinic site on longitudinal LVEF measures in our cohort, independent of ß-blocker use and other covariates (Table 3). As such, DMD patients receiving care at different clinics may be prescribed different medications to manage their CVD (Supplemental Table 1). The use of other medications (eg, ACE inhibitors, ARBs, etc.) in those patients not treated with BBs may suggest BB therapy was not indicated in these patients. Furthermore, this difference in approach to therapy selection and medication layering may alter the effectiveness of ß-blockers depending on their use as a first-line or additional therapy in our cohort. Additionally, the timing and efficacy of respiratory treatment may differ by clinic site as well. Previous literature has also demonstrated an attendant decline in respiratory and cardiac function in DMD patients, either from dependent or independent pathologies wherein more effective respiratory management has been correlated to improved cardiac function.46-48 This suggests the level of respiratory care and management may influence LVEF measures in DMD patients. These 3 factors may help to explain the contradictory findings regarding the influence of ß-blockers on LVEF measures in our cohort.
Other factors influencing longitudinal LVEF measures in DMD
Our model demonstrated a protective effect of corticosteroid-use on longitudinal LVEF measures for both genotype groups in our cohort (Table 3). Conversely, age had an inverse relationship with LVEF measures in our DMD patients. Firstly, it is not surprising that corticosteroid-use had a protective effect on LVEF, given that corticosteroid-use in DMD patients has been demonstrated to slow the progressive decline in cardiac function.49,50 Secondly, one would expect age to exert a negative influence on LVEF measures because DMD is a progressive disease whereby clinical prognosis deteriorates as patients age.
Methodological considerations
It must be acknowledged that there are inherent limitations in a natural history cohort. However, these limitations are mitigated by the fact that our cohort reflects current clinical management. Further, this study did not include preclinical markers of CVD in our cohort. Additionally, we were not able to include the random effects for height, weight, corticosteroid use, and ambulatory status in the model because there was not a sufficient amount of data to support the necessary degrees of freedom to appropriately model these effects.
Conclusion
The current study did not demonstrate an influence of patient ADRB1 genotype on longitudinal LVEF in our cohort. Despite previous literature suggesting a positive influence of ß-blocker use on cardiac function in DMD patients and of an ADRB1 genotypic difference in responsiveness to ß-blocker use, we did not observe this in our cohort. Specifically, the effect of ß-blocker therapy on LVEF in patients with DMD was not modified by ADRB1 genotype. Interestingly, our cohort did not demonstrate a positive influence of ß-blocker use on LVEF measures.
Supplemental Material
sj-docx-1-cic-10.1177_11795468221116838 – Supplemental material for Influence of β1 Adrenergic Receptor Genotype on Longitudinal Measures of Left Ventricular Ejection Fraction and Responsiveness to ß-Blocker Therapy in Patients With Duchenne Muscular Dystrophy
Supplemental material, sj-docx-1-cic-10.1177_11795468221116838 for Influence of β1 Adrenergic Receptor Genotype on Longitudinal Measures of Left Ventricular Ejection Fraction and Responsiveness to ß-Blocker Therapy in Patients With Duchenne Muscular Dystrophy by Eli F Kelley, Troy J Cross, Craig M McDonald, CINRG Investigators, Eric P Hoffman, Christopher F Spurney and Luca Bello in Clinical Medicine Insights: Cardiology
Footnotes
Acknowledgements
The authors are sincerely grateful for the patients that dedicated their time and agreed to participate in the collection of data used in this study. This study could not have been done without their participation. We would also like to thank all of the CINRG investigators whose tireless work and dedication made this research possible.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Heart, Lung, and Blood Institute (grant number 5T32HL105355-08).
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Conceptualization, Data Curation, Formal Analysis, Funding Acquisition, Methodology, Visualization, Writing – Original Draft Preparation, Writing – Review & Editing: EFK. Conceptualization, Data Curation, Formal Analysis, Methodology, Visualization, Writing – Review & Editing: TJC. Data Curation, Writing – Review & Editing: CMM. Data Curation: CINRG Investigators. Data Curation, Writing – Review & Editing: EPH. Data Curation, Writing – Review & Editing: CFS. Conceptualization, Data Curation, Methodology, Writing – Review & Editing: LB.
Data Statements
The data that support the findings of this study are available from The Cooperative Neuromuscular Research Group, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of The Cooperative Neuromuscular Research Group.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
