Abstract
Background:
There is limited data on the impact of comorbidity burden on clinical outcomes of patients undergoing cardiac implantable electronic devices (CIED) implantation.
Objectives:
Our aim was to assess trends in CIED implantations and explore the relationship between comorbidity burden and outcomes in patients undergoing de novo implantations.
Methods:
Using the National Inpatient Sample database from 2000 to 2014, we identified adults ⩾18 years undergoing de novo CIED procedures. Comorbidity burden was assessed by Charlson comorbidity Index (CCI), and patients were classified into 4 categories based on their CCI scores (CCI = 0, CCI = 1, CCI = 2, CCI ⩾3). Annual implantation trends were evaluated. Logistic regression was conducted to measure the association between categorized comorbidity burden and outcomes.
Results:
A total of 3 103 796 de-novo CIED discharge records were identified from the NIS database. About 22.4% had a CCI score of 0, 28.2% had a CCI score of 1, 22% had a CCI score of 2, and 27.4 % had a CCI score ⩾3. Annual de-novo CIED implantations peaked in 2006 and declined steadily from 2010 to 2014. Compared to CCI 0, CCI ⩾3 was independently associated with increased odds of in-hospital mortality, bleeding, pericardial, and cardiac complications (all P < .05). Length of stay and hospital charges increased with increasing comorbidity burden.
Conclusions:
CCI is a significant predictor of adverse outcomes after CIED implantation. Therefore, comorbidity burden needs to be considered in the decision-making process for CIED implant candidates.
Introduction
The utilization of cardiac implantable electronic devices (CIED) such as permanent pacemakers (PPM), cardiac resynchronization therapy (CRT), and implantable cardioverter-defibrillator (ICD) in the management of patients with severe heart failure and heart rhythm abnormalities are well established.1 -3
With the aging population, patients undergoing CIED implantations are older and have a higher comorbidity burden.4,5 In daily clinical practice among patients undergoing CIED implants, multiple comorbidities, whether cardiac or noncardiac, influence a patient’s clinical course synergistically rather than in isolation. The Charlson comorbidity index (CCI), a well-validated comorbidity burden measure, is one of the most used measures of comorbidity burden. 6 CCI was initially developed by Charlson et al 6 to provide an applicable tool based on the seriousness of comorbid conditions for risk stratification of mortality within 1 year of hospitalization. The initial comorbidity index comprised 19 categories and was adapted to 17 comorbid conditions by Deyo et al 7 to ICD-9-CM codes for administrative data use.
The CCI provides a mechanism for weighing the prognostic impact of cardiovascular and non-cardiovascular comorbidities based on their scores. 6 The CCI has proven its risk stratification value in predicting mortality outcomes in some diseases such as heart failure, ischemic stroke, and coronary artery disease.8 -10 Prior studies also revealed that CCI is a significant predictor of mortality, major adverse cerebrovascular and cardiovascular events.11,12 In a single-center study, renal dysfunction and diabetes were shown to be predictors of mortality following CRT implantation; these comorbid conditions are included in the CCI. 13
There are limited studies that have systematically examined trends of CIED implantation and the association of comorbidity index in relation to CIED patients’ outcomes. Thus, we analyzed de novo CIED implantation trends and evaluated the influence of CCI on clinical outcomes in CIED implantations.
Methods
Data source
The data was obtained from hospital discharges between 2000 and 2014 using the National Inpatient Sample (NIS) database, developed by the Healthcare Cost and Utilization Project (HCUP), which is sponsored by the Agency for Healthcare Research and Quality (AHRQ), a not-for-profit agency of United States (US) Department of Health & Human Services. 14 NIS represents a 20% stratified sample of all discharges from all United States community hospitals with approximately 8 million discharges per year. 14 Sampling weights provided by the NIS database allow for generalization to estimate national hospital discharge rates.
Inclusion criteria and clinical outcomes
Individuals ⩾18 years old that underwent de-novo CIED (PPM, CRT-D, CRT-P, ICD) implantations between January 2000 and December 2014 were identified using International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) procedure codes for PPM: 37.70, 37.71, 37.72, 37.73, 37.80, 37.81, 37.82, 37.83, CRT-P: 00.50, CRT-D: 00.51, and ICD: 37.94, 37.95, 37.96.
Comorbidity burden was determined by CCI. 7 CCI uses a scoring system with each identified comorbid condition as shown in Table 1 below weighted by a score ranging from 1 to 6 points based on its prognostic impact. 7 The assigned points were added to calculate a CCI score for each discharge record. Consequently, those without any comorbid condition according to CCI were assigned a CCI of 0, one comorbid condition with a weight of 1 were classified as CCI of 1, those with a comorbid condition with a weight of 2 and 2 comorbidities each weighting 1 were classified as CCI of 2. 7 Then, those with a totaled weighted points of comorbid condition of 3 or more were assigned a CCI of ⩾3. 7 The CCI scores were obtained, and the cohort were categorized into 4 groups based on their CCI score: 0, 1, 2, and ⩾3.
Deyo’s modification of Charlson comorbidity index (CCI).

Table obtained from Deyo et al. 7
Demographic characteristics collected from each hospital discharge include age, gender, race, median household income according to ZIP codes, and expected primary payer. 15 Hospital level characteristics are hospital region (Midwest, Northeast, Southern, Western region), location/hospital teaching status (rural, urban, nonteaching, urban teaching), and hospital bed sizes. 15
Hospital bed size indicates the number of acute care staffed beds in each hospital and the categories of hospital bed sizes vary according to the hospital region, location, and teaching status. 16 The criteria for classification of bed sizes into small, medium, and large are provided in Supplemental Table S1.
The primary outcomes of our study include in-hospital mortality, hospital costs, and length of hospital stay.
Secondary outcomes were procedural complications and included pericardial (composite of hemopericardium, tamponade, pericardiocentesis, acute pericarditis, and unspecified disease of the pericardium), cardiac, pulmonary (composite of pneumothorax, hemothorax, chest tube placement, and other iatrogenic pulmonary complications), vascular (composite of accidental puncture or laceration during a procedure and vascular complication requiring surgical repair), and bleeding complications (defined by hemorrhage/hematoma complicating a procedure or post-hemorrhagic anemia). Complications were identified using ICD-9 CM codes (Supplemental Table S2).
Statistical analysis
Continuous variables are presented as median and interquartile range, and categorical data are presented as percentages. Records were excluded from analysis if missing information on sex, race, income, insurance type, admission period (weekday vs weekend), length of hospital stay, and death status. Differences in sociodemographic and clinical characteristics across comorbidity groups were tested using the Rao-Scott Chi-square test and Kruskal-Wallis test. Based on our study objectives, (A) trends in CIED implantations over time were explored. Using logistic regression, a trend test was conducted from 2000 to 2014, with year as the sole predictor of implantation; (B) logistic regression was used to explore the association between the CCI comorbidities and the specified outcomes.
For all analyses, survey procedures incorporating NIS-specified weights were used. Due to records being sampled by hospitals rather than individuals, clustering of records within hospitals was considered in the analyses. All statistical analyses were conducted using SAS (v. 9.4, SAS Institute, Cary, North Carolina).
Results
A total of 3 103 796 de novo CIED procedures were identified from the NIS between 2000 and 2014.
CIED trends
We observed a stepwise increase in overall de novo CIED implantation from 2000 to 2006 (peak), followed by a steady decline in overall implantation from 2010 to 2014 (Figure 1a). NIS Data from 2000 and 2001 did not capture CRT-P/D implantations as CRT-P and CRT-D first received US Food and Drug Administration approval in 2001 and 2002, respectively. However, there was a general increase in CRT-P implanted per year from 2002 to 2014 (Figure 1d).

(a) Trends of all CIED implantations, 2000 to 2014, (b) annual trends of ICD implantation, 2000 to 2014, (c) annual trends of PPM implantation, 2000 to 2014, (d) annual trends of CRT-P implantation, 2000 to 2014, and (e) annual trends of CRT-D implantation, 2000 to 2014.
Patient characteristics
Figure 2 shows the distribution of CIED records by categorized CCI score: 22.4% had a CCI score of 0, 28.2% had a CCI score of 1, which is the most prevalent score, 22% had a CCI score of 2, and 27.4 % had a CCI score ⩾3 (Figure 2).

Distribution of categorized Charlson comorbidity index score.
Over the 15-year study, the proportion of patients with no comorbidity undergoing CIED implantation declined from 31.2% to 17.9%. However, the proportion of patients with CCI ⩾3 undergoing CIED procedures rose from 16.4% in 2000 to 39.8% in 2014 (Figure 3).
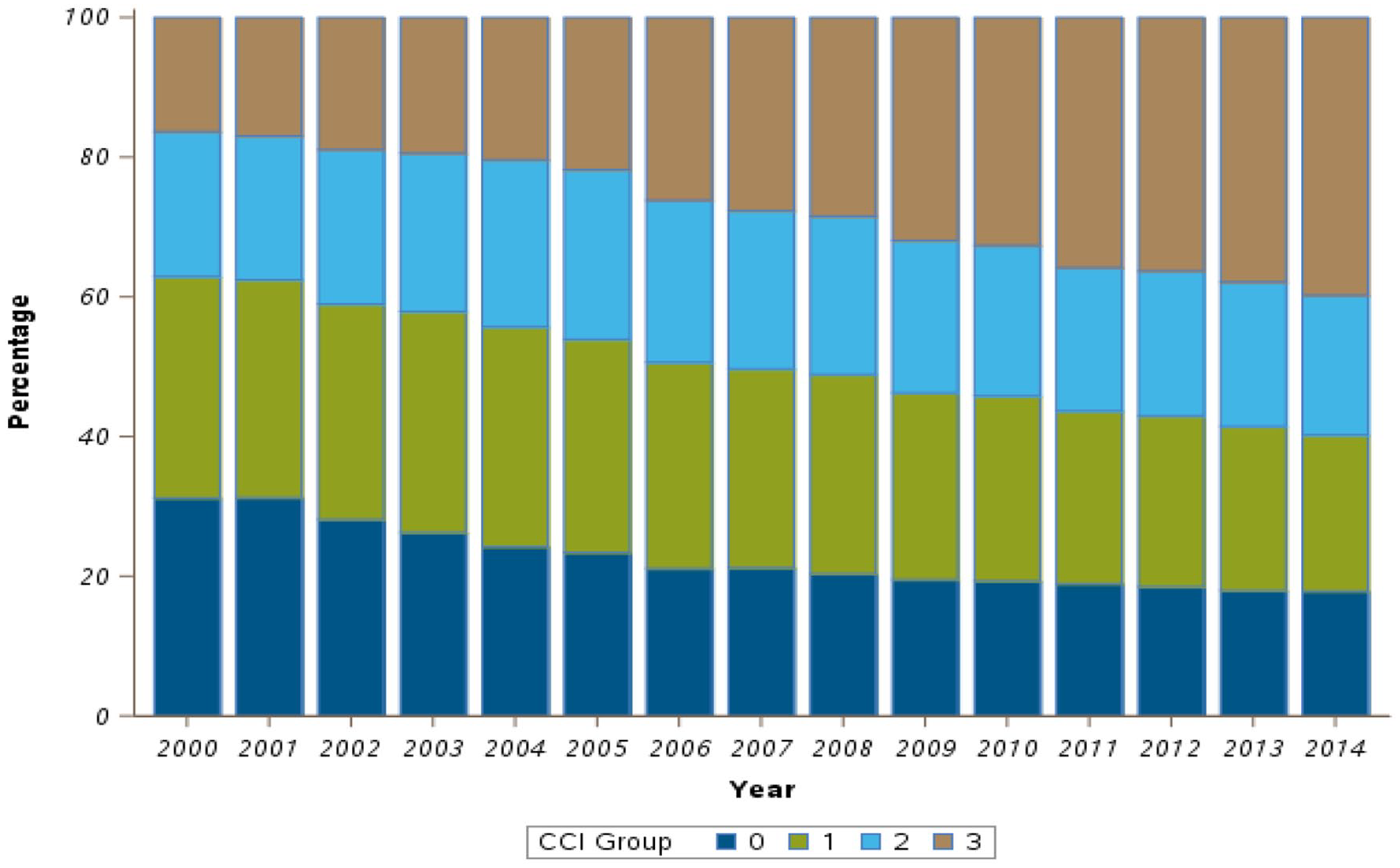
Distribution of categorized Charlson comorbidity index score, 2000 to 2014.
Sociodemographic and clinical characteristics for the included individuals categorized by CCI scores are illustrated in Table 2. The median age for subjects with no comorbidity was slightly higher than subjects with 1 or more comorbidity (76 vs 75 years, P < .001). The incidence of conditions such as cardiac arrest, ventricular tachycardia, ventricular fibrillation, and cardiogenic shock increased as the comorbidity burden increased. The most prevalent comorbidity in CCI ⩾3 group was congestive heart failure (CHF). The prevalence of CHF among patients with CCI of 1 was 41.7%, and it increased to 73.9% among patients with CCI ⩾3. At the same time, the most common non-cardiovascular comorbidity among the CCI ⩾3 group was chronic pulmonary disease.
Sociodemographic data of included individuals stratified by categorized Charlson comorbidity index (CCI) score.
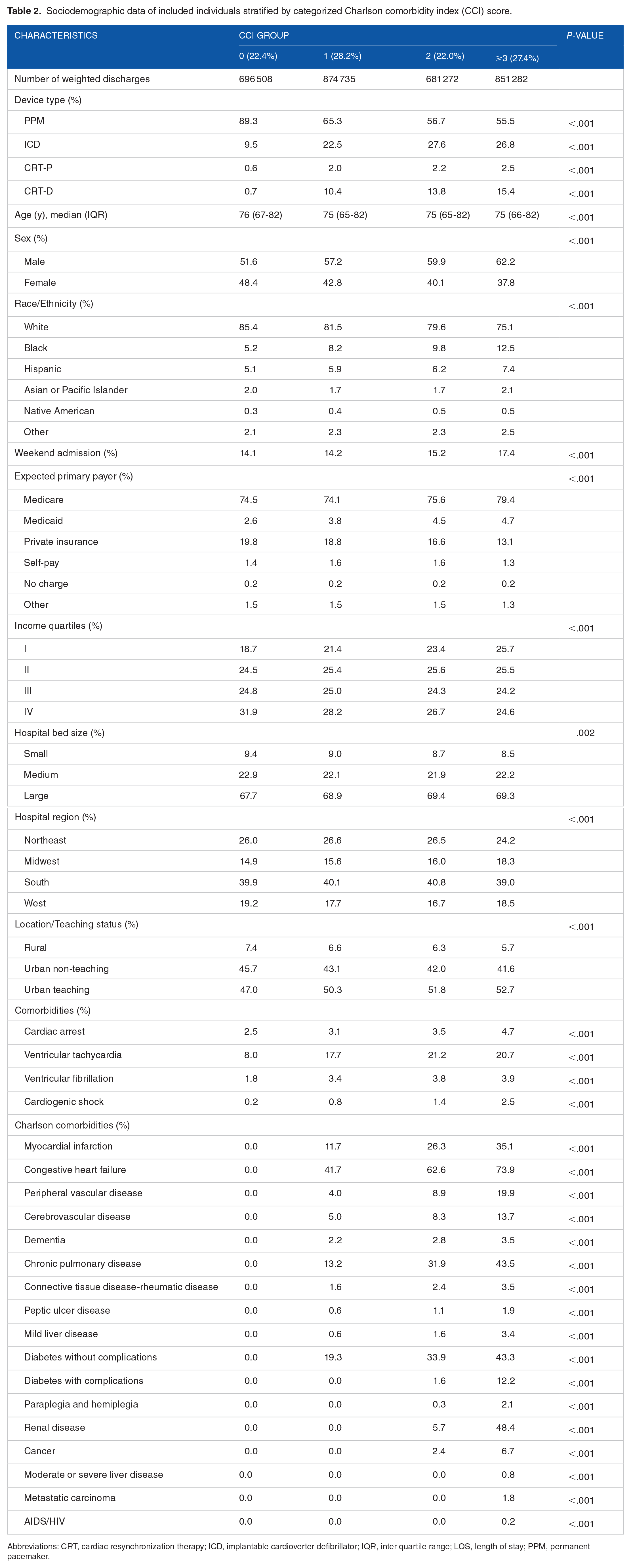
Abbreviations: CRT, cardiac resynchronization therapy; ICD, implantable cardioverter defibrillator; IQR, inter quartile range; LOS, length of stay; PPM, permanent pacemaker.
Outcomes
Median LOS increased 3-fold from 2 days as CCI score improved from 0 to ⩾3 (Table 3). The average total charges showed a stepwise increase with increasing CCI (CCI 0: $41 025, CCI ⩾3: $91 274, P < .001) (Table 3).
Outcomes according to categorized Charlson comorbidity index score.
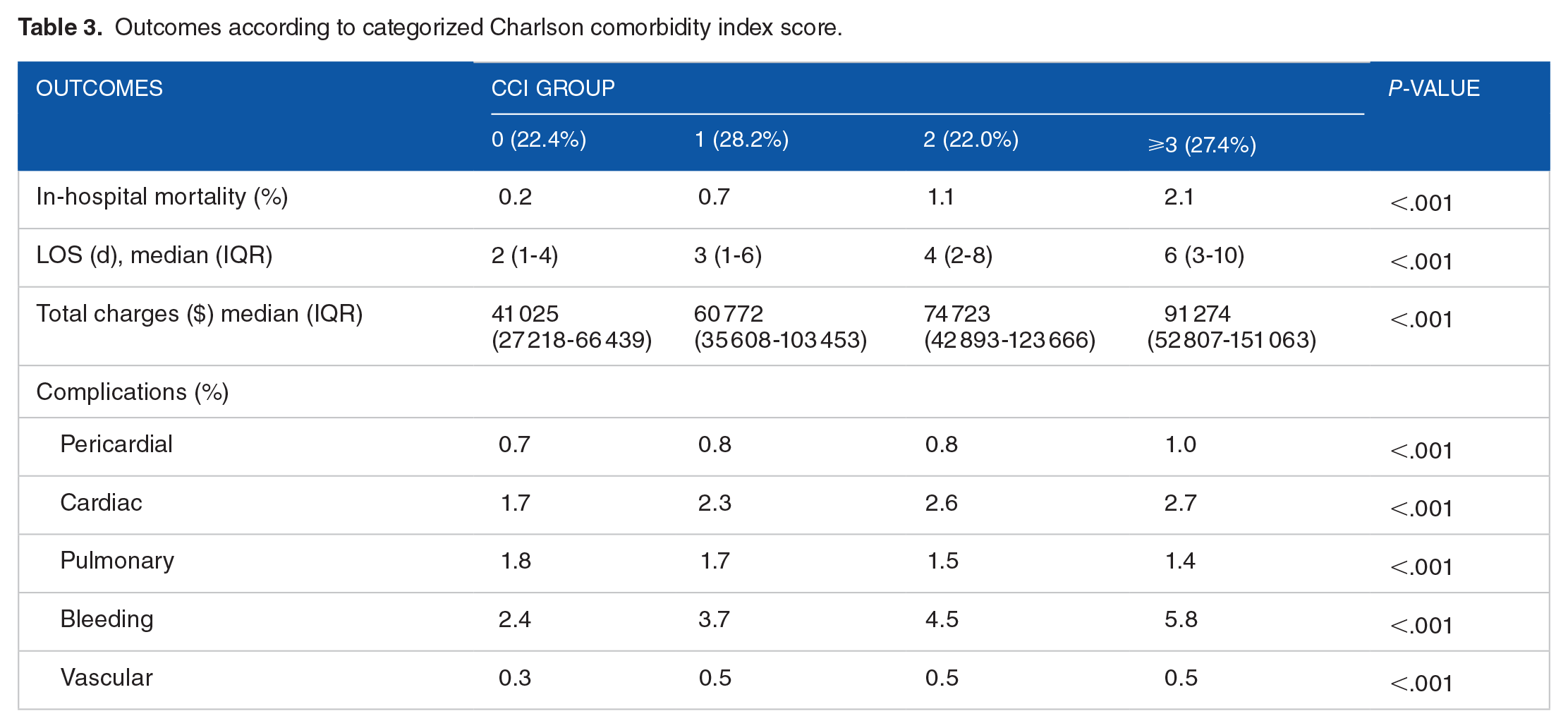
In-hospital mortality increased with increasing comorbidity burden from 0.2% for CCI = 0% to 2.1% for CCI score ⩾3 (Table 3). Similarly, there was an increase in rates of pericardial, cardiac, and bleeding complications for the whole CIED cohort as comorbidity burden increased except for pulmonary and vascular complications (P < .001) (Table 3).
In regression analysis, compared to the reference category of CCI = 0, subjects with CCI = 1 had almost 3 times increase in odds of mortality (OR: 2.79, 95% CI: 2.46-3.16), almost 5 times increase in odds of in-hospital mortality for CCI = 2 (OR: 4.61, 95% CI: 4.07-5.22) and 9-fold increase in odds of mortality for subjects with CCI ⩾3 (OR: 9.05, 95% CI: 8.03-10.18). In contrast, increased comorbidity burden was associated with reduced odds of pulmonary complications (CCI = 1, OR: 0.92, 95% CI: 0.88-0.97; CCI = 2, OR: 0.84, 95% CI: 0.79-0.89; CCI ⩾3, OR: 0.76, 95% CI: 0.72-0.81) (Table 4).
Association between categorized Charlson comorbidity index score and outcomes.

P < .05.
Discussion
Our study of over 3 million CIED discharge records is one of the first to systemically study the impact of comorbidity burden defined by CCI score in a national database in patients undergoing de novo CIED procedures. In addition, we reported trends in de novo CIED implantation over 15 years. The annual number of all index CIED implantations peaked in 2006 and trended down from 2010 to 2014. The percentage of patients with the greatest comorbidity burden (CCI ⩾3) increased significantly over the 15-year study. We also showed that CCI was associated with increased odds of inpatient mortality and peri-procedural complications except for pulmonary complications. Higher CCI score was also associated with increasing LOS (days) and total hospital charges.
We observed a 16% decline in CIED inpatient implantations yearly from 2010 to 2014, mainly driven by a decrease in PPM and ICD implantations. Similarly, in a study by Pasupula et al 17 on the trends in hospital admissions after CIED procedures, there was a decline in the total number of CIEDs implanted per year from 2010 to 2014. A possible reason for decreasing inpatient CIED implantations may reflect a shift toward outpatient implantations. 17 Secondly, there is a possibility that hospital systems began enforcing strict compliance with the Centers for Medicare and Medicaid guidelines considering the Department of Justice’s federal audit of ICD implantations violations and proof of non-evidence-based ICD implants in the literature.18 -20
This study showed that approximately 78% of patients undergoing new CIED implants had at least a CCI score of 1. Similar findings were observed in heart failure patients undergoing CRT implantations between 1999 and 2008 which revealed that approximately 80% of patients had at least 3 comorbidities. 21 According to a Centers for Medicare and Medicaid Services report, more than half of Medicare beneficiaries diagnosed with heart failure have 5 or greater coexisting chronic medical conditions. 22 Thus, this observation in our study reinforces that comorbidities such as heart failure hardly occur alone, and patients undergoing CIED implants often have multiple comorbid conditions.
The proportion of patients with the greatest comorbidity severity (defined by CCI ⩾3) increased profoundly by 143% from 2000 to 2014. In a prior analysis of CRT implantations using NIS data, the percentage of patients undergoing CRT implantations with 2 or more comorbidities according to the Deyo-CCI increased from 55.9% in 2003 to 70.2% from 2003 to 2013. 23
In regression analysis, we observed the most significant impact of comorbidity burden on in-hospital mortality after CIED implantation (OR: 9.05, 95% CI: 8.03-10.18). Each unit increase in CCI score was associated with worsening in-hospital mortality rates. Similar findings have been described in previous studies.24 -26 In a 13-year Danish nationwide study of ICD patients, a 1.6-fold to 5-fold increase in mortality rate from low to high comorbidity burden level was reported. 27 Another finding in this study was that increasing comorbidity burden was associated with increased length of hospital stay and consequently, increasing total hospital charges. Consistent with our findings, Swindle et al studied 26 887 patients after CRT and ICD implantations. The authors showed that increasing CCI score contributed significantly to the increased hospital stay and higher hospital charges, especially among CRT-D implants and older patients. 28
Increasing CCI score was also associated with a higher risk of all the complications studied except for pulmonary complications. Prior studies have also reported that preexisting comorbidities are associated with an increased risk of complications after device implantation.4,29
Higher CCI scores was associated with increased odds of bleeding complications. Predictably, many CIED implants candidates are on anticoagulation therapy and dual antiplatelet therapy for cardiac comorbidities. Some studies have linked the use of anticoagulation and antiplatelet therapy with a higher rate of bleeding complications after CIED implantation.30,31 Thus, the use of anticoagulation and antiplatelet therapy may partly explain the reason for increased bleeding complications observed in our study. Unfortunately, we could not assess the effect of these medications on bleeding complications in our study because NIS database lacks information on medication use. In another study using the NIS database, the authors reported that concomitant comorbidities of coagulopathy, renal failure, and history of congestive heart failure were associated with a higher risk of bleeding complications post-cardiac device implantation. 32 Thus, the high prevalence of congestive heart failure among patients with 1 or more comorbidities in our study may partially explain the observed association of increased odds of bleeding complications.
Limitations
This study has some limitations which need to be considered when interpreting our results. First, the NIS is a de-identified administrative database that uses ICD codes. Although, the use of ICD codes is widely used, it may be subject to coding errors and documentation variations.33,34
The NIS database lacks some details that may have contributed to reporting of inpatient complications. For example, data about the technique of CIED procedures, medication use (ie, anticoagulation use), echocardiographic variables for heart failure, operator volume, number of operators, and duration of procedure are not available in the database. Thus, the impact of several factors on in-hospital outcomes could not be evaluated. Therefore, we recommend caution when interpreting these outcomes concerning cardiac device implantation.
Additionally, only the comorbidities listed in the inpatient discharge records were used to calculate the CCI score without thoroughly examining the entire medical history of the subjects undergoing CIED implantation.
Also, since there has been an increase in outpatient procedures in the past 5 to 10 years and our study is mainly limited to inpatient procedures coupled with the fact that our study is primarily observational, there is a risk of selection bias. 23
Finally, NIS captures only in-hospital complications before discharge, so delayed complications and follow-up data could not be evaluated. Notwithstanding, the literature review showed that a significant number of complications occurs within 24 hours post-implantation, which increases the relevance of our results. 35
Conclusion
Our findings may assist physicians in carefully selecting heart failure patients with competing noncardiac morbidity for CIED to reduce LOS, in-hospital mortality, short-, medium-, and long-term mortality. Based on the increasing prevalence of comorbid conditions seen in patients undergoing CIED implantations, it is imperative to consider comorbidity burden in the decision-making process for these patients. Patients with higher comorbid burden generally have worse clinical outcomes. Therefore, effective strategies need to be put in place to minimize the risk of complications post-CIED implantation. Consequently, it is vital to identify information from these comorbidity burden scores that may be useful in risk stratification of CIED implant candidates.
Supplemental Material
sj-docx-1-cic-10.1177_11795468221108212 – Supplemental material for Impact of Comorbidity Burden on Cardiac Implantable Electronic Devices Outcomes
Supplemental material, sj-docx-1-cic-10.1177_11795468221108212 for Impact of Comorbidity Burden on Cardiac Implantable Electronic Devices Outcomes by Temitope Ajibawo, Oluwatimilehin Okunowo and Adeniyi Okunade in Clinical Medicine Insights: Cardiology
Footnotes
Author Contributions
Initiation of project, writing,editing and submission of manuscript, interpretation of analysis: TA; Writing and editing of manuscript: AO; Data analysis, editing of manuscript: OO.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
