Abstract
Aims:
To investigate the independent effect on depression of painless diabetic polyneuropathy, painful diabetic polyneuropathy, and general and diabetes-related comorbidities.
Methods:
In 181 patients, the presence of painless diabetic polyneuropathy, painful diabetic polyneuropathy, comorbidities and depression was assessed using the Michigan Neuropathy Screening Instrument Questionnaire, the Michigan Diabetic Neuropathy Score, nerve conduction studies, the Douleur Neuropathique en 4 Questions, the Charlson Comorbidity Index and the Beck Depression Inventory-II.
Results:
In all, 46 patients met the criteria of confirmed painless diabetic polyneuropathy and 25 of painful diabetic polyneuropathy. Beck Depression Inventory-II scores indicative of mild–moderate–severe depression were reached in 36 patients (19.7%). In a multiple logistic regression analysis (including age, sex, body mass index, being unemployed, duration, haemoglobin A1c, insulin treatment, systolic blood pressure, nephropathy, retinopathy, Charlson Comorbidity Index and painful diabetic polyneuropathy), female sex (odds ratio: 5.9, p = 0.005) and painful diabetic polyneuropathy (odds ratio: 4.6, p = 0.038) were the only independent predictors of depression. Multiple regression analysis, including Douleur Neuropathique en 4 Questions and Michigan Diabetic Neuropathy Score instead of painful diabetic polyneuropathy, showed that Douleur Neuropathique en 4 Questions, in addition to female sex, was a significant predictor of depressive symptoms severity (p =0.005).
Conclusion:
Painful diabetic polyneuropathy is a greater determinant of depression than other diabetes-related complications and comorbidities. Painful symptoms enhance depression severity more than objective insensitivity.
Introduction
The prevalence of depression has increased in diabetes with nearly twice the overall risk compared to non-diabetic control subjects.1,2 Moreover, in longitudinal studies, diabetes heightens the risk of incident depression by 24%. 3
While rates of depression are elevated in patients with chronic illnesses, in diabetes the excess risk has been mostly attributed to lifestyle and health-related behaviours 4 and to diabetes-related complications, such as macro- and micro-vascular disease, 5 including retinopathy, nephropathy and neuropathy.6,7
A particular link has been observed between depressive symptoms and diabetic polyneuropathy (DPN) that is said to be mediated by neuropathic symptoms, that is, feet insensitivity, pain and unsteadiness. 8 There is also convincing evidence that depression represents a substantial comorbidity of painful diabetic polyneuropathy (PDPN)9–11 with its severity depending on pain intensity. 12 PDPN, however, has been found to be associated with other general and diabetes-related comorbidities,12,13 previously not always taken into account when evaluating the relationship between PDPN and depression.
A depressive comorbidity, in either major or minor forms, can affect self-care, adherence to medical treatment and outcomes in chronic non-diabetic 4 and even more so in diabetic disease, with impact on quality of life, glycaemic control, risk of micro- and macro-vascular complications, morbidity and mortality, and health-care costs.14–18 Data from a range of settings suggest that the prognosis of both diabetes and depression – in terms of disease severity, complications, treatment resistance and mortality – is worse for either disease when they are comorbid than when they occur separately.4,14,16 Although the question of whether identification and treatment of depression might improve diabetes outcomes has not received a full answer yet, nevertheless it is of clinical relevance to clarify the factors involved in the complex relationship between diabetes and depression also in the specific setting of PDPN.
Thus, the objective of this study was to investigate the independent effects on depression of PDPN, non-painful DPN, and general and diabetes-related comorbidities, bearing in mind the role of possible confounders.
We attempted to address the following questions: (1) Is the association of depression with PDPN greater than with non-painful DPN? (2) Does this relationship persist after controlling for comorbidities and other variables associated with PDPN? (3) Is it possible to discriminate between the role of neuropathic pain and other neuropathic symptoms?
In exploring the determinants of depression with regard to the presence of neuropathic pain of PDPN, we used a biopsychosocial approach based on the view that physical illness is the result of the dynamic interaction of biological, physiologic, psychological and social factors, which may influence the clinical presentations and outcomes of disease. 19
Research design and methods
Patients
From May 2011 to November 2012, we prospectively evaluated patients attending the Diabetic clinic of the University Hospital of Tor Vergata (Rome, Italy). Inclusion criteria were the presence of type 1 or type 2 diabetes, a duration of at least 5 years in type 1 patients, and age between 18 and 75 years. Exclusion criteria were non-diabetic peripheral neuropathies or neuropathic pain due to causes different from DPN, non-neuropathic pain, advanced peripheral arterial disease, active foot ulcers, severe comorbidities (i.e. heart failure, recent cardiovascular events), estimated glomerular filtration rate <45 mL/min/1.7 m2, previously diagnosed obstructive sleep apnoea syndrome, serious psychiatric disorders and any other clinically relevant disease preventing neuropathy assessment or an adequate understanding of the questionnaires. Pain medications were not an exclusion criterion unless they determined a reduction in pain intensity under 3 on an 11-point scale. According to the selection criteria, 92 patients were excluded and 181 included.
The Ethics Committee of the University Hospital of Rome Tor Vergata approved the study. All participants provided written informed consent.
Clinical and metabolic parameters assessment
Socio-demographic characteristics (age, sex, marital status, employment status and educational background) were assessed as variables and potential confounders in the association between depression and neuropathic pain. A complete clinical history was recorded regarding diabetes, cardiovascular disease and any potential cause of polyneuropathy. A subject who smoked regularly at least one cigarette per day was considered a current smoker. Alcohol consumption was recorded. Subjects who took part in leisure-related physical activity for at least 1 h per week were considered physically active.
Height, weight, body mass index (BMI) and casual blood pressure (BP) were measured. Routine laboratory assessment was performed, including for haemoglobin A1c (HbA1c), cholesterol, triglycerides, serum creatinine and 24-h urinary albumin excretion. Micro- and macro-albuminuria were considered present with a 24-h albumin excretion of 30–299 or ⩾300 mg, respectively. The presence of non-proliferative or proliferative retinopathy was determined via ophthalmoscopic examination.
Neurological measurements
Neurological assessment was performed in a quiet room and included evaluation of neuropathic symptoms and deficits using the Questionnaire of the Michigan Neuropathy Screening Instrument (MNSI-Q) and the Michigan Diabetic Neuropathy Score (MDNS) 20 MNSI-Q consists of questions about feelings in legs and feet, and history of ulceration or amputation (0–13 score range); MDNS includes assessment at the dorsum of great toes of vibration, pin prick, and pressure sensation, and of muscle strength and reflexes in the upper and lower limbs (overall range: 0–47).
Nerve conduction studies were carried out on tibial motor and sensory nerve (non-dominant side), peroneal motor nerve (non-dominant side) and bilateral sural sensory nerves, using a Medtronic Keypoint standard electromyography (EMG) device (Medtronic, Skovlunde, Denmark) and a standardized technique. 21 The amplitudes of compound muscle action potentials and sensory nerve action potentials, distal motor latencies, motor and sensory conduction velocities and minimal F-wave latencies were measured and z-scores were calculated from values of healthy volunteers obtained through similar techniques. 21 Electrophysiological examination was considered abnormal if one abnormal attribute (among amplitude, conduction velocity, distal latency or F-wave latency) was found in at least two nerves. 22
The definition of confirmed DPN required the presence of electrophysiological abnormality plus neuropathic symptoms and/or signs. 23
Neuropathic pain was assessed on the basis of pain history and examination. Neuropathic pain intensity over the preceding 24 h was measured using a numerical rating scale (NRS). 24 The Douleur Neuropathique en 4 Questions (DN4) screening tool was also used. 25 DN4 consists of 10 items, 7 relating to pain description and to its associated abnormal sensations (DN4-interview), and 3 relating to a brief neurological examination in the painful area. 25 The cut-off value for diagnosis of neuropathic pain is a total score of 4 out of 10, while the DN4-interview is abnormal with a cut-off of 3.25,26
Definition of PDPN included the presence of DPN plus clinician-diagnosed chronic (>6 months) neuropathic pain, with symmetrical and distal distribution in the lower limbs, and of at least moderate severity (NRS ⩾ 3).23,27
Assessment of depression and comorbidities
Depression was evaluated using the Beck Depression Inventory II (BDI-II), a 21-item self-report instrument designed to assess the severity of depressive symptoms. 28 It is a revised version of the original BDI, updated in order to correspond to criteria from the Diagnostic and Statistical Manual of Mental Disorders (DSM). Each item measures the presence and severity of somatic or cognitive depressive symptoms, rated on a 4-point scale ranging from 0 to 3, to get a total score ranging from 0 to 63. The BDI-II has been validated as a sensitive, specific and predictive tool for measuring depression, 28 with categorical depression ratings of minimal (0–13), mild (14–19), moderate (20–28) and severe (39–63). 28 Due to its consistency with Diag-nostic and Statistical Manual of Mental Disorders, 4th Edition (DSM-IV) diagnostic criteria, it is often used for the diagnosis of depression. 29 The use of antidepressant medication was self-reported.
Medical comorbidities were assessed using the Charlson Comorbidity Index that predicts the 10-year mortality for a patient who may have a range of comorbid conditions. 30 This prognostic index assigns non-zero weights (on values of 1, 2, 3 or 6) to 19 conditions based on their risk of mortality to get a total score (ranging from 0 to 37) that predicts mortality. Clinical conditions and associated scores are as follows: 1 each for myocardial infarct, congestive heart failure, peripheral vascular disease, dementia, cerebrovascular disease, chronic lung disease, connective tissue disease, ulcer, chronic liver disease and diabetes; 2 each for hemiplegia, moderate or severe kidney disease, diabetes with end organ damage, tumour, leukaemia and lymphoma; and 3 each for moderate or severe liver disease; 6 each for malignant tumour, metastasis and acquired immunodeficiency syndrome (AIDS). 30 It is considered a valid and reliable method to measure comorbidity in clinical research with predictive value for outcomes, such as mortality, disability, readmissions and length of stay.30,31
Statistical analysis
Data are expressed as mean ± standard deviation. Descriptive statistics were used to characterize clinical variables of the studied patients. Unpaired Student’s t test and analysis of variance (ANOVA) as tests of significance for means and the χ2 test for categorical variables were used. Spearman coefficient correlations for non-normally distributed variables were used to evaluate the reciprocal relationships. Multiple logistic regression analysis was used to assess the clinical, metabolic and neurological variables that were predictors of depression.
A biopsychosocial model was used with the aim to determine the influence on the presence of depressive symptoms and their severity of biological factors (age, gender, BMI, BP and physical activity), disease factors (diabetes type, duration and control; insulin use; diabetic complications; neuropathic symptoms and signs; neuropathic pain and its sensory characteristics, that is, DN4 symptomatic items, and comorbidities as Charlson Comorbidity Index) and social factors (marital status, educational level and employment status).2,32 Subsequently, we assessed the associations and correlations of biological, disease-related and social variables with depression, and finally we included those variables found to be significantly associated with depressive symptoms in the multiple analyses.
We explored the hypothesis that neuropathic pain (both the presence of PDPN as a categorical variable and the DN4 score as a continuous variable) might be a major determinant of depression and considered whether other variables could play a confounding role in this relationship 33 with a particular attention to comorbidities.
Statistical analyses were done using the StatView V program (SAS Institute Inc., Cary, NC, USA) on a Macintosh iBook G4 computer. A value of p < 0.05 was considered significant.
Moreover, using the χ2 test, we calculated for the symptomatic DN4 items the respective sensitivity, specificity, positive predictive value, negative predictive value and the likelihood ratio for a positive result (LR+) – the ratio of sensitivity to (1 − specificity) – for the presence of depression.
Statistics/data analysis (STATA) 12.1 (StataCorp LP, College Station, TX, USA) was used for additional analyses and to calculate both multiple regression analysis and the variation inflation factor.
Results
The 181 selected patients had a mean age of 60.7 ± 11.5 years, BMI of 29.6 ± 5.0 kg/m2, diabetes duration of 14.5 ± 11.8 years and HbA1c of 7.2% ± 1.3%; 106 were males, and 33 had type 1 diabetes.
Clinical and neurological results
Among those studied, 46 (25.4%) satisfied the criteria of confirmed painless DPN (neuropathic symptoms or signs plus electrophysiological abnormalities), 25 (13.8%) for PDPN diagnosis (neuropathic pain due to DPN) and 110 (60.8%) did not have either. Three patients with PDPN used drugs for neuropathic pain (two pregabalin and one oxycodone) but without complete pain relief (NRS ⩾ 3). Table 1 shows the clinical parameters in the three groups of patients. Patients with painful and painless DPN exhibited a higher age, a longer diabetes duration, a higher BMI, a higher percentage of micro-vascular (retinopathy and nephropathy) and macro-vascular disease (peripheral and cardiovascular), a lower percentage of physically active people and of high school graduates, and higher scores of Charlson Comorbidity Index compared to patients without DPN.
Clinical characteristics of patients with painful diabetic polyneuropathy (PDPN), with painless diabetic polyneuropathy (DPN+) and without diabetic polyneuropathy (DPN−).
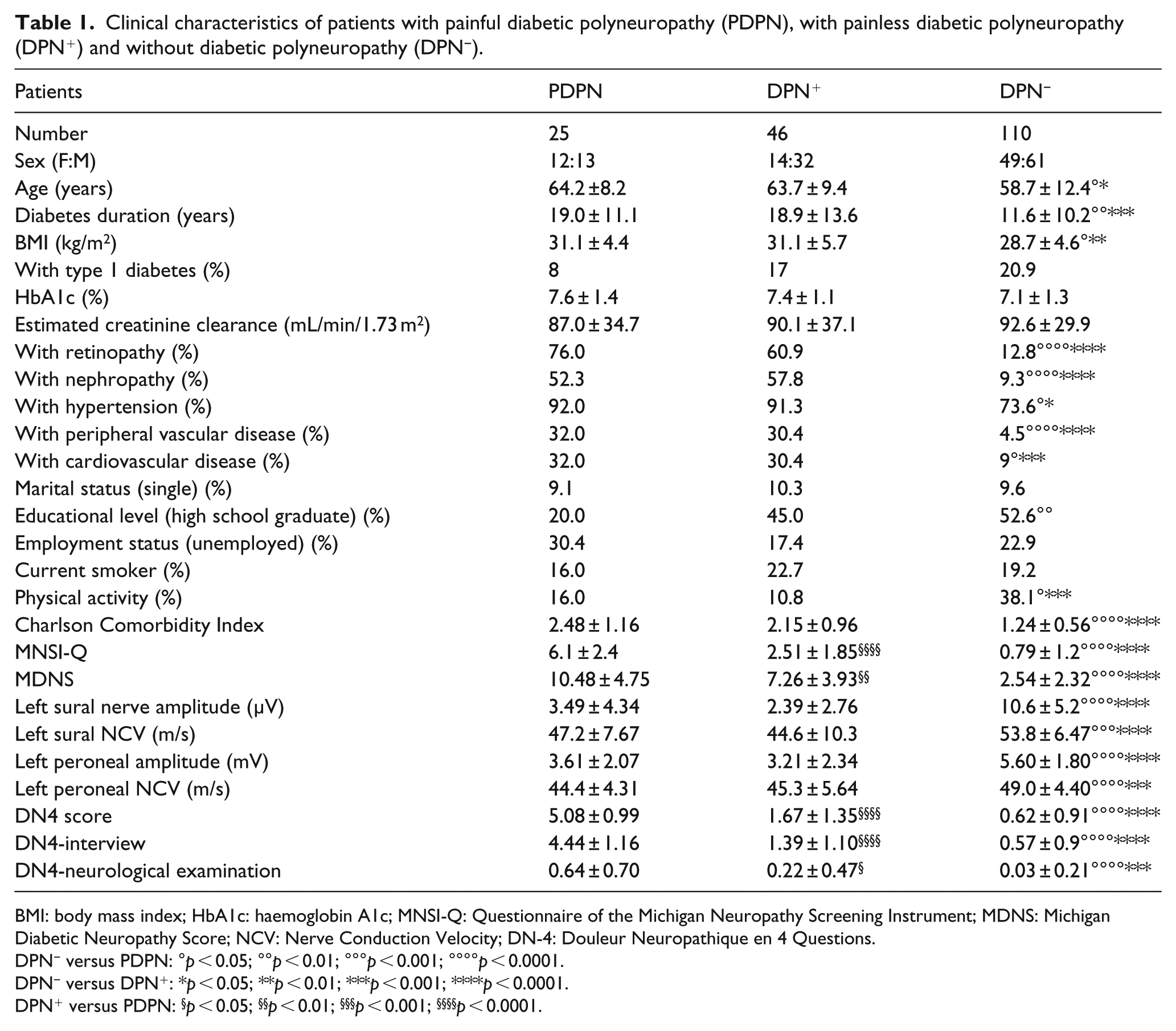
BMI: body mass index; HbA1c: haemoglobin A1c; MNSI-Q: Questionnaire of the Michigan Neuropathy Screening Instrument; MDNS: Michigan Diabetic Neuropathy Score; NCV: Nerve Conduction Velocity; DN-4: Douleur Neuropathique en 4 Questions.
DPN− versus PDPN: °p < 0.05; °°p < 0.01; °°°p < 0.001; °°°°p < 0.0001.
DPN− versus DPN+: *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
DPN+ versus PDPN: §p < 0.05; §§p < 0.01; §§§p < 0.001; §§§§p < 0.0001.
The only difference in neurological measurements between PDPN and DPN patients was represented by the higher scores in symptoms and signs, that is, MNSI-Q and MDNS, in the group with painful neuropathy (Table 1).
Depression and its determinants
BDI-II scores indicative of mild–moderate–severe depression (range: 14–63) were reached in 36 patients (19.7%). This condition was associated with female sex, being unemployed, presence of micro-albuminuria and retinopathy, greater insulin requirement, a higher HbA1c and a higher Charlson Comorbidity Index (Table 2). Seven patients reported to be under treatment with antidepressants: two with venlafaxine, two with paroxetine, one with fluoxetine, one with duloxetine and one with sertraline. Among them, four had minimal and three mild or moderate depressive symptoms on BDI-II, two had painless DPN, two PDPN and the last three did not have DPN.
Clinical characteristics of patients with and without depression.
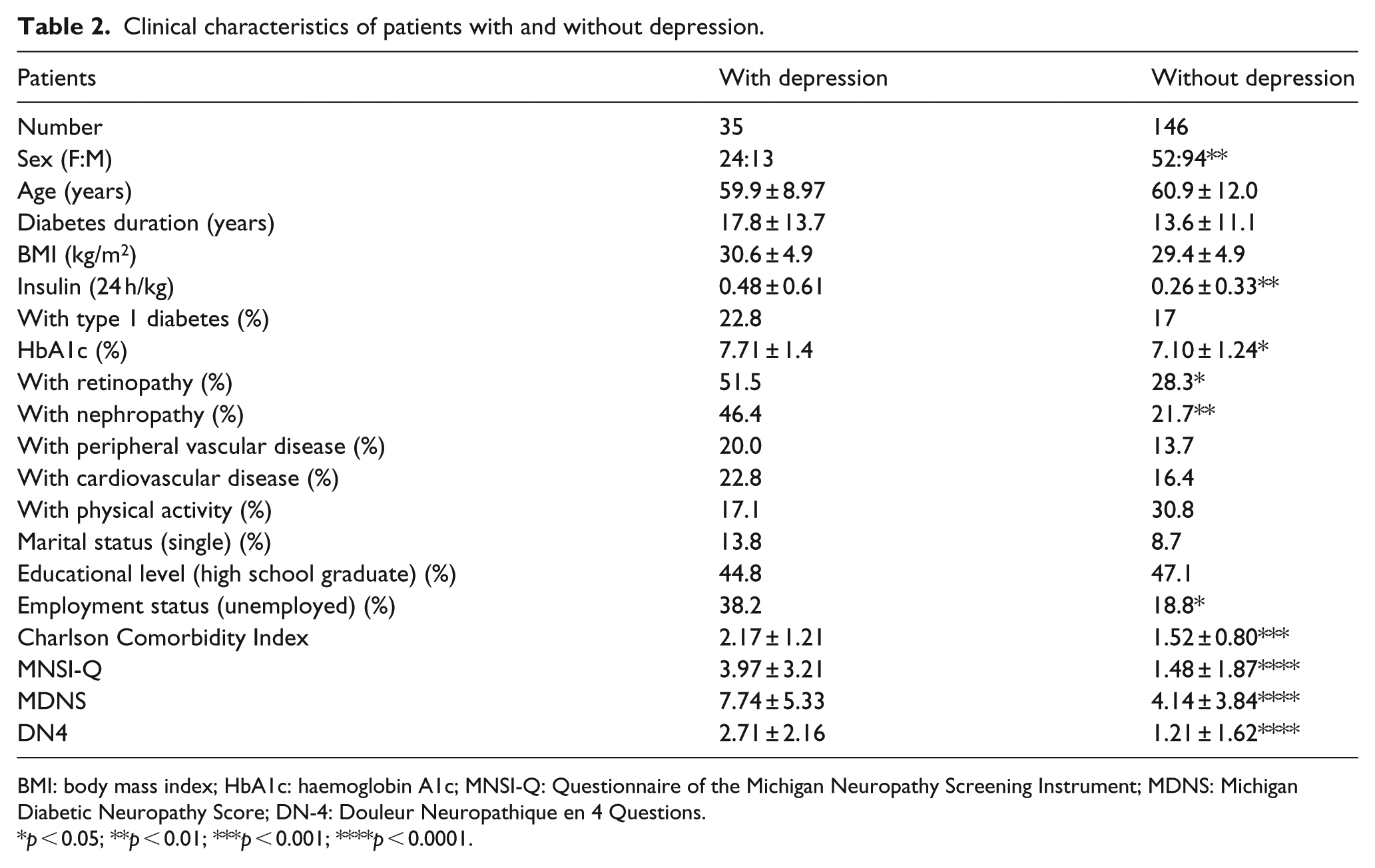
BMI: body mass index; HbA1c: haemoglobin A1c; MNSI-Q: Questionnaire of the Michigan Neuropathy Screening Instrument; MDNS: Michigan Diabetic Neuropathy Score; DN-4: Douleur Neuropathique en 4 Questions.
p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Depression was present in a higher percentage of patients with painless DPN (26%) and PDPN (44%) when compared to the non-neuropathic group (10%) (DPN+ vs DPN−: χ2 = 8.31, p =0.004; PDPN vs DPN−: χ2 = 17.26, p < 0.0001, respectively). There was no significant difference between painful and painless DPN despite the higher percentage in the PDPN group (χ2 = 1.79, p = 0.18) (Figure 1). However, the patients with neuropathic pain showed a higher BDI-II score (14.8 ± 10.3) than both those with non-painful DPN (9.9 ± 7.0, p = 0.018) and without DPN (5.9 ± 5.7, p < 0.0001) (Figure 1). All these differences remained significant after including age, sex, BMI and diabetes duration as covariates in the ANOVA analysis. Patients with depression also had a higher MNSI-Q, MDNS and DN4 score than those without depression (Table 2).
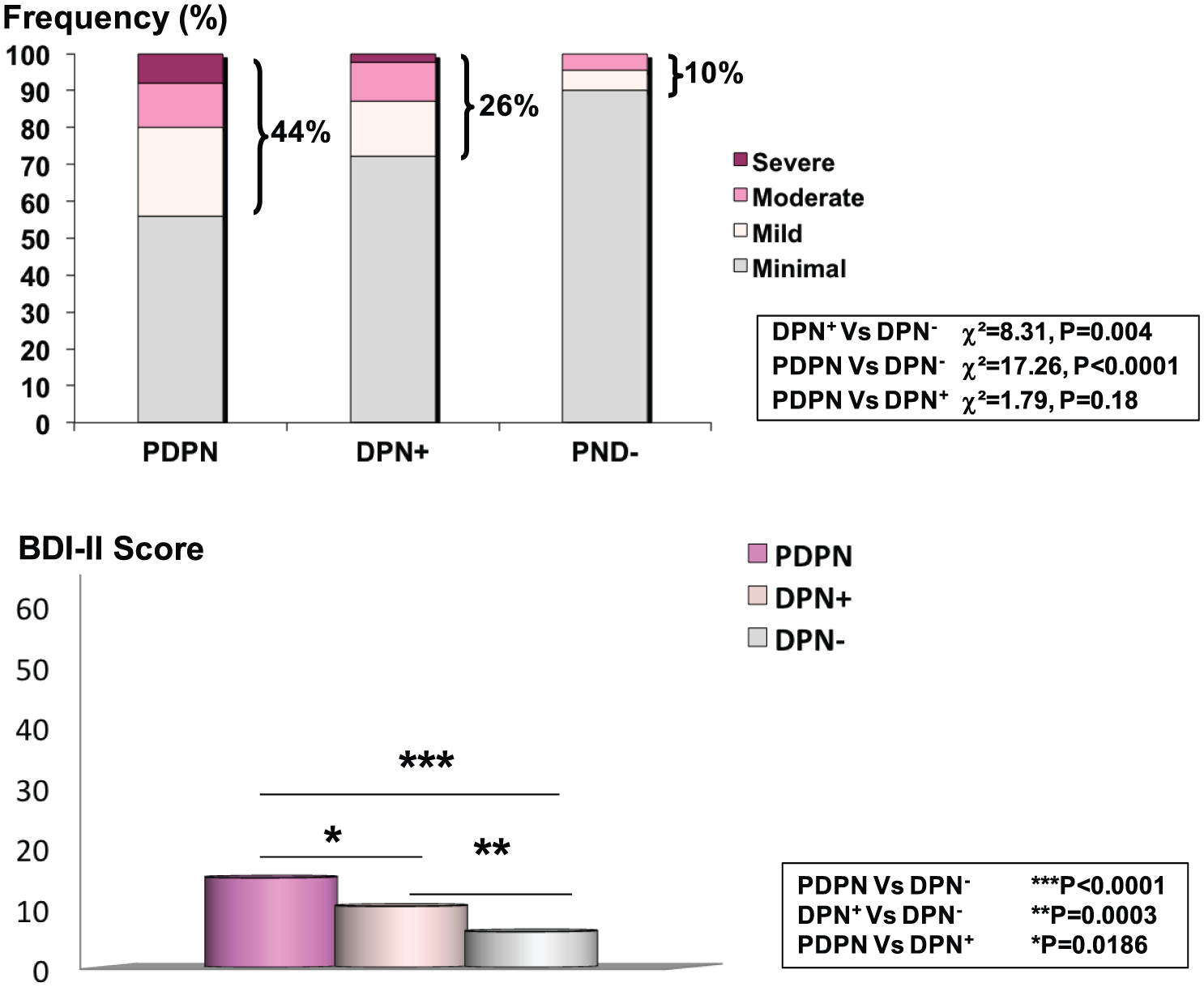
Frequency of depression (according to BDI-II score: minimal if 0–13, mild if 14–19, moderate if 20–28 and severe if 39–63), and mean values of BDI-II score in patients with PDPN, with painless DPN (DPN+) and without DPN (DPN−), after ANOVA adjustment for age, sex, BMI and diabetes duration.
The BDI-II score was related to a number of continuous clinical variables: diabetes duration (rho = 0.17, p = 0.019), HbA1c (rho = 0.28, p = 0.0002), insulin dose (rho = 0.22, p = 0.0031), Charlson Comorbidity Index (rho = 0.35, p < 0.0001) and neurological indexes such as sural nerve amplitude (rho = −0.36, p < 0.0001), MNSI-Q (rho = 0.45, p < 0.0001), MDNS (rho = 0.44, p = 0.0031) and DN4 (rho = 0.46, p < 0.0001) (Figure 2).
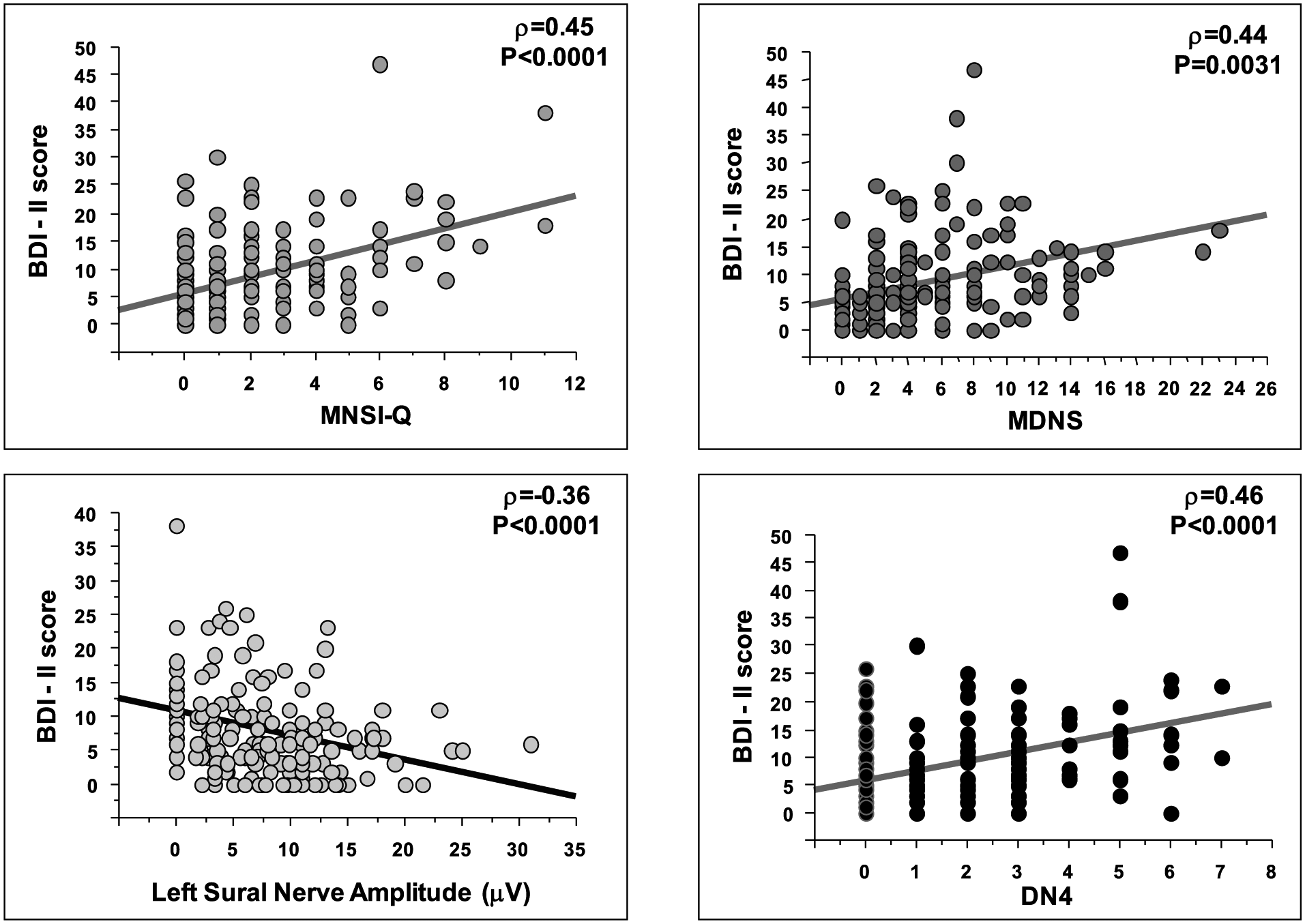
Spearman correlations of depressive symptoms severity (BDI-II score) with neurological variables (MNSI-Q, MDNS, sural nerve amplitude and DN4).
Thus, in a multiple logistic regression analysis, we aimed at identifying which clinical, metabolic and neurological variables predicted the presence of depression, and we included independent variables such as age, female sex, BMI, being unemployed, diabetes duration, HbA1c, insulin treatment, systolic BP, nephropathy, retinopathy, Charlson Comorbidity Index and PDPN. In this model that explained 26% of variance, the only independent predictors of depression were found to be female sex (odds ratio: 5.86, p = 0.005) and PDPN (odds ratio: 4.56, p = 0.038) (Table 3).
Predictors of depression in a multiple logistic regression analysis: odds ratio for having depression.
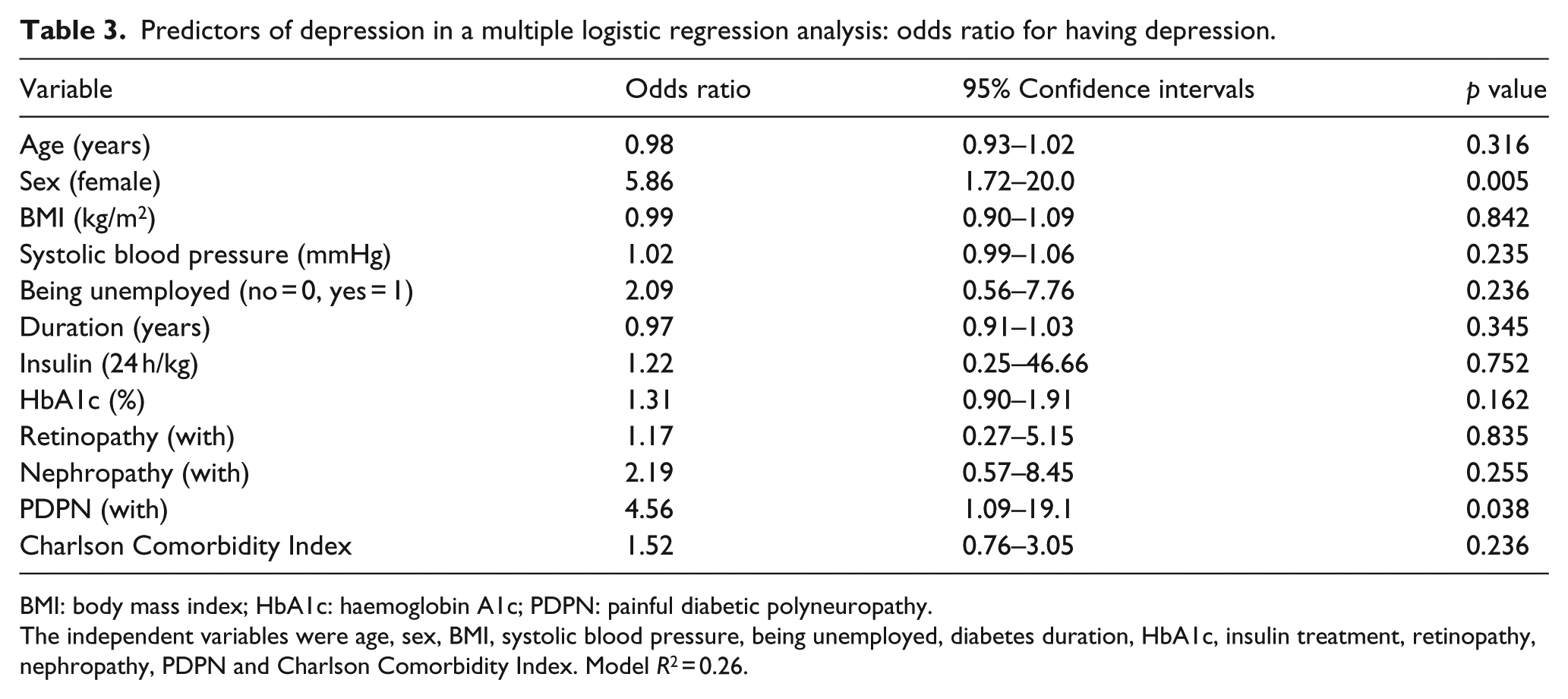
BMI: body mass index; HbA1c: haemoglobin A1c; PDPN: painful diabetic polyneuropathy.
The independent variables were age, sex, BMI, systolic blood pressure, being unemployed, diabetes duration, HbA1c, insulin treatment, retinopathy, nephropathy, PDPN and Charlson Comorbidity Index. Model R2 = 0.26.
Moreover, we used a multiple regression analysis to evaluate the relationship of BDI-II with the variables that were found to be related to BDI-II in simple regression analyses, and included them in the following order: DN4, female sex, being unemployed, diabetes duration, insulin treatment (24 h units/Kg), HbA1c, retinopathy, nephropathy, MDNS and Charlson Comorbidity Index. In this analysis, DN4 (in addition to female sex) was still significantly related to BDI-II (p = 0.005) (Table 4). Furthermore, we calculated the variation inflation factors of the variables included in this model and found that they ranged from 1.33 to 2.28, thus documenting the absence of collinearity between the predictors.
Multiple linear regression analysis to investigate the effects of neuropathic pain (as DN4) on BDI-II score after adjusting for sex, being unemployed, diabetes duration, insulin treatment, HbA1c, nephropathy, retinopathy, MDNS and Charlson Comorbidity Index.
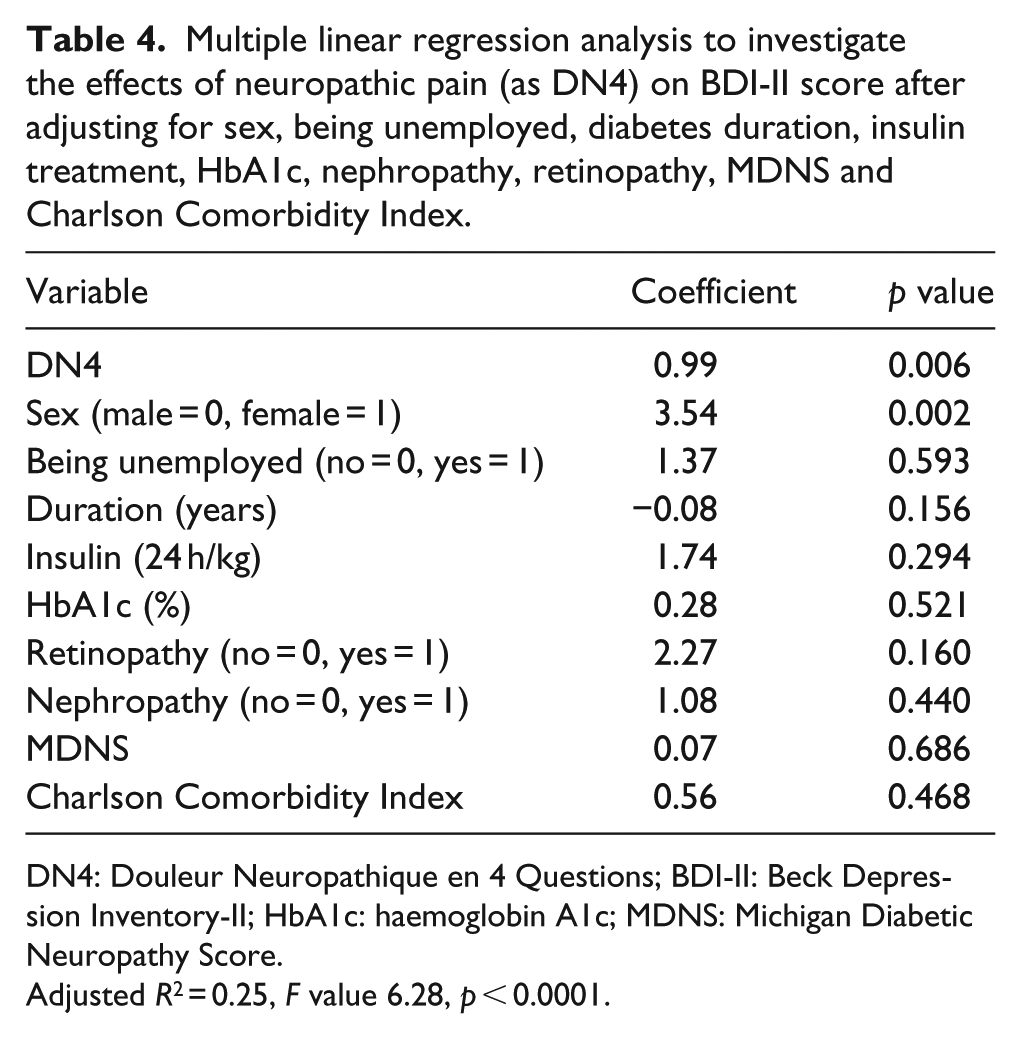
DN4: Douleur Neuropathique en 4 Questions; BDI-II: Beck Depression Inventory-II; HbA1c: haemoglobin A1c; MDNS: Michigan Diabetic Neuropathy Score.
Adjusted R2 = 0.25, F value 6.28, p < 0.0001.
Among the seven symptomatic items of DN4, those most likely to be associated with depression were ‘painful cold’ and ‘electric shock’ with an odds ratio of 9.2 and 6.9 and LR+ of 8.3 and 5.1, respectively, whereas the least likely items were ‘tingling’ and ‘numbness’, with an odds ratio of 2.2 and 2.8, respectively (Table 5).
Association of depression with symptomatic items of DN4.
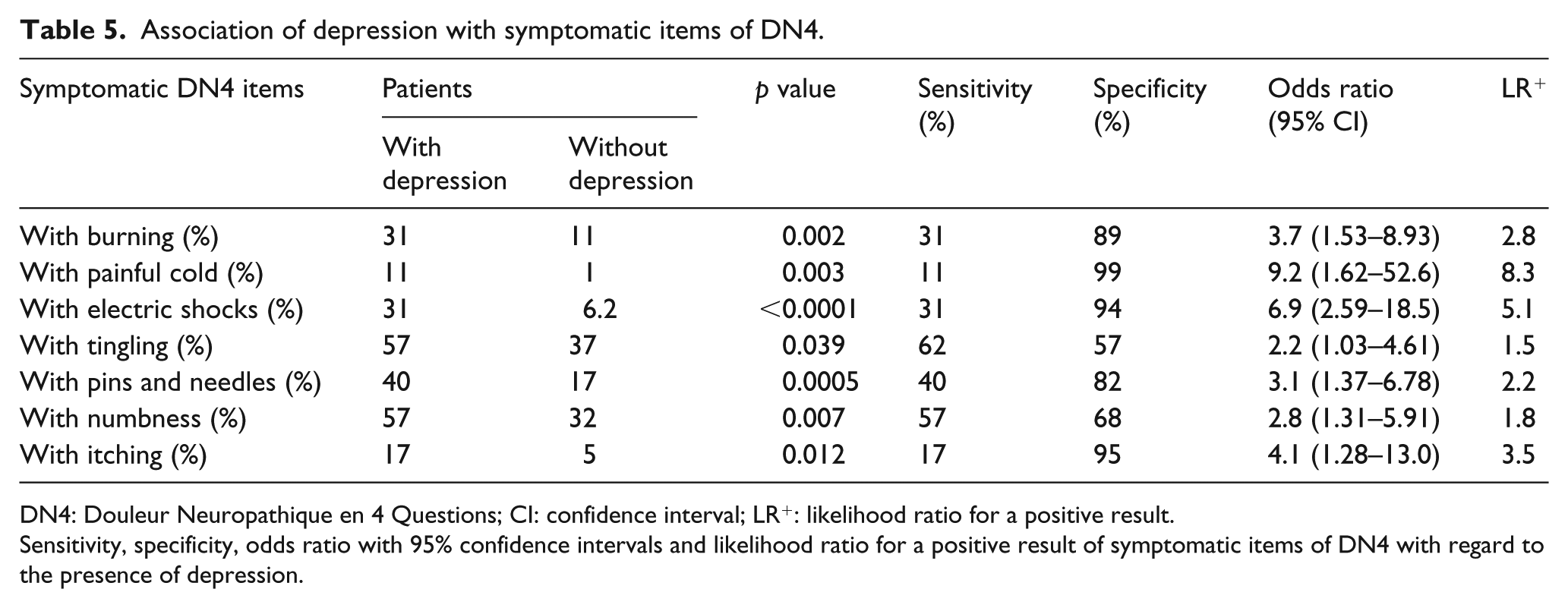
DN4: Douleur Neuropathique en 4 Questions; CI: confidence interval; LR+: likelihood ratio for a positive result.
Sensitivity, specificity, odds ratio with 95% confidence intervals and likelihood ratio for a positive result of symptomatic items of DN4 with regard to the presence of depression.
Discussion
This study was aimed at investigating the independent effect on depression of PDPN, non-painful DPN, and general and diabetes-related comorbidities, bearing in mind the role of possible confounders. The link between depression and diabetes and also between neuropathic pain and depression has been well documented.9–11 However, the complex interrelationship between PDPN and other comorbidities of diabetes and its complications9,13 makes it difficult to discriminate between the respective roles on depression of pain, neuropathy and other comorbidities. Therefore, we attempted to answer some specific questions and in doing so we used validated approaches to the diagnosis of neuropathic pain, DPN, depression and comorbidity burden.
Is the association of depression with PDPN greater than with non-painful DPN?
We found a confirmed diagnosis of painless DPN and PDPN in 25.4% and 13.8% of patients, respectively. These rates of prevalence correspond to what is reported in the literature in diabetic populations of a similar age and setting.13,34
Mild to severe depression according to BDI-II results was present in 19.4% of patients. This prevalence rate was slightly lower than in other studies. 35 A strict selection of patients with the exclusion of severe comorbidities, an accurate diagnosis of DPN and PDPN, and the use of BDI-II for depression screening might partially explain this finding. However, when using the same BDI-II tool, a similar proportion of BDI-II scores >14 (17.5%) was found in 458 type 1 patients. 36
Depression was significantly more common in patients with PDPN (44%) or painless DPN (22%) than in those without neuropathy (10%). These percentages were quite similar to those of 41.1% and 24.5% observed in patients with PDPN and with DPN, respectively, using the Hospital Anxiety and Depression Scale (HADS-D).6,37 Despite the lack of a significant difference between painless DPN and PDPN, the severity of depressive symptoms, seen through the BDI-II score, was higher in patients with PDPN compared to those with painless DPN, also after adjustment for sex, age, BMI and duration as covariates. These findings document a stronger relation between depression and painful DPN compared to painless DPN.
A meta-analysis confirmed the impact of diabetic complications on depression, and found an effect size for diabetic neuropathy (defined by a weighted r value of 0.28) greater than for retinopathy (r = 0.17) and lower than for sexual dysfunction (r = 0.32). 5 In a subsequent Dutch study among 570 type 2 patients, a high depression score, based on the Centre for Epidemiologic Studies Depression scale, was found in 52% of those with neuropathy compared with 31% of those without neuropathy (p < 0.001). 35 Moreover, Yoshida et al. 7 in 129 diabetic patients found a percentage of symptomatic depression (according to Zung’s Self-Rating Depression Scale) of 36.4%, and that diabetic neuropathy was associated with depression independently of many demographic and diabetes-related variables (odds ratio: 3.10). Moreover, using the HADS-D, Gore et al. 12 found moderate to severe symptoms of depression in 28% of 255 patients with PDPN.
Although the association between depression and both painless and painful DPN has been extensively described, to our knowledge, there are no studies so far that have evaluated concomitantly the respective impact of painless DPN and PDPN on depression. Van Acker et al. 38 showed that both PDPN and DPN independently affected quality of life, measured with short form 12 (SF-12) Health Survey, but they did not use a specific screening tool for depression. Takahashi et al. 39 in 1983 found in 17 patients with PDPN significantly higher depression scores (using Zung’s Self-Rated Depression Scale) than in 20 age-matched diabetic subjects with non-painful DPN. However, diagnostic criteria of PDPN were not well defined, with the possible inclusion of acute forms of painful neuropathy, and only patients with pain response to treatment were selected.
In addition, this study reports for the first time a significant correlation between BDI-II score and DPN measures, that is, symptoms, deficits, electrophysiological parameters and DN4. In a previous study, no correlation was found between the first version of BDI and the result of a non-validated self-questionnaire on neuropathic symptoms including pain. 40 On the other hand, the score of HADS-D was found increasingly higher in patients with mild, moderate and severe average pain intensity on Brief Pain Inventory. 12 Moreover, in 494 DPN patients, DPN degree – according to Neuropathy Disability Score and the vibration perception threshold – was significantly associated with the severity of depressive symptoms assessed with HADS, after checking for demographic and disease variables. 8 However, this relation was lost after the inclusion in the stepwise linear regression model of neuropathic symptoms, which were stronger predictors of depressive symptoms than neuropathic signs. 8 Finally, in a recent study in 121 diabetic patients with PDPN, the depressive subscore of HADS-D was related to pain intensity – measured using Neuropathic Pain Scale – in both univariate and multivariate analyses. 41
In conclusion, in our study, depressive symptoms were associated with both painless DPN and PDPN but were more severe in the painful form compared to the painless.
Does this relationship persist after controlling for comorbidities and other variables associated with PDPN?
Both depression in diabetes and PDPN occur together with a number of comorbidities. Depression in type 1 diabetes has been described to be associated with higher age (18), female sex (18), low values of household income, education level and employment status (18), with poor glycaemic control 35 and the presence of diabetic complications.18,35 In type 2 diabetes, other factors were also clinical correlates of depression, such as obesity and ethnicity. 35 On the other hand, 80% of 255 patients with PDPN had at least one chronic medical comorbidity, such as congestive heart failure, chronic obstructive pulmonary disease, coronary heart disease, nephropathy, peripheral vascular disease, retinopathy, in addition to neuropathy, anxiety, depression and sleep disturbance. 9 Other chronic painful and non-painful conditions have also been reported as common comorbidities of PDPN such as migraine, fibromyalgia, restless leg syndrome, chronic low back pain, osteoarthritis, irritable bowel syndrome, cognitive dysfunction and chronic fatigue.9,37
In our study, both painless and painful DPN, when compared to the absence of neuropathy, were associated with higher age, diabetes duration, BMI, higher percentage of retinopathy, nephropathy, peripheral vascular disease and cardiovascular disease, with a lower percentage of physically active people, and only for PDPN with a low educational level. On the other hand, depression was linked to female sex, insulin use, higher HbA1c, retinopathy and nephropathy, confirming previous observations.5,18,35 Thus, it is justified to suggest that the relation between depression and PDPN could be affected by the reciprocal association of these conditions with comorbidities. Therefore, to get a combined measure of comorbidities, we adopted the Charlson Comorbidity Index that is a widely used measure, 30 also validated and applied in diabetic patients. 42 In our study, Charlson Comorbidity Index was associated with painless DPN, PDPN and depression, and correlated with BDI-II score. However, in a multivariate analysis, the significant relationship between PDPN or BDI-II with Charlson Comorbidity Index lost its significance, and we managed to document that the relationship between neuropathic pain and depressive symptoms was independent of main comorbidities affecting survival. Vileikyte et al. 6 documented that the predictive role of DPN (i.e. signs and symptoms including pain) on the development of depression was independent of the number of diabetes complications and comorbidities. However, in that study DPN and not PDPN alone was considered, as neuropathic pain was just one neuropathic symptom, and self-reporting of comorbidities (i.e. no better detailed than ‘all medical conditions different from diabetes and its complications’) and diabetic complications were accepted.
After adjustment for multiple covariates, including diabetes-related and unrelated conditions associated with PDPN and depression, we found that PDPN together with female sex was an independent predictor of the presence of depression and DN4 was a main determinant of the severity of depressive symptoms. This is the first documentation of the role of PDPN on depression after taking into account their comorbidities in a structured way.
Is it possible to discriminate between the role of neuropathic pain and other neuropathic symptoms?
The DN4 is one of the most common screening tools for neuropathic pain, also specifically validated for PDPN, 26 which also contains questions about non-painful neuropathy symptoms (tingling, pin and needles, numbness, and itching). When analysing which of the seven symptoms of DN4 was most likely to be associated with depression, the results were ‘painful cold’ and ‘electric shocks’, whereas those with the lowest odds for depression were ‘tingling’ and ‘numbness’. This finding points to a prominent role of typical symptoms of neuropathic pain compared to other sensory or function loss symptoms. Vileykite et al. 6 showed a prevalent impact on depression of unsteadiness assessed using NeuroQol. DN4 does not investigate this particular symptom and thus we cannot add information on this finding in our population.
Strengths and limitations
The strength of this study was the choice to use specific instruments to evaluate the presence and severity of neuropathic pain, as well as validated modalities for neuropathic symptoms and signs, in addition to nerve conduction studies. The fact that we obtained a confirmed diagnosis of DPN based on clinical and electrophysiological assessment, 23 and an accurate diagnosis of PDPN, according to current guidelines, 27 is in our opinion worthy of consideration.
Another strength of our study was the adoption of BDI-II that is the most widely used, and is also considered valid and reliable screening instrument to evaluate depressive symptomatology 2 although we recognize that self-report questionnaires may overestimate prevalence of depression compared with diagnostic interview. 1
Moreover, we carefully evaluated the presence of diabetic complications, considered to be associated with depression, together with the other clinical characteristics of patients, including marital and employment status. This is the first time a structured assessment of comorbidities has been done with the use of the validated Charlson Comorbidity Index.
A limitation of the study was the lack of sleep assessment; therefore, we are unable to detect whether sleep-related complaints mediate the relation between pain and BDI-II. Moreover, the presence of other forms of mood disorders such as anxiety was not covered by our assessment.
Conclusion
In conclusion, we were able to demonstrate that depressive symptoms are associated with both painless and painful DPN, but to a greater degree of significance with PDPN, and that the severity of symptoms is higher in patients with PDPN than in those with non-painful DPN. The relationship between depression and PDPN persists also after controlling for comorbidities – assessed using a structured tool like Charlson Comorbidity Index – and other covariates of PDPN. Thus, diabetic peripheral neuropathic pain is a greater determinant of depression than other diabetes-related complications and comorbidities. Some painful symptoms (i.e. painful cold and electric shock) are stronger predictors of depression than tingling and numbness.
Thus, in order to be effective, the management of PDPN should be prompt and take into account the risk of depression and its possible multiple impacts on pain relief, patient’s self-management of diabetes, quality of life and prognosis.
Footnotes
Acknowledgements
Part of this study was presented at the 23rd Annual Meeting of the Diabetic Neuropathy Study Group of the EASD (NEURODIAB), 19–22 September 2013, Castelldefels, Spain.
Author’s contributions
C.D.A. designed the study; researched, extracted and analysed data, and wrote the manuscript. R.M., C.G., F.D.G., L.C., S.L., G.M. and G.A.M. contributed to the design, researched data, contributed to the discussion and revised the manuscript. D.L. contributed to the design, acquisition of data and to the discussion, and reviewed the manuscript. V.S. designed and conducted the study, researched and analysed data, and wrote the manuscript. All the authors approved the version to be published. V.S. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
