Abstract
Background:
Severe sepsis is commonly associated with mortality among critically ill patients and is known to cause coagulopathy. While antithrombin is an anticoagulant used in this setting, serum albumin levels are known to influence serum antithrombin levels. Therefore, this study aimed to evaluate the outcomes of antithrombin supplementation in patients with sepsis-associated coagulopathy, as well as the relationship between serum albumin levels and the effects of antithrombin supplementation.
Methods:
This retrospective study evaluated patients who were >18 years of age and had been admitted to either of two intensive care units for sepsis-associated coagulopathy. The groups that did and did not receive antithrombin supplementation were compared for outcomes up to 1 year after admission. Subgroup analyses were performed for patients with serum albumin levels of <2.5 g/dL or ⩾2.5 g/dL.
Results:
Fifty-one patients received antithrombin supplementation and 163 patients did not. The Cox proportional hazards model revealed that antithrombin supplementation was independently associated with 28-day survival (hazard ratio [HR]: 0.374, P = 0.025) but not with 1 year survival (HR: 0.915, P = 0.752). In addition, among patients with serum albumin levels of <2.5 g/dL, antithrombin supplementation was associated with a significantly lower 28-day mortality rate (9.4% vs 36.8%, P = .009).
Conclusion:
Antithrombin supplementation may improve short-term survival, but not long-term survival, among patients with sepsis-associated coagulopathy.
Introduction
Severe sepsis is a common cause of death among critically ill patients, 1 and is frequently associated with coagulopathy, which may manifest as thrombocytopenia. 2 Antithrombin is an anticoagulant used in this setting, although a phase III trial involving high-dose antithrombin for adults with sepsis and septic shock did not reveal a significant improvement in the 90-day mortality rate. 3 In contrast, recent large propensity score analyses have indicated that antithrombin supplementation may indeed decrease the mortality rate among patients with sepsis-associated coagulopathy.4–6 Nevertheless, most studies have not evaluated long-term outcomes, for example, a meta-analysis of 30 studies conducted by Allingstrup et al 7 only evaluated survival outcomes up to 90 days. Therefore, it remains unclear whether antithrombin treatment affects the long-term mortality rate among patients with sepsis-associated coagulopathy.
Other studies have revealed that serum albumin levels are associated with serum antithrombin levels.8,9 Moreover, Aibiki et al 10 reported that serum albumin levels were associated with post-administration serum antithrombin levels in critically ill patients, with significantly lower post-administration antithrombin levels in patients with serum albumin levels of <2.5 g/dL (vs ⩾2.5 g/dL). However, to the best of our knowledge, no studies have evaluated the association between serum albumin levels and antithrombin supplementation effects.
Disseminated intravascular coagulation (DIC) is a cause of coagulopathy in sepsis. Criteria for DIC have been suggested by the International Society on Thrombosis and Haemostasis (ISTH). 11 However, it may sometimes be difficult to distinguish DIC from other coagulopathies in patients with sepsis. Moreover, fibrin-related markers are needed to meet the DIC criteria suggested by the ISTH, and such markers may not be measured in all settings.
Therefore, we evaluated survival outcomes among patients with sepsis-associated coagulopathy who did or did not receive antithrombin supplementation and investigated the relationship between serum albumin levels and antithrombin supplementation effects without the use of DIC criteria.
Methods
Study population and setting
This retrospective study evaluated patients who were admitted to medical or surgical intensive care units (ICUs) at either of two tertiary referral hospitals in Japan during the period between January 2014 and December 2016. Institution A was a university hospital that used antithrombin aggressively, while Institution B was a public hospital that did not use antithrombin. The ethics review boards at each hospital approved the retrospective protocol (29-101 and zn180401) of this study. Informed consent for inclusion was obtained by the opt-out method.
Patients were considered to be eligible if they were >18 years of age, had been admitted to the ICU for infection, and had their platelet count decrease to ⩽100,000/µL during the first 7 days after admission. The researchers retrospectively examined the patient’s Sequential Organ Failure Assessment (SOFA) score; those whose SOFA scores increased by more than 2 points (Sepsis-3 criteria) were included. 12 The exclusion criteria were hematological malignancy, anticoagulant treatment before the admission, death within 24 hours after admission, extracorporeal membrane oxygenation, and Child-Pugh class C liver cirrhosis.
Treatment
The present study lacked a pre-defined indication for antithrombin supplementation; thus, it was selected at the discretion of the attending physician. The antithrombin group received antithrombin concentrate (Nihon Pharmaceutical Co. Ltd, Tokyo, Japan) at a typical dose of 1500 IU/day for up to 3 consecutive days, unless the patient died or treatment was discontinued by the attending physician.
Data collection and outcomes
The patients’ medical records were retrospectively searched to collect data regarding their demographic and clinical characteristics. The severity of illness was evaluated using the Acute Physiology and Chronic Health Evaluation (APACHE) II score and SOFA score, which were determined using the most aberrant clinical and laboratory results obtained during the first 24 h after ICU admission. Data were also collected regarding laboratory parameters (platelet count, prothrombin time-international normalized ratio [PT-INR], and albumin) from the ICU admission, as well as survival outcomes at 28 days and 1 year. The post-discharge survival outcomes were determined by contacting the patient or their family via telephone. In-hospital bleeding events were also recorded, with major bleeding defined as bleeding that was either fatal, associated with critical organ failure, or required the infusion of ⩾6 units of packed red blood cells within 24 hours. In our country, one unit of packed red blood cells has a volume that is approximately one-half of that used in other countries.
Statistical analysis
The groups that did and did not receive antithrombin were compared using the Mann–Whitney U test for continuous variables and Fisher’s exact test or the chi-square test for categorical data. Kaplan–Meier curves were compared using the log-rank test, and subgroup analyses were performed for patients with serum albumin levels of <2.5 g/dL and ⩾2.5 g/dL. A Cox proportional hazards model was used to examine the association between antithrombin supplementation and survival rate. All analyses were performed using IBM SPSS software (version 22.0; IBM Corp., Armonk, NY, USA).
Results
The study flowchart is shown in Figure 1. During the study period, 307 consecutive patients were potentially eligible, although 214 patients were ultimately included after applying the exclusion criteria. Fifty-one patients received antithrombin supplementation and 163 patients did not. The patients’ baseline characteristics are shown in Table 1. Relative to the group that did not receive antithrombin supplementation, the antithrombin group had significantly lower SOFA scores, serum albumin levels, and PT-INR values. There were no significant differences in age, sex, APACHE II score, platelet count, underlying diseases, or infection site (except for urinary tract infection).
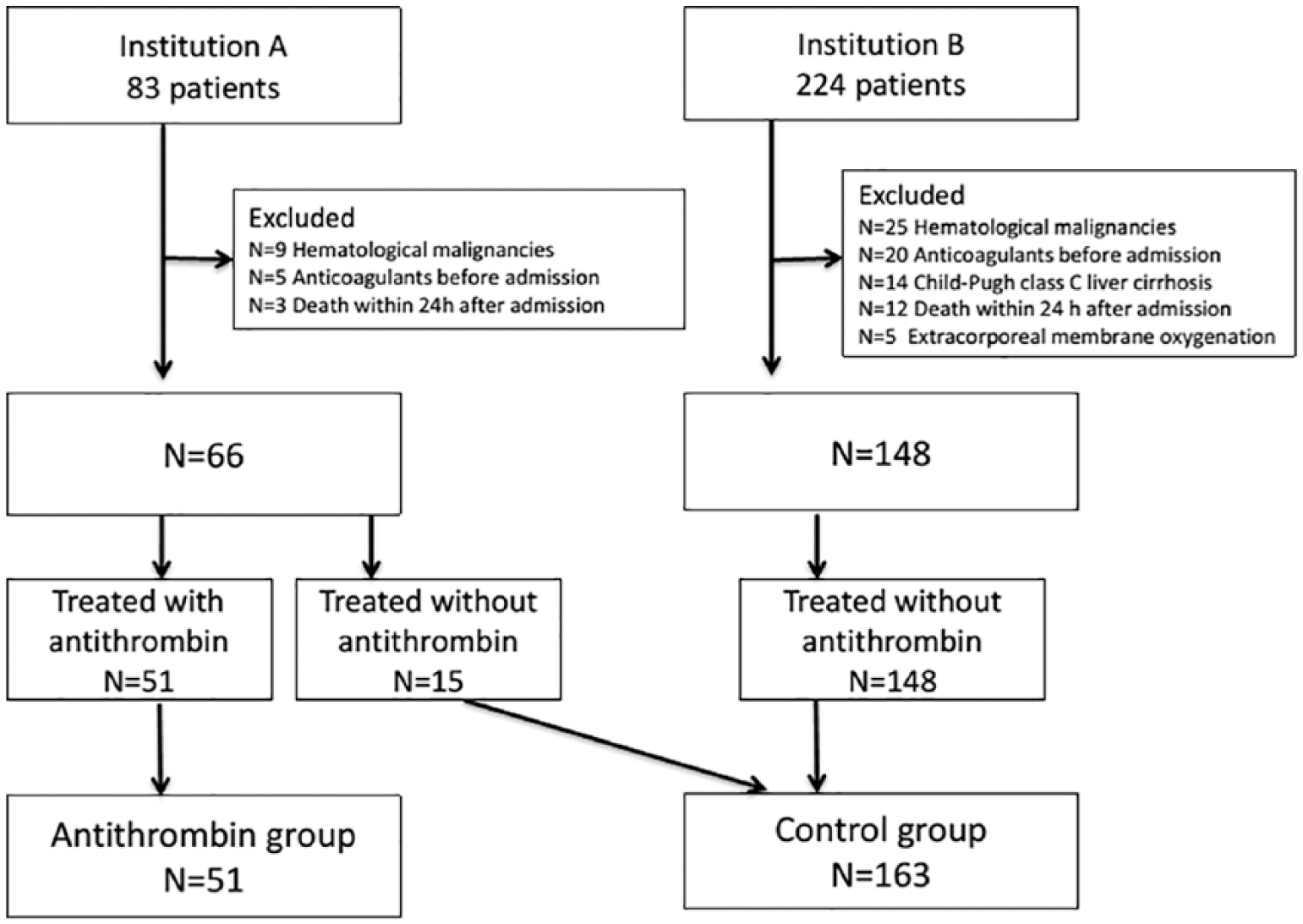
Study flowchart.
The patients’ baseline characteristics.

APACHE II: Acute Physiology and Chronic Health; Evaluation II; PT-INR: prothrombin time-international normalized ratio; SOFA: Sequential Organ Failure Assessment.
Data are shown as median (interquartile range) or number (percentage).
Table 2 shows the results of univariable analyses, which revealed that the antithrombin group had a significantly lower 28-day mortality rate, relative to the group that did not receive antithrombin. No significant differences were observed in the rates of 1 year mortality and major bleeding. Figure 2 shows the cumulative survival rates of the two groups during the follow-up period; the antithrombin group had non-significantly better cumulative survival during the early phase and poorer cumulative survival during the late phase, relative to the group that did not receive antithrombin (P = .964).
Univariable analysis of the outcomes.

Data are shown as number/total (percentage).

The log-rank P-value was .99.
The rates of follow-up to 1 year were 94% (48/51) in the antithrombin group and 87% (142/163) in the control group. The Cox proportional hazards model revealed that antithrombin supplementation was independently associated with 28 day survival (hazard ratio [HR]: 0.374, 95% confidence interval [CI]: 0.159–0.883, P = .025) but not with 1 year survival (HR: 0.915, 95% CI: 0.560–1.497, P = .752) (Table 3). Age, serum albumin level, and SOFA score were associated with the 1-year survival rate. In the subgroup analyses, patients with serum albumin levels of < 2.5 g/dL who received antithrombin had a significantly lower 28-day mortality rate than their counterparts who did not receive antithrombin (9.4% vs 36.8%, P = .009) (Table 2). There was also a non-significant trend toward an improvement in the survival curve for that subgroup (P = .303) (Figure 3A). Among patients with serum albumin levels of ⩾2.5 g/dL, antithrombin supplementation was not associated with improvements in the 28-day or 1-year mortality rates (Table 2); moreover, there was a non-significant trend toward a worsened survival curve among patients who received antithrombin supplementation (P = .416) (Figure 3B).
Multivariate Cox proportional hazards model for 28-day and 1-year mortality outcomes.

CI: confidence interval; HR: hazard ratio; PT-INR: prothrombin time-international normalized ratio; SOFA: Sequential Organ Failure Assessment.
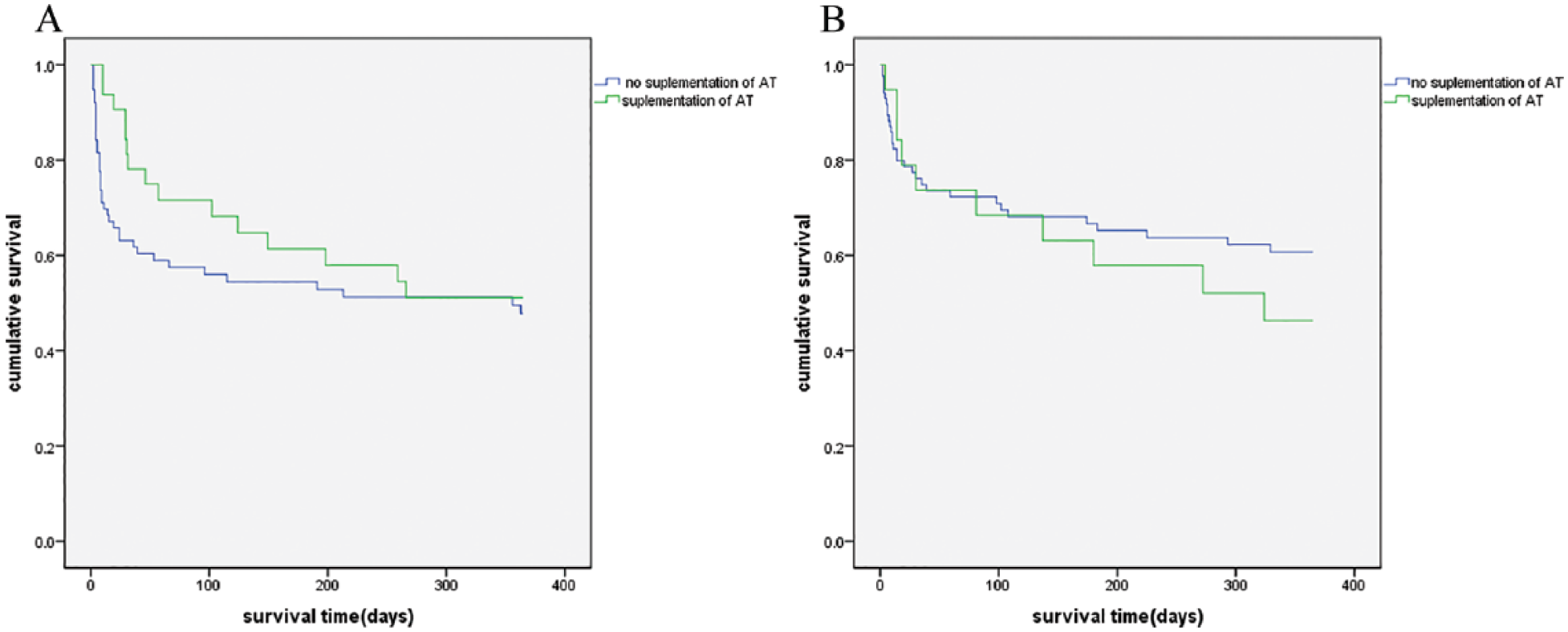
Kaplan–Meier survival curves are shown for patients in the antithrombin and control groups with serum albumin levels of (A) < 2.5 g/dL. The log-rank P-value was .345 and (B) ⩾ 2.5 g/dL. The log-rank P-value was .416.
Discussion
The findings of this study revealed that antithrombin supplementation was associated with a higher 28-day survival rate, although it did not affect the 1 year survival rate, and that the survival curves for the antithrombin and control groups crossed at approximately the 6 month mark. Interestingly, among patients with serum albumin levels of <2.5 g/dL, antithrombin supplementation improved the short-term survival rate, although it tended to be associated with inferior long-term survival among patients with serum albumin levels of ⩾2.5 g/dL. Thus, antithrombin supplementation may improve short-term survival but not long-term survival. Similarly, Hayakawa et al 6 reported that antithrombin treatment was associated with improved survival during the early period after ICU admission, which may be related to improved coagulation status, 13 although such improvement is not a definitive treatment for sepsis. Therefore, it appears that antithrombin does not improve the long-term survival rate among patients with sepsis.
Our findings also suggest that serum albumin levels at admission were influenced by antithrombin supplementation. Previous studies have indicated that serum albumin levels decrease because of reduced synthesis and/or enhanced consumption during sepsis14,15; notably, excessive albumin leakage from the capillaries significantly affects serum albumin levels in patients with sepsis. 16 Furthermore, other studies have suggested that antithrombin activity is related to vascular endothelial cells17,18; Iba and Saitoh 19 reported that it protected endothelial cells by binding to glycosaminoglycans and suppressing capillary leakage. Niederwanger et al showed increased survival with higher serum antithrombin levels in pediatric septic patients. They demonstrated that antithrombin optimized coagulation and served as a potential regulator of inflammatory processes and subsequent tissue damage. 20 Therefore, antithrombin supplementation may be more effective in patients with hypoalbuminemia, rather than in those with normal albumin levels. Taken together, our findings suggest that antithrombin has a diminished effect in patients with normal albumin levels, which indicates that antithrombin supplementation should be reserved for patients with reduced serum albumin levels.
Strengths and limitations
To the best of our knowledge, our study is the first to evaluate long-term outcomes (up to 1 year) among patients who received antithrombin treatment for sepsis-associated coagulopathy. In addition, it is the first to examine the effects of antithrombin supplementation with respect to serum albumin levels. However, this study had several limitations. First, we used a retrospective observational design and cannot confirm a causal relationship among the analyzed factors. Second, the antithrombin group was exclusively treated at Institution A and the control group was mainly treated at Institution B, which might have affected our findings. Third, a small proportion of patients could not be reached at the 1-year follow-up. Fourth, we did not use the ISTH criteria for DIC and did not monitor serum antithrombin levels, contrary to the approach used in previous studies.21,22 Thus, our findings might not be comparable to those of the previous studies. Nevertheless, our findings could facilitate creation of a simple parameter to guide antithrombin supplementation among patients with sepsis-associated coagulopathy (e.g. based on serum albumin levels), which could be particularly useful for institutions that do not routinely monitor serum antithrombin levels.
Conclusion
Among patients with sepsis-associated coagulopathy and hypoalbuminemia, antithrombin supplementation may improve short-term survival, but not long-term survival. However, our study had several limitations, and further studies are needed to conclusively evaluate this issue.
Footnotes
Acknowledgements
We gratefully acknowledge the devoted cooperation of the staff in the Shiga University of Medical Science and Kobe City Medical Center General Hospital.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Yutaka Eguchi has received transportation fees from Nihon Pharmaceutical Corporation. The remaining authors declare that there is no conflict of interest.
Authors’ Contributions
ME collected the data from institution A, analyzed the data, and drafted the manuscript. KF and YE supervised the study and revised the manuscript. AI and KA collected the data from institution B and revised the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
