Abstract
Xanthoma is a rare condition mostly caused by hyperlipidemia. The pathogenesis of gout is hyperuricemia, which is caused by a disorder of purine metabolism and/or a decrease in uric acid excretion. Xanthoma combined with gout is very rare. This case report presents magnetic resonance images of a case of xanthoma combined with gout infiltration of the Achilles tendon.
Introduction
Xanthoma is a rare disease mostly secondary to hyperlipidemia, particularly with high levels of triglyceride (TG) and total cholesterol (TC). There is a variety of clinical manifestations, one of which is the formation of papules or nodules on tendons. The lesions are multiple, symmetrical, and vary in size. The pathogenesis of gout is hyperuricemia caused by a disorder of purine metabolism and/or a decrease in uric acid excretion, causing deposition of urate crystals in various joints and the surrounding soft tissue. Its main clinical features are joint pain and/or the appearance of gouty nodules, which most often occur at the extremities of joints. Xanthoma combined with gout is very rare. It can be diagnosed using magnetic resonance imaging (MRI) in patients with hyperlipidemia and hyperuricemia. Here, we report on a case that manifested as both xanthoma and gout infiltration of the Achilles tendon and provide the features observed using MRI. We believe our report may help clinicians improve their diagnosis and treatment of xanthoma combined with gout infiltration of the Achilles tendon.
Case Report
A 33-year-old male patient presented with bilateral heel pain and inflammation in both Achilles tendons, which had started 26 months previously, due to unknown reasons. The pain was often greater after overactivity and was relieved after rest with no obvious nocturnal pain. Over the previous 2 months, the pain in his heels had become more intense and was not entirely relieved after rest, with pronounced enlargement of his heels. There was no definite family history of the symptoms or history of trauma. The patient had a history of gout for approximately 5 years, but had never been treated with drugs. His past control of uric acid levels was unknown. Physical examination demonstrated thickening and swelling of both Achilles tendons with soft tissue masses. Moreover, there were no adhesions of the enlarged Achilles tendons with the surrounding skin which had mild tenderness, medium hardness, and a clear boundary. The temperature of the surrounding skin was not high, and movement in both ankles was not significantly restricted.
Biochemical analysis of his blood revealed serum TG levels of 3.02 mmol/L, TC of 8.44 mmol/L, LDL (low-density lipoprotein) of 5.4 mmol/L, HDL (high-density lipoprotein) of 1.19 mmol/L, and uric acid of 470 µmol/L. The normal reference ranges are 0.10 to 1.80 mmol/L, 0.67 to 2.80 mmol/L, 2.10 to 3.30 mmol/L, 0.8 to 1.92 mmol/L, and 155 to 428 µmol/L for TG, TC, LDL, HDL, and uric acid levels, respectively.
Bilateral ankles were scanned using MRI, confirming that the right Achilles tendon and right peroneal longus tendon were thickened. The anteroposterior diameters of the middle Achilles tendon and the middle part of the peroneal longus tendon were approximately 1.6 and 1.3 cm, respectively. The left Achilles tendon was clearly thickened with an anteroposterior diameter of approximately 2 cm at the middle.
Sagittal slices demonstrated protrusion of the anterior edge of both Achilles tendons and the right peroneal longus tendon. T1-weighted images showed high signals with speckles, nodular, and grid shapes distributed in the Achilles tendon. Proton density-weighted (PDW) images showed that the corresponding regions still produced abnormal signals in a strip or grid (Figures 1–3).

(A, B) Images of the sagittal plane (T1-weighted image) showed thickened Achilles tendons with a high signal strip (white arrow).
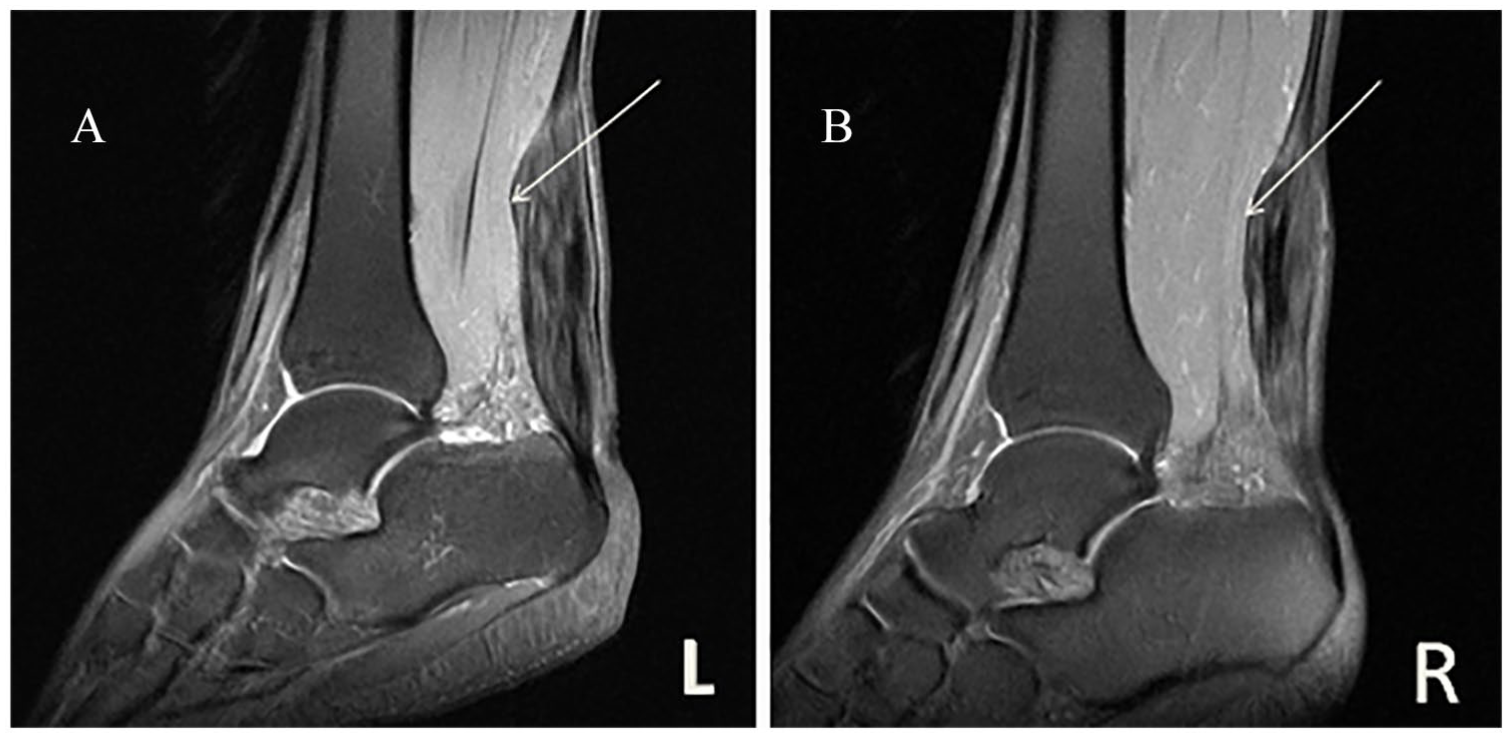
(A, B) Images of the sagittal plane (proton density-weighted) showed thickened Achilles tendons with a high signal strip, anterior border protuberance (white arrow), with no abnormal signal close to the subcutaneous tissues.

(A, B) Images of the axial plane (proton density-weighted) showed thickened bilateral tendons and right peroneal longus tendon with high (grid) and low (nodules) signal (white arrow).
Bilateral Achilles tendons were examined using ultrasonography, confirming that lower segment of bilateral Achilles tendons was thickened with inhomogeneous echo. Continuity of bilateral tendons was acceptable. The anteroposterior diameters of the left and right Achilles tendons were approximately 1.4 and 1.1 cm, respectively (Figure 4A and B).
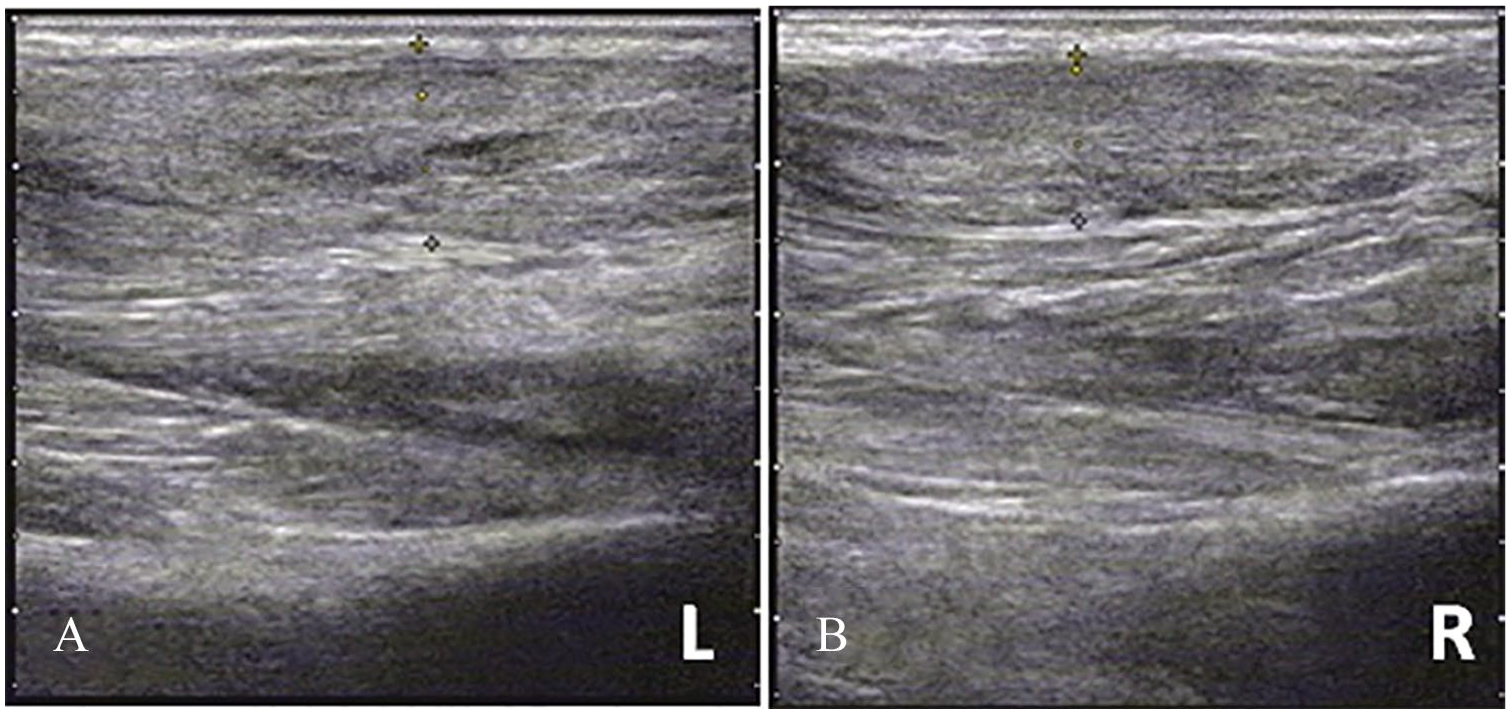
(A, B) Images of ultrasonography showed thickened bilateral tendons and inhomogeneous echo. Continuity of bilateral tendons was acceptable.
Exploration of both Achilles tendons and tumor resection was performed under spinal epidural anesthesia. Diffuse enlargement of the left Achilles tendon was discovered during surgery. There was close association between the tissue mass and the Achilles tendon. The surface of the tissue mass was dispersed into small, yellow nodules with unclear boundaries. After creating an incision in the Achilles tendon, adipose tissue that had infiltrated into the tendon and adipose nodules was observed distributed throughout the mass. Fat-infiltrated tissue and nodular adipose tissue were excised. Pathologic examination results showed that there was a large quantity of cholesterol crystals, clusters of foamy macrophages, foreign body giant cells, and fibrous hyperplasia within the mass. Amorphous urate crystals were observed in a number of nodules that were surrounded by multiple inflammatory cells and hyperplastic fibrous tissues. Combined with laboratory tests of hyperlipidemia and hyperuricemia, a diagnosis of xanthoma combined with gout infiltration of the Achilles tendon was made (Figure 5A and B).
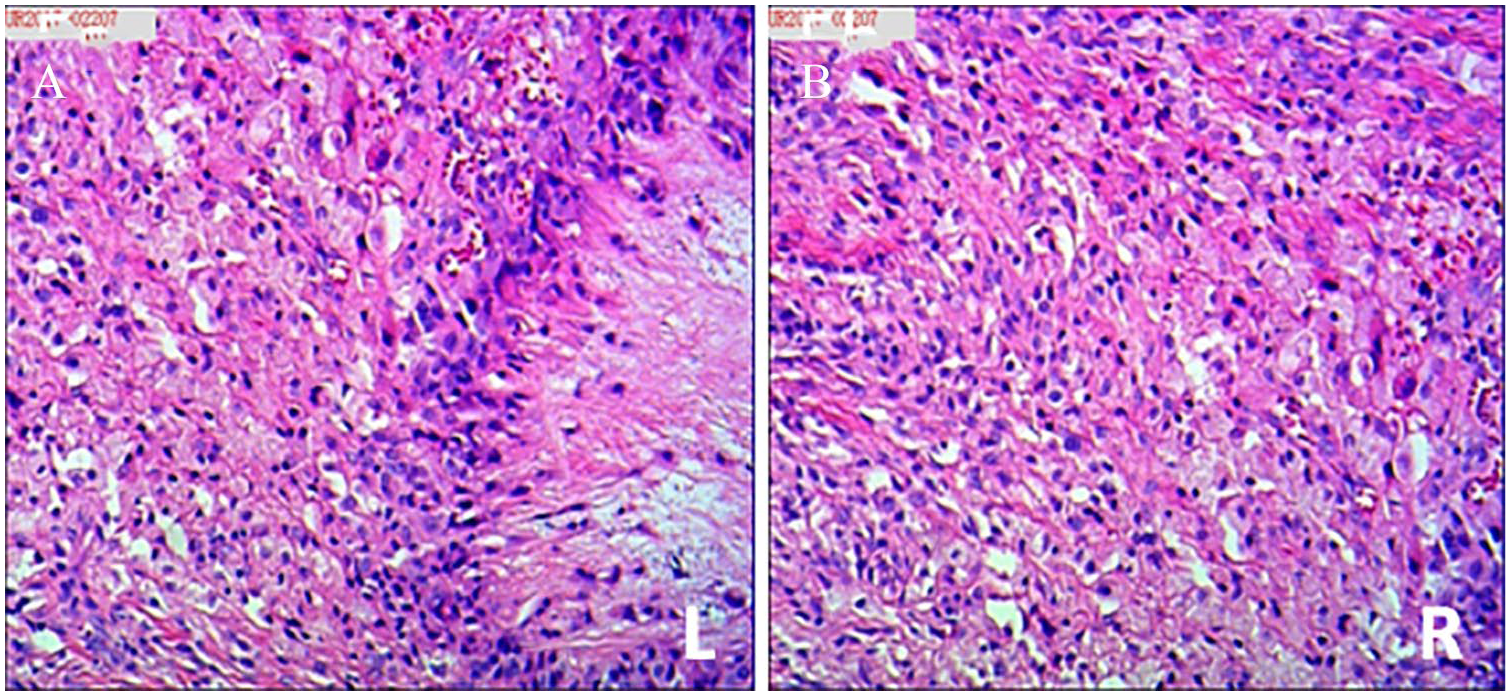
Pathologic examination of excised tissue indicated the presence of foam cells, gouty crystals, a large number of macrophages, and other inflammatory cells with proliferation of tendon sheath fibers. (HE×10) Hematoxylin and Eosin Stain.
Discussion
The Achilles tendon is commonly the location of tendinous xanthoma, in addition to tendons of the hand (extensor tendons) and those of the elbow.1,2 It is rarely observed in clinical practice and can easily be confused with neurofibromatosis. Gouty nodules can occur on any part of joints, including articular cartilage, synovial membranes, joint capsules, tendons, ligaments, or intraosseous or subcutaneous tissue. The joints of the extremities are the most common location.
It is likely that mechanical stress and extensive vascularization are essential factors in the formation of xanthomas. Furthermore, endothelial cells and macrophages are the principal contributors to the pathogenesis of tendinous xanthomas. Xanthoma of tendons occurs due to collagen fiber hyperplasia and macrophages becoming filled with cholesterol lipids.3–5 Tsouli noted that in most cases, tendinous xanthoma was associated with hyperlipidemia, although in rare cases, also in patients with normal blood lipid levels. 1 Plasma TG, TC, and LDL in this case were all higher than reference values. The mechanism of lipid deposition in tendons is not clear. At present, Rapp and other isotopes have been used to test the hypothesis that tendon xanthoma lipid principally comprises LDL from circulating blood, which transforms to oxidized low-density lipoprotein (OXLDL) at the tendon, which is then phagocytosed by local macrophages to form foam cells, which accumulate to form xanthoma. 3 , 6 The formation of tendon xanthoma when blood lipids are normal may be related to increased plasma monoclonal immunoglobulin or abnormal function of a number of lipoproteins. 7 In this case, there was no family history of the condition.
Sections of xanthoma are usually yellow. Xanthoma capsules are either intact or incomplete. They can penetrate into the tendon or are closely associated with the tendon. A large number of mononuclear cells with foamy cytoplasms were seen by microscopy. Inflammatory cells had infiltrated into some areas, and fibrous tissue and cholesterol crystals were observed locally. Gouty nodules are composed of crystalline or amorphous uric acid surrounded by inflammatory tissue, including histiocytes, lymphocytes, fibrocytes, and foreign-body giant cells. Blood supply to the gouty nodules is abundant, which essentially causes the formation of a granuloma due to the deposition of urate. The pathology of this case is consistent with xanthoma combined with gouty nodules.
Tendinous xanthoma occurs in tendons and ligaments, but mostly extensor tendons. The tissue mass is normally pliable with moderate mobility with a light yellow, brown red, or normal surface color, typically without pain. Recently, several international studies have shown that the anteroposterior diameter of the Achilles tendon is positively correlated with plasma cholesterol concentration and age.7,8 The clinical manifestations of gout can be divided into 4 stages, namely, asymptomatic hyperuricemia, acute gout, intermittent gout, and chronic gout stone. 9 Gouty nodules usually occur during the chronic gout stone stage, a clinical feature of gout. Typical gout nodules are commonly observed in joint capsule tendons and subcutaneous tissue in acromegaly. Because the patient in this case had a long history of gout, gouty nodules were diagnosed clinically before surgery. Xanthoma emerged as a differential diagnosis.
The Achilles tendon is generally thickened, with a convex front edge and an anteroposterior diameter >7 mm. Using an MRI spin echo sequence, thickening of the Achilles tendon was displayed as a high-signal fine mesh or strip, which was better defined in turbo spin echo (TSE) PDW images. 7 Dussault et al 10 believe that the uniformly sized, round, low-signal structures seen on magnetic resonance (MR) images probably represent collagen fibers, whereas the high-signal intensity between the collagen fibers represents infiltrating cholesterol-laden foam cells and an associated inflammatory response, but there is still a lack of histological evidence for this assertion. Bude et al 11 have presented characteristic MRI manifestation of Achilles tendon xanthoma, but the speckled or reticulated appearance, due probably to edema or inflammation or the tumor itself, could not be displayed. Magnetic resonance imaging is invaluable when no definite diagnosis exists or the need to exclude other lesions of the Achilles tendon cannot be achieved using other methods. 8 No qualitative diagnosis was achieved by ultrasound in this case, but MR examination showed bilateral Achilles tendon xanthoma. Furthermore, infiltration of the tendon by foam cells with attendant inflammatory changes can be detected as a signal abnormality on the MR images before tendon enlargement, so signal abnormality is more sensitive than physical changes to the Achilles tendon, but similar signal abnormalities can also be found in Achilles tendon injuries, degeneration, and tendinitis. 10 A comparison of the advantages of ultrasound and diagnostic MR requires a larger sample size combined with pathological follow-up. In this case, the anteroposterior diameters of the bilateral Achilles tendons were greater than 7 mm, and the tumor was clearly displayed on the MR images. Signals from gouty nodules on MR images are changeable and lack specificity. Long T2 signals are associated with protein components in gouty nodules. Short T2 signals are related to calcification, fibrous tissue, crystal, deposition of hemosiderin, and stability of protons in the gouty nodules. Magnetic resonance imaging suggested that this case was xanthoma. Because of the long history of gout, gout nodules were used as a differential diagnosis.
In conclusion, for patients with a long-term history of gout and poor control of uric acid, hyperlipidemia is accompanied by thickening of the Achilles tendon. Magnetic resonance imaging may play a more prominent role in diagnosing xanthoma combined with gouty nodules.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
YF is mainly responsible for collecting clinical and imaging data. At the same time, YF is also responsible for writing this case.
QLH is responsible for the feedback of the pathological results of this case and the preliminary revision of the manuscript.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique, is not under consideration by any other journal, and has not been published elsewhere. The authors confirm that they have permission to reproduce any copyrighted material. No author will receive monetary or other gain from publication of this article. The authors have presented this case for educational purposes only. Written consent was obtained from the patient or his relatives for publication of this study.
