Abstract
Traumatic brain injury is a major cause of death and disability in adults. This study investigated the effect of oral administration of amantadine on the neurological outcomes of patients with diffuse axonal injury (DAI) in the intensive care unit (ICU). This double-blind clinical trial was conducted in the ICU of Imam Hospital in Urmia. Patients with DAI were intubated and received mechanical ventilation in the ICU. They were divided into 2 groups: patients receiving amantadine (A) and placebo (P). The acquired data were analyzed using SPSS, P < .05 significant level. Findings showed no significant difference between the 2 groups in age and sex. There was no significant difference between the mean Glasgow Coma Scale (GCS) at the time of admission and discharge, and the mean Glasgow Outcome Scale (GOS) of the patients in 2 groups. No significant difference was observed in the duration of mechanical ventilation, hospitalization, and mortality in both groups (P > .05) in ICU. However, there was a significant difference between the mean GCS at the time of admission and discharge and death. Also, significant differences existed between the mean GOS in discharged and deceased patients (P = .001). This study showed no significant difference between the mean GCS at the time of admission and discharge and the mean GOS of the discharged patients and the mortality rate in the 2 groups. However, there were clear statistical differences between these variables in discharged and deceased patients. It is recommended that further studies are conducted with a larger sample size.
Introduction
Traumatic brain injury (TBI) is a major cause of mortality and long-term disability in people around the world under the age of 40 years. Its incidence ranges from 67 to 317 per 100 000 populations and its mortality rate varies from 4% to 8% in moderate injuries to 50% in severe injuries. By 2020, TBI will exceed many diseases as the major cause of death and disability (World Health Organization [WHO]). The main reason for a coma to occur following head trauma is not clear. One theory is based on the fact that following TBI, subcortical white matter may stimulate functional disconnection. Consequently, the metabolic activity between the cerebral cortex and the deeper regions of the brain may be interrupted or the link between the thalamic structures of the brain may be disconnected. This disconnection occurs due to diffuse axonal injury (DAI). The DAI is divided into 3 degrees: (a) injury to parasagittal white matter in the brain hemisphere, (b) regional injury to the corpus callosum in addition to the previous injury, and (c) injury to the cerebral crus (crus cerebri) in addition to the previous injuries. 1
Evaluation of the patients with TBI includes the primary clinical assessment based on airway, breathing, circulation, and disability (ABCD) approach and observation and neurological assessment to determine Glasgow Coma Scale (GCS). Prognosis for TBI depends on the severity of the initial injury. The initial GCS can somewhat define the initial neurological condition; however, it cannot accurately determine the prognosis. Medical or surgical treatments for TBI are based on the type of the injury.1,2
It should be mentioned that our understanding of the pathophysiological mechanisms of TBI has improved in the recent years; however, its pharmacotherapy has not improved significantly. 2 In the recent years, findings from limited studies have shown the role of dopaminergic neurotransmitter in improving levels of consciousness and adjusting sleep-wake cycles, although the molecular mechanisms of these processes are not fully understood.3,4 Initially, amantadine was used in the prevention and treatment of influenza. Later due to its effect on improving the symptoms of Parkinson disease (PD), Food and Drug Administration (FDA) approved it as an effective treatment for it. Amantadine is well absorbed orally, and about 90% is excreted unchanged from the kidneys. The time to peak plasma concentration ranges from 1 to 4 hours and its half-life is 15 hours in adults and 29 hours in elderly patients. The major positive effect of amantadine in reducing symptoms of PD is attributed to its dopaminergic effects.5,6 Some articles investigated the effect of amantadine on the dopaminergic system of the brain.
Different animal studies have also shown the effect of amantadine on increasing dopamine levels, especially in the frontal cortex.7,8 Amantadine could pass the blood-brain barrier (BBB) easily and is measurable in the cerebrospinal fluid (CSF). 9 Limited studies have demonstrated the beneficial effect of amantadine on improving levels of consciousness in patients with TBI. They suggested that amantadine was safe and resulted in a quicker improvement in patients with low levels of consciousness and reduced the rate of mortality due to TBI.10-13 Based on researchers’ knowledge, a similar study has not been conducted in Iran.
An increase in the incidences of TBI, the proven effect of amantadine on dopaminergic system and its possible role in improving the consciousness levels of patients motivated the researchers to conduct this study in the patients with TBI in intensive care unit (ICU) of Imam Khomeini Hospital, Urmia, Iran.
Method
Following the confirmation and approval of this double-blinded clinical trial study by the proposal Review Council of the Hospital and the University Ethics Committee, the study was conducted in 2016 aimed at investigating the effect of oral amantadine on the neurological outcomes of TBI patients admitted to the ICU. Diagnosis of TBI was done according to clinical manifestation and imaging by neurosurgeons before patients admitted to ICU. Sample size calculation was not performed, but the number of subjects was based on a previous study about this topic. 11 Patients were randomly assigned into 2 groups based on the random number table and researchers did not know which subjects were in the A or P groups. All patients with a DAI with GCS ⩽ 8 who were intubated and admitted to the ICU and received routine therapies were included in the study. Patients were divided into 2 groups: amantadine (A) and placebo (P).
In A group, patients received 100 mg capsules of amantadine orally, twice daily (from Amin Tehran Pharmaceuticals Company), and within 3 days, its dosage reached 200 mg twice daily. This treatment continued until the desired outcome, death, or complication. In P group, the patients received the placebo. Placebo was made by the drug manufacturer of starch in the form of amantadine tablets for identical appearance and shapes and was administered to patients.
Patients previously treated with amantadine or with a history of allergy to it were excluded from the study. Researchers filled the form containing demographic characteristics (age and sex), the mean duration of mechanical ventilation, length of ICU stay, GCS at the admission, discharge or in-hospital fatality, Glasgow Outcome Scale (GOS), and the frequency of mortality in patients. In addition, researchers obtained the consent from the primary caregivers before starting amantadine therapy. Patients’ information remained completely confidential, and the researchers remained loyal to the Declaration of Helsinki. It should be noted that amantadine therapy was stopped when the patients were extubated or when their GCS score was above 8 (GCS > 8).
Statistical Methods
Quantitative variables were presented as mean ± SD and qualitative variables as numbers (percentage). The mean age, GCS, GOS, duration of mechanical ventilation, and the length of hospitalization were compared in groups A and P using independent t-test. The frequency of sex and mortality was compared by Fisher exact test between 2 groups. Statistical analysis was performed using SPSS version 20 software. The P value <.05 was considered statistically significant.
Result
Of 33 patients in group A, 22 (66.7%) of them were men and 11 (33.3%) were women. Of 33 patients included in group P, 24 (72.7%) were men and 9 (27.3%) were women. Based on the Fisher exact test, there was no significant difference between the sex of the patients in 2 groups (P = .39).
The mean age of patients in the A group was 39.63 ± 13.08 years and the mean age in the P group was 35.84 ± 11.25 years. According to t-test, there was no significant difference between the age of the patients in 2 groups (P = .21).
In A group, the mean GCS at the time of admission was 5.72 ± 1.58, and in P group, the mean was 5.51 ± 1.64. Based on the results of the t-test, there was no significant difference between the 2 groups in GCS at the time of admission. In A group, the mean GCS at the time of discharge was 13.36 ± 2.02, and in P group, it was 13.03 ± 2.75. According to the t-test, there was no significant difference between the GCS in 2 groups at the time of discharge.
The mean GOS in the A group was 2.18 ± 1.21 and in P group was 2.15 ± 1.30. Results of the t-test suggested no significant difference between GOS in 2 groups at the time of admission (Table 1).
Comparison of mean and standard deviation of GCS and GOS at the time of admission and discharge in the 2 groups.

Abbreviations: GCS, Glasgow Coma Scale; GOS, Glasgow Outcome Scale.
The duration of mechanical ventilation in A group was 20.18 ± 15.87 days and 23.42 ± 16.33 days in P group. The t-test analysis showed no significant difference in the duration of mechanical ventilation between 2 groups. The mean length of hospitalization was 28.75 ± 17.38 days and 31.93 ± 18.29 days in both A and P groups, respectively. According to the t-test, there was no significant difference in the length of hospitalization between 2 groups (Table 2).

Of 33 patients in the A group, 31 patients (93.9%) discharged and 2 (6.1%) of them died. In P group, 30 (90.9%) patients discharged and 3 (9.1%) patients died. Results of the Fisher exact test showed that there was no significant difference in the outcome of patients with TBI (P = .5) in 2 groups. The mean GCS was 5.80 ± 1.52 at the time of admission in discharged patients and 3.40 ± 0.54 in deceased patients. The results of the t-test showed that GCS between discharged and deceased patients was statistically significant (P = .001). In other words, GCS was lower in dead patients at the time of admission. The mean GCS in discharged patients was 13.73 ± 1.52 and it was 6.60 ± 1.34 in deceased patients. Results of t-test analysis suggested that there was significant difference between GCS at the time of discharge and the outcomes of patients (P = .001). The mean GOS in discharged patients was 2.93 ± 0.98 and 1 for the deceased patients. Based on the t-test, there was significant difference between the GOS of discharged and deceased patients (P = .001). The mean duration of mechanical ventilation in discharged patients was 21.26 ± 16.49 days and it was 28.40 ± 7.43 days in the deceased patients. Results of t-test showed no significant difference between the duration of mechanical ventilation and the outcome of patients (P = .34). The GOS predicts the long-term course of rehabilitation to return to work and everyday life in TBI patients.
The mean length of hospitalization in discharged patients was 30.50 ± 18.39 days at the time of discharge and it was 28.44 ± 7.43 days in dead patients at the time of death. According to t-test, there was no significant difference between the mean length hospitalization and the outcome of patients (P = .80; Table 3).
Comparison of the mean and standard deviation of GCS at the time of admission, discharge and GOS, and the duration of mechanical ventilation and hospitalization based on the outcome of patients.
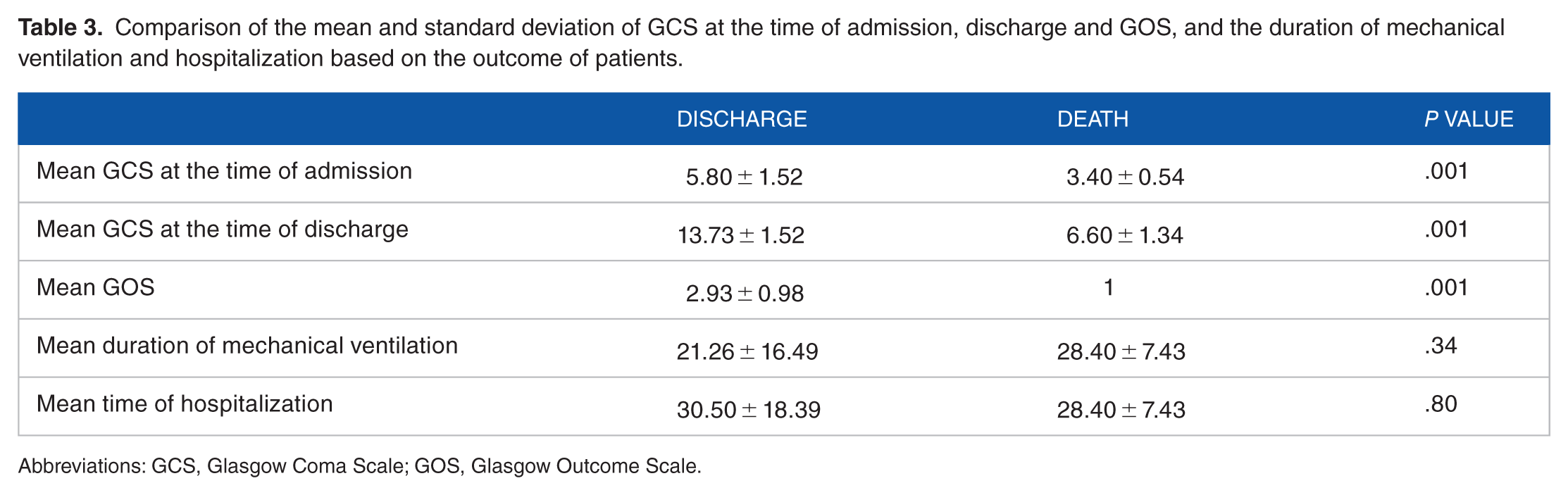
Abbreviations: GCS, Glasgow Coma Scale; GOS, Glasgow Outcome Scale.
Discussion
Hospitalized patients with traumatic DAI under mechanical ventilation in ICU of Imam Khomeini Hospital in Urmia were investigated in this study. In this study, patients were divided into 2 groups: amantadine (A) and placebo (P). There was no significant difference between 2 groups in age, sex, duration of mechanical ventilation, duration of hospitalization in the ICU, mortality, and mean GCS at the time of admission and discharge. However, significant difference was observed in GCS at the time of admission, discharge, or death. Also, significant difference was observed in GOS at the time of discharge or death between discharged and deceased patients.
Neuroprotection in TBI with various drugs conducted in animal and human models.14-17 Only a few studies have investigated the effect of amantadine after TBI. In 2012, Giacino et al 9 did a similar study. They examined the effect of amantadine and placebo in 184 patients who were in a vegetative state in placebo-controlled trial and stated that during the 4-week treatment period, recovery was significantly faster in A group than in P group.
In another study in Slovakia conducted by Saniova et al, 10 patients started receiving Amantadine on day 3 of hospitalization. Their findings suggested that in patients treated with amantadine, the outcome GCS was higher and the fatality rate was lower than in the group treated with standard therapy alone.
In a study conducted by Meythaler et al, 11 patients who received amantadine for 6 weeks showed an improvement in mini-mental status. They concluded that the patients with DAI seemed to improve more rapidly, while they were on amantadine. They considered that amantadine had a profound effect on the improvement of dopamine and N-methyl-D-aspartate (NMDA) receptors damaged in DAI. For sample size, this study involved a smaller sample than that of Giacino et al 9 and Saniova et al 10 and larger than the study conducted by Meythaler et al. 11 Unlike the aforementioned studies, there were no significant differences between A and P groups in GOS-GCS, mortality, duration of mechanical ventilation, and length of hospitalization in ICU. However, the mean GCS and GOS were significantly different in dead and discharged patients. In justifying these findings, it should be noted that the mechanisms, receptors, and their pass ways such as reticular activating system (RAS), cortex, and the interconnections involved in the consciousness of people are still not fully understood. The severity of the injuries and trauma, as shown in the case of the dead patients, is one of the most important issues in this regard. The effects of amantadine on dopamine and NMDA receptors are proven and probably other important mechanisms that are damaged during the trauma affect the outcome of injured patients. A 7 score increase in GCS in the discharged patients in A and P groups was a conflicting finding and authors could not find an explanation for it. Taking patients to health centers, treating them faster, and preventing the added injuries can be considered as a factor in its increase. In patients in A and P groups who died, little increase in GCS score was observed, which shows the severity of the trauma.
In 2004, Green et al 12 conducted a retrospective study in pediatric patients with TBI in the United States. They reported that amantadine group showed a greater improvement, and they suggested that it may be effective. In 2005 study, Leone and Polsonetti 13 suggested that following a TBI, amantadine is a reasonable option for improving cognition and reducing agitation; however, confirmatory evidence for the efficacy of it is necessary.
Both of the aforementioned studies acknowledge the potential impact of amantadine on neural functions in patients with TBI and DAI and recommend validating tools. Perhaps their intention is related to the complexity of physiological mechanisms involved in the neurological functions, including patient consciousness. This study, with its short time interval and low sample size, was not able to show a clear difference between A and P groups in combination with conventional and standard treatments for DAI. Two studies also examined the effect of memantine, an analog of amantadine, on animal and human samples. Ito et al 18 examined the effect of memantine on animal models. They indicated that memantine can protect against transsynaptic neuronal degeneration in the murine brain (superior colliculus) after retinal injury. In fact, they studied the protective effect of memantine on retinal cells in mice. In a 2010 study in Korea, Kim et al19,20 studied changes in cerebral glucose metabolism in patients with posttraumatic cognitive impairment after memantine therapy (20 mg/day) on 17 patients. They stated that the prefrontal and the parietal cortical structures after memantine treatment result in cognitive improvement in the setting of TBI. In this study, researchers treated 33 patients with 400 mg amantadine (200 mg twice daily). In spite of the difference in GCS at the time of admission, discharge, or death, and GOS of the discharged or deceased patients, there were no statistical differences between control and study groups. To investigate the effect of amantadine better, we suggest studying the process of enhancing GCS in both groups with a larger sample size.
Our study had a few limitations such as impossibility of the patients’ follow-up after discharge, a lack of clear consensus regarding radiological diagnosis of DAI, and a small sample size. We measured the GOS of the patients exactly before discharge, because the goal of our study was not a long-term follow-up and the patients were not available after discharge. We also knew the limitations of magnetic resonance imaging (MRI) in diagnosis of DAI. Currently, the capacity to radiological diagnose of DAI is a topic of study using tractography and diffusion tensor imaging (DTI). These new measures have not accessible in our hospital. We also suggest that further investigations in this topic will perform with enough sample sizes.
Based on the findings of this study, amantadine does not have any significant effect on the consciousness level of patients with TBI.
Conclusion
Finally, according to the results of previous studies and this study, it can be stated that amantadine and other neuroprotective drugs are still used routinely in some centers in patients with DAI and low levels of consciousness. This occurs in spite of a lack of a scientific foundation and with many challenges concerning their effectiveness. Further studies, with a larger sample size, are recommended.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
RA- Design of study, Review of Literatures, Statistical Consulting. MAVH- Corresponding Author, Help to Design of study, Data collection. AK- Review of Literatures, Analysis of Data. AM- Review of Literatures, Analysis of Data. MMSG- Data Collection, Literature review. GAM- Statistical Analyses, Final English Edit, Help to manuscript submission.
