Abstract
In the perinatal and adult forebrain, regionalized neural stem cells lining the ventricular walls produce different types of olfactory bulb interneurons. Although these postnatal stem cells are lineage related to their embryonic counterparts that produce, for example, cortical, septal, and striatal neurons, their output at the level of neuronal phenotype changes dramatically. Tiveron et al. investigated the molecular determinants underlying stem cell regionalization and the gene expression changes inducing the shift from embryonic to adult neuron production. High-resolution gene expression analyses of different lineages revealed that the zinc finger proteins, Zic1 and Zic2, are postnatally induced in the dorsal olfactory bulb neuron lineage. Functional studies demonstrated that these factors confer a GABAergic and calretinin-positive phenotype to neural stem cells while repressing dopaminergic fate. Based on these findings, we discuss the molecular mechanisms that allow acquisition of new traits during the transition from embryonic to adult neurogenesis. We focus on the involvement of epigenetic marks and emphasize why the identification of master transcription factors, that instruct the fate of postnatally generated neurons, can help in deciphering the mechanisms driving fate transition from embryonic to adult neuron production.
New neurons are generated throughout life in two specific regions of the mammalian brain: the hippocampus and the olfactory bulb (OB). In the OB, large numbers of interneurons are permanently issued from stem cells residing in the ventricular/subventricular zone (V/SVZ) lining the forebrain ventricles. From here, they migrate via the rostral migratory stream (RMS) into the OB where they differentiate as interneurons.
These postnatal and adult generated interneurons exhibit considerable phenotypic diversity at several levels. First, they are heterogeneous at the neurotransmitter level. Indeed, it has been shown that neurons using exclusively GABA, neurons that use both GABA and dopamine, and also very few glutamatergic neurons are generated and integrated. Second, they show varying final locations in the OB. Although most OB interneurons remain in the deep positioned granule cell layer, a substantial fraction integrates in the superficial glomerular layer. Finally, adult-born neurons show a wide spectrum of morphologies, projection patterns, and targets. 1
Lineage studies demonstrated that the diversity of OB interneurons is closely tied to their spatial origin in the stem cell compartment. For example, interneurons generated by progenitors of the dorsal part of the V/SVZ will predominantly integrate in the superficial layers of the OB and express subtype markers such as calretinin (CR), tyrosine hydroxylase, or the transcription factors (TFs) TBR1/2. In contrast, OB neurons produced along the lateral aspect of the ventricle are purely GABAergic and integrate in deeper layers of the OB. The discovery that neuronal heterogeneity is determined by their site of origin led to the notion that the stem cell niche represents a cellular mosaic in which populations of stem cells in defined dorsoventral and anteroposterior positions are predetermined to produce specific neurons for the OB. 2 This, in turn, implies that molecular determinants, for example, differentially expressed TFs, underlie early fate specification and ultimately neuronal function and connectivity.
Gene Expression in Space and Time
Tiveron et al. 3 set out to systematically identify and functionally characterize such fate determinants based on high-resolution gene expression analyses. Up to now, gene expression analyses performed in the V/SVZ-RMS-OB neurogenic system relied either on microdissection of tissues 4 or on cell sorting based on expression of a limited set of membrane markers5,6 that define specific differentiation stages. However, in this neurogenic system, such approaches present several limitations. Although the stem cell compartments of the dorsal and lateral lineages are physically separated, these regions harbor progenitors at different differentiation stages (neural stem cells [NSCs], transit amplifying progenitors and migrating neuroblasts). In addition, in the RMS and the OB, both lineages are intermingled and cannot be easily distinguished, let alone isolated.
To overcome these limitations, Tiveron et al. performed a lineage tracing approach based on targeted brain electroporation. Previous work demonstrated that in vivo brain electroporation can be used to transfect DNA-based vectors, 7 or even messenger RNA, 8 into the different stem cell compartments surrounding the ventricle. Over the following hours and days, relatively homogeneous and timed cohorts of cells pass through the different differentiation stages in the V/SVZ and the RMS to finally integrate as OB interneurons. Using green fluorescent protein expression followed by microdissection, dissociation, and fluorescence-activated cell sorting, the group isolated such cohorts after 8 hours, 1, 2, 4, 7, and 14 days, thus covering the entire neurogenic sequence from stem cells to neurons. This approach was performed in parallel for the dorsal and the lateral neuronal lineage. Subsequent microarray analyses generated a unique high-resolution picture of gene expression in space (dorsal versus lateral stem cells) and in time (from stem cells to neurons).
In a first analytic step, the authors were particularly interested in TFs specifically expressed and regulated in the dorsal lineage because this progenitor pool accounts for most of the interneuron subtype diversity in the OB. Moreover, the dorsal stem cell niche undergoes considerable molecular and structural rearrangements 9 during the transition from embryonic to postnatal neurogenesis and produces remarkably different neuronal subtypes before and after birth: first glutamatergic projection neurons for the developing cortex and later mainly GABA/dopaminergic interneurons for the OB. Therefore, Tiveron et al. compared the expression dynamics of selected candidate factors in the embryo and at postnatal stages.
These analyses led to the description of 4 categories of TFs based on their expression (Figure 1):
First, TFs that are expressed in the embryonic pallium and whose expression pattern and dynamics are maintained after birth in progenitors of the dorsal V/SVZ. This class is, for example, represented by Emx1 and Emx2.
Second, TFs that are expressed in both embryonic and postnatal progenitors but whose expression pattern changes after birth. A striking example of this category is Pax6. Pax6 is transiently expressed during corticogenesis where it controls the generation of glutamatergic projection neurons. However, after birth, most of the dorsal V/SVZ progenitors maintain its expression all along the neurogenic sequence. In this context, Pax6 is indispensable for the generation of dopaminergic OB neurons. 10
Third, TFs that in the embryo are confined to subpallial NSCs but that start to be expressed in the dorsal lineage after birth. This is the case for the Dlx1/2 TFs, which are expressed in the embryonic ganglionic eminences where they represent major determinants of cortical interneuron fate. 11 At postnatal stages, both TFs are induced during migratory stages in the dorsal lineage. 10 Importantly, it has been demonstrated that Dlx2 interacts with Pax6 to specify dopaminergic interneurons of the OB. 12
Fourth, TFs that show de novo expression in postnatal NSCs. This is the case for Zic1 and Zic2, which in the embryo are restricted to medial aspects of the ventricular wall. During postnatal and adult stages, Zic1/2 appear in the dorsal stem cell compartment, coincident with the shift of neuron production from glutamatergic projection neurons to GABAergic and dopaminergic interneurons.
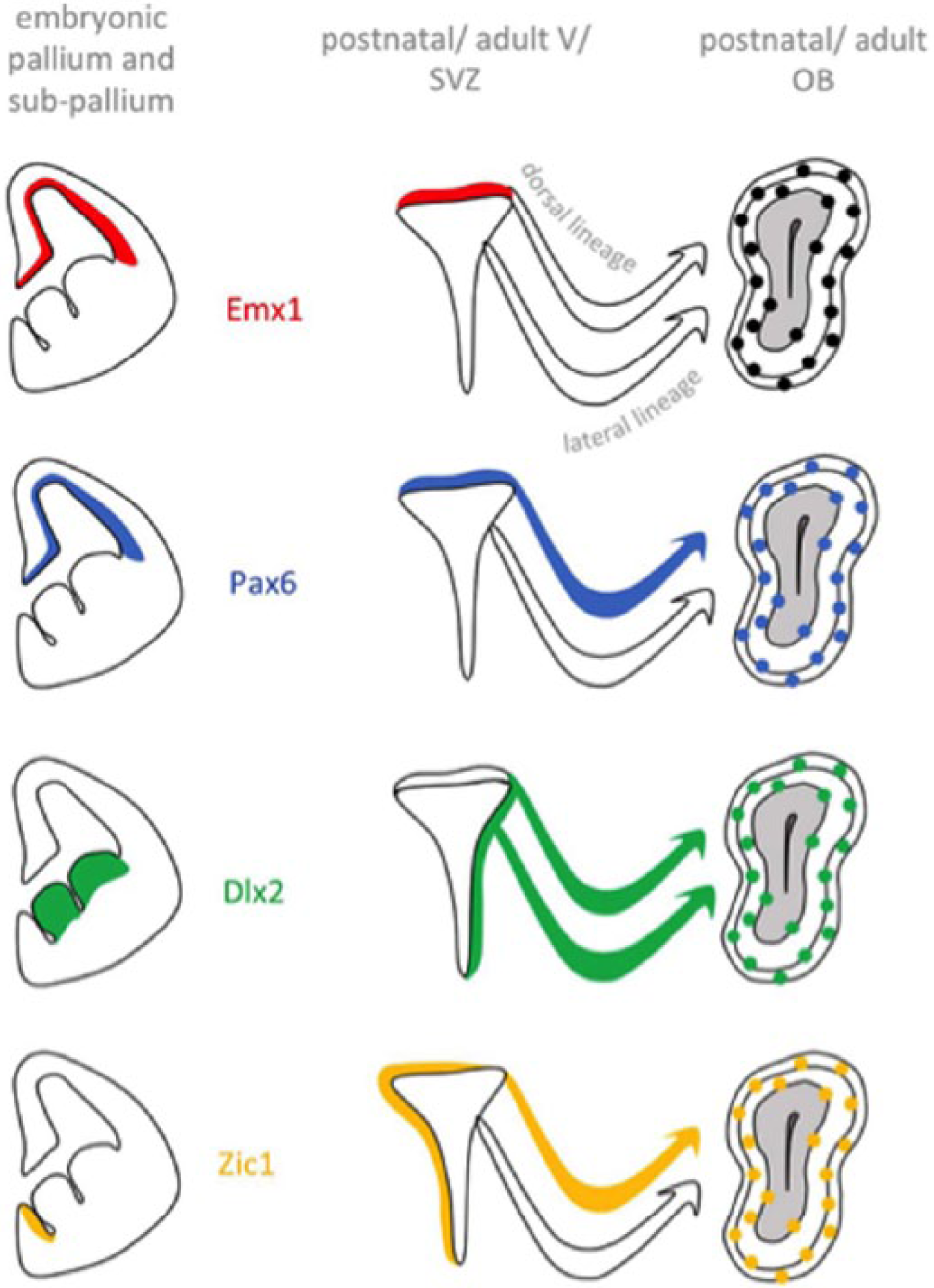
Examples of 4 categories of transcription factors classified based on their respective expression pattern in the embryo versus postnatal dorsal V/SVZ. V/SVZ indicates ventricular/subventricular zone; OB, olfactory bulb.
Zic Proteins as Molecular Fate Determinants
Based on these results, Tiveron et al. aimed at deciphering the functional implication of Zic1 and Zic2 in the control of neuronal differentiation of dorsal progenitors. Using a pan-Zic antibody, 13 the authors first revealed that Zic proteins are postnatally induced in a mediolateral gradient within the dorsal V/SVZ but are absent from the lateral V/SVZ (Figure 2). By combining electroporation-based lineage tracing with Zic immunostaining, the authors demonstrated that the proteins are expressed in about 50% of all cells in the dorsal stem cell compartment and maintained in migrating neuroblasts and mature neurons. Further analyses with subtype-specific markers revealed that Zic protein is highly expressed in most of the CR-positive OB interneurons but excluded from dopaminergic neurons. 9 Based on this observation, the authors hypothesized that Zic1/2 act as factors promoting the generation of this CR-expressing OB interneuron subtype.
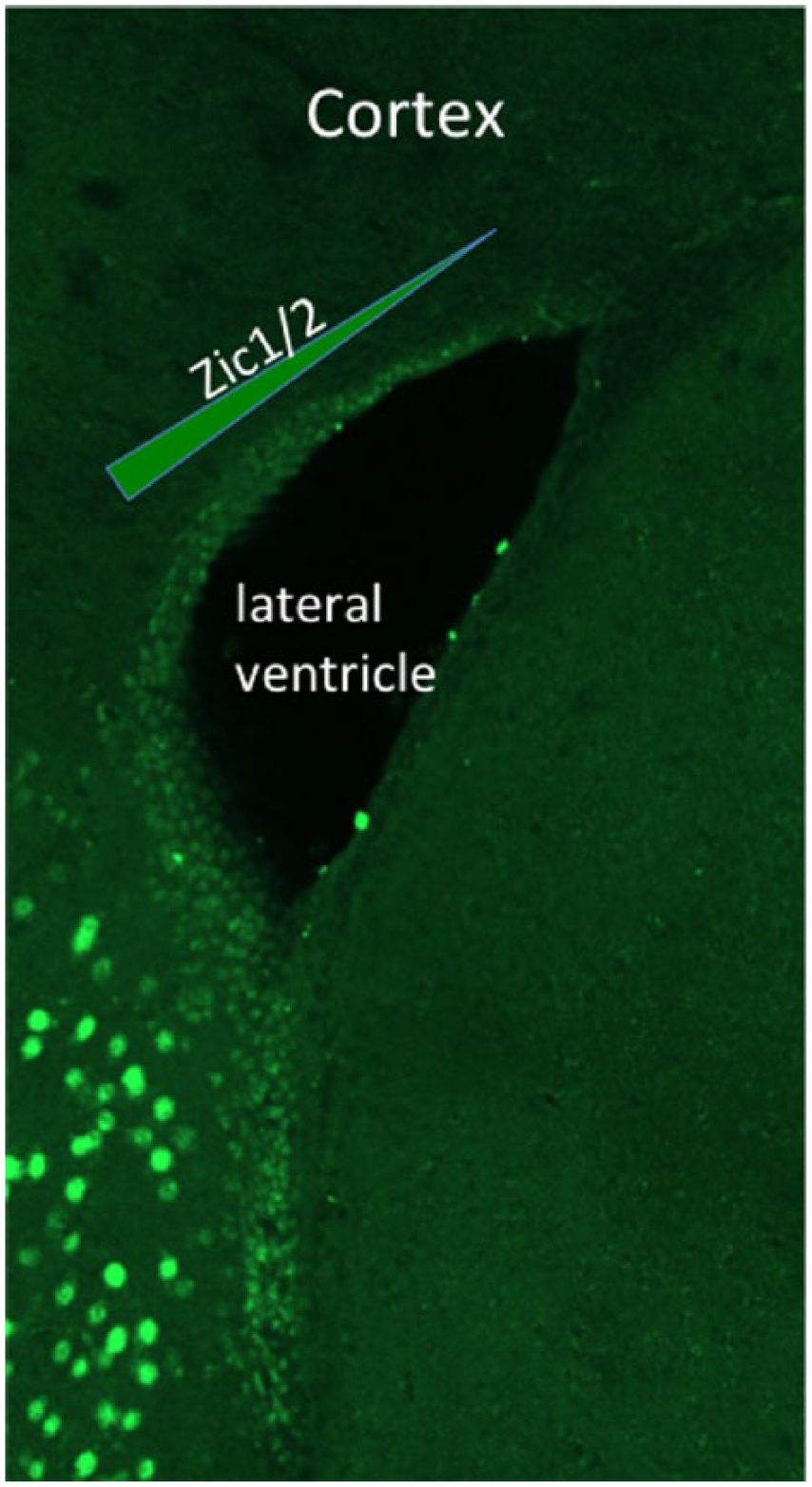
Zic immunostaining of the adult mouse forebrain, showing a mediolateral gradient of Zic1/2 expression in the dorsal wall of the subventricular zone.
Again, in vivo brain electroporation was used to investigate the validity of this scenario. Indeed, Zic1 or Zic2 overexpression favored the generation of CR-positive interneurons at the expense of dopaminergic neurons. Short hairpin RNA–mediated knockdown led to the opposite effect. Altogether, these results demonstrated that both Zic proteins act in the postnatal forebrain as inducers of CR expressing neurons while repressing dopaminergic fate. Interestingly, the anti-dopaminergic function of Zic proteins is evolutionary conserved and already present in Caenorhabditis elegans. 3
Intrinsic Determinants versus Extrinsic Cues
One of the most striking features of the V/SVZ-RMS-OB neurogenic system is represented by the radical change in neuronal subtype production by forebrain stem cell compartment that occurs around birth. What underlies the origin of the modifications in the expression pattern of these master TFs? Indeed, expression of some of these factors may be under the control of upstream TFs. However, for genes at the very top of the genetic cascade, the appearance of additional mechanisms seems likely. Work of several teams in the OB neurogenic system pointed to chromatin remodeling factors as important players in the control of fate shifts. For example, the polycomb protein Bmi1 is involved in the maintenance of NSCs, 14 whereas the trithorax-related protein Mll1 drives NSCs into neuronal differentiation. Interestingly, Dlx2 represents the main target of Mll1, which induces histone methylation marks at the Dlx2 locus that are necessary for transcription. Furthermore, the interaction of Mll1 with the Dlx2 locus is crucial for progenitors to give rise to OB interneurons. 15 Based on these data, it is likely that epigenetic events lie at the origin of expression changes of specific TFs which, in turn, lead to the emergence of new traits of V/SVZ progenitors at birth.
At the cellular level, novelty can either occur as an intrinsic program within the cell (cell autonomously) or may originate from extracellular cues (non–cell autonomously). In the V/SVZ-RMS-OB neurogenic system, this point has been addressed in several studies. The presence of a cell-autonomous program was demonstrated in a study where V/SVZ progenitors from distinct subregions were isolated and then re-grafted at different locations within the stem cell niche. 2 The authors observed that the neuronal subtypes generated by these progenitors show fates representing their place of origin rather than the region in which cells were grafted. However, other studies showed that the environment of NSCs also plays a role in neuronal fate determination and that, for example, morphogens regulate adult OB neurogenesis. In particular, the genetic ablation of the Shh pathway in the most ventral V/SVZ progenitors prevented their capacity to generate calbindin positive interneurons in the glomerular layer of the OB. 16 Likewise, the Wnt signaling pathway was shown to control the neurogenic capacity of progenitors located in the dorsal part of the V/SVZ. 17
Conclusions
During neural development, stem cells pass through successive stages that change the phenotype of the neurons they produce. It is now evident that both intrinsic and extrinsic cues are instructing the NSC niche. Clearly, more experimental work is needed to understand their intricate relationship as players in neuronal fate determination, in general, and specifically the acquisition of novel traits after birth. The identification of new key TFs such as Zic1/2 and a deeper insight into their temporal and spatial expression dynamics will pave the way for the comprehension of the molecular events at the basis of these transformations. Finally, a better understanding of these processes will not only elucidate how novelty is generated by V/SVZ progenitors but will also be relevant to neurodevelopmental processes in general.
Footnotes
Acknowledgements
The authors acknowledge the France-BioImaging/PICsL infrastructure (ANR-10-INSB-04-01) and the IBDM animal facilities.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: H.C. received funding from Agence National pour la Recherche (ANR) and Fondation pour la Recherche sur le Cerveau (FRC).
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AA, MCT, HC and CB wrote the paper. CB provided Images. AA performed artwork.
