Abstract
The favorable outcomes achieved with modern mitral valve repair techniques redefined the role of mitral valve replacement. Various international databases report a significant decrease in replacement procedures performed compared with repairs, and contemporary guidelines limit the application of surgical mitral valve replacement to pathology in which durable repair is unlikely to be achieved. The progressive paradigm shift toward endoscopic and robotic mitral valve surgery is also paralleled by rapid developments in transcatheter devices, which is progressively expanding from experimental approaches to becoming clinical reality. This article outlines the current role and future perspectives of contemporary surgical mitral valve replacement within the context of mitral valve repair and the dynamic evolution of exciting transcatheter alternatives.
Keywords
Introduction
The rapid development, favorable impact, and simplification of durable mitral valve repair techniques redefined the modern role of mitral valve replacement.1–5 The significant decrease in mitral valve replacement procedures performed in developed countries parallels modern international guidelines that strongly advocate mitral valve repair whenever possible.6–9 In addition, the increasing reports of experimental catheter-based device implantation are appealing to both clinicians and patients. 10 This article outlines the current role and future perspectives of contemporary surgical mitral valve replacement within the context of mitral valve repair and the dynamic evolution of exciting transcatheter alternatives.
Indications for Mitral Valve Replacement
Current guidelines limit mitral valve replacement to irrepairable valve pathology that will result in poor durability outcomes, especially in patients unlikely to tolerate future reinterventions. The presence of significant annular calcification; valvular dystrophic, inflammatory, or infective changes; subvalvular thickening or fusion; and progressive cardiomyopathy warrant primary mitral valve replacement to avoid the adverse operative outcomes associated with heroic attempts at repair that eventually result in replacement. 11 The persistently high incidence of rheumatic valvular disease with subsequent mitral valve stenosis in developing countries favors mitral valve replacement if primary percutaneous mitral valve balloon valvuloplasty is unavailable or clinically contraindicated. Aggressive annular decalcification and attempts at repair are reported with inconsistent long-term outcomes.12,13
Advanced valvular cardiomyopathy, age, and debilitating comorbidities are associated with poor mitral valve surgical outcomes, and it is reported that a substantial portion of severely symptomatic mitral valve patients are prohibited from surgical intervention by institutional heart teams for these reasons.14,15 The progressive clinical application of transcatheter replacement devices will most likely offer therapeutic alternatives to these patients and redesign current guidelines and recommendations for the generic approach to mitral valve disease.
Current Prosthesis Types and Selection
The surgical replacement of a stenotic or insufficient mitral valve is based on the premise that the prosthesis type chosen will have a beneficial impact on cardiac function and quality of life within the context of perioperative risks and long-term prosthesis complications.
In modern practice, 7 mechanical,16–23 6 stented biological porcine,24–28 and 1 bovine pericardial prostheses 29 are available and approved for clinical use, which are classified and illustrated in Table 1 and Figure 1, respectively. The technical specifications and hemodynamic profiles of each device are well described and should be integrated in the prosthesis selection process. Mechanical valves are generally acquitted from structural failure 30 but require lifelong anticoagulation with associated thromboembolic and bleeding risks. Annular pannus formation can result in leaflet dysfunction and may require reintervention if significant. Biological valves are inevitable subjected to structural degeneration and may require future reintervention.
Current Food and Drug Administration–approved mitral valve prostheses.
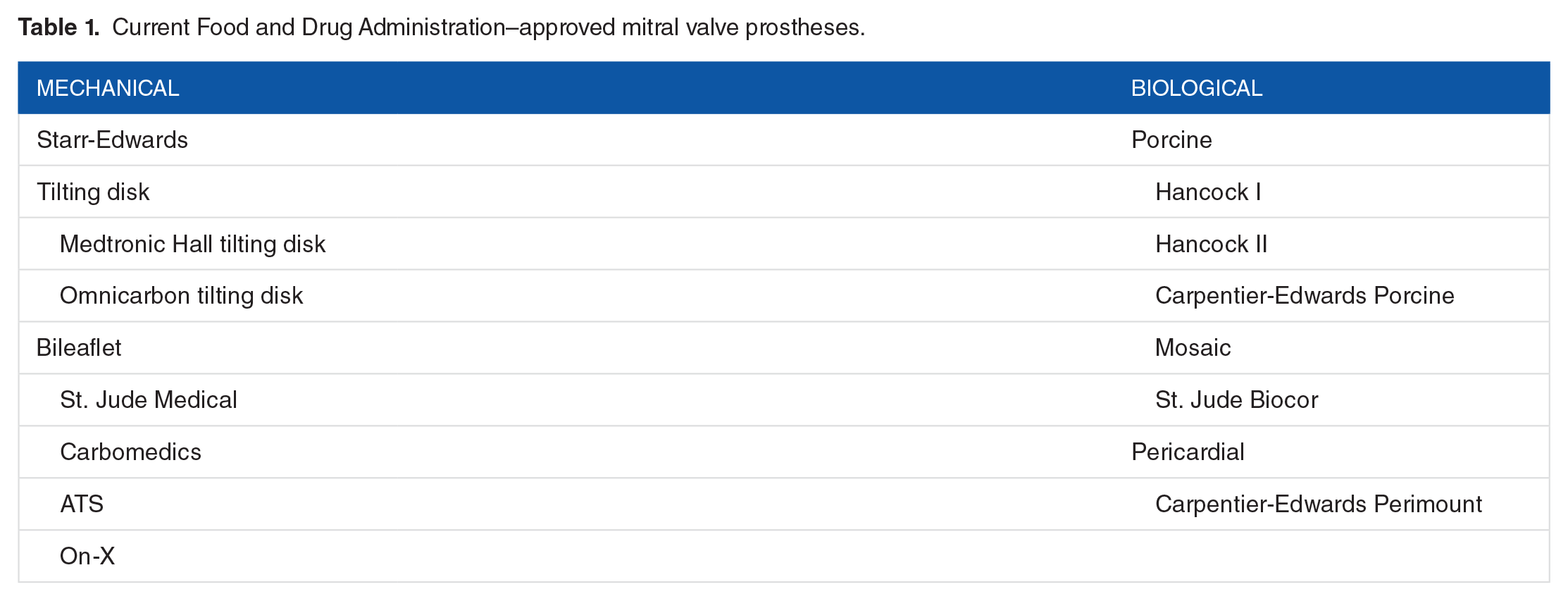
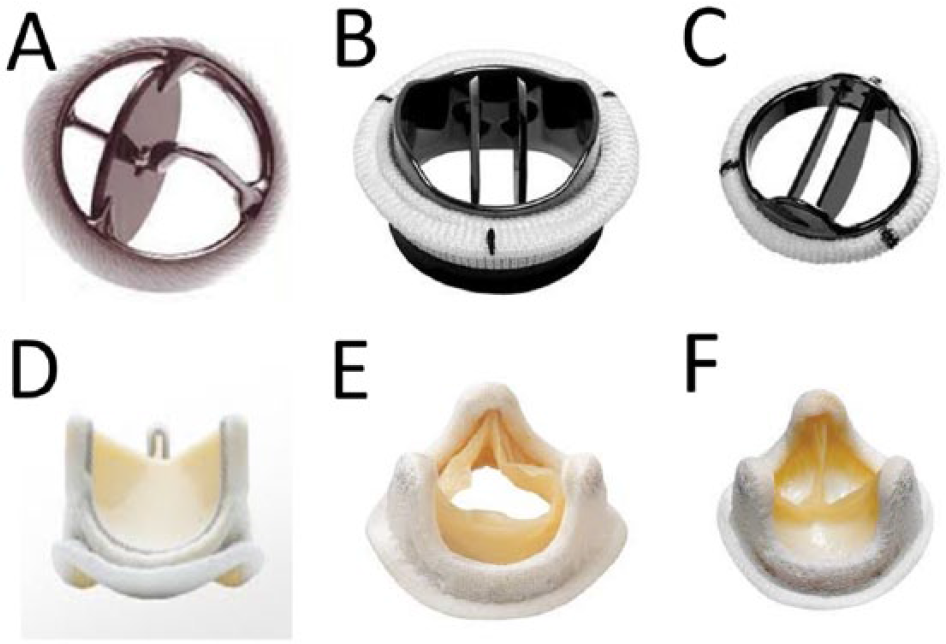
Examples of current Food and Drug Administration–approved mitral valve prostheses: (A) Medtronic Hall tilting disk, (B) On-X, (C) St. Jude Medical, (D) Carpentier-Edwards Perimount, (E) Mosaic, and (F) Hancock II.
Current international recommendations for bioprosthetic implantation include the following: informed patients who refuse mechanical valves, when safe therapeutic mechanical valve anticoagulation levels are unlikely to be achieved or contraindicated due to bleeding risks, when mechanical valve thrombosis occurs despite sufficient anticoagulation, when pregnancy is contemplated in young women, when future reinterventions can be performed at low risk and patients with life expectancy judged to be less than the presumed durability of the bioprosthesis. Even though current guidelines advocate bioprosthetic implantation in patients older than 70 years, continuous improvements in valve durability technology suggest possible safe and effective implantation at a younger age. 31
A mechanical prosthesis is recommended according to the desire of the informed patient if there are no contraindications for long-term anticoagulation, if risk factors for accelerated structural valve deterioration are absent, in patients already on anticoagulation as a result of having a mechanical prosthesis in another valve position, in patients younger than 65 years, in patients with a reasonable life expectancy for whom future redo valve surgery would be at high risk, and in patients already on long-term anticoagulation due to high risk of thromboembolism.
The choice of prosthesis should be individualized within the context of patient expectations, values, and health care preferences. The advent of transcatheter mitral valve devices will offer exciting alternatives to patients currently deemed unfit for surgery and will undoubtedly redefine the indications for mitral valve replacement in the future.
Technical Considerations and Operative Complications
Median sternotomy is still considered to be the most commonly used incision for mitral valve surgery, which allows access and exposure through Sondergaard’s groove, a transseptal incision, or by a transatrial oblique approach. 32
Minimal access, endoscopic (Figure 2) and robotic approaches are becoming increasingly established as excellent surgical alternatives.33–35 However, the current device costs in an unfavorable economic climate, in addition to significant learning curves in an era of uncompromising quality control, limit its application to centers of excellence with established experience. The technical setup of endoscopic port access surgery is well described. An anterolateral right mini-thoracotomy over the fourth intercostal space is used as working port for primary and redo surgery. Underlying lung adhesions are carefully released from the incision site, diaphragmatic surface, and pericardium to allow focused target access in redo cases.36,37 Venous drainage is usually established through the right internal jugular and femoral vein, and a femoral artery cannula with Y-arm is used for arterial flow and an endo-aortic for aortic occlusion and delivery of cold antegrade crystalloid cardioplegia.38–40 All guidewires and cannulae are positioned under transesophageal echocardiography guidance and preoperative aorta-iliac-femoral-axis evaluation either by an additional iliac artery contrast injection during cardiac catheterization or by magnetic resonance angiography usually performed in all patients.
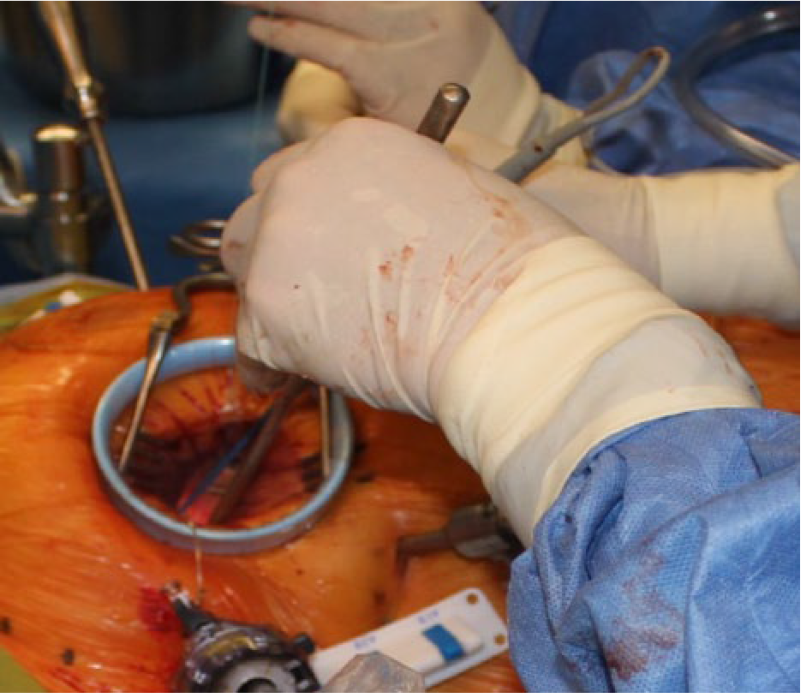
Endoscopic mitral valve surgery using long-shaft instruments and peripheral cannulation.
Native subvalvular apparatus should be preserved or reinforced to maintain left ventricular geometry and function.41–43 Extreme calcification of the posterior annulus may require radical removal, which partially detaches the left atrium from the left ventricle.
Everting or noneverting sutures should be placed into the annulus with attention to avoid injury to the noncoronary aortic valve leaflet, circumflex coronary artery, and atrioventricular conduction tissue located in close proximity. Endo-aortic balloon occlusion is frequently used in endoscopic mitral valve surgery and care must be taken to avoid puncture if the device migrates into the noncoronary aortic valve sinus during the procedure. It is generally advocated to use noneverting stitches for all prosthesis in the mitral position to identify and confirm proper seating. Leaflet mobility should be assessed to ascertain no entrapment by subvalvular structures.
It is generally recommended that mechanical prosthesis should be oriented in an anti-anatomic fashion 36 and bioprosthetic strut location oriented such that contact with the ventricular wall and impingement on the left ventricular outflow tract are avoided.
Left ventricular rupture occurs in 1% of procedures and can occur at the level of the annulus, papillary muscles, or mid-ventricular zones. 44 It is associated with aggressive decalcification and endocardial disruption that result in the intermyocardial fiber dissection of blood with subsequent reported mortality of 50%. Immediate recognition and replacement of the valve with dissection tract incorporation is required.
Postoperative Outcomes, Long-term Follow-Up, and Complications
Operative mortality associated with isolated mitral valve replacement is reported to range between 4% and 7% and is influenced by age, premorbid valvular cardiomyopathy, and other comorbidities.45,46 Lower operative mortalities were reported with minimally invasive approaches. 47 There are no differences in 10-year survival between mechanical and biological valves when patient characteristics are taken into account, which is reported to range between 50% and 60%.48,49
Thromboembolism is the most common postoperative complication of both bioprosthetic and mechanical valves and occurs at a rate of 1.5% to 2.0% per patient-year and is significantly increased in chronic atrial fibrillation and large left atrial size. 50 All mitral valve prostheses require postoperative anticoagulation, with lifelong vitamin K antagonists recommended for all patients with a mechanical prosthesis or bioprostheses who have other indications for anticoagulation.8,9,51 Current guidelines suggest target international normalized ratio (INR) levels according to prosthesis thrombogenic risk. Carbomedics, Medtronic Hall, St. Jude Medical, and On-X are regarded as low risk and require mean INR levels above 2.5, whereas all other bileaflet valves are considered medium risk for which INR levels above 3 are suggested. Lillehei-Kaster, Omniscience, Starr-Edwards, Bjork-Shiley, and other tilting disk valves are classified as high risk and require mean INR levels above 3.5. Patient-related risk factors are also considered and include mitral or tricuspid valve replacement, previous thromboembolism, atrial fibrillation, mitral stenosis of any degree, and left ventricular ejection fraction less than 35%. The presence of one or more patient risk factors requires a target INR level increase by 0.5. Current guidelines8,9 recommend the use of vitamin K antagonists for the first 3 months after implantation of a bioprosthesis. The low level of evidence pertaining to current bioprosthetic anticoagulation management is reflected by reports that suggest similar thromboembolic incidence for vitamin K antagonists and acetylsalicylic acid. 52 Bleeding rates related to the use of vitamin K antagonists are more frequent with mechanical valves, which are reported to be 2% to 4% per patient-year, of which the majority occur within the first year following surgery.
The addition of low-dose aspirin should be considered in patients with concomitant atherosclerotic disease and in patients with a mechanical prosthesis after thromboembolism despite adequate INR. 8 New oral anticoagulants, including factor 10 inhibitors, are currently not recommended as substitutes for vitamin K antagonists.8,9
In case of valve thrombosis, thrombolytics may be used to treat mitral prosthetic thrombosis in the absence of cardiogenic shock. If thrombolysis fails, or if there is hemodynamic compromise, valve replacement is required. 53
Prosthetic valve endocarditis risks are similar for both types and are reported to be 1.5% to 3% for the first year and 3% to 6% within 5 years. Long-term endocarditis risk is 0.2% to 0.35% per patient-year thereafter and appears to be slightly higher with mechanical valves. Endocarditis prophylaxis and management of prosthetic valve endocarditis are extensively described in specialized guidelines. 54
Prosthetic valve degeneration is the most significant complication of bioprosthetic valves. The 10-year freedom from clinically significant structural valve degeneration associated with biological valves is reported to be 78%, 89%, and 100% when implanted in patients younger than 60 years, between 60 and 70 years, and older than 70 years, respectively.55,56 Annual echocardiographic follow-up is recommended after the first 5 years following implantation to detect early signs of structural valve degeneration, regurgitation, or features of progressive stenosis, which include calcification, leaflet stiffening, and reduced effective orifice area. Reoperation is warranted in symptomatic patients with severe regurgitation or significant transprosthetic gradient increase and should be considered in asymptomatic patients with significant prosthetic dysfunction, provided that they are at low risk of perioperative complications. Prophylactic replacement of a bioprosthesis that is older than 10 years and without structural deterioration may be considered if an operative intervention is required on another valve or on the coronary arteries. Percutaneous balloon interventions should be avoided in the treatment of stenotic left-sided bioprostheses. Treating bioprosthetic failure by transcatheter valve-in-valve implantation is feasible in patients considered to be inoperable or at high risk 57 but is not an established alternative to surgery.
Patient prosthesis mismatch can occur when the indexed geometric orifice area is less than 1.5 cm2/m2 and may warrant replacement if high gradients and symptoms persist despite optimal medical therapy. 58
Paravalvular leak is reported to occur in 1.5% of the patients.59,60 It can be avoided by selecting a prosthesis with a large sewing ring in heavily calcified or poor-quality annular tissue. The use of pledgeted, noneverting mattress sutures and reinforcing the annulus with Teflon strips are reported.61,62 Reoperation is recommended if diagnosed early postoperatively, if related to endocarditis, or if associated with hemolysis requiring repeated blood transfusions or symptoms. Transcatheter closure is feasible, but reports that confirm consistent efficiency are limited at present. 63 It may, however, be considered in heart team–determined high-risk or inoperable patients.
Future Perspectives: Transcatheter Mitral Valve Replacement
The rapid development of catheter-based replacement devices and the continuous changes in patient expectations provide exciting prospects for the future treatment of mitral valve disease.
The feasibility of transcatheter mitral valve replacement was demonstrated on June 12, 2012, when Lars Sondergaard implanted the first-generation CardiAQ valve system (CardiAQ Valve Technologies, Inc., Irvine, CA, USA), through transfemoral-transseptal access in an inoperable 86-year-old patient. 64 Four transcatheter mitral valve systems65–67 that have subsequently been implanted in humans (Figure 3) and are in current clinical use include the second-generation CardiAQ valve system (Edwards Lifesciences, Irvine, CA, USA), Tiara valve (Neovasc Inc., Richmond, Canada), Tendyne valve (Tendyne Inc., Roseville, MN, USA), and Twelve valve (Medtronic, Minneapolis, MN, USA).
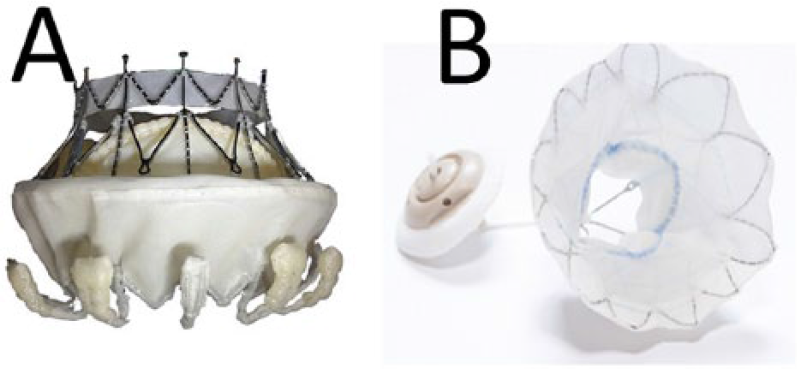
Examples of percutaneous mitral valve replacement prosthesis: (A) CardiAQ and (B) Tendyne.
These valves consist of nitinol self-expanding frames, bovine pericardial leaflets (Tendyne, however, is porcine), and a fabric-sealing skirt (CardiAQ consists of a pericardial skirt) and are delivered through direct transapical access. CardiAQ can also be delivered by transfemoral-transseptal access. Other devices, such as HighLife (HighLife SAS, Paris, France), Caisson (Caisson Interventional, LLC, Maple Grove, MN, USA), and M-Valve (Boston Scientific, Marlborough, MA, USA) are in preclinical development and will attempt to further offer innovative design solutions to overcome the challenges of catheter-based mitral valve replacement. 68
Conclusions
The role of mitral valve replacement is under continuous reevaluation and is at present limited to irrepairable valves or patients at high risk for future reinterventions. Successful outcomes are determined by meticulous perioperative risk assessment, prosthesis selection, anticoagulation management, and long-term clinical surveillance in well-informed and compliant patients. Endoscopic and robotic surgical approaches introduced attractive alternatives to conventional sternotomy access and are progressively becoming favored as the preferred surgical approaches by heart teams worldwide. Transcatheter mitral valve implantations are now a clinical reality and will undoubtedly redefine the role of mitral valve replacement in the near future.
Footnotes
Peer review:
Three peer reviewers contributed to the peer review report. Reviewers’ reports totaled 533 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Both authors contributed equally to this manuscript and apply the principles outlined in their daily mitral valve practices.
