Abstract
Background
Mitral valve disease (MVD) in children is highly variable, including such etiologies as complete atrioventricular septal defects, left-sided obstructive lesions, parachute mitral valve, valve dysplasia, mitral cleft, prolapse, and rheumatic MVD.1–3 Mitral valve disease can occur in isolation or within complex congenital heart diseases such as hypoplastic left heart syndrome or Shone's complex. 4 Mitral valve replacement (MVR) poses considerable morbidity and mortality in the pediatric population; 5 thus, mitral valve repair is performed whenever possible, particularly in very young patients. 6 However, MVR in some cases is inevitable, with mechanical mitral valve prostheses most commonly utilized due to their availability in small sizes (down to 15 mm) and increased durability compared with bioprosthesis, particularly in children.7,8
Although studies investigating the outcomes of children undergoing mechanical MVR (m-MVR) have examined specific age groups, such as those less than 2 years of age,2,9–11 or less than or equal to 5 years,3,12,13 these studies have been limited by small sample sizes and individual center experience and have not examined the spectrum of outcomes in neonates, infants, and children. The Kids’ Inpatient Database (KID) provided an opportunity to examine age-related characteristics and outcomes of children undergoing m-MVR across many centers.
Methods
Patient Population, Variables, and Outcomes
The KID was utilized to identify patients who underwent m-MVR (aged ≤ 18 years) in 2009, 2012, 2016, and 2019 (from January 1 to December 31 of each year). Patients with univentricular physiology who underwent m-MVR were excluded (n = 13). The KID is one of the Healthcare Cost and Utilization Project (HCUP) databases. Kids’ Inpatient Database is the largest publicly all-payer claim-based database in the United States. It includes data from approximately three million pediatric discharges each year representing approximately 20% of US inpatient hospitalizations across the country. The KID uses information from approximately 1,000 hospitals on the basis of size (small, medium, and large), teaching status (rural, urban nonteaching, urban teaching), and region (Northeast, Midwest, South, West), representing the overall hospital population in the United States. As per the KID website (https://hcup-us.ahrq.gov/kidoverview.jsp), KID data are being released every three years from 1997 through 2012; then every 3 years beginning with 2016. The KID was not available for 2015 because of the transition from ICD-9-CM to ICD-10-CM/PCS coding. The Institutional Review Board exempted the study as patients’ information is extracted from a national deidentified database.
A retrospective cohort study of admissions for m-MVR in patients ≤18 years of age was performed. Kids’ Inpatient Database was queried for The International Classification of Diseases-Ninth Revision (ICD-9) and Tenth Revision (ICD-10) codes to identify index procedure, demographics, hospital characteristics, associated procedures, and perioperative complications. Chronic lung disease in baseline characteristics included patients with ventilator-dependent respiratory failure, primary or secondary hypertension, and reactive airway disease. Respiratory complications in the perioperative period included diagnoses such as acute respiratory failure, requirement of mechanical ventilation > 48 h, pneumothorax, chylothorax, pneumonia, or pleural effusion. We used the following ICD-9 and ICD-10 codes for chronic kidney disease (CKD); ICD-9 codes for CKD; 585.1 Stage 1, 585.2 Stage 2, 585.3 Stage 3, 585.4 Stage 4, 585.5 stage 5, 585.6 end-stage renal disease, and 585.9 CKD, unspecified, and ICD-10 codes for CKD; N18.1 stage 1, N18.2 stage 2, N18.3 stage 3, N18.4 stage 4, N18.5 stage 5, N18.6 end-stage renal disease, and N18.9 CKD, unspecified.
The primary endpoint was in-hospital mortality within age groups (neonates < 1 month, infants 1-12 months, and older children). Secondary endpoints included perioperative complications, length of hospital stay (LOS), and nonhome discharges within age groups.
Statistical Analysis
Statistical analyses were performed using SPSS Software Version 28.0.0 (IBM Corporation). The entire cohort was divided into three groups according to age, neonates < 1 month, infants 1 month to 1 year, and older children and adolescents (1-18 years). Categorical variables were presented as counts and percentages, and continuous variables were reported as medians with interquartile range (IQR) or mean with standard deviation where appropriate. One-way analysis of variance was used to compare continuous variables if the continuous variable is normally distributed, and Kruskal-Wallis was performed for non-normally distributed variables. Comparisons between categorical variables were performed using χ2 test, and Fisher exact test was utilized when cell counts were less than 5. A P value <.05 was considered statistically significant.
Results
Demographics, Baseline Characteristics, and Indications
A total of 2,773 pediatric patients underwent MV procedures (repair and replacement), and of those 500 patients (18%) underwent m-MVR. The proportion of m-MVR involving patients who underwent MV procedures increased from 17.3% (110/637) to 30.8% (141/457). Demographics, baseline characteristics, and indications of surgery are depicted in Table 1. There is substantial variation among the US regions regarding m-MVR in neonates, with the South having the highest volume and Northeast having the lowest volume. Previous cardiac surgery is more common in older children and infants compared with neonates (55% vs 38.2% vs 25%, P < .002). Preoperative long-term anticoagulation is higher in older children compared with infants and neonates (27% vs 7.9% vs 5%, P < .01). Five patients required extracorporeal membrane oxygenation (ECMO) before surgery, of whom two were neonates.
Demographics, Baseline Characteristics, and Indications of Children Undergoing m-MVR.
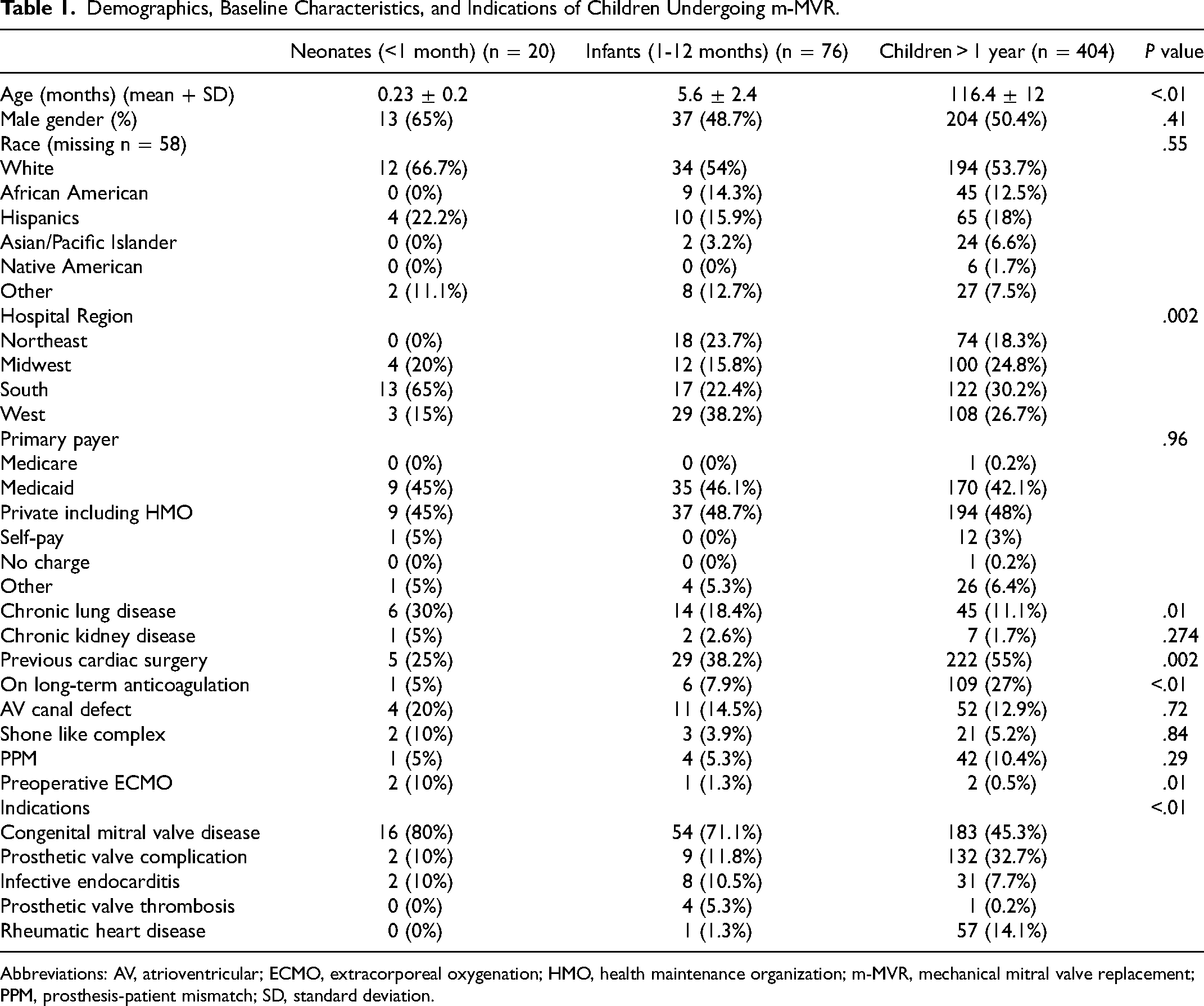
Abbreviations: AV, atrioventricular; ECMO, extracorporeal oxygenation; HMO, health maintenance organization; m-MVR, mechanical mitral valve replacement; PPM, prosthesis-patient mismatch; SD, standard deviation.
Operative Information
Operative data are shown in Table 2. Concomitant procedures occurred in 119 patients (23.8%), and there was no difference in distribution across the groups. Sixty-one patients (12.2%) had double mitral and aortic valve replacement, and this is comparable between the groups. ECMO was needed on the day of surgery or following days in 19 patients (3.8%), and was more frequently used in neonates and infants.
Operative Data.

Abbreviation: ECMO, extracorporeal membrane oxygenation.
Perioperative Complications and In-Hospital Mortality
The overall in-hospital mortality was 4.8% and was significantly higher in neonates and infants (10% vs 11.8% vs 3.2%, P = .003, Table 3). Figure 1 shows the timing of mortality during hospital stay. There was no significant difference between patients who received double mitral and aortic valve replacement during the same hospitalization compared with patients who had isolated MVR or MVR with other procedures (8.2% vs 4.3%, P = .185). Complications, including renal and respiratory, are more common in neonates and infants (Table 3). Complete heart block requiring pacemaker implantation after surgery occurred in 47 patients (9.4%), and the incidence is similar across the three groups. As expected, the LOS was longer in neonates (57 days [IQR, 24.8-90] vs 29.5 days [15.5-61] in infants vs 10 days [7-18] in older children and adolescents, P < .01). Transition into short-term/skilled nursing facilities or requiring home health care is higher in neonates and infants compared with their older counterparts (40% vs 36.8% vs 13.1%, P < .01).
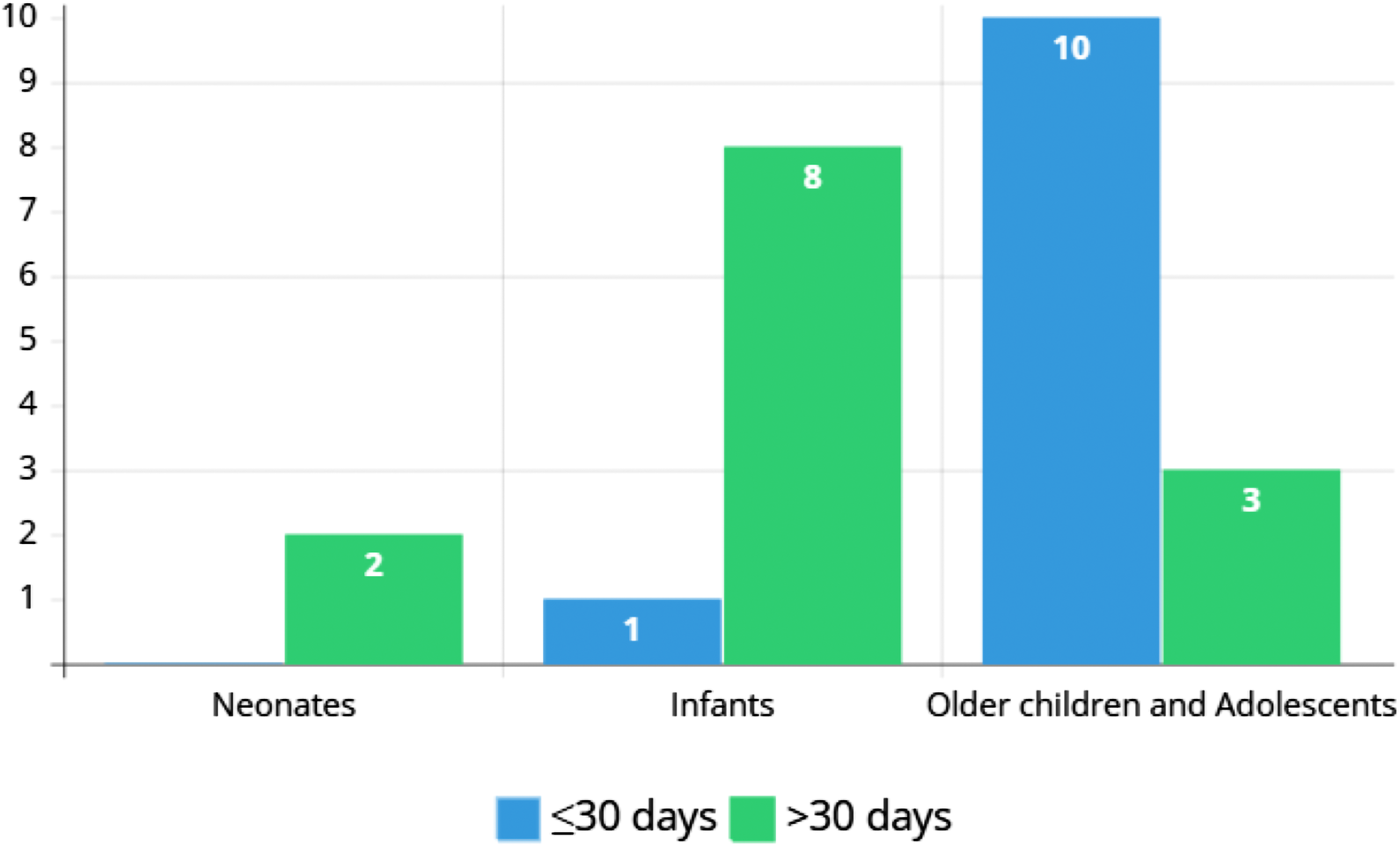
Timing of in-hospital mortality in neonates, infants, and older children undergoing mechanical mitral valve replacement (m-MVR).
In-Hospital Mortality and Perioperative Complications.
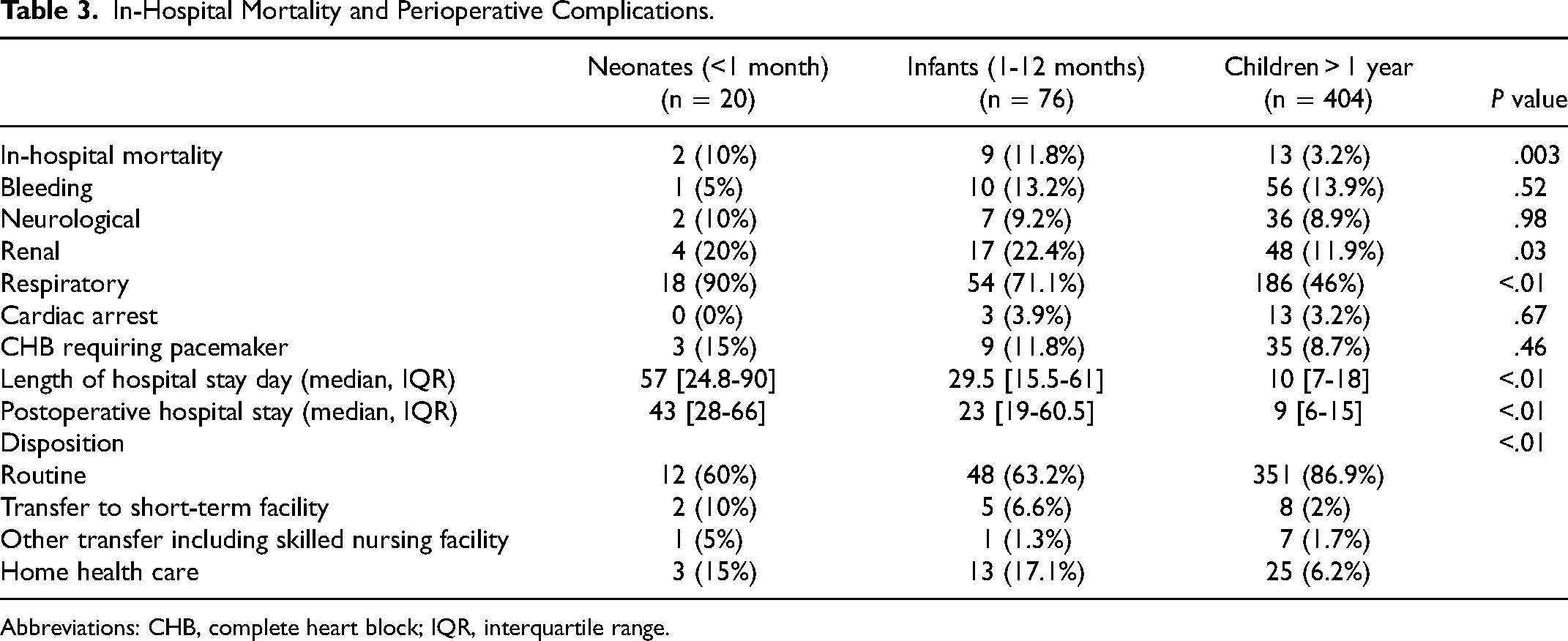
Abbreviations: CHB, complete heart block; IQR, interquartile range.
Discussion
The principal findings of this large national survey of m-MVR in the pediatric population are the following: (1) m-MVR was increasingly performed among pediatric patients undergoing MV interventions between 2009 and 2019. (2) There was a regional variation regarding m-MVR in neonates. (3) In-hospital morbidity and mortality were higher in neonates and infants compared with older children and adolescents. (4) The incidence discharge to rehabilitation facilities or need for home health care was substantial in neonates and infants.
In our study, we classified patients undergoing m-MVR into neonates, infants (1 month to 1 year), and older children > 1 year to concentrate on neonates and infants. There are a number of challenges when m-MVR is performed in a young child. First, the discrepancy between the native mitral valve annulus and prosthesis size; the smallest available prosthesis may be too large for the annulus size of native valve. This can lead to “high” valve placement in the left atrium, coronary complications, and prosthesis-patient mismatch (PPM). Furthermore, as the young child grows, PPM occurs with progressive cardiac stress and the potential for cardiac dysfunction over time.
Second, neonates and infants are usually in critical condition at the time of MVR with high respiratory support and significant elevation of left atrial pressure despite the maximal medical treatment. This deconditioning, including chronic ventilator support and back pressure on the lungs, has consequences in their recovery from surgery as well as rehabilitation. Historically, younger age at time of surgery was a major risk factor for in-hospital morbidity and mortality in comparison to older children and adolescents; early mortality in children <2 years was reported between 11% and 42% in small observational studies.2,9,14,15 A recent large multicenter study by Ibezim and associates found that early mortality in patients <2 years was as high as 25% (36/144 patients). 8 In-hospital mortality in our investigation in neonates and infants was 11% (11/96 patients) demonstrating improved early mortality in the most recent era. Tierney et al investigated 118 children <5 years who underwent MVR and showed a significant decline in operative mortality from decade to decade, from 41% (1976-1982) to 5% (1994-2006). 13 Although this is encouraging, it was unclear how survival has improved over time specifically in neonates and infants. The Mayo Clinic experience has shown a temporal decrease in early mortality with time in patients <2 years (14% [1993-2000], 9% [2001-2010], and 0% in 2011-2019]); however, this study was limited by a relatively small sample size in this younger age group. 14 The authors attributed the improved outcomes in the last decade to early intervention, improved surgical experience, improvement in perioperative care, and implementation of updated anticoagulation protocols.
The 15-mm m-MVR from St Jude Medical was approved recently by the US Food and Drug Administration and has been utilized as the smallest available m-MVR. 16 A multicenter study from the Netherlands reported 17 neonates and infants (median age 3.2 months) who had this prosthesis placed in the mitral position and were followed up for more than 10 years. 17 The study showed 6% early mortality (1/17 patients), mitral re-replacement was necessary in 11 patients (65%) with median of 2.9 years between the initial m-MVR and mitral re-replacement. Major adverse events including thromboembolism were low. The results from the prior experience were validated by another study performed by Eltayeb and colleagues that investigated seven infants who underwent 15 mm mechanical prosthesis. In this study, there were no operative deaths, and the mean time to re-replacement was 23 months. 18 The stented bovine jugular vein graft valve (Melody valve, Medtronic) has emerged as an encouraging substitute when surgically implanted. 19 However, left ventricular outflow tract obstruction is a particular concern with this approach.20,21
This national survey found that there is a national variation regarding the use of mechanical prosthesis in neonates, with the Northeast having the lowest incidence. Tierney et al from Boston Children's Hospital demonstrated a significant decline in MVR in infants and young children during the last 25 years, whereas balloon mitral valve dilation and mitral valve repair have expanded dramatically. 13 Another study from the same institution supports delaying early intervention in infants to allow early atrioventricular valve remodeling and development in other MV pathologies and reported increased incidence of MV repair. 4 The increase in MV repair was explained by the substantial advances in preoperative imaging and improvement in surgical techniques. During the same time frame of the current study, our data showed that 1,255 neonates and infants underwent mitral valve repair, and the in-hospital mortality was 4.4% which was lower than the in-hospital mortality of m-MVR in neonates and infants (11/96 patients, 11.5%).
In-Hospital Morbidity
The incidence of ECMO use following congenital heart surgery in children ranges from 3.2% and 8.4%.22–24 A recent meta-analysis investigating outcomes of patients undergoing ECMO following congenital heart surgeries by Wu and colleagues found an estimated in-hospital mortality as high as 57%, and bleeding was the most common complication in 47%. 25 The pooled overall incidence of neurological events and renal failure was 47% and 39%, respectively. Consistent with the literature, our investigation demonstrated that the incidence of patients who required intra- or postoperative ECMO due to cardiopulmonary compromise was 3.8%. Extracorporeal membrane oxygenation instituted on the same day of surgery was more often in neonates and infants compared with older children (P = .04). Mortality was as high as 47.4% (9/19 patients) in patients who required intra- or postoperative ECMO, and four patients were infants.
Pacemaker implantation after m-MVR poses significant morbidity and mortality. 26 A morphological study performed by Berdajs et al revealed that the atrioventricular node artery passes in close proximity to the mitral valve annulus in 23% of patients, and its damage potentially leads to the development of atrioventricular block. 27 Our investigation showed that 9.4% of patients had complete heart block requiring pacemaker implantation, and distribution was similar across the three groups. Our results also revealed that there was no difference between patients who had first m-MVR and patients who had a history of prior cardiac surgery. Tierney and associates demonstrated that supra-annular prostheses have lower risk of pacemaker insertion than annular placement in the post m-MVR period; however, patients continued to be at risk when prosthesis was re-replaced later. 13
Also, important findings from our study are the relatively high prevalence of substantial morbidities including acute respiratory failure, acute kidney injury, significant bleeding, and neurologic complications. Additionally, m-MVR in children is associated with prolonged hospitalization, particularly in young patients, with significant rates of home healthcare services.
Limitations
This study presents contemporary outcomes analysis in children with m-MVR providing valuable insights into a group of patients that will be a subject for further investigations in the era of emerging new mitral valve therapies. It represents “real world” outcomes and cross-section of all types of centers across the United States. However, our study has a number of limitations; first, KID is a national database that is derived from data of hospital claims with no access to patients’ medical records; consequently, it limits our analysis to the provided variables, reducing the ability to obtain detailed data. Additionally, we were not able to definitively determine the sequence of hospital complications throughout the hospital course and whether they occurred before or after index surgery. This should be taken into consideration when interpreting the data. Secondly, the utilized codes for diagnoses and procedures can be subject to reporting errors. However, the KID attempts to reduce the potential errors by using internal and external quality control methods; additionally, the major procedures codes are highly accurate because they are the main methods for reimbursement for the hospitals, so it is very unlikely to have coding error for m-MVR that could hamper our results. The error rates for HCUP database (nationwide inpatient sample) diagnoses and procedures codes in the 2003 database were 0.08% and 0.01%, respectively. 28 Finally, KID reflects a snapshot of hospital admission, and specific long-term data beyond hospital discharge are not available.
Conclusion
Mechanical MVR is increasingly performed during the most recent years. In-hospital mortality and morbidity remain higher in neonates and infants compared with older children and adolescents with prolonged hospitalization and higher rates of nonhome discharges. New emerging small mechanical prosthesis, bioengineered valves, newer mitral valve repair techniques, and transcatheter MV interventions offer a reasonable hope to improve in-hospital outcomes in neonates and infants.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
