Abstract
Background:
In recent years, there has been a growing interest in exploring the potential contribution of tryptophan (TRP) metabolism via the kynurenine (KP) and serotonin (SP) pathways in Glioblastoma (GBM) biology. This study aims to address the association between pre-operative peripheral blood levels of TRP, kynurenine (KYN), 5-hydroxy-tryptophan (5-HTP), and serotonin (5-HT) and relevant oncological outcomes in GBM IDH-wt patients.
Methods:
This is a single-center, retrospective clinical study. Serum from 62 adult patients undergoing maximal safe resection of newly diagnosed glioblastoma WHO-grade 4 IDH-wt (GBM) and n = 27 healthy controls were analyzed. The variables of interest were dichotomized via maximally selected rank statistics. Kaplan Meier and Cox multivariate regression analysis were conducted to explore the single contributions of these parameters in building a predictive model of overall survival (OS) and progression-free survival (PFS) in these patients.
Results:
The mean baseline serum levels of 5-HT, KYN, and 5-HTP were significantly lower in GBM when compared to n = 27 healthy individuals (P < .001). Patients with 5-HT <78 ng/mL had a median OS of 14.4 months compared to 22.5 months in patients with increased levels (P = .01). Shorter OS was observed in patients with KYN <18 ng/mL (9.8 vs 17.5 months, P = .002), KYN/TRP <2.55 (11.4 vs 17.1, P = .002), 5-HTP/TRP <0.89 (11.5 vs 17.6 months, P = .02), and 5-HT/TRP <5.78 (13.4 vs 19.1 months, P = .002) compared to patients with high levels. Shorter PFS in patients with 5-HT <78 ng/mL (P = .04), KYN <18 ng/mL (P = .02), 5-HT/TRP <5.78 (P = .001), KYN/TRP <2.55 (P = .005). Reduced KYN, 5-HTP, and 5-HT were independent predictors of poor OS.
Conclusions:
This study highlights an intriguing association between the degradation of TRP along the KP and SP and median survival times in GBM. Decreased KYN, 5-HTP, and 5-HT levels were associated with shorter OS.
Introduction
Glioblastoma (GBM) is the most common, fast-growing, and aggressive malignant primary CNS tumor, with a survival time of about 15 months. 1 The incidence of GBM ranges from 3.19 cases to 4.17 per 100 000 person-years, with a peak incidence in people over 40 years of age. 2 Current GBM treatment is multimodal and has not been substantially changed since 2005, despite remarkable efforts in neuro-oncological research. It consists of maximal safe resection surgery, followed by concurrent adjuvant radiotherapy and chemotherapy. 1 Glioblastoma cells share with other malignant tumors the migratory capability, mainly through infiltration and invasion of surrounding brain parenchyma, thus making it difficult to distinguish the tumor cell mass from the surrounding healthy tissue. The understanding of specific molecular mechanisms underlying GBM growth is of strategic relevance to conceiving and developing new therapeutic approaches. Along the genetic background, the tumoral microenvironment plays a crucial role in sustaining GBM cells’ proliferation, energy supply, and immune escape. This microenvironment encompasses a set of cellular and molecular components that form the context in which the tumor initiates, proliferates, and eventually infiltrates surrounding normal tissue and actively participates in communication that ultimately supports tumor progression. 3 Recent evidence suggests a pivotal role of tryptophan (TRP) metabolism via the kynurenine (KP) and serotonin (SP) pathways in this intricate peritumoral interplay.4,5
The KP is the major route of TRP catabolism leading to the production of the essential pyridine nucleotide, nicotinamide adenine dinucleotide (NAD+), along with several downstream metabolites of kynurenine (KYN). Around 1% of TRP is used for the synthesis of serotonin (5-HT) along the SP, 6 see Figure 1. The indoleamine 2,3-dioxygenase (IDO-1 and IDO-2) and tryptophan 2,3-dioxygenase (TDO-2) are the first and rate-limiting enzymes of the KP transforming TRP into formyl-KYN rapidly converted into KYN. 6 Particularly, IDO-1 and TDO-2 expression is likely to play a role in mediating brain tumor immune evasion via inhibiting tumor-specific immunity and driving malignant progression. 7 In physiological conditions, the peripheral KYN/TRP ratio mostly reflects TDO activity, while in inflammatory or tumoral conditions, IDO activity is likely to prevail.6,8 Hyperactivity of the KP may confer tumor survival advantages by increasing the availability of NAD+ for DNA repair, assisting in cell replication and maintenance of high metabolic activity, ultimately promoting tumor cell viability and proliferation. 9 Moreover, the up-regulation of KP enzymes inhibits tumor-specific immunity through a combined effect of depletion of TRP from the tumor microenvironment, a reduction of cytotoxic T-cell infiltrates, and a significant increase in the immunosuppressant T-regulatory (T-reg) phenotype. 10

Tryptophan metabolism via the kynurenine and serotonin pathway. Adapted from Comai et al. 6
The first step in the SP is the hydroxylation of TRP by the rate-limiting enzyme, tryptophan 5-hydroxylase (TPH), to 5-hydroxytryptophan (5-HTP). Changes in circulating TRP levels have been associated with an imbalance in its availability for the synthesis of 5-HTP and 5-HT in both the periphery and the central nervous system.6,11 5-HT is known to mediate glioma cell growth and migration,12,13 while its role in neuroimmune circuits, interorgan communication, and inflammation and immunity associated with glioma is a new emerging research field. The exact mechanism and receptors promoting these events in glioma cells are still largely unknown.
The KP and SP metabolites can be easily measured in blood and might be promising hallmarks of TRP metabolic derangements in GBM. While the KYN/TRP plasma ratio is used as an indirect marker of KP pathway activation,14 -16 the 5HTP/TRP and 5-HT/TRP plasma ratios might be a proxy of SP activity.6,17
Materials and Methods
Study design, patient selection, and data retrieval
This is a single-center, retrospective, observational clinical study designed to assess TRP metabolism via KP and SP in GBM patients. We included patients with adequate clinical follow-up who underwent maximal safe resection of newly diagnosed glioblastoma WHO grade 4 at IRCCS San Raffaele Hospital, Milan, Italy, between 2019 and 2022. Pediatric patients (<18 years old) and patients showing unresectable disease, undergoing biopsy only, IDH-mutated tumors, and recurrent GBM were excluded from the current analysis.
Diagnoses were originally performed according to the 2016 or 2021 WHO criteria, depending on the time of surgery. We only included “primary” (IDH 1-2 wild-type) GBM tumors. In detail, between 2019 and 2021, we only enrolled patients presenting a diagnosis of IDH-wt GBM through immunohistochemical detection of IDH1/IDH2 mutations. After 2021, only GBM IDH-wt, according to the latest WHO classification, was included. Pathological and molecular findings such as MGMT promoter methylation status, Ki-67 index, and p53 expression were reported. These parameters were included in the multivariate analysis to help identify the independent role of the study metabolites in predicting survival, given their well-known role in GBM. Indeed, MGMT promoter methylation is associated with better response to alkylating agents like temozolomide, the Ki-67 index reflects the proliferative activity of the tumor, and p53 expression is linked to tumor suppression and genomic stability.18,19
The TERT mutation was not routinely tested before 2021 in our center. Therefore, only a few patients had the mutation status revealed in this cohort.
Tumoral volumes were calculated on preoperative MRI imaging using cranial planning Brainlab software (Munich, Germany, 2021) on fluid-attenuated inversion recovery (FLAIR), T2-weighted (T2), and T1-weighted post-contrast sequences (T1-CE). The extent of resection (EOR) was calculated based on RANO criteria 20 by 2 independent neurosurgeons on postoperative MRI scans performed for radiotherapy planning or immediate postoperative CT/MRI scans when available.
Baseline, adjuvant therapies, and follow-up clinical data were retrospectively retrieved from clinical records and included age, sex, and performance status.
Serum from n = 27 healthy volunteers was collected and analyzed as control samples. The control group had a higher female/male ratio (2 vs 0.55, P = .01) and younger age (42 ± 11 vs 64.2 ± 10, P < .001) compared to the GBM group.
Blood sampling and processing
Preoperative peripheral blood samplings were routinely performed upon hospital admission, before any treatment, within 24 hours of surgery. Specimens were collected and stored in EDTA vacutainer venous blood collection tubes and separated by centrifugation (1500g, 15 minutes). The serum was then separated and stored at −80°C until the High-Performance Liquid Chromatography (HPLC) analysis was performed.
Analysis of serum levels of TRP, KYN, 5-HTP, and 5-HT
According to a well-validated method in our laboratory, 21 the separation and quantification of TRP, 5-HTP, and 5-HT were done with a Shimadzu LC-10AD HPLC system equipped with a Shimadzu RF-10AXL fluorometric detector set at excitation and emission wavelengths of 279 and 320 nm, respectively. Briefly, the chromatographic method consisted of an Apollo C18 (5 μm 250 mm × 4.6 mm) column (Sepachrom Mega Srl, Milan, Italy) and a mobile phase at a flow rate of 1 mL/min composed by 5% of a mix milliQ water/acetonitrile (5% water, 95% acetonitrile) and 95% of a mix milliQ water/methanol (90% water, 10% methanol) acidified with orthophosphoric acid to a pH of 3.5. KYN was quantified using the same mobile phase, a Robusta C18 (5 μm 250 mm × 4.6 mm) column (Sepachrom Mega Srl), and a Shimadzu SPD-10A UV–VIS detector set at 360 nm.
Statistical analysis
Statistical analysis was conducted using R Core Team (2022), using survival (Therneau T, 2023), ggsurvfit (Sjoberg D, 2023), maxstat (Hothorn T, 2017), and ggplot2 (Wickham H, 2016) packages. Categorical variables are reported as absolute numbers and percentages, whereas continuous variables are reported as mean and standard deviation or median and interquartile range (IQR). The difference in baseline characteristics and the unadjusted univariate analyses were performed using the t-test or Mann-Whitney (U-test) in accordance with the normality of the distribution and Chi-square or Fisher’s exact test, where appropriate. Pearson’s correlation test was used to infer associations between demographics and serum biomarkers.
The continuous variables of interest (TRP, KYN, 5-HTP, 5-HT, 5-HT/TRP, KYN/TRP, 5-HTP/TRP) were dichotomized using the maximally selected rank statistics. The Kaplan-Meier method was used to estimate OS and PFS in the study population using the newly dichotomized variables. Log-rank tests analyzed differences between groups. Multivariate Cox regression analyses were used to detect variables associated with increased overall survival times. The results of all tests are presented as P-values, and statistical significance was set as a probability value of .05 (95% confidence interval).
Results
Patients and pathological characteristics
A total of 65 patients affected by IDH-wt GBM were originally screened. N = 3 cases with perioperative mortality (<1 month) were excluded: 2 suffered respiratory complications, one postoperative hemorrhage. Overall, 62 patients were included in the final analysis. The mean age was 64.2 ± 10.5 years. Most of the patients were male (n = 40, 65%) and aged <65 years old (n = 34, 54%). Globally, patients displayed a good functional status (KPS >80) in 60% of cases. Patients have been clinically followed up for a median of 13.7 months. See Table 1 for a summary of the baseline characteristics of included patients and lesions. Assessment of MGMT promoter status was available in 57 patients (92%) and revealed promoter hypermethylation in 39% of cases. The quantitative analysis of ki67 and p53 expression (positive cells) reported a mean of 28% ± 16 and 17% ± 22, respectively. The mean T1-CE volume was 28.1 cm3 ± 21.6. The extent of resection (EOR) was reported as complete or near total in 47 (76%), subtotal or partial in 15 (24%).
Baseline characteristics of included patients and lesions characteristics.
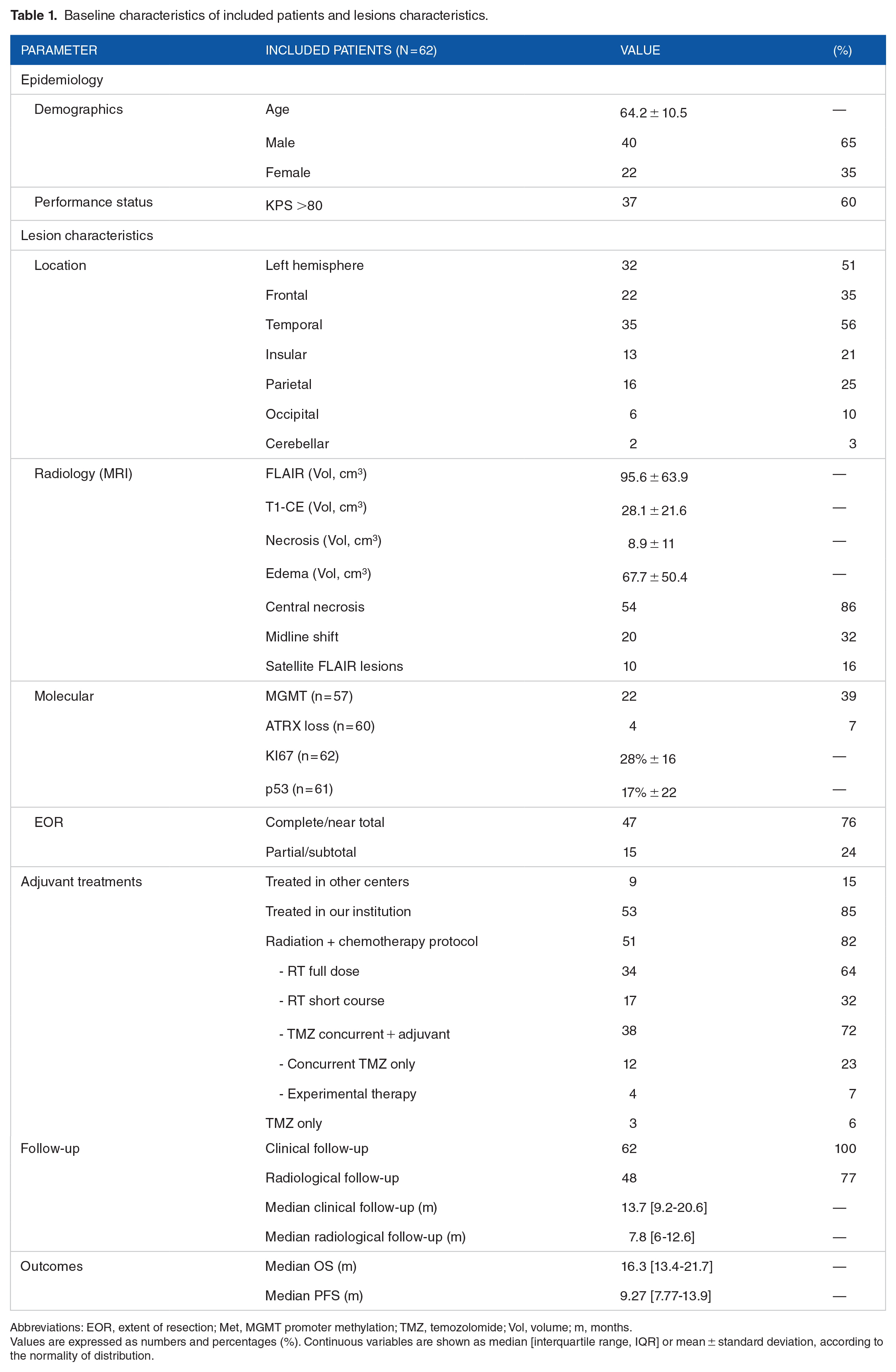
Abbreviations: EOR, extent of resection; Met, MGMT promoter methylation; TMZ, temozolomide; Vol, volume; m, months.
Values are expressed as numbers and percentages (%). Continuous variables are shown as median [interquartile range, IQR] or mean ± standard deviation, according to the normality of distribution.
Post-operative data on adjuvant radiotherapy (RT) and chemotherapy with temozolomide (TMZ) was available for the n = 53 (85%) patients treated in our Institutional radiotherapy and oncological services. Among them, n = 51 (94.6%) underwent radiation + chemotherapy protocol, n = 34 with full-dose (60 Gy, 30 fractions), and n = 17 with short-course (40 Gy, 15 fractions) RT regimens. N = 38 patients received concurrent + adjuvant TMZ regimen, while n = 12 only received the concurrent schedule without additional TMZ. Additional n = 4 patients received RT + experimental therapy.
Baseline blood markers
Pearson correlation analyses revealed no association between age and TRP (r = .03, P = .77), KYN (r = −.17, P = .16), 5-HTP (r = .16, P = .21), and 5-HT (r = −.16, P = .20). Similarly, sex was not correlated to TRP (r = −.05, P = .69), KYN (r = −.007, P = .95), 5-HTP (r = −.14, P = .25, and 5-HT (r = .14, P = .25).
Mean baseline serum levels of 5-HT (76.9 ng/mL ± 45.9 vs 326.8 ng/mL ± 85.3, P < .001), KYN (61.2 ng/mL ± 33 vs 465 ng/mL ± 132.4, P < .001), KYN/TRP (6.40 ± 3.95 vs 44.9 ± 17.3, P < .001), 5-HT/TRP (7.86 ± 4.51 vs 31.2 ± 11.5, P < .001), and 5-HTP/TRP (1.93 ± 2.16 vs 6.99 ± 3.77, P < .001) were significantly lower in patients with GBM than in controls. A tendency for lower TRP levels in GBM than in controls (9.95 µg/mL ± 2.36 vs 10.9 µg/mL ± 2.51, P = .06) was also observed. See Table 2 and Figure 2.
Baseline blood markers comparison between GBM and healthy controls.

Values are expressed as mean ± standard deviation.
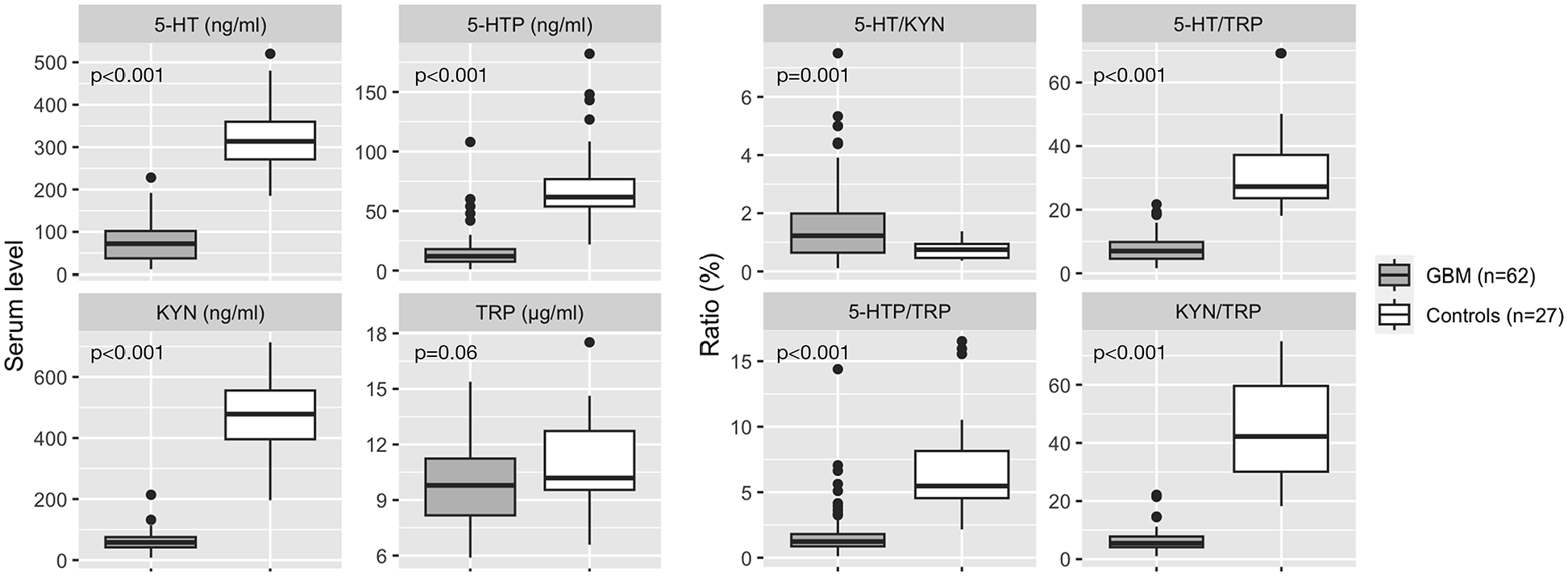
Comparison of GBM versus healthy controls baseline peripheral KP and SP metabolites. Boxplots represent median and interquartile ranges.
Overall survival (OS) analysis
Pre-operative laboratory parameters
The serum marker values were dichotomized using the maximally selected rank statistics for OS. The retrieved threshold for survival analysis was: TRP 10.6 µg/mL (P = 1.00); 5-HT 78 ng/mL (P = .14); KYN 18 ng/mL (P = .67); KYN/TRP <2.55 (P = 1.00); 5-HTP/TRP 0.89 (P = .43); 5-HT/TRP <5.78 (P = 1.00).
The median OS in the whole cohort of GBM patients was 16.3 months (95% CI: 13.4-21.7). Patients with 5-HT <78 ng/mL had a median OS of 14.4 months (95% CI: 11.4-17.6) compared to 22.5 months (95% CI: 17.7-27) in patients with 5-HT >78 ng/mL, P = .01. Shorter OS was recorded in patients with KYN <18 ng/mL (9.8 months, 95% CI: 11.4-17.6 vs 17.5 months, 95% CI: 14.4-22.5, P = .002), KYN/TRP <2.55 (11.4 months, 95% CI: 10.4-15.6 vs 17.1 months, 95% CI: 14.0-28.6, P = .002), 5-HTP/TRP <0.89 (11.5 months, 95% CI: 10.9-20.6 vs 17.6 months, 95% CI: 14.4-32.4, P = .02), and 5-HT/TRP <5.78 (13.4 months, 95% CI: 11.4-15.6 vs 19.1 months, 95% CI: 14.0-28.6, P = .02). See Figure 3.

Kaplan-Meier curves for overall survival (OS) analysis. (A) 5-HT (ng/mL) levels high versus low. (B) KYN levels (ng/mL) high versus low. (C) 5-HT/TRP ratio high versus low. (D) KYN/TRP ratio high versus low.
Age, sex, performance status, MGMT methylation status
Patients older than 65 years had a median OS of 14.0 months (95% CI: 10.5-16.4), compared to 20.6 months (95% CI: 17.6-32.4, log-rank test P = .03) in younger patients. The median OS for the patients with low (<70) and high (⩾80) KPS were 16.1 (95% CI: 12.9-22.5) and 16.3 (95% CI: 10.5-22.4), respectively (log-rank test P = .90). Survival analysis for sex and MGMT methylation status did not reveal OS differences in our cohort of patients (P = .70 and P = .50, respectively), see Figure S1.
Progression-free survival (PFS) analysis
The median PFS in the whole cohort was 16.3 months (95% CI: 13.4-21.7). Patients with 5-HT <78 ng/mL had a median PFS of 8.6 months (95% CI: 7.1-12.6) compared to 15.3 months (95% CI: 9.3-21.4) in patients with 5-HT >78 ng/mL, P = .04. Shorter PFS was detected in patients with KYN <18 ng/mL (7.1 months, 95% CI: 4.70-11.3 vs 9.3 months, 95% CI: 8.63-15.4, P = .02), KYN/TRP <2.55 (5.9 months, 95% CI: 3.77-9.8 vs 11 months, 95% CI: 8.63-15.4, P = .005), and 5-HT/TRP <5.78 (7.77 months, 95% CI: 4.70-10.4 vs 12.3 months, 95% CI: 8.63-17.8, P = .001), see Figure 4.
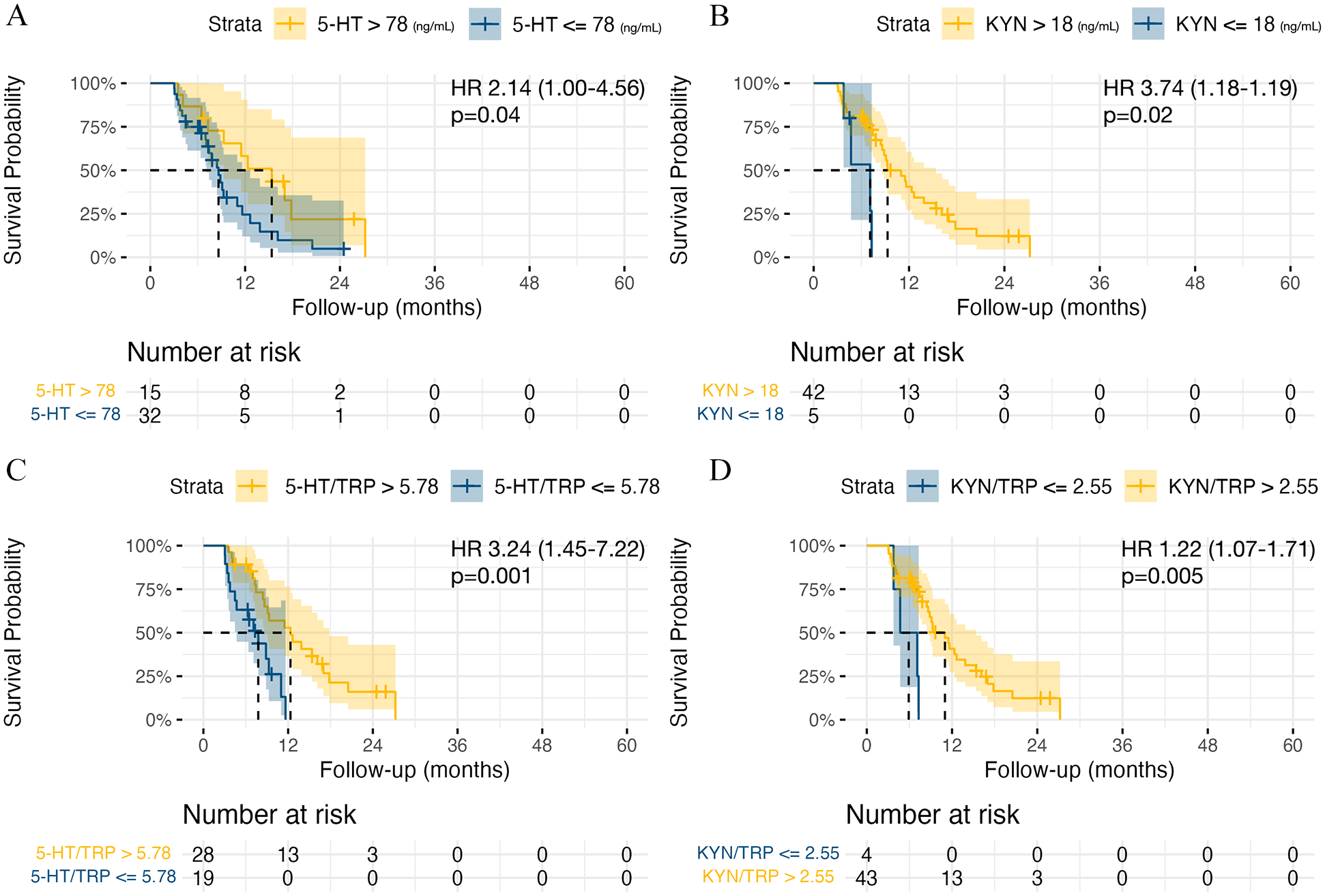
Kaplan-Meier curves for progression-free survival (PFS) analysis. (A) 5-HT levels high versus low. (B) KYN levels high versus low. (C) 5-HT/TRP ratio high versus low. (D) KYN/TRP ratio high versus low.
Cox regression analysis
Multivariate Cox regression analysis of OS accounting for demographics, tumoral characteristics, EOR, and adjuvant therapies (R2 = 75%, P < .001) confirmed increased age (HR: 1.09, 95% CI: 1.02-1.15, P = .005), mean platelet volume (MPV, HR: 2.70, 95% CI: 1.40-5.22, P = .002), and Ki67 (HR: 62.9, 95% CI: 3.47-1142, P = .005) being associated with worse OS. Contrarily, increased baseline 5-HT (HR: 0.98, 95% CI: 0.97-0.99, P = .03), KYN (HR: 0.97, 95% CI: 0.94-0.99, P = .02), and 5-HTP (HR: 0.88, 95% CI: 0.81-0.96, P = .005) were independent predictors of increased OS. Increased 5-HT levels were also associated with longer PFS (HR: 0.97, 95% CI: 0.96-0.99, P = .001), together with increased 5-HTP (HR: 0.91, 95% CI: 0.85-0.97, P = .004). Increased MPV predicted worse PFS (HR: 2.69, 95% CI: 1.25-5.79, P = .001), Table 3.
Multivariate Cox regression analysis of overall survival (OS) and progression-free survival (PFS) controlling for demographic and lesional parameters.
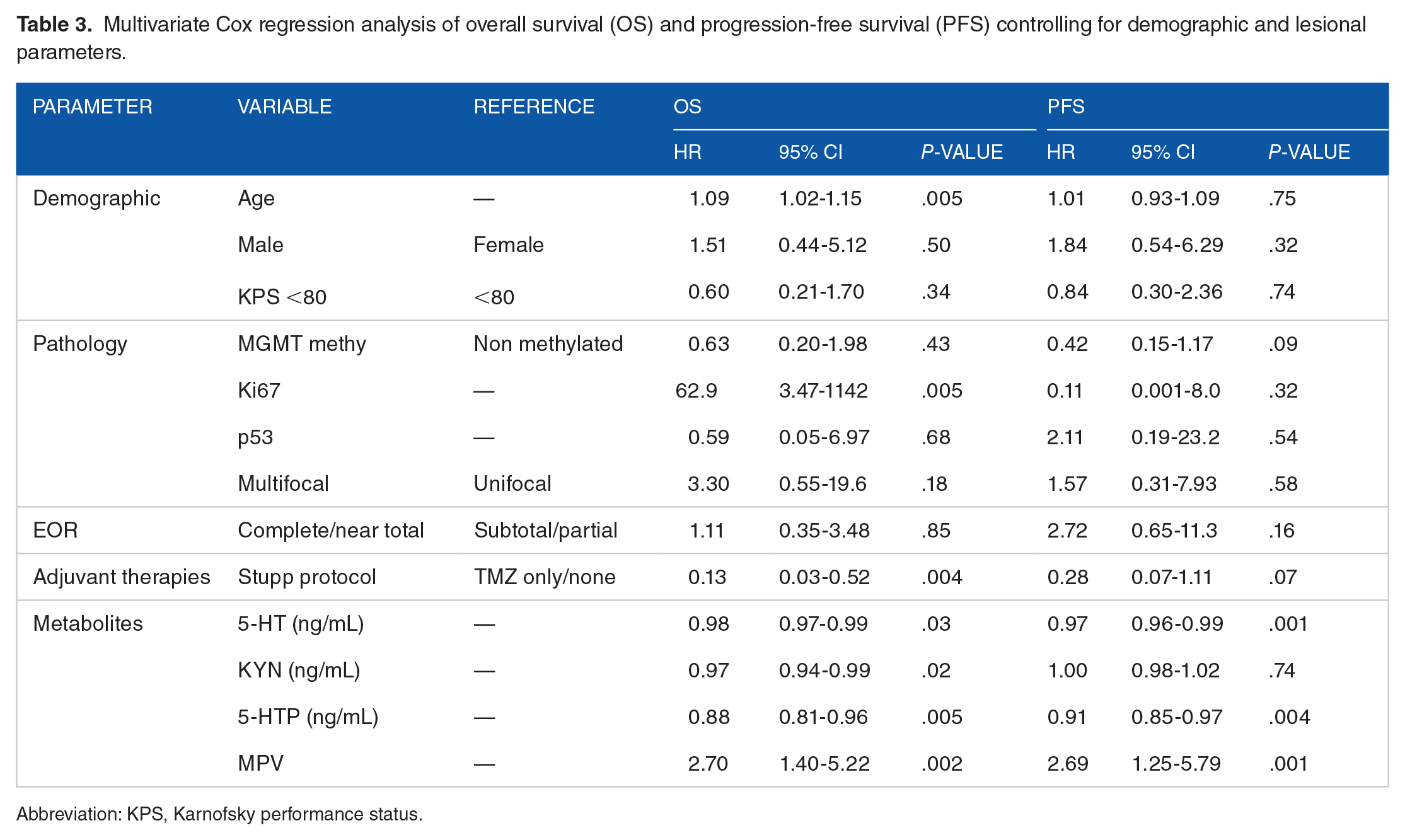
Abbreviation: KPS, Karnofsky performance status.
Discussion
This study revealed that TRP metabolism is significantly altered in GBM patients, as reflected in a reduction of serum levels of KYN, 5-HTP, and 5-HT compared with healthy controls. Greater decreases are associated with worse OS and are independent risk factors for decreased survival. Additionally, lower 5-HTP and 5-HT are associated with decreased PFS.
Tryptophan metabolism via the Kynurenine pathway in GBM
The KP has received increasing attention in cancer pathology due to its implication in tumor progression and immune modulation.22,23 Its involvement has been described in different tumors, including primary brain, breast, lung, colon, and renal cancer. The induction of the rate-limiting enzymes initiating the KYN pathway (IDO-1/2 and TDO-2) is the key aspect of KP activation in GBM. Tumoral cells express IDO1 due to the loss of tumor-suppressor gene Bin1 14 or when identified by tumor-infiltrating immune cells and exposed to pro-inflammatory cytokine IFN-γ.24,25 Concerning other enzymes of the KP, it has been shown that the kynurenine aminotransferases (KAT) are downregulated, 4 while the quinolinate phosphoribosyl-transferase (QPRT) that catalyzes the conversion of quinolinic acid (QA) to nicotinic acid mononucleotide, the final step in the biosynthesis of NAD+ coenzyme, is hyperactive, thus conferring tumor survival advantages, assisting in cell replication, and maintaining high metabolic activity. 9 The up-regulation of key KP enzymes has been observed in other cells comprising the tumoral microenvironment (particularly the tumor-infiltrating macrophages and other antigen-presenting cells) and is known to inhibit tumor-specific immunity through a combined effect of depletion of TRP26,27 and direct activity of KP intermediates (ie, KYN, 3-HAA, and 3-HK) that are associated with a reduction of CD8+ cytotoxic T-cells infiltrate and a significant increase in the immunosuppressant CD4+/CD25+/FOXP3+ (Tregs) phenotype. 10 Particularly, TRP starvation is known to promote T-cell autophagy and anergy, 27 while KYN potently binds to cytoplasmic aryl hydrocarbon receptor (AhR), which promotes a crude reduction of CD8+ T-cell infiltration.28,29 All these elements contribute to the peri- and intra-tumoral accumulation of immunosuppressive T-regs that may worsen GBM overall prognosis.10,30 Higher KP activity is also associated with resistance to treatments such as immune checkpoint inhibitors (anti-PD-1), ipilimumab (anti-CTLA-4), NAD+ inhibitors, and chemotherapeutic agents such as cisplatin and paclitaxel.31,32
To sum up, KP hyperactivity is thought to promote tumoral persistence by skewing the KP away from the neuroprotective branches of the pathway, increasing the tumor’s ability to produce NAD+ to support cellular metabolism and proliferation, and mediating brain tumor immune evasion and resistance to therapies. Over-activation of KP is known to increase the consumption of TRP by glioma cells and a substantial modification in downstream KP substrate concentrations within cells and the peritumoral micro-environment. The final changes in local concentrations of substrates are partially reflected in peripheral blood through the brain-blood barrier. Only a few authors have analyzed the peripheral levels of KP substrates in GBM models and patients and found the KYN/TRP ratio as a promising biomarker for predicting the clinicopathologic status of tumors to GBM progression. Adams et al 4 characterized the KP in plasma from GBM patients (n = 18) and demonstrated an overall reduction of TRP (−61%) and KYN (−37%) when compared to healthy individuals. They found increased KYN/TRP (+188%) in their cohort of patients due to the more consistent decrease in TRP in relation to KYN. Little information was given regarding the nature of controls and the timing of sampling, and the study analyzed a non-homogeneous cohort of GBM (naïve and recurrent), IDH-mutated and -wt, and other neuronal/glial malignancies. A recent report by Zhai et al 33 reported a significant reduction in serum KYN levels (−70%) and a slightly smaller decrease in TRP levels (−68%), resulting in decreased KYN/TRP ratio (−7%) in a small cohort series of GBM patients (n = 10) compared to controls. Interestingly, KYN and TRP levels returned to values comparable to those of controls 10 weeks post-surgery. Following the postoperative surge, an elevated KYN/TRP ratio was found to be predictive of poorer OS. The authors suggested that the late increase in the KYN/TRP ratio could be linked to the induction of IDO-1/2 or TDO2 by treatments such as chemo/radiotherapy and/or dexamethasone or to systemic IDO-1 induction associated with immune activation upon recurrence. Accordingly, Panitz et al, 34 analyzing a cohort of 43 recurrent GBM patients, observed a slightly more pronounced reduction in serum TRP levels (−18%) relative to KYN (−14%) compared to controls. They also noted that the magnitude of reduction decreased with the number of enzymatic steps between TRP and its metabolites, with the smallest reduction observed for downstream products. The authors proposed that this could be related to the overall TRP availability, which controls the levels of its systemic metabolites. In our study, which includes the largest homogeneous cohort of naïve-treatment IDH-wt patients, we observed a trend for TRP reduction (−10%) accompanied by a pronounced KYN depletion (−87%) and a corresponding decrease in the KYN/TRP ratio (−86%), before any treatment. This substantial reduction in KYN levels and the relatively lower KYN/TRP ratio in treatment-naive GBM aligns with the findings by Zhai et al 33 before disease recurrence. This data suggests that the extent of TRP metabolism dysregulation may vary depending on the stage of the disease and the treatments being administered, such as chemotherapy or radiotherapy. Notably, we found that a greater reduction in KYN levels before treatment was associated with poorer prognosis, underscoring the central role of TRP metabolism in GBM. Downstream of KYN, Adams et al 4 observed that both the neuroprotective compounds kynurenic acid (KYNA) and picolinic acid (PIC) levels were decreased in the serum of GBM patients, while Kesarwani et al 35 observed that quinolinic acid (QUIN) accumulates in GBM and contributes to immune tolerance state by polarizing macrophage toward an anti-inflammatory and tumor-supportive phenotype (M2-like).
Collectively, these data suggest that pre-operative KYN and TRP levels potentially reflect the extent of KP dysregulation in GBM and might exert a prognostic role in predicting improved survival times for patients offered surgery and adjuvant treatments. KP dysregulation has received increasing attention in cancer GBM and is under active investigation to develop molecules aiming to modulate its activity. Our study represents the largest cohort (n = 62) supporting the involvement of KP in GBM pathology and the ongoing efforts to target this pathway as a potential therapeutic approach.
Indeed, Preclinical models have shown that inhibiting the KP can attenuate GBM progressions. IDO1 inhibitors, such as INCB024360, have garnered increasing attention, 36 and subsequent studies have demonstrated that combining IDO1 inhibition with immune checkpoint inhibitors could significantly enhance therapeutic efficacy. 37 One study found that dual inhibition of IDO-1 and TDO with PVZB3001 suppressed tumoral growth and decreased the KYN/TRP ratio. 38 Building on these promising preclinical findings, clinical trials are now investigating the efficacy of IDO-1 inhibitors in GBM. Several IDO1 enzyme inhibitors or KP modulators are currently being evaluated in clinical trials (for a detailed review, see Tang et al 39 ). Notable trials include Epacadostat in conjunction with radio/chemotherapy and bevacizumab (NCT03532295), BMS-986205 with nivolumab/radiotherapy or chemoradiotherapy (NCT04047706), and indoximod combined with TMZ, radiation therapy (NCT025027), and bevacizumab (NCT02052), which are currently recruiting recurrent GBM patients. Interestingly, in a phase 1 trial, the IDO-1 inhibitor PF-06840003 demonstrated disease control in 47% of patients (n = 8). 40 These promising results highlight the importance of further investigating KP dysregulation in GBM patients and suggest that evaluating TRP metabolites could aid in the identification of patient cohorts most likely to benefit from targeted interventions aimed at these pathways.
Tryptophan metabolism via Serotonin pathway in GBM
The SP, the other major metabolic pathway of TRP, has been far less investigated in GBM pathology compared to the KP. In this work, we observed significantly reduced systemic 5-HT levels in treatment-naïve GBM patients. To date, no other studies have addressed the peripheral levels of 5-HT in GBM. The critical step in 5-HT synthesis is highly dependent on TRP availability, and changes in TRP are known to impact both the systemic and brain 5-HT production.11,14,29 Thus, it can be hypothesized that the observed reduction in TRP levels could influence the overall 5-HT synthesis.
The exact role of 5-HT in GBM remains unexplored for several aspects. 8 5-HT has been implicated in the modulation of several cancer-related pathways, including PI3K/AKT and MAPK, which are critical for cell survival and proliferation. 41 Moreover, various 5-HT receptors are expressed on GBM cells, suggesting that they may influence tumor behavior via receptor-mediated mechanisms. However, only a limited number of studies have examined the expression of 5-HT receptors in GBM models. For example, the 5-HTR5A was found to be downregulated in GBM compared to low-grade gliomas, and its stimulation enhanced GBM autophagy and apoptosis. 22 Conversely, the 5-HTR7A was identified as promoting IL-6 release by U373 GBM cells, thereby facilitating tumoral progression via an autocrine pathway. 23 Additionally, 5-HT may modulate the tumor microenvironment, potentially influencing the immune response to tumors. 5-HT has been shown to affect the behavior of macrophages and T-cells, which play critical roles in tumor immune surveillance. Although 5-HT signaling has been explored in T cells, the precise mechanism by which it influences T cell differentiation and functions remains unclear. Given that the specific pattern of 5-HT receptor expression involved in GBM progression has not been fully characterized, the extent and nature of SP dysregulation in GBM remain uncertain.
In this study, reduced systemic 5-HT levels were associated with poorer OS despite standard-of-care treatments. This result might be explained by the specific pattern of 5-HT receptors in GBM cells, which could promote a more aggressive course of the disease when systemic 5-HT levels are reduced. Alternatively, systemic platelet activation could account for this reduction. Peripheral 5-HT is primarily synthesized in enterochromaffin cells, released into the bloodstream, and absorbed by circulating platelets, forming a potent 5-HT reservoir. In the thrombotic milieu of tumors, platelet aggregation frequently occurs, leading to the release of large amounts of 5-HT, which may result in higher local concentrations than those in the bloodstream. Interestingly, systemic platelet activation has been linked to GBM progression and angiogenesis.42,43 In this study, elevated MPV further confirmed its independent predictive role for poorer OS. In summary, 5-HT appears to play a multifaceted role in GBM biology, influencing tumor growth in the tumor microenvironment and potentially offering new avenues for therapeutic intervention. Most clinical studies on 5-HT and cancer have focused on the relationship between tumors and depressive disorders, as well as the use of antidepressants. Depression and psychological distress are common in cancer patients, 44 and in those with primary malignant brain tumors, it has been associated with a poorer OS.45,46 This has led to the hypothesis that selective serotonin reuptake inhibitor (SSRI) might positively affect the survival of patients with GBM. Caudill et al 47 reported a survival benefit with the use of SSRIs, though this finding was not replicated in a larger retrospective study (n = 497). 48 These data raise the question of whether the negative impact of low serum levels of 5-HT on OS and PFS in GBM is an indirect effect of metabolic imbalance toward KP, a direct consequence of systemic depletion of 5-HT, possibly including its downstream metabolite melatonin, or both. Additional studies are necessary to clarify the role of TRP metabolism in GBM. The lack of clarity regarding which 5-HT receptors are involved complicates our understanding of how 5-HT influences tumor behavior, whether it promotes or inhibits growth, and how it might be leveraged therapeutically. Further research is necessary to delineate these pathways and determine how they could be exploited in treating glioblastoma.
Limitations
The main limitations of this study include the monocentric design, the relatively small sample size, the absence of comprehensive molecular analysis (eg, full MGMT methylation profile, TERT, and EGFR mutations), and the lack of assessment of downstream metabolites of KYN. The study did not include a longitudinal analysis of the biomarkers following surgery and treatment and did not provide a sex-matched control cohort.
Conclusions
This study unveils an intriguing connection between KP activation, TRP consumption, and a relative reduction in 5-HT synthesis, potentially providing GBM cells with a survival advantage. Additional studies are needed to further analyze this interrelation and to identify the individual contributions of KP, SP, platelets activation, and immune infiltrate in the progression of GBM. In particular, the analysis of downstream metabolites of the KP will help to have a better biological framework for linking TRP metabolism to GBM. This approach could lay the groundwork for exploring the therapeutic potential of interventions targeting these systems, thereby enhancing therapeutic approaches for GBM patients.
Supplemental Material
sj-tif-1-try-10.1177_11786469241312475 – Supplemental material for Preoperative Peripheral Blood Serotonin and Kynurenine Levels Are Associated With Oncological Outcomes in Glioblastoma IDH-wt Patients
Supplemental material, sj-tif-1-try-10.1177_11786469241312475 for Preoperative Peripheral Blood Serotonin and Kynurenine Levels Are Associated With Oncological Outcomes in Glioblastoma IDH-wt Patients by Silvia Snider, Filippo Gagliardi, Pierfrancesco De Domenico, Stefano Comai, Antonella Bertazzo, Sofia Nasini, Benedetta Barzon, Angela Ruban, Francesca Roncelli and Pietro Mortini in International Journal of Tryptophan Research
Footnotes
Author Contributions
All authors contributed to the study’s conception and design. Silvia Snider (SS) and Filippo Gagliardi (FG) contributed equally to this work. PDD, SS, SC, and FG performed material preparation, data collection strategy, and analysis. SC, AB, SF, and BB analyzed tryptophan metabolites via serotonin and kynurenine. The first draft of the manuscript was written by SS, FG, and PDD, and all authors commented on previous versions. All authors read and approved the final manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The non-profit association “Amici di Tosco,” Merate (Italy)—94038420132, funded this research.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was performed in line with the principles of the Declaration of Helsinki. Data were retrospectively collected after approval from the internal Ethics Committee (internal protocol ID: NCH 02-2020) granted on 13th July 2022. Upon hospital admission, each patient signed written consent for the treatment and collection and analysis of personal data and specimens stored in IRCCS San Raffaele institutional Biobank, Milan, Italy (internal protocol ID: NCH 06-2020, internal Ethics Committee approval 15th July 2020).
Patient Consent for Publication
Informed consent for publication of clinical data was obtained from all individual participants included in the study.
Data Availability
The datasets generated during and analyzed in this study are available from the corresponding author upon reasonable request. Present data are original; they will be accessed based on journal policies.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
